Abstract
Introduction
The most accepted theory regarding the mechanism by which laser irradiation enhances enamel acid resistance is the reduction of bound carbonate, when the enamel surface is heated in the range of 100–400°C. 16 –18 Nevertheless, the modification of organic matter has been reported as one of the mechanisms in laser-induced caries prevention (organic blocking theory). 19
On the other hand, there is a high caries prevalence on occlusal surfaces of permanent molars. 20,21 Although this surface is considered a risk area for the development of caries lesions, there are no previous reports in the literature that assess the effects of Er:YAG laser irradiation on these surfaces, research having been carried out only on smooth surfaces. Consequently, the purpose of this study was to evaluate in vitro the acid resistance and the structural changes of the occlusal enamel surface of permanent molars after subablative Er:YAG laser irradiation.
Materials and Methods
Tooth selection and sample preparation
The study protocol was reviewed and approved by the Research and Ethics Committee at the Autonomous University of the State of Mexico. All subjects enrolled in this research signed an informed consent. Thirty unerupted permanent third molar teeth extracted from 14 patients for therapeutic reasons were stored in 0.1 (w/v) thymol solution at 4°C until the experiment was begun. The crown was separated from the root in each tooth by means of a diamond disc (BesQual, New York, NY) mounted on a low-speed motor (Brasseler, Savannah, GA) under distilled water irrigation to prevent dehydration. The crown was fixed to a glass slide with thermoplasticized epoxy resin (Allied, Rancho Dominguez, CA). Afterwards, a diamond wheel (South Bay Technology, Inc., San Clemente, CA) mounted on a cutter (South Bay Technology, Inc.) was employed to obtain the samples under constant irrigation. Mesiodistal cuts were made to eliminate lingual and buccal surfaces from the crown, separating the central portion of the occlusal surface to include the fissure. Each of these slices was also sectioned to obtain four samples 1.5 mm wide × 4 mm in length (one for each group). The samples were rinsed with deionized water and dried at room temperature. Finally, the surface of each sample, except the occlusal region, was coated with an acid-resistant varnish.
Er:YAG laser irradiation
An Er:YAG laser system (Lumenis OPUS DUO™ Er: YAG + CO2, Yokneam, Israel) was used to irradiate the samples. The main experimental parameters of the laser utilized during the study were wavelength fixed at 2.94 μm, energy pulse from 100 to 150 mJ, pulse repetition rate of 10 Hz, pulse duration of 250–400 μsec, and an exit tip diameter from 1.0 to 1.3 mm. One hundred twenty occlusal enamel samples were divided into four groups (n = 30), as follows: Group I was the control (no laser irradiation); Group II was irradiated at 100 mJ (12.7 J/cm2) with a sapphire tip 10,13 of 1.0 mm in diameter; Group III was irradiated at 100 mJ (7.5 J/cm2) with a sapphire tip of 1.3 mm in diameter; and Group IV was irradiated at 150 mJ (11 J/cm2) with a sapphire tip of 1.3 mm in diameter. Energy levels were calibrated with the calipers of the equipment, and the energy delivered was measured periodically with a power meter (LaserMate-P, Coherent Co., Santa Clara, CA). The irradiation was manually performed 8,13 in one direction such that the tips were smoothly scanned perpendicular to the samples' enamel surface while distilled water was sprayed (5.0 mL/min) to reduce heating. Each sample was irradiated only once during a time period of 18 sec while the tip-sample distance was kept fixed at 1 mm. A sheet of stainless steel (23 mm × 5 mm × 0.5 mm) was fixed to the top of the laser handpiece to carry out this task. At that tip-sample distance, the exit tip and the laser beam had the same diameter, as corroborated with a laminated infrared sensor screen (Lumitek International, Inc., Ijamsville, MD).
Atomic absorption spectrometry
After laser treatment, the specimens were placed in a lactic acid solution (0.1 M, pH 4.8) 7 and incubated at 37°C in 100% humidity for 24 h. Then, the samples were removed and rinsed with deionized water in the test tube to get rid of Ca residues from the surface. This procedure was followed in order to obtain the Ca concentration from the samples by atomic absorption spectrometry (Varian SpectrAA, Mulgrave, Australia).
