Abstract
Outpatient care following nonhospitalized traumatic brain injury (TBI) is variable, and often sparse. The National Academies of Sciences, Engineering, and Medicine’s 2022 report on Traumatic Brain Injury: A Roadmap for Accelerating Progress highlighted the need to improve the consistency and quality of TBI care in the community. In response, the present study aimed to identify existing evidence-based guidance and specific clinical actions over the days to months following nonhospitalized TBI that should be prioritized for implementation in primary care. In systematic literature searches, 17 clinical practice guidelines met our eligibility criteria and an additional expert consensus statement was considered highly relevant. We extracted 73 topics covered by one or more existing clinical practice guidelines. After removing redundant and out-of-scope topics, those deemed essential (not requiring prioritization), 42 topics were subjected to a prioritization exercise. Experts from the author group (n = 14), people with lived experience (n = 112), and clinicians in the community (n = 99) selected and ranked topics they considered most important. There were areas of agreement (e.g., early education was ranked highly by all groups) and discordance (e.g., people with lived experience perceived diagnostic tests/investigations as more important than the other groups). We synthesized the prioritization survey results into a top-10 list of the highest priority clinical actions. This list will inform implementation efforts aimed at improving post-acute care for nonhospitalized TBI.
Introduction
Traumatic brain injury (TBI) remains a leading cause of disability across the life span. 1 The majority of people with TBI require minimal to no inpatient hospital care. Outcomes from nonhospitalized TBI are variable, with persistent symptoms and disability in up to one-third of patients. 2 Despite considerable scientific advances, diagnosis, prognostication, treatment, and equitable care access pose ongoing challenges. Recognizing this, the U.S. Department of Defense funded the National Academies of Sciences, Engineering, and Medicine (National Academies, hereafter) to conduct a study to identify barriers and possible solutions for improving TBI care and research. After reviewing the scientific literature and hearing testimony from people with lived experience (PWLE) and others involved in TBI prevention, care, recovery, and research, the committee outlined eight recommendations in their 2022 report Traumatic Brain Injury: A Roadmap for Accelerating Progress. 3 The National Academies subsequently established a Forum on TBI to facilitate implementation of these recommendations. An Action Collaborative on TBI Care was convened under the auspices of this Forum to compile and disseminate best practices and resources on symptom management, care models, patient/family/provider materials, and other information aimed at enhancing recovery in the period after TBI.
Action Collaborative members’ work focused initially on adults with TBI who do not present to an emergency department or are discharged directly from the emergency department (i.e., nonhospitalized), but also applicable to people with TBI who are able to care for themselves at discharge from acute care. Most cases of nonhospitalized TBI would fulfill criteria for mild TBI. 4 Because the National Academies TBI Roadmap report 3 called for replacement of the “inaccurate and misleading” (pg. 5) convention to trichotomize TBI severity into mild, moderate, and severe, and the subsequent National Institutes of Health initiative to propose a new classification system, 5 the Action Collaborative elected to not use the term mild TBI.
One of the key recommendations in the National Academies TBI Roadmap report 3 was to “reduce unwarranted variability and gaps in administrative and clinical care guidance to ensure high-quality care for TBI” (pg. 6). Toward this end, members of the Action Collaborative on TBI Care formed a Working Group in December 2022 to review the landscape of current clinical practice guidelines (CPGs) for managing TBI and to identify and synthesize the top-priority recommendations from those documents, at least some of which could be applied in primary care. The CPG Working Group focused on the post-acute phase (the days to ∼6 months following injury) because this period was perceived to have the greatest variability and gaps in care, and complement efforts toward management of TBI as a chronic disease. 6 Members of other Action Collaborative Working Groups are currently (1) developing updated patient-facing education materials, (2) proposing core elements of an interdisciplinary post-acute TBI clinic, and (3) identifying goals for TBI follow-up care.
Here we describe the CPG Working Group’s process for identifying priority clinical actions for outpatient management of nonhospitalized TBI. The process involved extracting topics from existing CPGs; conducting prioritization surveys with experts, clinicians, and PWLE; and a final consolidation by consensus of CPG Working Group members. Identifying top priority clinical actions can inform the development of guidelines for implementation in primary care settings.
Methods
Identifying existing CPGs
The CPG Working Group was aware of recently performed systematic searches that had broader criteria than considered necessary for the present purpose. We decided it would more efficient to screen these search results rather than conduct a new (redundant) systematic search. We obtained the search results containing all included and ineligible articles from Sander et al, 7 and all included articles from systematic reviews performed by Berrigan et al 8 (updated to April 2021) and Lithopoulos et al 9 (updated to October 2022).
