Abstract
Abstract
Cortical gyrification, as a specific measure derived from magnetic resonance imaging, remains understudied in mild traumatic brain injury (mTBI). Local gyrification index (lGI) and mean curvature are related measures indexing the patterned folding of the cortex,ml which reflect distinct properties of cortical morphology and geometry. Using both metrics, we examined cortical gyrification morphology in 59 adult males with mTBI (n = 29) versus those without (n = 30) mTBI in the subacute phase of injury (between 2 weeks and 3 months). The effect of IQ on lGI and brain-symptom relations were also examined. General linear models revealed greater lGI in mTBI versus controls in the frontal lobes bilaterally, but reduced lGI in mTBI of the left temporal lobe. An age-related decrease in lGI was found in numerous areas, with no significant group-by-age interaction effects observed. Including other factors (i.e., mTBI severity, symptoms, and IQ) in the lGI model yielded similar results with few exceptions. Mean curvature analyses depicted a significant group-by-age interaction with the absence of significant main effects of group or age. Our results suggest that cortical gyrification morphology is adversely affected by mTBI in both frontal and temporal lobes, which are thought of as highly susceptible regions to mTBI. These findings contribute to understanding the effects of mTBI on neuromorphological properties, such as alterations in cortical gyrification, which reflect underlying microstructural changes (i.e., apoptosis, neuronal number, or white matter alterations). Future studies are needed to infer causal relationships between micro- and macrostructural changes after an mTBI and investigate potential sex differences.
Introduction
Mild (mTBI) traumatic brain injury (TBI), the most common form of TBI, 1 is a serious public health issue. 2 Defined as transient impairment to mental functioning, which may or may not include loss of consciousness, it is caused by biomechanical forces acting on the brain. 3 Despite spontaneous recovery in the majority of cases, 3 a significant minority experience persistent functional problems.4,5 Although neuroimaging has been used post-mTBI to understand the neurobiology of injury, it is not currently diagnostic or prognostic for mTBI. Rather, other protocols, such as the Glasgow Coma Scale (GCS), 6 clinical history, and information regarding the injurious event, are used for diagnostic purposes. 7
Magnetic resonance imaging (MRI) studies have extensively contributed to our understanding of mTBI through reports of structural8–10 and functional 11 dysregulation. The assessment of cortical gyrification—a calculation of cortical architecture derived from MRI—has been limited in mTBI.12,13 Cortical gyrification refers to the convex and concave patterning of the cerebral cortex, which begins pre-natally and intensifies during the third trimester, 14 at which point the cortex is transformed from a lissencephalic into a gyrencephalic structure. Mirroring other metrics of cortical gray matter (volume, thickness, and surface area), cortical gyrification exhibits an inverted-U developmental trajectory, peaking at ∼2–3 years of age,15,16 and then declining sharply during childhood and adolescence 17 and gradually decreasing through adulthood.18,19 Moreover, recent imaging studies suggest that gyrification is a potential marker for age-related brain and cognitive decline beyond midlife. 20
Environmental factors may contribute to cortical gyrification morphology (i.e., meditation 21 and physical training 22 ); however, mTBI can be framed as an insidious environmental factor that may also impact gyrification and plasticity—given that neurotrauma is associated with inflammation/edema, 23 apoptosis, 24 and demyelination—presenting as alterations to gray10,25 and white matter. 26 To date, two studies12,13 have examined cortical gyrification in mTBI using separate morphological metrics of gyrification: local gyrification index (lGI) and mean curvature. lGI quantifies the degree of local gyrification by estimating the amount of the unexposed surface hidden within sulci and has been used in studies of neuropsychiatric27,28 and neurodevelopmental disorders. 29 Mean or extrinsic curvature (computed as the average of the two principal curvatures: maximal, K1 and minimal, K2 curvatures) captures the extrinsic qualities of a surface 30 (i.e., degree of sharpness 31 ) and is a related, but distinct, property of cortical geometry when compared with lGI. 32 Rather, lGI is akin to intrinsic or Gaussian curvature (product of the two principal curvatures), with the difference being that it captures a wider area (50-mm diameter). 32 Although mean curvature and lGI are related measures of the gyrification of the cortex, it is important to consider their methodological differences when determining cortical morphology and geometry.