Energy dispersive X-ray spectroscopy
After acid dissolution, 48 samples (12 per group) were dried at room temperature and attached to a testing ring using an adhesive carbon paper (SPI Supplies, USA) to determine the weight percentages (% wt) of Ca, phosphorus (P), oxygen (O), and chlorine (Cl) by energy dispersive X-ray spectroscopy (EDS) (ThermoNoran Superdry, Thermo Scientific, Waltham, MA).
Scanning electron microscopy analysis of the enamel surface
To prevent charge effects on the samples, a thin gold layer (vacuum evaporator, JEOL, Tokyo, Japan) was deposited on the surface of four samples (one per group) to be observed by scanning electron microscopy (SEM) (JSM-5300; JEOL).
Statistical analysis
All data were analyzed using the SPSS 13.0 for Windows statistical package (SPSS Inc., Chicago, IL, USA). The measurements were analyzed using the Kolmogorov–Smirnov test at a p ≤ 0.05 level of significance to assess the distribution of the data. One-way analysis of variance (ANOVA) was used to determine differences in the concentration of calcium among the groups. The Kruskal–Wallis test was performed to compare the percent by weight of the different elements, with a level of significance designated as p ≤ 0.05.
Results
Atomic absorption spectrometry
The data dispersion was similar among all groups, as shown in Fig. 1. The acid solution of Group IV showed the highest concentration of released Ca from the enamel while the lowest corresponded to Group I (control). No statistically significant differences were found (p = 0.997), as shown in Table 1.
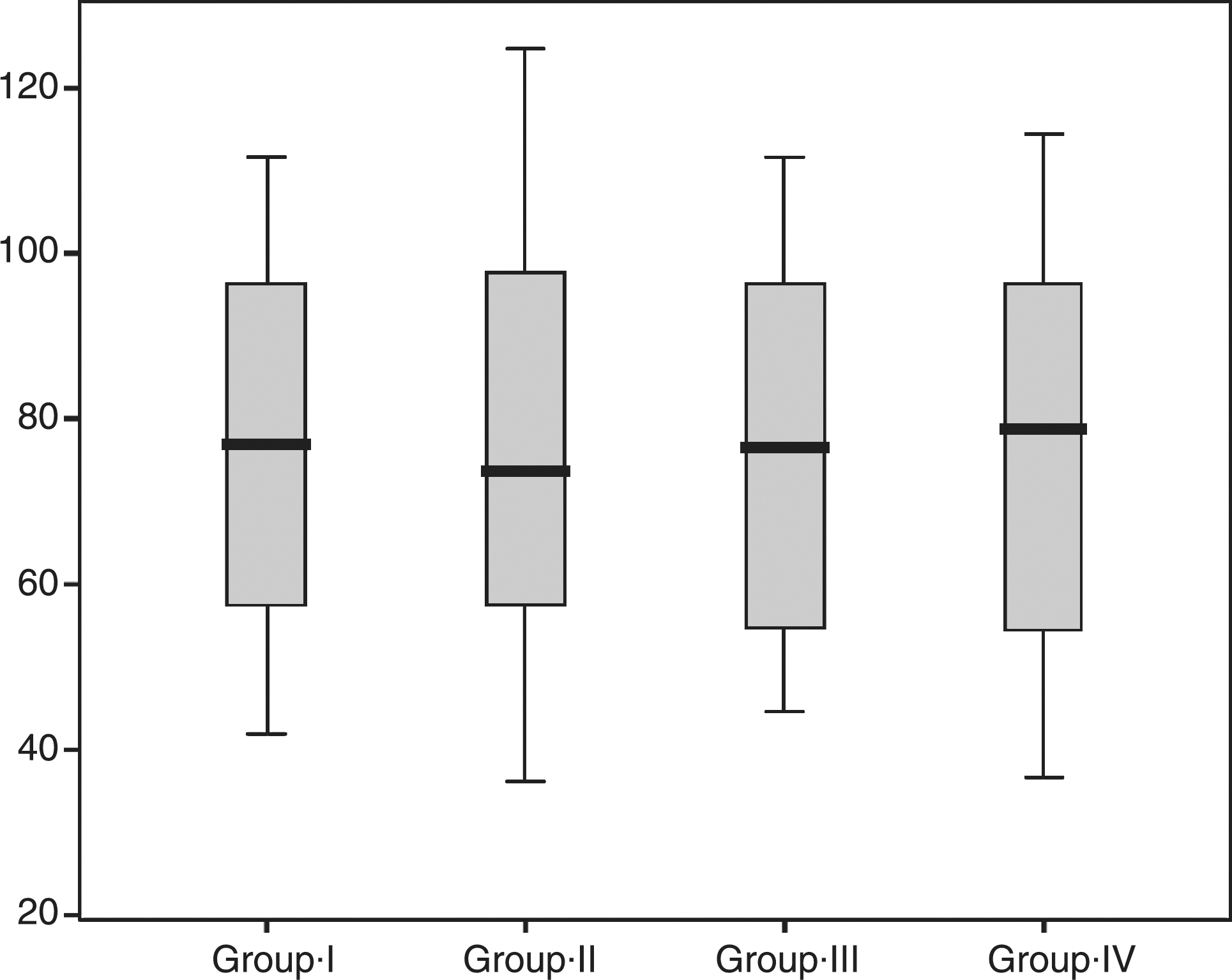
Box plots depicting Ca concentration in acid solution for control and Er:YAG irradiated groups. Data correspond to minimum, maximum, and median values.
