Abstract
There is increasing recognition of phenotypic variability in pediatric asthma, providing the opportunity for a more personalized approach to therapy. Increasingly biologic therapies, in particular those targeting the “allergic” (or T helper 2) pathway, are being considered for children with severe asthma. However, there is a great deal of variability in the extent these biologic therapies have been studied in children, as well as efficacy of results thus far. The goal of this article is to review the mechanism of action, efficacy, and potential predictive and monitoring biomarkers of the biologic medications focusing on the pediatric population, in an effort to establish a more personalized approach to asthma in the pediatric population for the 21st century.
Introduction
A
Many children with asthma have evidence of poor asthma control, defined based on symptom control, frequency and severity of exacerbations, and airflow limitation.4,5 In the United States, 57% of children with asthma had an asthma exacerbation in 2008; 209 children in the United States died from asthma in 2016. 6
Recently released survey data from the United States note that asthma carries a “significant economic burden” with an estimated cost of $81.9 billion in 2013. 7 The TENOR (Epidemiology and Natural History of Asthma: Outcomes and Treatment Regimens) observational study has noted significantly higher rates of direct and indirect healthcare utilization, including significantly more days of school/work missed (P < 0.001) due to poor asthma control compared with well-controlled asthmatic patients. 8
In contrast to poorly controlled asthma, severe asthma is identified based on the necessity of high-dose inhaled corticosteroid (ICS) therapy and a second controller therapy in the previous year, or systemic corticosteroids for 50% of the year, to prevent asthma from being uncontrolled or that remains uncontrolled despite these medications. 9 Despite optimization of all other factors, about 5%–10% of the asthmatic population continue to have severe asthma, accounting for ∼50% of asthma healthcare utilization in North America. 4
There is increasing recognition that asthma is an umbrella term for various phenotypes, and an understanding of these phenotypic differences may allow a more personalized treatment approach to the severe asthmatic. One of these phenotypic pathways is thought to be the type 2 pathway (T helper 2 Th2), or the “atopic” pathway, which involves immunoglobulin(Ig)-E production, and release of cytokines (including interleukin [IL]-4, IL-5, IL-13). Some of the biomarkers specific for the Th2 pathway include peripheral blood and sputum eosinophilia or bronchoalveolar eosinophil counts, fractional exhaled nitric oxide (FeNO) levels, and serum periostin levels.4,10
Biologic therapies are increasingly being considered in patients with severe asthma. There are now biologic therapies targeting several of the mediators of the Th2 pathway (IgE, IL4, IL5, IL13, and others) as well as the “danger signal” pathway (eg, thymic stromal lymphopoietin [TSLP]).
The goal of this review is to highlight the mechanism of action, efficacy, and predictive biomarkers for the biologic medications, highlighting the work done in the pediatric (age 18 and younger) population whenever possible.
