Abstract
Phage therapy shows promise for treating mycobacterial infections, though clinical trial data on efficacy are still needed. We review preclinical studies assessing the efficacy and safety of phage therapy in mycobacterial diseases, focusing on in vitro, ex vivo, and in vivo models to inform clinical trial design. Most studies addressed Mycobacterium tuberculosis (M. tb), with phage D29 commonly used. Of all studies reporting activity, 91% (20/22) reported reduced mycobacterial counts. Some phages (4/7 assessed) also demonstrated activity against intracellular mycobacteria. Animal models (seven studies) showed efficacy and safety of four phages assessed, in infection models for M. tb, Mycobacterium ulcerans, Mycobacterium avium, and Mycobacterium abscessus, respectively. Substantial variability in results could be explained by experimental designs used. To advance clinical trials, we recommend standardized preclinical experiments on phage susceptibility, intracellular activity, and sampling from infection sites. This preclinical evidence supports further exploration of phage therapy for mycobacterial infections in clinical trials.
Introduction
Tuberculosis (TB) has returned as the most frequent infectious cause of death worldwide. 1 In 2022, an estimated 10.6 million people developed active TB globally, and a total of 1.3 million people died. 1 The current first-line treatment regimen, introduced 40 years ago, is still effective. 2 Yet its success is threatened by the emergence and spread of antimicrobial resistance, with nearly a half million people diagnosed with multidrug resistant (MDR) TB in 2023.1 New drugs and regimens were approved for MDR-TB, including BPaL-M (bedaquiline, pretomanid, linezolid, and moxifloxacin).3–5 However, resistance to newly approved drugs is emerging and will increase with wider implementation of these regimens.6,7 New therapeutic options are needed to treat drug-resistant TB, shorten regimens for all types of TB, and reduce relapse rates. Pulmonary disease due to nontuberculous mycobacteria (NTM) is increasing globally, and treatment options remain limited and complex with considerable toxicity and poor chances of success. 8
Bacteriophages (phage) are viruses that can infect and lyse bacteria. Phage therapy was used prior to the availability of antibiotics and has continued to be used in certain areas of the world. 9 The antimicrobial resistance crisis has sparked renewed interest in phage therapy. 10 Although safety of phage therapy is well-established, progress in this field is hampered by a lack of high-quality clinical efficacy data. 11 In terms of clinical trials, Phase 1 studies with healthy volunteers will be of limited value given the favorable safety profile of phage therapy, provided that a high-quality sterile product is used, and pharmacokinetics will be significantly affected by the presence of the target bacterium.
Numerous case reports and case series show that phage therapy can be effective.11–14 However, so far clinical trials have failed to show efficacy of phage therapy, potentially due to shortcomings in trial designs including lack of phage susceptibility testing and use of low-quality phage products.15–17 To date, there are no clinical trials with phage therapy for mycobacterial infections. Mycobacteriophages have been successfully used for treatment of cases with refractory NTM disease.18–22 Mycobacteriophage administration was well tolerated, and no adverse reactions resulted from administering phage to 20 patients for prolonged periods. NTM show strong variations in phage susceptibility, resulting in the need for individualized phage treatment, if lytic phages are available at all.23,24 In particular, smooth morphotypes of Mycobacterium abscessus tend to be resistant to phage. 24 The genotypic and phenotypic diversity of Mycobacterium tuberculosis (M. tb) is relatively low making this bacterium an ideal candidate for development of one phage treatment strategy that can be used in almost all patients with TB.25,26 Such a combination of mycobacteriophages was recently developed, 27 showing strong lytic activity in vitro against strains of the major human-adapted lineages of M. tb complex. 27 This phage combination includes mycobacteriophages from different genetic clusters to minimize chances of resistance development.
Before testing the efficacy of such phage combinations in humans, some key questions need to be addressed using preclinical models. The activity of phage treatment depends on whether phages can reach the target bacteria and establish a productive infection. 28 Mycobacteria can adapt to challenging environments, including intracellular, hypoxic, and acidic milieus; therefore, it is important to understand phage activity in these environments. Although models for these environments exist, studies in human samples from the site of disease can provide the most helpful information to translate model findings toward clinical use. In vivo disease models can inform administration strategies, and stability data are needed for each administration method, including intravenous, nebulized, or topical administration. We summarize current preclinical evidence on the efficacy and safety of phage therapy in mycobacterial infections.