SD, standard deviation.
EDS evaluation
The experimental data obtained by EDS for all groups regarding % wt of Ca, P, O, and Cl are displayed in Table 2. They show similar content for each element but the value for P from Group IV was observed to be slightly higher. In any case, no statistically significant differences were found (p = 0.402).
SEM surface analysis
Group I control (no laser irradiation) micrograph had exposed prisms caused by acid dilution, as shown in Fig. 2. All irradiated groups showed craters (Figs. 3a, 4a, 5a) as well as homogenous irradiation areas (Figs. 3b, 4b, 5b).

Scanning electron microscopy (SEM) micrograph representative of samples from the control group showing exposed prisms due to enamel acid dilution. ( × 2000). Scale bar = 10 μm.
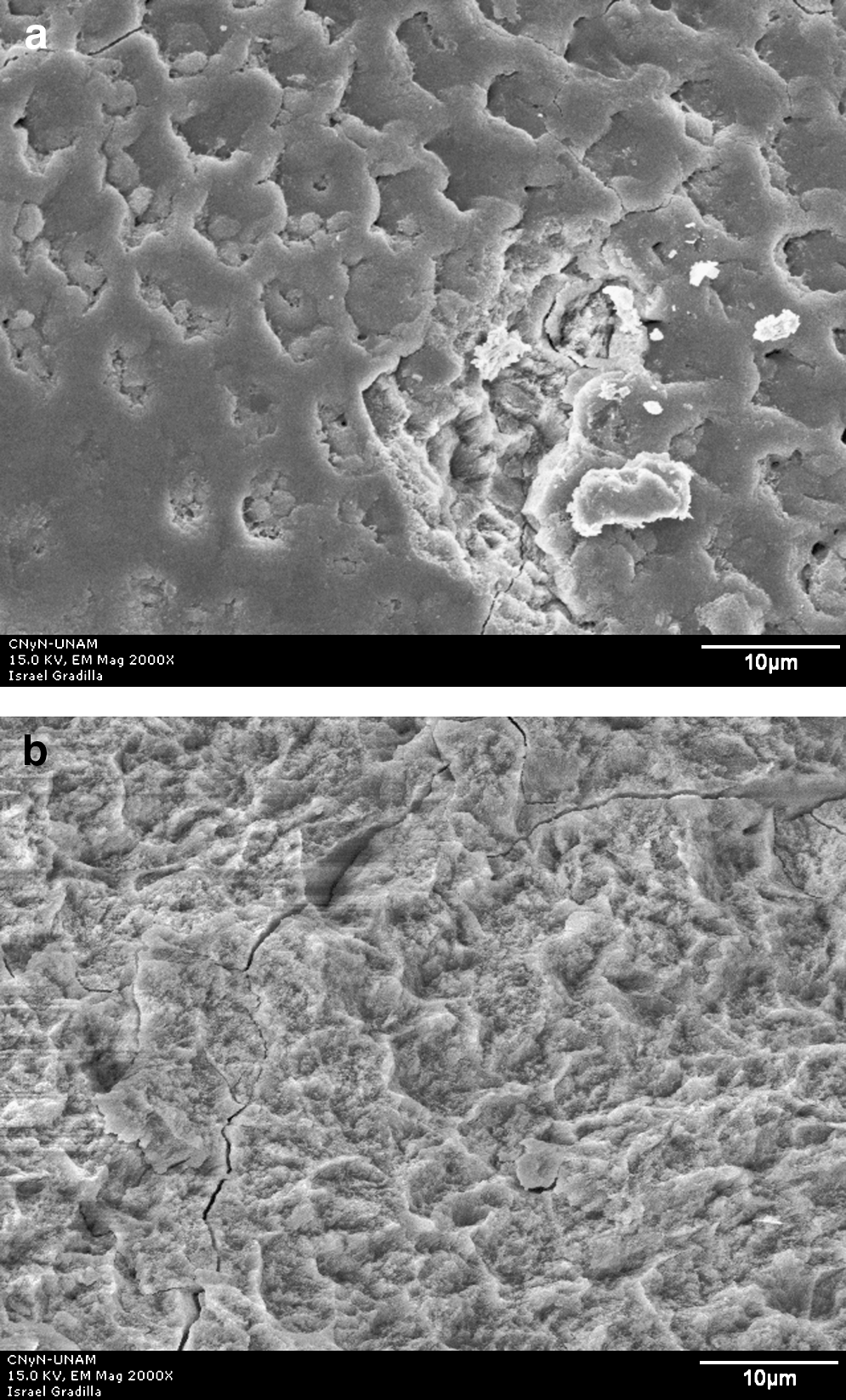
SEM micrograph of enamel surface after Er:YAG laser irradiation at 100 mJ/10 Hz and 12.7 J/cm2 and acid dissolution. (
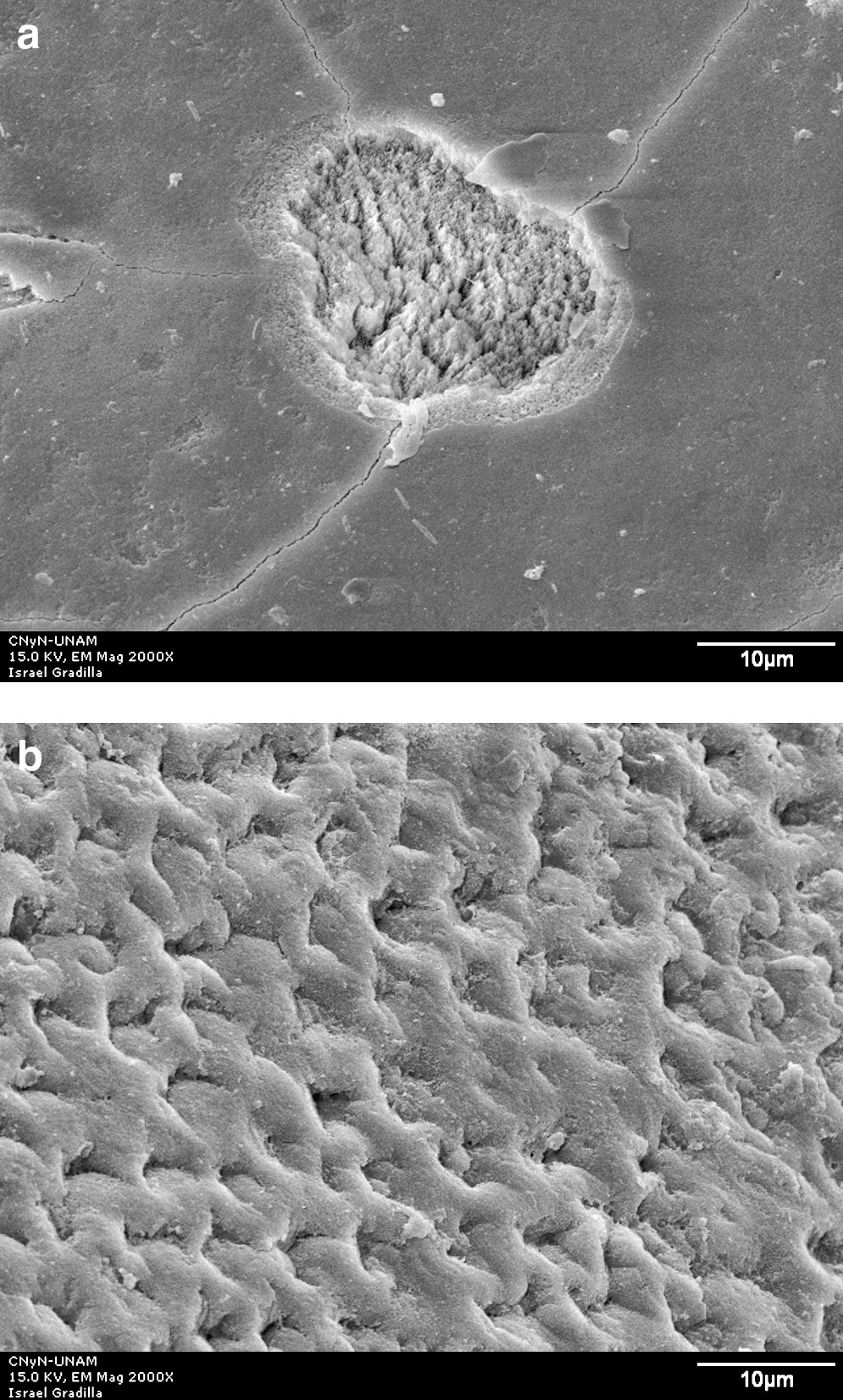
SEM micrograph of enamel surface after Er:YAG laser irradiation at 100 mJ/10 Hz and 7.5 J/cm2 and acid dissolution. (
