The purpose of this study was to determine whether pranlukast, a leukotriene receptor antagonist,
attenuates exercise-induced bronchospasm (EIB) in children with asthma. The study
was designed as a randomized, multicenter, double-blind, placebo-controlled two-period
crossover trial. The subjects were 17 stable asthmatic children aged 7–14 years old (mean,
11.0 years) with a history of typical symptoms of EIB. They received dry syrup of pranlukast
or placebo both after supper on the first day and after breakfast on the second day
of the study in each period. All exercise challenges were performed on a bicycle ergometer
and spirometry measurements of pulmonary function (FVC, FEV1.0, PEF, MMF,
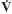 50 and
50 and
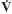 25) were collected pre-and post-exercise. Compared with placebo, pranlukast significantly
inhibited the reduction of pulmonary function after exercise. Consequently, this study
demonstrated that pranlukast, a leukotriene receptor antagonist, is a useful drug in childhood
asthma management. (Pediatr Asthma Allergy Immunol 2005;18[1]:5–11.)
25) were collected pre-and post-exercise. Compared with placebo, pranlukast significantly
inhibited the reduction of pulmonary function after exercise. Consequently, this study
demonstrated that pranlukast, a leukotriene receptor antagonist, is a useful drug in childhood
asthma management. (Pediatr Asthma Allergy Immunol 2005;18[1]:5–11.)