Methods
PubMed and Embase were searched between January 5, 2024, and January 24, 2024, to identify available preclinical evidence for phage therapy for mycobacterial infections, with an updated search performed on March 21, 2025. We defined a search strategy based on a predefined population, intervention, comparator, outcome model in collaboration with a clinical librarian (AT). The “population” was defined as preclinical models of mycobacterial infections. Defined interventions included mycobacteriophages, control no mycobacteriophages, a buffer without mycobacteriophages or an antimycobacterial drug. Outcomes of interest were efficacy of phage-mediated killing of mycobacteria in disease-relevant conditions, and for animal studies, adverse events associated with mycobacteriophage therapy (detailed search strategy in Supplementary Data). Titles and abstracts were screened using Zotero. We included peer-reviewed articles published before March 15, 2025 in English, German, French, Italian, or Dutch, which reported original data on efficacy of phage-mediated killing of mycobacteria in specific conditions, ex vivo samples or animal models, and/or safety of phage therapy in animal models. GenBank reports and articles exclusively reporting in vitro mycobacterial susceptibility of phages in conventional culture conditions were excluded. The literature search was complemented by cross-referencing of published reviews and included articles. Data were extracted using Microsoft Excel.
Results
A total of 55 articles were included after critical review of selected articles and cross-referencing (Fig. 1). Figure 2 shows an overview of mycobacterial hosts and phages assessed in respective preclinical assays. Forty-four studies relating to phage treatment are discussed here (in vitro, ex vivo, and in vivo studies, respectively), as well as results of 11 studies relating to treatment with phage-related bacterial products such as lysins.
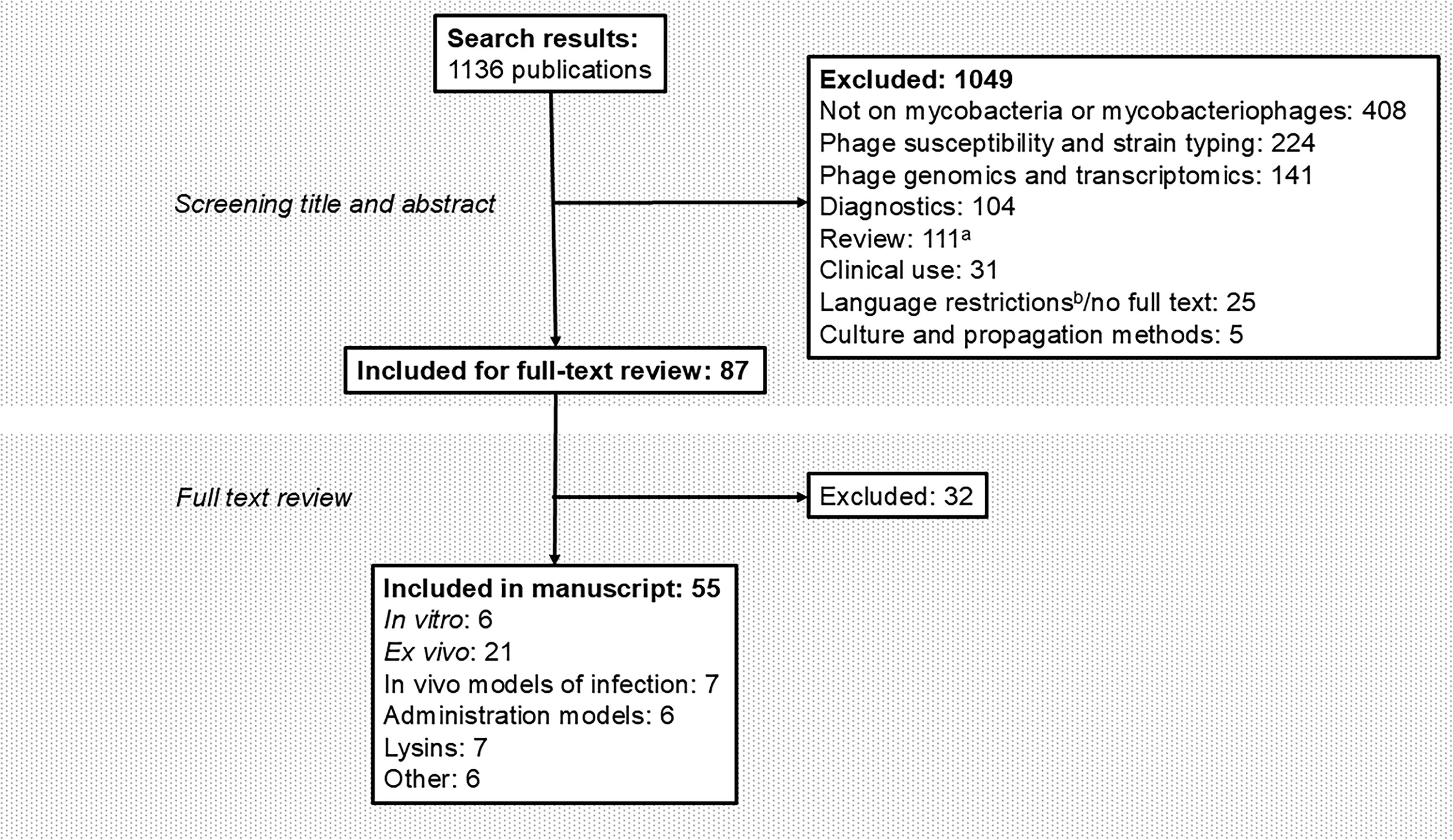
Overview of literature search and article selection. Figure illustrating the flow of article selection. aSelected reviews were used for cross-referencing. bA total of 19 articles were excluded based on language restrictions, of which 10 in Chinese and 7 in Russian.
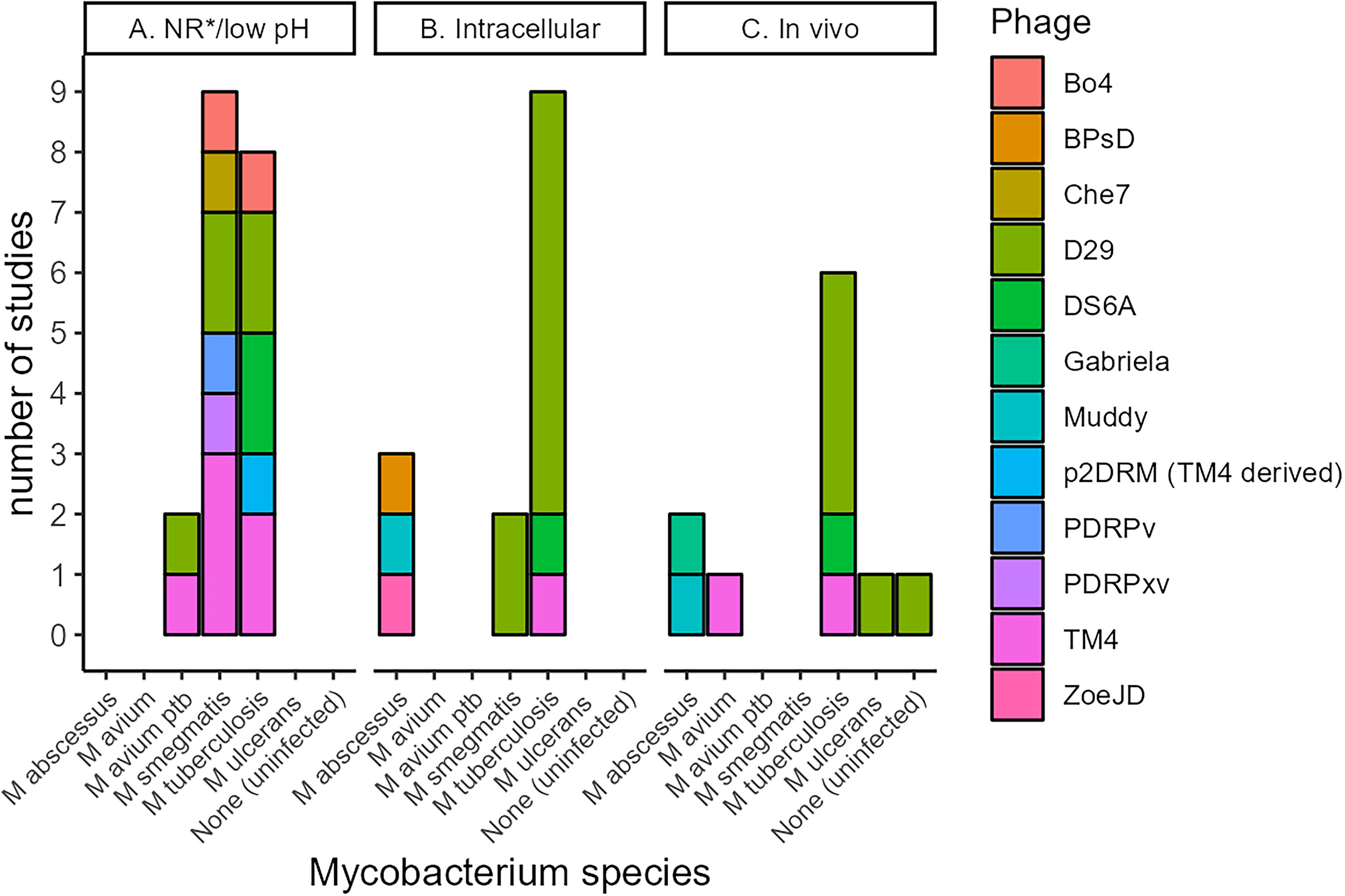
Overview of preclinical assays, mycobacterial hosts and phages. Overview of mycobacterial hosts and phages assessed in preclinical assays involving nonreplicating mycobacteria
In vitro studies
Nonreplicating mycobacteria
Mycobacteria can survive for extended intervals in a nonreplicating, dormant state—a state with reduced susceptibility to many antibiotics. A nonreplicating state of mycobacteria can be induced in vitro using culture in media with low pH, culture in hypoxic conditions, exposure to drugs, or a stationary phase culture. A total of six studies reported on efficacy of phages on nonreplicating mycobacteria or mycobacteria in media with low pH (Table 1, Fig. 2A); lytic activity of phages varied among phages used and respective conditions. Phages used were D29, TM4, Bo4, Che7, PDRPv, PDRPxv, and DS6A.29,30,32–34 For M. smegmatis in media with low pH, Bo4 and a combination of phages, including D29, TM4, Bo4, Che7, PDRPv, PDRPxv, and DS6A demonstrated lytic activity. 