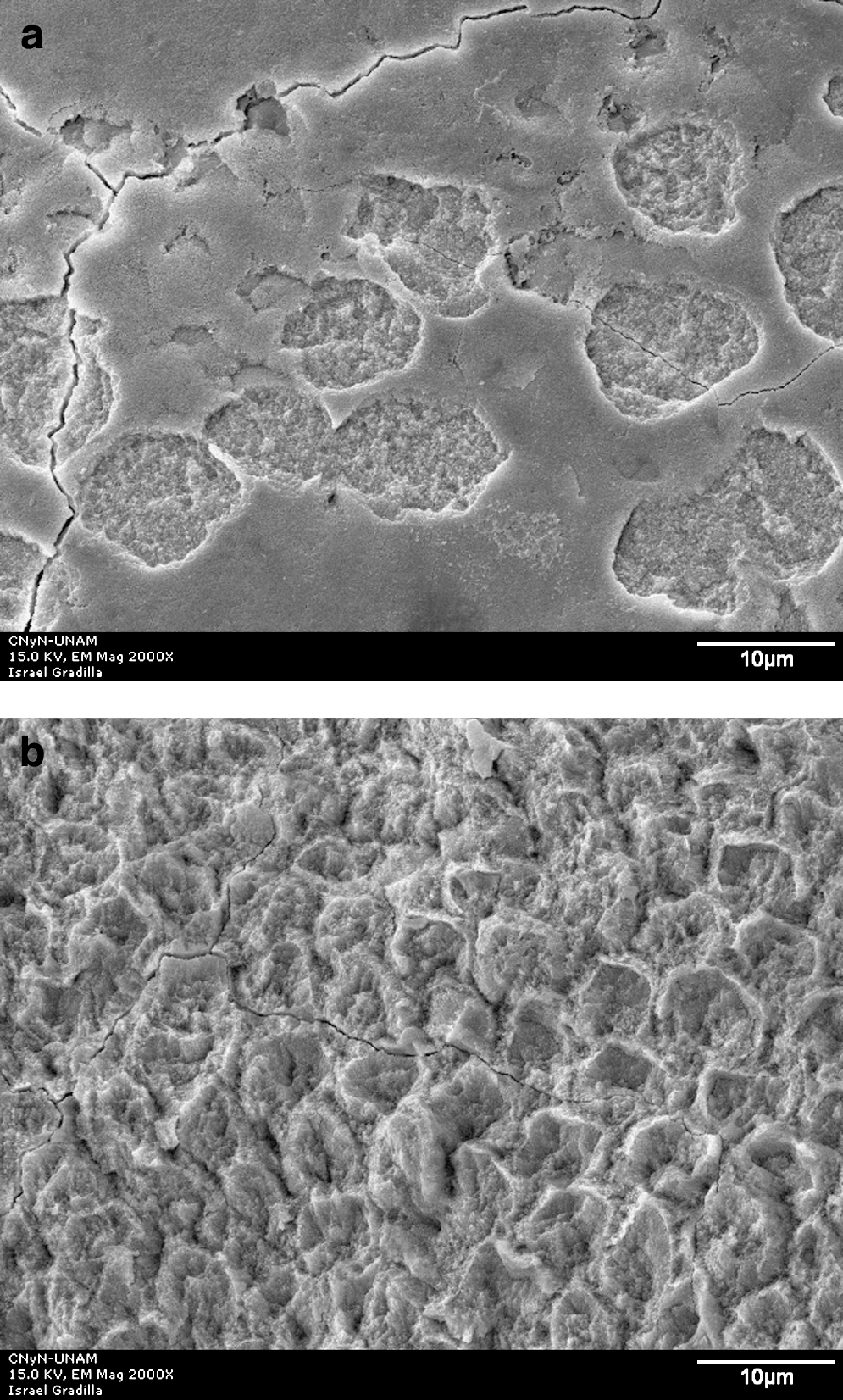
SEM micrograph of enamel surface after Er:YAG laser irradiation at 150 mJ/10 Hz and 11 J/cm2 and acid dissolution. (
Discussion
Several studies 4 –9 have demonstrated that enamel irradiation with Er: YAG laser produces changes that make the enamel resistant to developing caries lesions. In this experiment we used three different energy densities achieved through variations in the diameter of sapphire tip and output energy.