In this study, we investigated cortical gyrification morphology in a cohort of male adults with and without mTBI using both the lGI and mean curvature measures to assess associations between lGI and mTBI severity during the acute/subacute phase and functional symptomology. Previous studies using this cohort have determined changes to structural morphology in acute/subacute mTBI, using various indices including decreased cortical volume and cortical thickness. 10 Given the accepted association between cortical thickness and neuronal number,33,34 and mean curvature as a reliable measure of underlying white matter atrophy, 35 we predict that mTBI will result in a reduction in gyrification and curvature and provide a better understanding of morphological dysregulation in the cortex after mTBI. To test this prediction, we examined 1) lGI group differences with and without controlling for the effects of mTBI severity, symptoms, and IQ and 2) mean curvature group differences.
Methods
Participants
Adult males (N = 59; 29 mTBI, 30 control) 20–45 years of age were recruited for this study. Participants with mTBI were recruited from a level 1 trauma center (Sunnybrook Health Sciences Centre, Toronto, Ontario, Canada) in Toronto, with a diagnosis made by a physician. Inclusion criteria were: an mTBI within the previous 3 months (mean time post-injury at scan = 33.33 days), no reported history of mTBI, and a normal head computed tomography scan at admission. Symptomatology was not a prerequisite for inclusion, but when present was limited to loss of consciousness of no more than 30 min, post-traumatic amnesia, alterations of consciousness, and/or confusion of no more than 24 h and GCS >13 in the first 24 h after injury. 6
Control group participants were recruited from the local community and through flyers posted at the hospital. Exclusion criteria included: self-reported history of neurological, psychological, and psychiatric disorders and previous concussive TBI that resulted in a transient alteration of mental function. Exclusion criteria for both groups included taking anticonvulsant medications, benzodiazepines, and gamma-aminobutyric acid antagonists, contraindication to MRI, or gross neurostructural abnormalities and/or significant artefacts in their MRI scan.
This study was approved by and conducted in accordance with the research ethics boards of The Hospital for Sick Children (Toronto, Ontario, Canada) and Sunnybrook Health Sciences Centre. Informed written consent was obtained from all participants.
Cognitive-behavioral assessment
Intelligence levels were assessed using Wechsler Abbreviated Scale of Intelligence (WASI). 36 mTBI symptomology and severity were assessed using the Sports Concussion Assessment Tool 2 (SCAT2). 37 These assessments were administered to all participants on the day of MRI scanning. For our brain-behavior analyses, we obtained the “total symptom” and “symptom severity” scores from SCAT2. The symptom score refers to the total number of symptoms a participant has, and a maximum possible score would be 22, whereas the severity score refers to the severity of the symptoms a participant has—obtained through the summation of all scores—yielding a maximum score of 132.
Neuroimaging data acquisition parameters
T1-weighted images were obtained using a 3 Tesla Siemens Trio MRI scanner (with a 12-channel head coil, MAGNETOM Tim Trio; Siemens AG, Erlangen, Germany) at the Hospital for Sick Children. The sequence was a three-dimensional (3D) sagittal magnetization-prepared rapid gradient echo with repetition time = 2300 ms, echo time = 2.96 ms, inversion time = 900 ms, and flip angle = 90 degrees. A field of view of 240 × 256 mm with 192 slices yielded 1-mm isotropic voxels. Motion restriction and stabilization of the head during imaging were attained with foam padding.
Image processing
FreeSurfer software version 6.038 was used for image processing and reconstruction, as described in a previous work. 39 Briefly, T1-weighted images were registered to MNI305 atlas before intensity normalization, skull stripping, and white matter segmentation steps. A cortical surface mesh was computed for each individual scan. Pial (gray matter/cerebral spinal fluid boundary) and white (gray-white matter boundary) surfaces were then differentiated. Using spherical registration, individual scans were registered to FreeSurfer's template space, called fsaverage. Last, cortical segmentation based on the Desikan-Killany atlas was performed.