30 For M. smegmatis and Mycobacterium avium subspecies paratuberculosis in hypoxic conditions, TM4 was able to establish a productive infection (infection with phage replication), whereas D29 was not. 34 There was no report on lytic activity. TM4 was able to productively infect M. smegmatis in stationary phase; a mycobacteriophage combination including D29, TM4, Bo4, Che7, PDRPv, PDRPxv, and DS6A demonstrated lytic activity.30,33 A dual reporter phage engineered from TM4 demonstrates ability to infect M. tb persister cells. 29 A recent study found that D29, TM4 and DS6A were active against M. tb in hypoxic conditions created in plastic vacutainers and acidic conditions at a pH of 5. 31
In Vitro and Ex Vivo Experiments with Mycobacteriophage for Mycobacteria Infections
Table showing preclinical evidence from in vitro and ex vivo assays with phages and mycobacteria in different conditions, including nonreplicating mycobacteria, intracellular mycobacteria and mycobacteria in sputum.
BMDM, bone-marrow-derived macrophages; CF, cystic fibrosis; CFU, colony-forming units; DNA, deoxyribonucleic acid; INH, isoniazid; LORA, low-oxygen recovery assay; MAB, Mycobacterium abscessus; MOI, multiplicity of infection; M. tb, Mycobacterium tuberculosis; SCFM, synthetic cystic fibrosis sputum medium; TEM, transmission electron microscopy.
In summary, insight into phage dynamics in nonreplicating mycobacteria remains limited. Variability in results reported could be due to altered lytic activity of phages against nonreplicating mycobacteria, potentially caused by the lack of phage replication in dormant state mycobacteria.