The CPG Working Group created a priori inclusion/exclusion criteria for existing CPGs based on an adapted version of National Guideline Clearinghouse definition of a CPG 10 and additional criteria for alignment with the scope of TBI Forum’s Action Collaborative on TBI Care (see Supplementary Data S1). Two independent raters (pairs of CPG Working Group members) screened titles and abstracts, applying the eligibility criteria. Another pair of independent raters reviewed full-text reviews, with discrepancies resolved by a third (M.T.B.). We searched for additional CPGs and recent updates of eligible CPGs from the reference lists of published reviews of CPGs 11,12 and forward citation tracking of eligible CPGs. The number of articles excluded at each stage is reported in Figure 1.
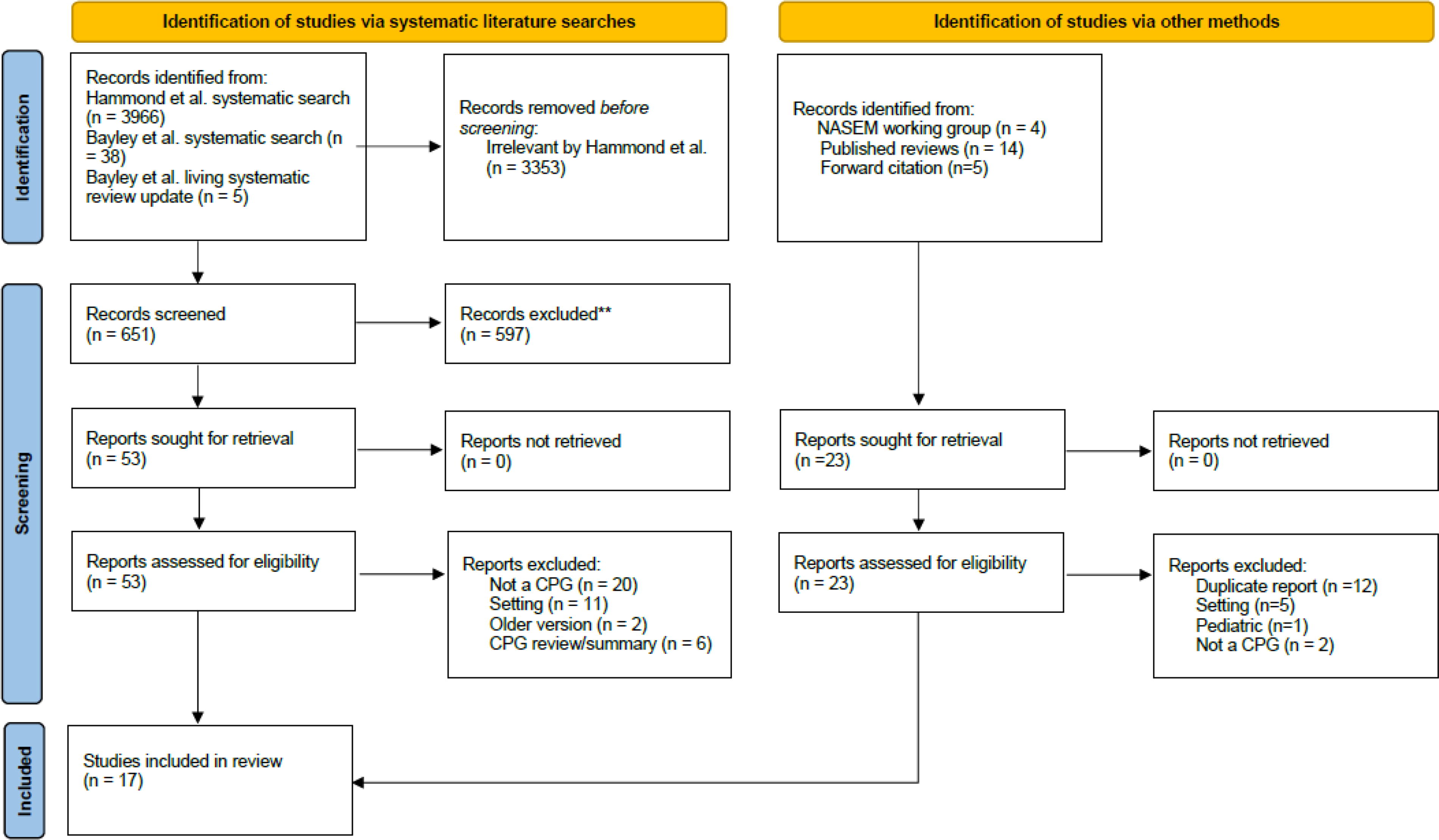
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram. CPG, clinical practice guideline; NASEM, National Academies of Sciences, Engineering, and Medicine.