Quality control
Quality control on FreeSurfer output was performed based on ENIGMA Cortical Quality Control Protocol 2.9 (April 2017; http://enigma.ini.usc.edu) through visual inspection of gray-white matter segmentation and cortical labeling. Manual troubleshooting was performed when needed. Three participants (2 mTBI, 1 control) were excluded, resulting in 27 mTBI and 29 controls in the final sample (Table 1).
Participant Demographics
SD, standard deviation; SA, surface area (mm2); eTIV, estimated total intracranial volume (mm3); mTBI, mild traumatic brain injury; N.S., no significant differences between groups as indicated by t-tests.
Local gyrification index and mean curvature
lGI was computed on FreeSurfer by quantifying the degree of gyrification locally at thousands of vertices across each hemisphere. 32 lGI is a 3D measure of the ratio between the area of an estimated circular region of interest (ROI; 25-mm radius) on the pial surface and the area of the corresponding ROI on the outer surface (i.e., lGI of 5, highly folded cortex; lGI of 1, a smooth cortex).
Mean curvature was computed by default on FreeSurfer after the execution of the “recon-all” command. At each vertex on the surface, mean curvature was calculated as the average of the two principal curvatures, K1 and K2, maximum and minimum curvatures, respectively.
Statistical analyses
To compare lGI and mean curvature values between groups, we undertook a whole-brain approach and conducted general linear models (GLMs) on the Query, Design, Estimate, Contrast (QDEC) application on FreeSurfer, utilizing the DODS (different offset, different slope) design matrix. For lGI analyses, we controlled for the effect of surface area given the strong positive correlation between the lGI measure and surface area, 40 and we did not apply any smoothing. 41 For mean curvature analyses, we controlled for the effect of estimated total intracranial volume (eTIV), similar to previous curvature studies,31,42,43 and applied a 20–full width at half maximum smoothing kernel. The effect of age was controlled for in both lGI and mean curvature analyses, and all covariates were demeaned. Last, Monte-Carlo z-simulations (p < 0.05) correction was used.
To examine the relation of IQ with lGI, we repeated our lGI main analyses (with the identical procedures described above) on the subset of participants with available IQ scores (23 mTBI, 22 controls). Briefly, including this covariate resulted in minimal changes to the between-groups contrast without the covariate, showing that IQ played a minimal role in the results. To explore the association between mTBI severity and symptoms with lGI, we undertook a whole-brain approach and conducted GLMs on QDEC (DODS) while controlling for the effects of diagnosis, age, and surface area (latter two demeaned). No smoothing was applied, and Monte-Carlo (p < 0.05) correction was used.
Results
Participant demographics
Independent t-tests conducted on the whole sample (n = 56) indicated no significant group differences in age, surface area, or eTIV (Table 1).
Mild traumatic brain injury is associated with increased frontal and reduced temporal local gyrification index
GLMs yielded significant group differences with greater bilateral lGI in mTBI compared to controls in the frontal lobes (Fig. 1A; left: cluster 1 peak: lateral orbitofrontal, cluster 2 peak: superior frontal; right: cluster peak: pars orbitalis; all ps = 0.0001), but reduced lGI in mTBI in a cluster located in the left temporal lobe (Fig. 1A; cluster 3 peak: middle temporal; p = 0.0012).

(
We examined lGI in participants with available IQ scores (WASI; n = 23 mTBI, n = 22 controls; Fig. 1B). In the left hemisphere, we found greater lGI in mTBI compared to controls in the lateral and medial orbitofrontal cortex (cluster 1 peak: lateral orbitofrontal; p = 0.0001) and posterior cingulate gyrus (cluster 3 peak: superior frontal; p = 0.0002; note: this cluster does not extend to the medial superior frontal lobe as it did in the main analysis). Reduced lGI in mTBI was observed in the temporal lobe, but unlike the main analysis, this effect was then shifted superiorly to include the middle (cluster 4 peak: middle temporal; p = 0.0002) and superior temporal gyri extending to the insular region (cluster 2 peak: superior temporal; p = 0.0001; Fig. 1B), rather than the inferior temporal gyri (Fig. 1A). We found greater lGI in mTBI in the right medial and lateral prefrontal cortices (cluster 1 peak: rostral anterior cingulate; p = 0.0089); however, these clusters were much smaller than those identified in the main analysis. A new right hemisphere cluster was identified in the lateral temporal lobe (cluster 2 peak: middle temporal; p = 0.03420) depicting reduced lGI in mTBI. Significant positive associations between lGI and IQ and interaction effects are reported in the Supplementary Materials (Supplementary Fig. S2).