Ex vivo studies
Intracellular mycobacteria
During infection and disease, mycobacteria reside intracellular in macrophages, escaping host immune responses. For the use of phage therapy for intracellular infections like mycobacteria it is essential to assess whether phages can enter these cells and infect and lyse intracellular bacteria. A total of nine studies reported on the efficacy of phage-mediated killing on intracellular mycobacteria (Table 1, Fig. 2B).36–42,44,49 Most studies reported on the efficacy of phage D29. Other phages included DS6A, TM4, BPsΔ33HTH-HRM10 (BPsΔ) and ZoeJΔ45 (ZoeJΔ), and Muddy.
Most, but not all, studies demonstrated an effect of phage D29 on M. tb in infected murine macrophages with a decrease in colony-forming units (CFUs).37–39 The effect was stronger when liposomal coated D29 was used. 38 Two studies reported no effect of D29.40,49 No cytotoxic effects were noted.42,44 Other studies reported no effect of phage TM4 on CFU of M. tb and M. avium; however, when TM4-infected M. smegmatis was used as a “Trojan Horse” vector, TM4 was able to reduce intracellular CFU of both mycobacterial species.36,40 Phage-mediated killing of intracellular M. abscessus was assessed in differentiated THP-1 macrophages, lung epithelial cells (A549) and murine bone-marrow-derived macrophages. 41 For phages BPsΔ and ZoeJΔ, phage uptake was demonstrated and associated with a reduction in M. abscessus CFU. For phage Muddy, which has a strong lytic effect on the extracellular M. abscessus strain used, phage uptake was demonstrated, but there was no effect on M. abscessus CFU after 48 h of incubation. 41
One study assessed the effect of phage D29 on granulomas derived from peripheral blood mononuclear cells infected with M. tb. 50 A decrease in CFU was reported for the D29 treated granulomas versus the control, with an increased effect for D29 coated in liposomes.
Variation in bactericidal activity of phages against intracellular mycobacteria may be explained by phage-dependent characteristics including sensitivity to low pH, receptors for infection of mycobacteria, or variable effects on nonreplicating mycobacteria. Strategies for increased intracellular uptake of phages, like liposomal coating, may be beneficial for increased activity.
Mycobacteria in samples from site of disease
No studies were found evaluating the bactericidal activity of phages on mycobacteria in human samples from the site of disease. When it comes to pulmonary infections, phage efficacy may be impaired by proteins and other molecules in sputum. Only one study reported on efficacy of phage-mediated lysis of a combination of 131 phages on M. abscessus in synthetic cystic fibrosis sputum medium and showed that M. abscessus in this medium was resistant to phage infection. 35
Although there are no studies published on phage killing of mycobacteria in human sputum samples, there is a body of literature on phage-based diagnostics for the detection of mycobacteria (mostly M. tb) in human sputum samples. As these assays were developed as ex vivo diagnostics on clinical isolates or decontaminated sputum, it is difficult to extrapolate to inhaled phage therapy. However, the success of these assays to detect mycobacteria is to an extent also dependent on the ability of phage to infect mycobacteria in sputum.51–63 Most diagnostic assays use D29 (FastPlaqueTB, Actiphage) derived phages to infect and detect M. tb in decontaminated sputum samples.
Mycobacterial reporter phages have been created using genetic modification. This is usually from TM4—a lytic mycobacteriophage active against all M. tb strains tested so far, using Luciferase Reporter Phages (LRPs) or fluorescent phage technologies. 64 LRPs offer opportunity for accelerated phenotypic drug susceptibility testing.54,55 Evidence on their performance on direct sputum samples remains limited. One study reported on the sensitivity of phage ϕ2GFP10 on M. tb spiked sputum where it detected 90% of mycobacteria. 58
For all diagnostic assays combined, sensitivity for detection of M. tb in decontaminated sputum samples ranged from 58% to 90%51–61 ; lower than when these assays are applied directly on culture isolates.62,63 The phage assay was affected by an inhibitory activity inherent to sputum, which was inactivated by sodium hydroxide (used for sputum decontamination) or heat treatment. 61 As inhalation is an attractive administration strategy of pulmonary mycobacterial infections, increased understanding of the efficacy of phage on mycobacteria in human sputum samples that have undergone minimal processing before applying phage is essential.