Future understanding of the successes and limitations of these medications should also assist in the overall management of asthma and the design of new medications to address unmet needs. From the available literature, it is becoming clear that these medications affect, to varying degrees, rate of exacerbations, pulmonary function, medication reduction, and overall symptom control. Although the data available are increasing in the adult population, it is extremely limited in the adolescent proportion of these studies and even more limited in those <12 years of age.
This review provides an overview of the information available for each medication currently approved with an emphasis on what is known in adolescents and children. We also indicate where potential predictive and monitoring biomarkers are available. Predictive biomarkers are those that permit an estimate of likelihood of a medication having a positive response. Biomarkers can also assist with monitoring response to offer an early indication of benefit for the intervention. A perspective on other medications on the horizon will also be included with the available information on ongoing studies.
Biologic Therapies
Anti-IgE medications: omalizumab and ligelizumab
Omalizumab and ligelizumab (in development) are both humanized anti-IgE monoclonal antibodies that bind circulating IgE, causing decreased IgE levels, inhibition of IgE binding with its receptors, and downregulation of IgE receptors on mast cells, basophils and dendritic cells (Table 1). 11 Omalizumab binds to free IgE, but not to IgE bound to mast cells (which would, through cross-linking, result in allergic mediator release). This results in decreased release of inflammatory mediators related to the allergic response. In a phase 1 study, ligelizumab has been shown to be more potent at suppressing free IgE and IgE bound to mast cells and basophils than omalizumab. 12
CRTh2, chemoattractant receptor-homologous molecule expressed on T helper 2 cells; FEV1, forced expiratory volume in 1 second; IgE, immunoglobulin-E; IL, interleukin; IV, intravenous; N/S, nonsignificant; OCS, oral corticosteroid; PGD2, prostaglandin D2 receptor; SC, subcutaneous; QoL, quality of life; TSLP, thymic stromal lymphopoietin.
There have been several observational and randomized controlled trials on the use of omalizumab in the pediatric population. In contrast, the data on ligelizumab are limited to a single phase 2 study in adults.
Omalizumab
Omalizumab is the only biologic therapy for asthma to be studied in children as young as 5 years of age and one of the only biologic therapies for asthma approved for pediatric use in children 6 years and older. There have been 2 observational studies13,14 and 4 randomized double blind placebo-controlled trials (RDBPCTs)15–18 of omalizumab in the pediatric asthmatic population (Figs. 1, and 2). All the studies but one 13 looked specifically at the atopic population, largely defined by aeroallergen sensitization,14–18 and the majority15–18 recruited children with IgE levels of 30–1,300 IU/mL. Omalizumab was administered subcutaneously every 2–4 weeks and dose was calculated based on manufacturer instructions (body weight and serum IgE dependent).13–19
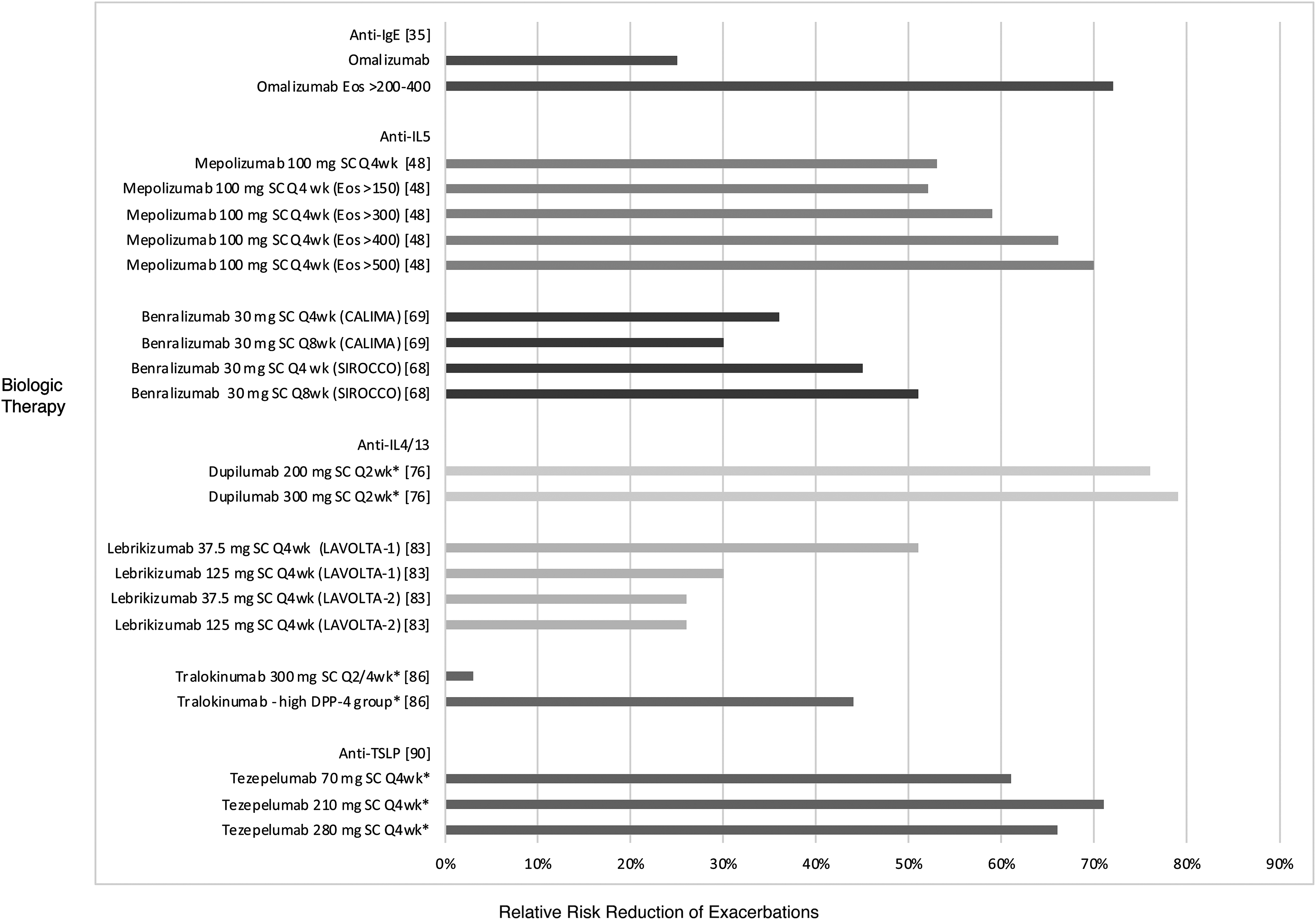
Relative risk reduction of exacerbations with biologic therapies in phase 2B*/3 studies. Adapted with permission from Lacy P and Nair P, in The human eosinophil. Wintrobe's Clinical Hematology (14th ed). Eds Greer JP, Arber DA, Glader B, Lost AF, Means RT, Paraskevas F, Rogers GM, Foerster J. Lippincott Williams & Wilkins, Philadelphia, PA, 2018 (in press).
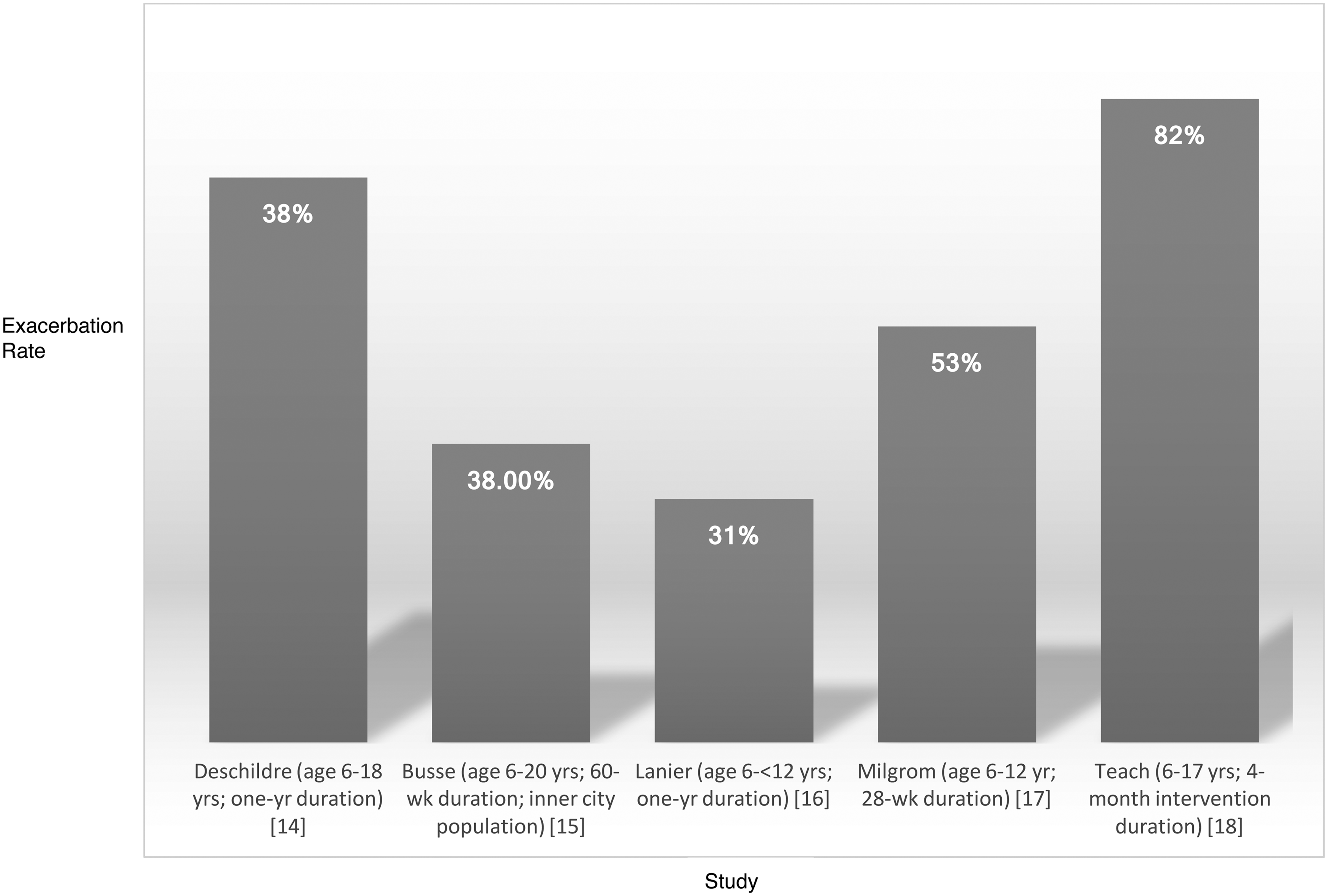
Decrease in exacerbation rate (%) with omalizumab compared with placebo. Calculated as [placebo exacerbation rate—omalizumab exacerbation rate]/placebo exacerbation rate × 100.