Mild traumatic brain injury severity and symptoms are associated with increased frontal and reduced temporal and parietal local gyrification index
To determine associations of lGI with mTBI severity (SCAT2; N = 47; mTBI, n = 27; control, n = 20), we further examined group differences in lGI with severity included in the model. We found greater lGI in mTBI in a left orbitofrontal cluster (Fig. 1C; cluster 1 peak: lateral orbitofrontal; p = 0.0001) and reduced lGI in mTBI in a left temporal lobe cluster (cluster 3 peak: middle temporal; p = 0.0071). The significant medial frontal lobe cluster no longer reached significance; however, a significant cluster was found in the superior parietal lobe (cluster 2 peak: precuneus; p = 0.0001) depicting reduced lGI in mTBI. Right hemisphere analysis showed greater lGI in mTBI in clusters located in the frontal lobe (cluster 1 peak: rostral anterior cingulate, p = 0.0001; cluster 2 peak: pars orbitalis, p = 0.03490). Significant associations between lGI and mTBI severity are reported in the Supplementary Materials (Supplementary Fig. S1A).
We also examined group differences in lGI (n = 47) with symptom number included in the model. Greater lGI in mTBI was observed in a left orbitofrontal cluster (Fig. 1D; cluster 1 peak: medial orbitofrontal; p = 0.0001). However, the significant temporal lobe and medial superior frontal lobe clusters in the main analysis did not reach significance. Instead, reduced lGI in mTBI was observed in a superior parietal cluster (cluster 2 peak: precuneus; p = 0.0001). In the right hemisphere, greater lGI in mTBI was found in clusters located in the frontal lobe (cluster 1 peak: rostral anterior cingulate, p = 0.0001; cluster 2 peak: rostral middle frontal, p = 0.0225). Significant associations between lGI and mTBI symptoms are reported in the Supplementary Materials (Supplementary Fig. S1B).
A significant decrease of lGI with age was found across groups in two clusters covering the left frontal and parietal lobes (Fig. 2; cluster 1 peak: superior frontal, p = 0.0001; cluster 2 peak: pars opercularis, p = 0.013) and two clusters covering all lobes, except the occipital on the right hemisphere (cluster 1 peak: superior frontal, p = 0.0001; cluster 2 peak: superior temporal, p = 0.0033; Fig. 2). No significant group-by-age interaction effects were observed.
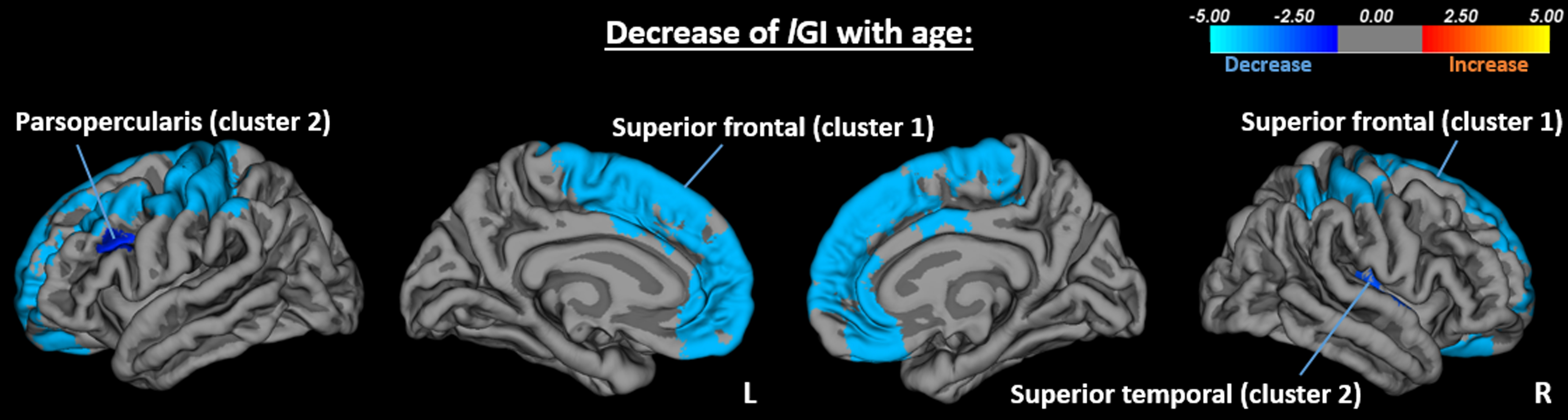
Decrease of lGI with age in left (cluster 1 peak: superior frontal, p = 0.0001; cluster 2 peak: pars opercularis, p = 0.013) and right (cluster 1 peak: superior frontal, p = 0.0001; cluster 2 peak: superior temporal, p = 0.0033) hemispheres.