In vivo studies
Phage-mediated killing in animal models of mycobacterial infections
Over the last 30 years, six studies reported on safety and efficacy of phage therapy in animal models of mycobacterial infections.49,50,65–69 Five studies were done in mice and one in zebrafish (Table 2, Fig. 2C). Phage D29 was most frequently used for M. tb; others were DS6A (for M. tb), TM4 (for M. avium), Muddy and Gabriela (for M. abscessus), and D29 was used in an animal model of Buruli ulcer (Mycobacterium ulcerans). Phages were administered intranasally, endotracheal, subcutaneous or intravenously.
Mycobacteriophage for Treatment and Prevention of Mycobacterial Infections in Animal Models
Table showing evidence for phage therapy from animal models of mycobacterial disease.
BALF, bronchial alveolar lavage fluid; bd, twice daily; CFTR, cystic fibrosis transmembrane conductance regulator; DLN, draining lymph node; dpi, days post-infection; IFNg, interferon-γ; IL, interleukin; IV, intravenous; pfu, plaque-forming units; TNFα, tumor necrosis factor-α.
There were no phage-related adverse effects in any of the studies.43,50,65–69 Liposomal D29 reduced CFU in lungs of mice 35 days post-infection with M. tb. 50 A recent study reported a significant reduction in lung and spleen M. tb CFU of mice treated with DS6A. 49 Another recent study explored the use of inhaled administration with a dry-powder inhalation containing D29 and TM4, this study found no significant decrease in CFU in lungs or spleens of mice treated with phage. 40 Intravenously administered M. smegmatis infected with TM4 reduced M. avium CFU in the spleens of mice without causing adverse effects. 65 However, M. avium resistance to TM4 was reported in 23% of mice after a single dose of treatment. For Buruli ulcer, a single subcutaneous dose of phage D29 reduced pathology, ulceration, and M. ulcerans CFU in a mouse footpad model. 69 One study in zebrafish reported reduced M. abscessus CFU and abscess formation with phage Muddy with synergism with rifabutin, while there was no effect with control phage Gabriela. 67
Aside from studies mentioned above, one study evaluated the preventive effect of nebulized phage. Intranasally nebulized D29 had a preventive effect for pulmonary TB; a single dose 30 min prior to challenge with M. tb decreased the M. tb pulmonary burden at 24 h and 3 weeks post challenge. 66
Limited evidence from in vivo models of mycobacterial disease supports further research that can inform dosing strategies within treatment regimens combined with antibiotics.
Mycobacteriophage administration methods
Six studies focused specifically on administration methods of phages, particularly via nebulizers (Table 3).70–73 The use of jet nebulizers caused a significant reduction of phage titer and damage to phages (phages D29 and Maco2);70–72 the vibrating mesh nebulizer performed best (phage D29). 70 Phage Fulbright could successfully be incorporated in a nanofiber wound dressing; phages were stable and antimycobacterial activity (against M. smegmatis mc2155) was observed on phage-containing fibers in vitro. 73 One study evaluated aerosol delivery of the anti-M. tb phage FionnbharthΔ45Δ47. Aerosol delivery dispersed phage across all lung lobes, was well tolerated and did not induce robust neutralizing humoral immunity, in contrast to IV administration which was associated with increasing neutralizing total IgG and IgA responses over time.
Administration Methods for Mycobacteriophages
Preclinical evidence for different administration methods of mycobacteriophages.
These findings support further investigation of nebulized administration of phages, provided the nebulizer has been tested for its impact on phage stability.
Lysins and preclinical models of mycobacterial infections
An alternative approach to utilize phages in the treatment of mycobacterial infections could be phage-encoded enzymes, lysins. To overcome the barrier of the mycobacterial cell wall, including the mycolic arabinogalactan and peptidoglycan layers, mycobacteriophages encode two distinct types of lysins; Lysin A and Lysin B. 75
Four studies reported on the activity of lysins against intracellular mycobacteria, including M. smegmatis and M. tb (H37Rv), with three studies reporting significant bactericidal activity or lysins A and B derived from different phages (Supplementary Table).76–79
Two studies evaluated lysins as treatment of mycobacterial infections in mouse models, with one reporting bacteriostatic activity in the mouse footpad model of M. ulcerans and one reporting bactericidal activity against M. abscessus in severe combined immunodeficiency (SCID) mice.80,81
Conclusion
Preclinical evidence for phage therapy against mycobacterial infections remains limited. However, published studies show efficacy of phage-mediated lysis in in vitro conditions and in animal models of infection, including efficacy in disease-relevant challenging conditions such as nonreplicating and intracellular mycobacteria. There are no reports adverse events in in vivo studies of phage therapy, adding to favorable safety data from clinical use of phage therapy.11,14,22 These data provide a basis for further exploration of phage therapy in mycobacterial diseases.