Pediatric data
Brodlie et al. conducted a 16-week uncontrolled therapeutic trial of omalizumab in 34 consecutive children (aged 5–16 years) with oral corticosteroid (OCS)-dependent asthma and noted that omalizuab use decreased median daily prednisolone requirements (median daily dose reduced from 20 to 5 mg, P < 0.0001), improved quality of life (P < 0.0001), and asthma control (P = 0.0001). 13
An observational study of 104 children with poorly controlled asthma (aged 6–18 years) by Deschildre et al. noted that omalizumab as add-on therapy significantly reduced exacerbation rates (OCS use) by 72% (P < 0.0001), hospitalization rates by 88.5% (P < 0.001), ICS dose by 30% (P < 0.001), and improved asthma control (P < 0.0001) over a year of therapy. 14
An RDBPCT by Busse et al. of omalizumab versus placebo in addition to standard therapy in 419 children and young adults (age 6–20 years) with persistent uncontrolled asthma in the Inner-City Anti-IgE Therapy for Asthma (ICATA) study found omalizumab in addition to guideline-based therapy reduced symptoms by 24.5% (P < 0.001), exacerbations (need for OCS) (48.8% versus 30.3%; P < 0.001), and hospitalizations (6.3% versus 1.5%; P = 0.02) despite significantly lower doses of ICS (% step levels 4–6; P < 0.001) and long-acting beta-agonists (LABA) (% prescribed; P = 0.003) therapy for a 60-week duration. 15 A post hoc analysis noted the effect of omalizumab on decreasing seasonal asthma exacerbations in the spring and fall compared with placebo (P < 0.001).
Lanier et al. performed an RDBPCT of 627 children aged 6 to <12 years old with poorly controlled asthma and noted that omalizumab use for a 1-year period significantly reduced significant asthma exacerbations (worsening symptoms requiring doubling of baseline ICS dose and/or systemic steroids) by 31% versus placebo (P = 0.007), and overall exacerbations by 43% (P < 0.001) versus placebo. 16
An RDBPCT by Milgrom et al. of 334 children aged 6–12 years with moderate-to-severe asthma noted that omalizumab use resulted in a significant reduction in ICS dose (median reduction 100% versus 66.7%; P = 0.001), increased frequency of complete ICS therapy removal (55% versus 39%; P = 0.004), and decreased asthma exacerbations requiring doubling of ICS or OCS use (18.2% versus 38.5%; P < 0.001), although there was little change in asthma symptom scores versus placebo. 17
The PROSE (Preventative Omalizumab or Step-up Therapy for Fall Exacerbations) study by Teach et al. noted that in 478 children (aged 6–17 years) with ≥1 asthma exacerbations in the year before enrollment, omalizumab use initiated 4–6 weeks before return to school lowered the fall exacerbation rate (OCS use or hospitalization) compared with placebo (odds ratio [OR] 0.48; 95% CI: 0.25–0.92) but not compared with an ICS boost (OR 0.73; 95% CI: 0.33–1.64). 18 In subgroup analysis of children who experienced an exacerbation during the run-in phase, omalizumab was significantly more effective than either placebo (OR 0.12; 95% CI: 0.02–0.64) or ICS boost (OR 0.05; 95% CI: 0.003–0.98) in preventing exacerbations versus placebo.
A 1-year real-life multicenter study of pediatric allergy and pulmonology tertiary centers in Italy noted a significant reduction in asthma exacerbations with omalizumab (P < 0.001) compared with the previous year, and a 96% reduction in hospitalizations. 19
Adolescent and adult experience
These results are consistent with studies including both adolescents (12 years and older) and adults, which have demonstrated omalizumab efficacy in improving asthma outcomes, including symptom scores,20,21 quality of life,22–24 asthma exacerbations,21–26 emergency department (ED) visits and asthma-related hospitalizations,21,23,25,27 ICS and rescue inhaler requirements, 28 and sputum eosinophil levels compared with placebo. 24
A recent Cochrane review of omalizumab use in adults and children with asthma found that omalizumab use compared with placebo reduced asthma exacerbations (OR 0.55; 95% CI: 0.42–0.60), reduced hospitalizations (OR 0.16; 95% CI: 0.06–0.42), and increased the likelihood of withdrawing ICS therapy (OR 2.50; 95% CI: 2.00–3.13). 29
Tertiary prevention has been examined in several of the pediatric studies although effects have been modest. Both Deschildre and Brodlie's studies noted nonsignificant improvements in forced expiratory volume in 1 second (FEV1) with omalizumab use (4.9% improvement; P = 0.02, and 0.15 L improvement; P = 0.1, respectively)13,14 and Milgrom's study noted no significant improvement in FEV1 in the omalizumab group. 17 The adolescent/adult studies have equally been mixed with predominantly nonsignificant improvements noted in FEV1. 24
Omalizumab is Food and Drug Administration (FDA) approved for moderate-to-severe asthma in patients aged 6 years and older with environmental allergies (positive skin testing or serum-specific IgE to perennial aeroallergen).
Predictive biomarkers
Several of the pediatric studies have examined predictive markers of omalizumab response although results have been conflicting. Busse et al. found that children with allergy/exposure demonstrated the greatest benefit from omalizumab (71.2% reduction in exacerbations). 15 In contrast, Deschildre et al.'s study did not find any effect modifier on omalizumab response with allergic comorbidities nor any relationship between IgE level and outcomes. 14 In their study, age >12 years was the only significant factor associated with good response (53.5% versus 76%; P = 0.02), whereas Brodlie et al.'s subgroup analysis showed no improvement based on age. 13 Teach et al.'s study noted recent asthma exacerbation to be most predictive of omalizumab response. 18 A post hoc analysis of the ICATA study found significantly greater response to omalizumab in exacerbation reduction with increased asthma severity, as defined by a lower prebronchodilator FEV1 (<90% predicted) or hospitalization in the past 6 months, 30 whereas another post hoc analysis noted a significantly greater response among children with elevated baseline eosinophil counts of ≥300/μL. 31 Exacerbation rates were reduced with omalizumab in the <300/μL eosinophil group by 5% (95% CI: −48% to 41%, P = 0.83), whereas in the ≥300/μL group exacerbations were reduced by 39% (95% CI: 15%–56%, P = 0.004) compared with placebo in adolescents and adults. 22
The Composite Asthma Severity Index (CASI) has been developed to determine asthma severity by examining daytime and night-time symptoms, albuterol use, lung function, controller medications, and exacerbations.32,33 The CASI scores focus on 5 domains, providing a composite score that can be used to indicate the change in burden of asthma with an intervention. 32 Recently in a post hoc analysis, it has been identified as a useful tool for identifying children most likely to achieve a favorable response to omalizumab. 34
In the adolescent/adult studies, high levels of type 2 inflammatory markers 10 such as FeNO levels,22,26 eosinophilia,15,22,26 periostin levels, 22 and IgE levels 23 have predicted improved response to omalizumab, although once again studies are mixed with not all studies consistently identifying these markers as predictive. 25 A recent study on pooled data from 2 RDBPCTs of omalizumab response noted clinical markers of asthma severity (history of emergency asthma treatment, hospitalization, FEV1 < 65% predicted, requirement for LABA use, or beclomethasone >600 μg/day) and higher baseline blood eosinophil levels (≥300/μL) to be predictive of omalizumab response. 35 In addition, in contrast to the theory that Th2 markers are predictive, a recent small study noted baseline levels of CXCL12 (chemokine C-X-C motif ligand 12) and IL10 to be predictive of omalizumab response. 36
Monitoring biomarkers
In Milgrom et al.'s study, there was a reduction in free serum IgE in the omalizumab group by 95%–99%, with greatest reductions observed during the first few days after dosing. 17 No reduction was noted in the placebo group. Ideally, a reduction in free serum IgE would be a useful monitoring biomarker but this assay is not readily available in clinical laboratories. In the adolescent/adult studies, omalizumab has been shown to significantly reduce FeNO, alveolar nitric oxide levels, and sputum eosinophil levels as well. 24 To date, these have not been correlated with the level of clinical response or to differentiate responders from nonresponders.