Mean curvature
No significant main effects of group or age were found in either hemisphere (Fig. 3). However, a significant group-by-age interaction effect was observed, showing an increase of mean curvature with age in the mTBI group, but a decrease of mean curvature with age in the control group, in a cluster covering parts of the right medial occipital and temporal lobes (peak: lingual; p = 0.0059).

Mean curvature group-by-age interaction effects (peak: lingual; p = 0.006).
Discussion
In this study, we used two distinct metrics of cortical gyrification to understand alterations in neuromorphology after mTBI in adult males. A striking observation was that of atypical lGI in mTBI in the frontal and temporal lobes—two highly susceptible regions in mTBI44,45—which demonstrates both dysregulated gyrification and microstructural disparities in these cohorts. The mechanisms of cortical gyrification in early brain development is yet to be elucidated, with theories highlighting the role of mechanical forces or biological factors that dictate the emergence of the characteristic gyri and sulci of the cerebral cortex. At a cellular level, increased gyrification is found in regions that have undergone higher rates of neurogenesis (and thus contain abundant cortical progenitors) compared to regions that have undergone lower rates of neurogenesis. 46 As such, altered gyrification in persons with mTBI likely reflects alterations to neuronal populations, supported by histological examinations in pre-clinical mTBI models. 24
Alternatively, the tension-based hypothesis of cortical gyrification proposes that mechanical tension along axons in early development results in strongly interconnected regions being pulled toward one another whereas less-interconnected regions separate, resulting in the emergence of gyri and sulci. 47 This is reflected by associations between gyrification and underlying structural connectivity. 48 Thus, cortical gyrification atypicalities in mTBI could reflect white matter structural connectivity disruptions, which we know is present in mTBI. 26 Further, mTBI-evoked neuroinflammation can cause cerebral edema and/or damage to axonal cytoskeleton architecture 23 disrupting white matter microstructure, resulting in altered structural connectivity. Thus, the atypical macrostructural alterations in gyrification observed in persons with mTBI may reflect underlying microstructural and/or cellular consequences of brain injury. However, it is important to note that there is still a lack of consensus regarding the theory that best accounts for cortical gyrification.49,50
Key observations of our study were region-specific increases and decreases in lGI in mTBI. These findings reflect aspects of Wilde and colleagues' (2021) mTBI study 13 ; however, study population differences between our studies may account for differences in our observations. Wilde and colleagues' study sample included male and female adolescents (mean age = 14 years), whereas our cohort included adult males (mean age = 29 years). These contrasting findings indicate that age at injury influences both developmental effects 51 and recovery trajectories, 52 as well as structural morphological outcomes. For instance, it may be that pediatric mTBI affects gyrification differently compared to mTBI occurring during adulthood, reflecting dynamic changes in gyrification throughout the life span.18,19
In support of previous observations (Wilde and colleagues), we found that greater lGI in the left temporal lobe was associated with reduced mTBI severity (and symptoms). This is in support of our findings of reduced lGI in the lateral temporal lobe in mTBI relative to controls. Further positive associations between lGI-mTBI severity (and symptoms) were found in the left parietal lobe. Interestingly, with the inclusion of symptom or severity, we observed an additional left superior parietal cluster of reduced lGI in mTBI that overlapped with the cluster showing positive associations between lGI-mTBI severity and symptoms. As such, mTBI is generally associated with atypical gyrification in the left superior parietal lobe. Inclusion of symptom scores resulted in gyrification atypicalities merely in the left frontal and parietal lobes and left temporal cluster, showing that reduced lGI in mTBI no longer reached significance.