There are important technical drawbacks that limit the interpretation and extrapolation of preclinical phage therapy studies. For studies focused on mycobacterial killing and its quantification, phage carryover in culture media is a significant and hard to tackle risk. The use of phage inactivation buffer is essential to interpret whether the phage effect occurs in specific conditions studied (e.g., intracellular or hypoxic), or when mycobacteria are cultured for quantification. For intracellular studies, only two mention the use of phage inactivation buffer,41,44 and none of the studies on nonreplicating mycobacteria describe measures taken to minimize phage carryover. Another important measure to pick up phage carryover is to plate serial dilutions for CFU quantification. In many cases, it was unclear to what extent that was done due to poor description of microbiologic methods. For examination of phage effects on nonreplicating mycobacteria, none of the studies found reported a confirmation of the nonreplicating state of mycobacteria through methods such as RNA sequencing or Nile Red staining.
Important gaps in our knowledge of preclinical phage therapy studies still exist. More evidence is needed on phage efficacy on intracellular and nonreplicating mycobacteria, taking these technical issues into account. Phage activity against intracellular mycobacteria may be improved through liposomal coating of phages. Although few studies reported on combinations of phage with antitubercular drugs rifampicin and isoniazid,27,34 more evidence is needed on phage-antibiotic interactions as future regimens will most likely combine antibiotics with phage. There is some evidence supporting different administration strategies, including the testing of phage stability with different nebulizers and the stability of a dry-powder formulation of phage. The recent publication on immune responses evoked by phage with nebulized versus IV administration 74 is an important addition as immune responses to phage with the formation of neutralizing antibodies have been associated with impaired efficacy of phage therapy.14,19 Future studies should explore dosing regimens with different administration strategies of phage, combined with antibiotics comparing different dosing schemes, to evaluate efficacy as well as immune responses to phage. More studies are needed to assess phage efficacy on mycobacteria in clinical samples, including sputum given nebulized phage therapy is an attractive option for pulmonary disease.
This review has not addressed a number of important questions, such as the need for more efficient phage susceptibility tests, especially slow-growing organisms such as M. tb, and the potential impact of phage therapies on phage-based TB diagnostics.
This review summarizes preclinical evidence of phage therapy for mycobacterial infections, adding to the equipoise for well-designed, proof-of-concept clinical trials that are urgently needed. It appears unnecessary to put every new therapeutic phage candidate through preclinical toxicity studies as is required for new drugs that, other than phages, have the potential to interact with human metabolism. Preclinical work with phages should focus on efficacy under different biological conditions, combined with relevant antibiotics, with a range of pathogens and application methods. This evidence will serve as a support to researchers developing phage therapy for mycobacterial conditions.
Footnotes
Acknowledgments
The authors thank Alice Tillema, clinical librarian, Radboud University Medical Center in Nijmegen, The Netherlands, for input in search strategy development.
Authors’ Contributions
S.J.: Conceptualization (lead); writing—original draft (lead); formal analysis (lead); writing—review and editing (equal); funding acquisition (lead). S.E.L., C.A.G.B., R.N.C., J.v.I., and G.F.H.: Writing—review and editing (equal); funding acquisition (equal). C.M., B.R., and M.P.T.: Writing—original draft (supporting); writing—review and editing. A.H.D.: Writing—review and editing (lead); funding acquisition (lead).
Author Disclosure Statement
None of the authors has any conflicts of interest to declare.
Funding Information
European Union’s Horizon 2020-Marie Sklodowska-Curie Personal Fellowships (grant agreement No. 101063247 (Phage-TB); S.J.); Bill and Melinda Gates Foundation, INV-060943.