Advantages and limitations
Omalizumab is one of the few biologic therapies with data in the pediatric population, and studies in this population consistently show a benefit in particular in atopic children, although the extent of a disease-modifying effect is unclear. There are concerns about potential side effects of omalizumab, such as malignancy and anaphylaxis risk. Although 2003 analysis of pooled clinical trial data reported a malignancy risk of 0.5% in omalizumab-treated patients (compared with 0.2% of control subjects), 37 the Epidemiologic Study of Xolair, a prospective cohort of 7,857 adolescents and adults, noted no increased malignancy risk with omalizumab compared with control (hazard ratio [HR] 1.09, 95% CI: 0.87–1.38). 38 There is also an ∼0.2% risk of anaphylaxis with omalizumab. 39
Efficacy in other disorders
Omalizumab has been approved in adults for use in chronic spontaneous urticaria, and this potential benefit is also being studied in children, 40 and is being studied in severe atopic dermatitis in adults and children,40,41 as an addition to oral immunotherapy in children and adults 42 as well as in the treatment of allergic rhinitis and chronic rhinosinusitis in adults. 43 In addition, omalizumab has been shown to prevent anaphylaxis in an adolescent with systemic mastocytosis 44 and to be effective in the management of cutaneous mastocytosis in the pediatric population. 45
Future needs
Studies are ongoing in omalizumab, including the Preventing Asthma in High Risk Kids study, which is an RDBPCT to determine whether 2 years of omalizumab in children aged 2–3 years will prevent progression to persistent asthma (NCT02570984). With regard to the concerns about anaphylaxis and development of cancer, the experience to date has been reassuring, but with each level of younger age introduction, these factors will have to be monitored. Because there have been reports of anaphylaxis and at unexpected times in relation to duration of treatment and time after dose, it is likely that office administration will continue to be required. In addition, the optimal duration of omalizumab treatment and whether it produces a long-lasting effect remain to be determined.
Ligelizumab
In a phase 2 double-blind parallel-group trial in 37 adults with mild allergic asthma, ligelizumab elicited a 3-fold greater provocative concentration of allergen, causing a 15% decrease in FEV1 (PC15) compared with omalizumab (P = 0.10) and 16-fold greater than placebo (P = 0.0001) at 12 weeks. 46 However, wide variability in response was noted from a <2- to >500-fold change in bronchial allergen challenge responses. Ligelizumab was administered subcutaneously at doses of 24, 72, or 240 mg every 2 weeks. So far, no studies have been done in children or adolescents, and there is no data on phenotypic response. It does not appear that further studies are planned with ligelizumab. Total IgE increased in this study for both ligelizumab and omalizumab. Basophil surface IgE was reduced in the higher doses of ligelizumab by up to 99%. Ligelizumab is currently in clinical trials for chronic spontaneous urticaria in adults. 47
Anti-IL-5 medications
IL5 is a cytokine that recruits eosinophils from the bone marrow and promotes both the activation and longevity of these cells. 11 There are 3 anti-IL-5 biologic therapies that have been trialed in adolescent and adult asthma—mepolizumab, reslizumab, and benralizumab (Fig. 1). None of these biologic therapies have been trialed in children under the age of 12 years. Mepolizumab and reslizumab are humanized monoclonal antibodies against IL5, whereas benralizumab is a humanized monoclonal antibody against the IL5 receptor. The studies performed with the anti-IL5 medications have been largely limited to eosinophilic asthma under the assumption that eosinophilia is predictive of response to these medications, although the criterion for inclusion for level of eosinophilia has varied slightly between studies. Although some data are available in adults, less information is available with adolescents and studies are in progress in children <12 years of age.
Mepolizumab
Adolescent and adult experience
The Mepolizumab as Adjunctive Therapy in Patients with Severe Asthma (MENSA) study randomized 576 adolescents and adults (aged 12–82 years) with severe eosinophilic asthma (serum eosinophils ≥150 cells/μL at study entry or ≥300 cells/μL at some time in the previous year) and frequent exacerbations to mepolizumab at a dose of 100 mg subcutaneous (SC) or 75 mg intravenous (IV) every 4 weeks or placebo. 48 Asthma exacerbations requiring OCS were reduced 47% and 53% (P < 0.001)) with IV and SC mepolizumab compared with placebo, respectively; asthma exacerbations requiring an ED visit were reduced 32% and 61% with IV and SC mepolizumab (P = 0.30 and P = 0.02 compared with placebo, respectively); asthma exacerbations resulting in hospitalization were reduced 39% and 69% with IV and SC mepolizumab (P = 0.33 and P = 0.03 compared with placebo, respectively); and asthma symptoms were significantly improved (P < 0.001) with both IV and SC mepolizumab compared with placebo. No data specific to the adolescent population included in this study are accessible from the published studies.
The Steroid Reduction with Mepolizumab (SIRIUS) randomized 135 adolescents and adults with severe eosinophilic asthma (6-month history of OCS requirement; blood eosinophil levels of either ≥150 cells/μL during the optimization phase or ≥300 cells/μL during the 12-month period before screening) to mepolizumab (100 mg SC every 4 weeks) or placebo in addition to their maintenance asthma medication and a tapering regimen of OCS. 49 Mepolizumab significantly increased the likelihood of oral steroid reduction with an overall OR of 2.39 (95% CI: 1.25–4.56; P = 0.008). Despite this reduction in OCS dose, there was also a significant reduction in asthma symptoms (between-group difference, −0.52 points; 95% CI: −0.87 to −0.17; P = 0.004) and asthma exacerbations (doubling of OCS dose, hospital admission, or ED visit) (rate ratio [RR] 0.68, 95% CI: 0.47–0.99; P = 0.04) in the mepolizumab group. Although adolescents and adults were recruited, the study patients were largely adult in age (age range 16–74 years).