Inclusion of IQ yielded similar results to the main analysis with the addition of a right lateral temporal lobe cluster depicting reduced lGI in mTBI compared to controls. Additionally, the left temporal lobe cluster depicting reduced lGI in mTBI in the main analyses shifted superiorly to encompass the insular region, supporting other observations of atypicalities in the insular region post-mTBI such as gray matter volume. 53 In agreement with findings of a positive association between gyrification and intelligence level in normative literature,54,55 we also found a positive association between IQ and lGI across groups.
Cortical gyrification undergoes dynamic changes throughout the life span, and in agreement with the widely reported findings of age-related decrease of lGI after toddlerhood,18,19 we also found a decrease of lGI with age across groups. The absence of significant group-by-age interaction effects may suggest a similar trajectory across groups in the age range of our cohort.
In contrast to the previous observation of increased mean curvature in mTBI compared to controls, 12 we found no group differences or a main effect of age, with the exception of an increase of mean curvature with age in the mTBI group, but a decrease in the control group, in a right hemisphere cluster (Fig. 3). The null findings in our analyses may be explained by biological and methodological factors. For instance, no difference in mean curvature may exist between adult males with and without mTBI or that dysregulation is present but weak, or that a high degree of individual variability or small sample size of our cohort results in insufficient statistical power to detect small effects. King and colleagues (2016) included a larger sample (n = 85) compared to our study (n = 56), which may have captured more subtle characteristics of cortical curvature 31 when compared to lGI.
Our study has several limitations, and the results should be interpreted with caveats. These include a small sample size, with a relatively weakly powered design to detect small effect sizes. It is also impossible to say whether these alterations result from the primary injury, are attributable to secondary or tertiary neurochemical cascades, whether they reflect a true pathological biomarker of mTBI, or are related to some other comorbid state, or even if they are purely epiphenomenological. It is also impossible to deduce whether these gyrification abnormalities existed in the pre-injurious state. With these data here, these issues are essentially unanswerable, and we hope to address these in future studies; for example, with a prospective study that uses pre-season imaging in contact sports to image changes directly after mTBI and compare against clinical groups with similar symptoms (e.g., post-traumatic stress disorder), but without TBI.
Conclusion
In sum, our study presents a comprehensive understanding of cortical gyrification in adult males with mTBI through two complementary metrics of cortical gyrification. Our work is the first to identify cortical gyrification atypicalities in adults with mTBI in support of previous findings in pediatric cases. Future large-scale longitudinal studies, including female participants, are needed to investigate the nexus between cortical gyrification and microstructural properties to infer causal relationships between micro- and macrostructural changes after a concussion and investigate sex effects. Future longitudinal studies, with multiple time points, would shed light on patterns of recovery, which are not directly associated with symptomology. Such conclusions cannot be made with our current cross-sectional study design and need to be empirically investigated by future longitudinal efforts.
Footnotes
Authors' Contributions
Avideh Gharehgazlou: Formal analysis, Methodology, Software, Validation, Visualization, Writing—original draft. Rakesh Jetly: Funding acquisition, Writing—review and editing. Shawn G. Rhind: Funding acquisition, Writing—review and editing. Amy C. Reichelt: Writing—review and editing. Leodante Da Costa: Participant recruitment, Writing—review and editing. Benjamin T. Dunkley: Data curation, Funding acquisition, Project administration, Investigation, Supervision, Writing—original draft, Writing—review and editing.
Funding Information
This study was funded by grants from Defence Research and Development Canada (DRDC), the Canadian Institute for Military and Veteran Health Research (CIMVHR), and the Innovation for Defence Excellence and Security (IDEaS) Program.
Author Disclosure Statement
No competing financial interests exist.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