Due to the high prevalence of dental caries on occlusal surfaces, 20,21 we evaluated the potential preventive effect of irradiation of these surfaces by Er:YAG laser. Our results are in accordance with those reported by Apel et al., 13 who used subablative energy on the labial surface of bovine front teeth, but without using water as a coolant system. The use of water irrigation with a constant energy density while subablating the samples in our study was based on observations in pilot studies. These studies showed a rough and coarse enamel surface under visual inspection and stereoscopy when optimal energy densities and no water cooling system were used, as suggested by Liu et al. 15 Hossain et al., 7 however, reported a lower percentage of caries prevention when water was used as a cooling system. The percentage for dental caries prevention obtained in previous reports 6,12,15 ranged from 32% to 50%. However, there were variations in the energy density, pulse rate, and the time periods when the enamel surface was irradiated.
In our study, the % wt of Ca, P, O, and Cl on the enamel surface were similar for all groups. The results of Ca % wt were consistent with the concentration of the same element in the acidic solution, suggesting that the resistance to acid dissolution is not reached under these experimental conditions. It is worth mentioning that there are no previous reports in the literature that establish the concentration of several elements on the enamel surface after irradiation by an Er:YAG laser.
Regarding the morphological changes observed by SEM, the craters and cracks were similar to those reported in other studies. 9 –11 The use of a high energy density during laser irradiation probably enhances the roughness of the enamel surface. The lack of smoothness on the enamel surface would not promote the prevention of caries, because the rough surface seems to be more prone to the accumulation of bacteria or because of the massive demineralization along the resultant cracks, as was also indicated by Apel et al. 14 Thus, laser irradiation might increase the risk for the development of caries lesions. Kantorowitz et al., 22 reported that demineralization occurs around microfractures when the enamel is irradiated with a CO2 laser but the surfaces between these fractures seem to be totally resistant to demineralization. This fact highlights the need to understand the nature of these fractures and how to avoid their undesirable effect on the potential use of lasers in oral preventive care.
Cecchini et al. 9 found that Er:YAG laser irradiation at lower energies can decrease enamel solubility without severe alterations of the tooth structure, even under the cooling effect of water flow. Liu and Hsu 18 suggested the reduction of carbonate and modification of organic matter as mechanisms for dental caries prevention by Er:YAG laser irradiation. They reported no apparent morphological changes, such as cracks or craters, in contrast to our observations by SEM. Consequently, additional research on this field is necessary to correlate chemical and morphological changes on enamel surfaces irradiated by Er:YAG laser for dental caries prevention.
Conclusion
The results of this study suggest that the use of subablative Er:YAG laser irradiation did not increase acid resistance of the occlusal enamel surface as expected. Morphological microscopic changes, including craters and cracks, were observed on the irradiated enamel surface.
Footnotes
Acknowledgments
We thank Israel Gradilla from Centro de Nanociencias y Nanotecnología, Universidad Nacional Autónoma de México (UNAM) for technical assistance, and David Villanueva Jurado and Gerardo Guadarrama Álvarez, Maxillofacial Surgeons from the School of Dentistry, Universidad Autónoma del Estado de México (UAEM) for their help in obtaining the samples. This project was financed by UAEM and UNAM.
Author Disclosure Statement
The authors declare that no competing financial interests exist.