The Dose Ranging, Efficacy, and Safety with Mepolizumab (DREAM) study randomized 621 asthmatics of age 12–74 years with recurrent severe exacerbations and evidence of eosinophilic inflammation (based on sputum eosinophil counts ≥3%, FeNO level ≥50 parts ppb,, blood eosinophil level of ≥0.3 × 10 9 /L, or deterioration of asthma control with ≤25% reduction in ICS or OCS) to 3 different doses of mepolizumab (75, 250, 750 mg IV every 4 weeks) or placebo and noted a significant 39%–52% reduction in asthma exacerbations (OCS use) (P < 0.0001), as well as severe exacerbations requiring ED or hospital admissions with meplizumab. 50 However, no significant improvement in asthma symptom scores or quality of life was noted. Although adolescents and adults were recruited, only adults were randomized in the trial.
Mepolizumab is FDA approved as add-on maintenance therapy in patients who are 12 years and older with eosinophilic asthma.
Predictive biomarkers
The 3 randomized controlled trials above all included only patients with eosinophilic asthma on the basis that an antibody that reduces eosinophil levels would be most effective in eosinophilic asthma. This is supported by studies showing no clinical benefit or improvement in FEV1 with mepolizumab in moderate persistent asthmatics in the absence of eosinophilia.11,51,52 The DREAM study noted that reductions in the rate of clinically significant exacerbations with mepolizumab varied based on baseline blood eosinophil count and number of exacerbations in the year before study commencement. MENSA also noted in exploratory multivariate modeling that the only covariate identified that influenced efficacy of mepolizumab on exacerbations was blood eosinophil count at screening, with enhanced response to mepolizumab noted in patients with a blood eosinophil count of ≥500 cells/μL.
Monitoring biomarkers
Mepolizumab has been shown to decrease serum eosinophil counts46,50,51 and sputum eosinophil counts50,51 compared with placebo. It would be useful to identify other biomarkers that could differentiate the pharmacologic response from the clinical response and serve as indicators of beneficial effect.
Advantages and limitations
To date, there are no studies on children <12 years old, and although many studies included adolescents, the study populations were predominantly adult (median age 51 years, minimum age 12 years in pooled trials).53,54 In addition, the studies are limited to predominantly eosinophilic asthma. Studies are conflicting regarding a disease-modifying effect. DREAM noted no improvements in FEV1, SIRIUS noted a nonsignificant trend toward greater changes from baseline in the FEV1 before and after bronchodilation, whereas MENSA noted a significant improvement in baseline and postbronchodilator FEV1 with both IV and SC mepolizumab compared with placebo (P = 0.03 and P = 0.003/0.004, respectively). In addition, side effects such as herpes zoster infection have been noted in clinical trials. 55
Efficacy in other disorders
Mepolizumab has demonstrated efficacy in the treatment of pediatric eosinophilic granulomatosis with polyangiitis, 56 and in adults it has shown efficacy for eosinophilic chronic obstructive pulmonary disease, 57 aspirin-exacerbated respiratory disease, 58 and chronic rhinosinusitis with nasal polyposis. 59 Mepolizumab and benralizumab are being actively investigated for the treatment of hypereosinophilia 60 and eosinophilic esophagitis. 61
Future needs
Data on children <12 years are required. Studies are ongoing in this population, including an RDBPCT of children aged 6–17 years of age examining exacerbation rates with mepolizumab compared with placebo in addition to standard asthma care in children who live in an urban area (NCT03292588).
Reslizumab
Adolescent and adult experience
Two RDBPCTs have been conducted on reslizumab in adolescents and adults with eosinophilic asthma. A phase 3 trial of 374 adolescents and adults (aged 12–75 years) with inadequately controlled eosinophilic asthma (blood eosinophil count ≥400 cells/μL) on medium-dose ICS by Bjermer et al. randomized patients to placebo, infusions of 0.3 mg/kg reslizumab, or 3.0 mg/kg every 4 weeks for 16 weeks. 62 This study noted improved asthma control (change in ASUI 0.051; 95% CI: 0.012–0.089; P = 0.0094 and 0.047; 95% CI: 0.009–0.085; P = 0.016 for 0.3 mg/kg and 3 mg/kg reslizumab, respectively) compared with placebo. However, the quality of life measure trended to improve in the 0.3 mg/kg dose of reslizumab but was only significant for the higher dose (AQLQ score change 0.28; 95% CI: −0.036 to 0.59; P = 0.08 and 0.36; 965% CI: 0.05 to 0.67; P = 0.02 for 0.3 and 3 mg/kg reslizumab, respectively). Only 5% of study participants were in the adolescent age range but specific results are not reported for this age group.
Two duplicate RDBPCTs (phase 3) in adolescents and adults (age 12–75 years) with uncontrolled eosinophilic asthma (serum eosinophils ≥400 cells/μL) on medium-to-high dose ICS therapy noted significant reduction in asthma exacerbation frequency (study 1: RR 0.50; 95% CI: 0.37–0.67; study 2: 0.41; 95% CI: 0.28–0.59; both P < 0.0001) with reslizumab (3.0 mg/kg IV every 4 weeks), as well as improvements in time to exacerbation (OCS use, doubling of ICS, or asthma-related emergency treatment), asthma control, and quality of life. 63
A meta-analysis of reslizumab compared with placebo in the treatment of eosinophilic asthma in adolescents and adults noted reslizumab to decrease asthma exacerbations (OR 0.46; 95% CI: 0.35–0.59; P < 0.00001), blood eosinophil counts (standardized mean difference [SMD] = −475.62; 95% CI: −528.41 to −422.93; P < 0.00001), improve asthma control (SMD = −0.26; 95% CI: −0.36 to −0.16; P < 0.00001), and improve FEV1 (standardized mean difference = 0.16; 95% CI: 0.10–0.23; P < 0.00001) compared with placebo, supporting a disease-modifying effect as well. 64
Reslizumab is FDA approved for add-on therapy in adults aged 18 and older with eosinophilic asthma.
Predictive biomarkers
As with mepolizumab, the presence of eosinophilia as a biomarker for reslizumab efficacy was demonstrated in an RDBPCT of reslizumab in adult asthmatics, which demonstrated no clinically significant effect on either lung function or symptom control in patients unselected for baseline eosinophil levels. 65 However, in Bjermer et al.'s 62 study, baseline blood eosinophil levels did not influence magnitude of improvement in FEV1, although the number of patients in the sensitivity analyses was small.
Monitoring biomarkers
Reslizumab has been shown to decrease blood eosinophil counts compared with placebo in several randomized controlled trials, but it has not been correlated with the beneficial effect or responder/nonresponder populations.62,64,65
Advantages and limitations
Studies are limited to eosinophilic asthma, and no studies have been done to date on children under the age of 12 years old. In addition, side effects, such as elevation in creatine phosphokinase levels, have been noted in clinical trials. 66
Efficacy in other disorders
Reslizumab is being studied for use in eosinophilic esophagitis in children. 67
Future needs
Studies are ongoing on the use of reslizumab in the adolescent population such as an RDBPCT of reslizumab compared with placebo in adolescents and adults with uncontrolled eosinophilic asthma, examining the efficacy of reducing asthma exacerbations (NCT 02452190). Studies in the pediatric population are required.
Benralizumab
Adolescent and adult experience
Two RDBPCTs (phase 3) of adolescents and adults aged 12–75 years with severe uncontrolled asthma on high-dose ICS and LABA therapy (SIROCCO and CALIMA) enrolling patients at 677 research centers internationally with similar study designs have noted benralizumab to significantly improve exacerbation rates and asthma symptoms compared with placebo in patients with blood eosinophil counts of ≥300 cells/μL (who were stratified for inclusion in primary analysis).68,69 In the SIROCCO study, patients with lower blood eosinophil counts had reduced exacerbation rates (use of OCS, ED visit, and inpatient hospital stay due to asthma) with benralizumab (30 mg SC every 4 or 8 weeks) (P = 0.047) and improvement in asthma symptoms (P = 0.4), but other endpoints were not significant and the treatment effect was “smaller.” 68 Although the SIROCCO and CALIMA studies recruited adolescents and adults, the majority of patients randomized were adults. Some studies have noted an improvement in FEV1 with benralizumab,68,69 although in others no improvement in FEV1 was noted. 70
An RDBPCT of 369 adults with severe asthma requiring OCS use noted that benralizumab use (30 mg SC either every 4 weeks or every 8 weeks) reduced median final oral glucocorticoid dose from baseline by 75%, compared with a 25% reduction in the placebo group (P < 0.001). Benralizumab use reduced the annual exacerbation rate (increase in OCS, ED visit, and hospitalization) by 55% versus placebo when administered every 4 weeks (P = 0.003) and by 70% when administered every 8 weeks (P < 0.001). No significant effect on FEV1 was noted. 71
Benralizumab is FDA approved for patients with severe asthma aged ≥12 years, who have an eosinophilic phenotype.
Predictive biomarkers
Increased blood eosinophil levels (>300 cells/μL) have been identified in some studies as a biomarker of benralizumab efficacy.68,69,72 However, a pooled analysis of SIROCCO and CALIMA study data supported the use of benralizumab in patients with blood eosinophil counts ≥150 cells/μL 73 and a newly released subsequent pooled analyses extended the efficacy irrespective of eosinophil count, although noting that the higher the eosinophil count the greater the benefit. 74
Monitoring biomarkers
Benralizumab has been shown to decrease blood eosinophil counts, but studies are not available to show differentiation between responders and nonresponders in the magnitude of reduction in blood eosinophils.69,70
Advantages and limitations
There are a small number of studies thus far that are limited to the adult and adolescent population. Studies in children, and further studies in the adolescent population, are required.
Efficacy in other disorders
None has been studied to date.
Future needs
Studies are ongoing, with the MIRACLE study randomizing adolescents and adults with uncontrolled asthma despite moderate-to-high dose ICS plus LABA therapy to benralizumab versus placebo and measuring exacerbation rates as primary outcome measure. Patients are stratified based on eosinophil count (NCT03186209). The eosinophil level most predictive of response remains to be determined, and no studies have been done yet with benralizumab in asthmatic children <12 years of age.
Anti-IL4 and IL13
The IL4 cytokine is an essential cytokine to T helper 2 (Th2) cell polarization, whereas the IL13 cytokine is associated with periostin production in the bronchial epithelial cells, ultimately resulting in smooth muscle contraction, mucus production, airway remodeling and hyper-responsiveness, and goblet cell hyperplasia. 52 IL13 also works with IL4 to result in IgE production. The IL4 receptor (alpha subunit) is critical for both IL4 and IL13 signal transduction. There has been one RDBPCT on the use of an anti-IL4 alpha receptor antibody (dupilumab) and the studies on the anti-IL13 biologic tralokinumab are limited to 2 phase 2 studies (Fig. 1). The data are more robust for the anti-IL13 biologic lebrikizumab although results are inconsistent in terms of lebrikizumab's efficacy.
Dupilumab
Dupilumab is a human monoclonal antibody to the alpha subunit of the IL4 receptor, thereby blocking the activity of IL4 and IL13.
Adolescent and adult experience
An RDBPCT of dupilumab use in 104 adults (18–65 years) with moderate-to-severe asthma and high blood or sputum eosinophil counts (>300 cells/μL; >3%, respectively) on medium-to-high dose ICS plus LABAs resulted in an 87% reduction in asthma exacerbations (reduction of 30% or more in peak expiratory flow, 6 or more additional reliever inhalations, OCS use, and hospitalization) (44% versus 6%; OR 0.08; 95% CI: 0.02–0.28; P < 0.001) and increased time to asthma exacerbation (HR 0.10; 95% CI: 0.03–0.32; P < 0.001) despite stopping LABA and ICS therapy while on dupilumab (300 mg SC once a week) compared with placebo. 75 There was noted improvement in FEV1 as early as the second week of treatment, which was maintained for the 12 weeks of the study (P < 0.001). An RDBPCT of dupilumab (300 mg SC every 2 weeks for 24 weeks) in 210 adults with OCS-dependent asthma noted a 59% (95% CI: 37%–74%) decrease in severe exacerbations in the dupilumab group despite a −70.1% reduction in OCS (P < 0.001). 76 An RDBPCT of 1902 adolescents and adults (aged 12 years and older) with uncontrolled asthma noted that dupilumab (200 or 300 mg SC every 2 weeks) reduced severe exacerbations by 47.7% compared with pacebo (P < 0.001) while increasing FEV1 compared with placebo (P < 0.001). 77
Dupilumab has been FDA approved for adolescents and adults aged 12 years and older with moderate-to-severe asthma as add-on maintenance therapy.
Predictive biomarkers
There are few data on biomarkers that may predict improved response. A phase 2B RDBPCT by Wenzel et al. with 769 adults on medium-to-high dose ICS and LABA therapy randomized to dupilumab (200 or 300 mg SC every 2–4 weeks) versus placebo noted significant efficacy in improving FEV1 as well as decreasing severe exacerbations, irrespective of baseline eosinophil levels, suggesting that this may not be a specific biomarker. 78 A commentary on this study did note that IL5 is more specific to eosinophilia than IL4, and that other biomarkers such as FeNO or serum periostin levels may be more predictive, but these have not been studied to date. 79
Monitoring biomarkers
Dupilumab has been shown to reduce FeNO levels75,78 and other levels of Th2 inflammation (TARC, eotaxin-3, and IgE). 75
Advantages and limitations
There are a small number of studies thus far that are limited to the adolescent and adult population. Studies in children, and further studies in the adolescent population, are required.
Efficacy in other disorders
Dupilumab has been extensively studied in the treatment of atopic dermatitis in adults and children80,81 and is being investigated as a therapy in adults with eosinophilic esophagitis (NCT 02379052).
Future needs
Further studies are required to determine the reproducibility of this result, as well as to assess its efficacy in children and adolescents. Studies are ongoing with an RDBPCT of dupilumab in children aged 6 to <12 years with uncontrolled persistent asthma (NCT02948959).
Lebrikizumab
Lebrikizumab is a humanized monoclonal antibody against IL13.
Adolescent and adult experience
A phase 2 RDBPCT by Corren et al. of lebrikizumab (250 mg SC once a month) in 219 adults with asthma uncontrolled on medium-to-high dose ICS therapy noted a significant improvement in FEV1 after 12 weeks of lebrikizumab (5.5% points higher than placebo; P = 0.02) although no significant change was noted in several other secondary efficacy outcomes including asthma control (P = 0.85), with subgroup analysis demonstrating that adults with high periostin levels had significantly improved FEV1 with lebrikizumab (8.2% points higher than placebo; P = 0.03) compared with the low-periostin subgroup (1.6% points higher than placebo; P = 0.61). 82
A phase 2 RDBPCT by Noonan et al. of 212 adults aged 18–65 years not receiving ICS therapy noted no significant improvement in FEV1 with multiple different doses of lebrikizumab (125, 250, or 500 mcg SC monthly) compared with placebo even with stratification based on periostin levels, although there was a reduced risk of treatment failure in all lebrikizumab dose groups compared with placebo (HR, 0.21; 95% CI: 0.09–0.47; P < 0.001). 83
Pooled data from 2 RDBPCTs on multiple doses of lebrikizumab in adults with uncontrolled asthma despite medium-to-high ICS use and a second controller found that lebrikizumab use (37.5, 125, and 250 mg SC every 4 weeks) reduced the rate of asthma exacerbations (OCS use or hospitalizations), which was more pronounced in the periostin-high subgroup (60% reduction with lebrikizumab compared with periostin-high placebo group; 95% CI: 18%–80%) compared with the periostin-low subgroup (5% reduction with lebrikizumab compared with periostin-low placebo group; 95% CI: −81% to 47%) and an improvement in FEV1 that was once again more pronounced in the periostin-high group (9.1% improvement from baseline versus placebo in periostin-high group versus 2.6% improvement versus placebo in the periostin-low group). 84
Replicate phase 3, RDBPCTs (LAVOLTA I and LAVOLTA II) of 2 concentrations of lebrikizumab (37.5 or 125 mg SC every 4 weeks) in adults with uncontrolled asthma (1,081 patients in LAVOLTA I and 1,067 in LAVOLTA II) found that lebrizikumab reduced exacerbation rates (OCS use or hospitalization) in high biomarker (periostin/serum eosinophil) adults compared with placebo in both the 37.5 mg group (RR 0.49; 95% CI: 0.34–0.69; P < 0.0001) and in the 125 mg dose group (RR 0.70; 95% CI: 0.51–0.95; P = 0.0232). 85 In LAVOLTA II, exacerbation rates were also reduced in the high biomarker patients compared with placebo in the 37.5 mg dose (RR 0.74; 95% CI: 0.054–1.01; P = 0.06) and in the 125 mg dose (RR 0.74; 95% CI: 0.54–1.02; P = 0.06) groups although not significantly.
Predictive biomarkers
Periostin levels have emerged from the mentioned studies as a biomarker strongly correlated with lebrikizumab efficacy. Noonan et al.'s study found no difference in FEV1 improvement based on periostin levels, suggesting as noted in a recent review that interaction of IL13 with ICS might contribute to response 52 as the Noonan study did demonstrate a similar effect on IL13 activity (with decreases in serum IgE, FeNO levels, and blood eosinophil numbers) with lebrikizumab as other studies, such as Corren et al.'s study. It has been pointed out, however, that as periostin is a product of bone turnover, there may be higher baseline levels of this biomarker in children, which could limit its applicability as a biomarker of efficacy in the pediatric population. 52
Monitoring biomarkers
Lebrikizumab has been associated with a decrease in FeNO compared with placebo (P < 0.001), with greater reductions in FeNO in the high periostin groups. 82
Advantages and limitations
There are a small number of studies thus far that are limited to the adult population. Studies in children, and further studies in the adolescent population, are required. To date, further evaluation of lebrikizumab in asthma has been stopped due to the failure to meet endpoint in 1 adult study.
Efficacy in other disorders
Lebrikizumab is being studied in the treatment of adults with moderate-to-severe atopic dermatitis. 86
Future needs
Studies on adolescents and younger children are required.
Tralokinumab
Tralokinumab is a human monoclonal antibody against IL13. A phase 2 RDBPCT of multiple doses of tralokinumab (150, 300, or 600 mg SC every 2 weeks) versus placebo in 194 adults aged 18–65 years with moderate-to-severe asthma noted no significant improvement in asthma control (P = 0.375), FEV1 (P = 0.072) with tralokinumab although there was a significant decrease in beta-agonist use (P = 0.02). 87
A phase 2 RDBPCT of tralokinumab in 452 adults (aged 18–75 years) with severe uncontrolled asthma noted that tralokinumab use (300 mg SC every 2 or 4 weeks) did not result in significant percentage change in annual asthma exacerbation rates (OCS use) versus placebo (2 weeks: 6%; 95% CI: −31% to 33%; P = 0.71 and 4 weeks: −2%: 95% CI: −46% to 29%; P = 0.90). 88 Prebronchodilator FEV1 was significantly increased in the tralokinumab every 2 weeks group compared with placebo (change from baseline 7.3%; 95% CI: 2.6%–12.0%: P = 0.003) but not in the tralokinumab every 4 weeks group (1.8%; 95% CI: −2.9% to 6.6%; P = 0.45). Subgroup analysis identified adults with a high baseline serum dipeptidyl peptidase-4, or high serum periostin levels had improved FEV1, asthma control, and exacerbation rates compared with placebo.
Fevipiprant
Fevipiprant is a competitive antagonist to chemoattractant receptor-homologous molecule expressed on T helper 2 cells (CRTh2). CRTh2 is a prostaglandin D2 receptor that mediates inflammatory effects largely through its production by allergic cells such as mast cells. Results with fevipiprant have been inconsistent and limited to phase 2 studies. One study of 157 adults with mild-to-moderate allergic asthma who were weaned off ICS and LABA therapy noted no difference between fevipiprant (500 mg orally once daily) and placebo in FEV1 (P = 0.41) or asthma control (P = 0.11) between the groups. 87 However, a subgroup analysis of adults with more severe airway obstruction (FEV1 < 70% predicted) did demonstrate some significant improvements in both symptoms (P = 0.009) and lung function (P = 0.002) compared with placebo. Another phase 2 RDBPCT of 61 adults with moderate-to-severe asthma and elevated sputum eosinophil count (≥2%) noted a reduction in airway eosinophilic inflammation (P = 0.0014) in the fevipiprant (225 mg twice a day orally) group. 90
At this time, no studies have been complete in children and adolescents, and the biomarker profile of patients most likely to respond to fevipiprant remains unknown. However, several studies are randomizing adolescents and adults with moderate-to-severe asthma to fevipiprant versus placebo to determine efficacy (NCT03215758, NCT03226392, NCT02563067, and NCT0255568).
Tezepelumab
Tezepelumab is a human anti-TSLP monoclonal immunoglobulin that prevents binding of TSLP with its receptor, preventing TSLP-initiated inflammatory responses through activation of dendritic cells and mast cells. There has only been one proof-of-concept RDBPCT of tezepelumab (700 mg IV once a month) in 31 adults with allergic asthma that noted attenuation of both the early and late asthmatic responses, with a 45.9% smaller decrease in FEV1 during the late phase response after 12 weeks of treatment (P = 0.02) as well as significantly decreased blood and sputum eosinophils both before and after allergen challenge (Fig. 1). 91
A phase 2 RDBPCT of tezepelumab in adults with uncontrolled asthma despite medium-to-high ICS and LABA therapy noted significant reductions in exacerbation rates (P < 0.001) with tezepelumab (70 mg every 4 weeks, 210 mg every 4 weeks, or 280 mg every 2 weeks), independent of baseline serum eosinophil levels, as well as an improvement in FEV1 (P = 0.002). 92 Limitations are similar to fevipiprant, as there is a paucity of studies, no studies have been done in the pediatric population, the biomarker profile of those most likely to respond remains unknown, and it is unclear whether there will be a disease-modifying effect with this medication. No ongoing studies at this time with anti-TSLP biologics are being done in children or adolescents; studies remain limited to adults (NCT02698501).
Conclusion: Current Guidelines and Future Directions
Several international asthma guidelines now include the use of omalizumab and anti-IL5 medications for patients with severe asthma, including children and adolescents. The National Heart, Lung and Blood Institute's Expert Panel 3 (NHLBI EPR3) guideline recommends the use of omalizumab as adjunctive therapy in patients ≥12 years old who have atopic severe persistent asthma inadequately controlled with high-dose ICS and LABA therapy (ie, step 5 or 6). 93
The Global Initiative for Asthma (GINA) 2017 update recommends phenotype-guided add-on treatment with omalizumab in patients ≥6 years with severe allergic (elevated IgE) asthma and mepolizumab or reslizumab in patients ≥12 years with eosinophilic asthma as step 4 therapy. 94
The Canadian Thoracic Society (CTS) recommends omalizumab in patients ≥6 years with severe asthma (inadequately controlled on high dose ICS and at least 1 other controller), sensitization to ≥1 perennial aeroallergen, and serum IgE levels between 30 and 1,300 IU/mL (6–11 years) or 30–700 IU/mL (≥12 years). 9 The CTS also recommends anti-IL5 therapies in adults with severe eosinophilic corticosteroid-dependent asthma in an attempt to decrease/withdraw OCSs.
Although there is expanding evidence for biologic therapies in the pediatric population for some medications, such as omalizumab, the evidence remains limited to adults for the majority of this class of medications. Although most of the evidence is supportive of the role of these medications in severe asthma, however, asthma in childhood is quite different from asthma in adulthood, and results from adult studies may not be extrapolated to the pediatric population. For example, there is a higher prevalence of asthma in male children, whereas adult females have more severe and persistent disease. 1 Adult onset asthma may vary phenotypically from pediatric onset asthma 1 with pediatric asthma more commonly allergic in nature. 95
Many other questions remain, such as the utility of currently available biomarkers (with issues such as significant fluctuation of serum eosinophil levels and FeNO) as well as whether the biologics are as effective in all pediatric populations, as disease prevalence and severity are more common in the multiple race, African American, and American Indian children than it is in Caucasian children. 1 In addition, tolerability of parenteral medications by children and their families remains to be seen, although fevipiprant is an oral medication.
Moving forward, as studies continue to define the role of these biologic medications in the pediatric population, there is the emergence of an individualized approach to the asthmatic child, with the goal of decreasing the burden of asthma severity in childhood and advancing application to disease modification and prevention strategies. Long-term studies will be important to determine whether the medication has an effect on preventing ongoing loss in pulmonary function or improving pulmonary function, perhaps as an indicator of the reversal of airway remodeling. To date, the most experience in children with severe asthma has been with omalizumab, which is now approved to 6 years of age in some countries. Therefore, if the serum IgE is within limits for the age considered, it is usually considered as the first choice. The clinical impact appears to be on reducing the risk of exacerbations and also the possibility of reducing medication requirements. Next, 2 forms of anti-IL5 are available for use in some countries for adolescents of 12 years of age, specifically mepolizumab and benralizumab. A blood eosinophil count >300 cells/μL and perhaps 150 cells/μL is associated with an increased likelihood of achieving a favorable response on reducing exacerbations. More information and potential approval of medications, such as dupilumab, is anticipated, wherein experience in adults and adolescents shows effect on reducing exacerbations, improving pulmonary function, and the absence of significant adverse effects, such as anaphylaxis. 96 Some biologics are being considered for home administration. It will be important to assure efficacy and low risk to allow this be done safely and also important to assess adherence to treatment with home administration.
In addition, the NIAID Inner City Asthma Consortium and the NHLBI Precision Interventions for Severe and/or Exacerbation Prone Asthma (PrecISE) Network are conducting and designing studies to obtain more information on the use of biologics in children as well as adults and developing information on predictive and monitoring biomarkers. This information should be very helpful in personalizing the selection of these medications for children and also for evaluating their potential in altering the course of the disease when it begins in childhood.
Overall, it will be important to define predictive and monitoring biomarkers to assess the likelihood a patient will respond to a biologic and also to determine whether there is a favorable response to continue the biologic. This will provide a profile of those patients most likely to receive a favorable response and also to discontinue a medication if there is low likelihood of a response. Long-term studies will be needed to determine whether the biologic prevents progression of the disease and whether there is a chance for the biologic to further prevent or even reverse airway damage that has already occurred. While new medications are being introduced that have an opportunity to significantly impact the disease, benefit versus risk must be monitored to determine what favorable response patients are most likely to show.
Footnotes
Acknowledgment
No honorarium, grant, or other form of payment was given to write the article. No funding was received from any source.
Author Disclosure Statement
E.M.A. has received moderator fees from Novartis. A.B.B. has consulted to and/or received honoraria from AstraZeneca, Boegringer Ingelheim, and Novartis, and is a member of the GINA Scientific Committee and co-author of the GINA global strategy for asthma management and prevention. S.J.S. has consulted for Aerocrine, Astra Zeneca, Boehringer-Ingelheim, Daiichi Sankyo, Glaxo Smith Kline, Genentech, Novartis, Roche, and Teva and has received research support from the National Institutes of Health, the National Heart, Lung, and Blood Institute, GlaxoSmithKline, and the Colorado Department of Public Health and Environment Colorado Cancer, Cardiovascular, and Pulmonary Disease Program. He is also a former member of the Global Initiative for Asthma Scientific Committee and the National Asthma Education and Prevention Expert Panel 3.