The systematic search process yielded 17 eligible CPGs. The CPG Working Group decided to include the Concussion in Sport consensus statement 13 because it was expected to have a major impact on clinical practice even though it did not meet our definition of a CPG and does not identify itself as a CPG. This addition raised the total to 18 eligible CPGs (listed in Supplementary Data S1).
Identified topics covered in eligible CPGs
Each of the 18 eligible CPGs was assigned to a CPG Working Group member (one extractor per CPG). The extractors’ task was to complete a spreadsheet to mark which topics were covered in each CPG. The co-chairs (N.D.S. and K.L.) drafted an initial seed list of topics in CPGs and invited extractors to add topics covered in the CPG but not included in the seed list.
The extraction process identified 70 topics covered in eligible CPGs. Of these, three were determined to cover multiple distinct elements of care, and so were separated, resulting in a total of 73. This list of 73 topics was circulated to all CPG Working Group members, who were invited to add topics that they believed should be covered in a CPG on nonhospitalized TBI but were not covered in previous CPGs. Seven new topics were added through this mechanism. Of the complete list of 80 topics, 19 were removed because they were out of scope (e.g., involved pre-hospital or hospital care) and another 16 were removed because they were redundant or could be integrated within another topic (e.g., return to driving was incorporated into return to activity).
Of the remaining 45 topics, three were excluded from the subsequent prioritization process because the CPG Working Group considered them essential, leaving 42 topics (Supplementary Data S1). These three essential topics were as follows: (1) confirm a diagnosis of TBI, (2) determine the severity of the TBI, and (3) determine if the patient requires assessment at an emergency department for possible life-threatening complications. In a later CPG Working Group meeting, after publication of the 2023 update of the National Institute for Health and Care Excellence (United Kingdom) guideline on TBI, 14 which included a new recommendation (#1.4.12) on abuse/neglect as a possible cause of TBI, the CPG Working Group decided to add screening (and possible safeguarding) for abuse/neglect as a fourth essential topic. Finally, even though screening for social determinants of health had been included in the prioritization process, the CPG Working Group decided to add it as a fifth essential topic.
Public prioritization survey
To elicit input from the public, the CPG Working Group designed an anonymous survey modeled after the type used for “interim prioritization” in James-Lind Alliance Priority-Setting Partnerships, for example, see Ref. 15 The survey asked respondents to self-identify as (1) a person who sustained a TBI, (2) a caregiver for a person with TBI, or (3) a health professional who provides TBI care. Respondents were then instructed to select up to 10 topics (from a list of 42) that are “most important” to include in a CPG for TBI and in a second step, rank order their selections. The survey included lay definitions of TBI and CPG. Finally, respondents entered their demographic characteristics. With branching logic, patients and caregivers responded to additional questions about the injury, most problematic symptoms, and care received. A draft of the survey was circulated to lay reviewers at the U.S. Department of Defense and a patient focus group involved in other activities of the Action Collaborative on TBI Care to get feedback and recommendations to improve readability. A copy of the final survey is provided in Supplementary Data S1. The survey was reviewed by the Institutional Review Board of the University of California, San Francisco (IRB#: 23-39445, Reference#: 387719) and determined to be exempt.
The survey was programmed into Qualtrics and distributed by email by staff and members of the TBI Forum to a list of professional and patient associations/organizations (primarily within the United States and Canada) compiled by the CPG Working Group. CPG Working Group members also shared the survey invitation within their professional networks and encouraged snowball sampling and social media promotion. Advertisements highlighted that the public survey was intended for “adults with TBI who do not require acute hospital care (mild TBI/concussion)” or “individuals with lived experience of mild TBI, including patients and their family members.” The English-language survey opened on November 14, 2023, with an advertised deadline of December 15, 2023. A Spanish-language version was open from February 22 to March 24, 2024 and then because of low engagement, extended to April 21, 2024, with additional promotion.
Expert prioritization survey
CPG Working Group members were invited by email to complete a copy of the same survey designed for the public, but with minor changes. This survey differed from the public survey in that respondents were explicitly asked to consider the following factors for determining the “importance” of topics: (1) prevalence of the problem/symptom, or the proportion of patients the topic applies to, (2) underuse, overuse, or misuse of the advice, diagnostic test, or intervention, (3) concerns about practice variation, (4) costs associated with different practice options, (5) concerns (other than cost) about inequities in access to or delivery of diagnostic tests, interventions, or information, (6) likelihood that the guideline recommendation will be effective in influencing practice, (7) if consistently implemented, likelihood that the guideline recommendation will improve the quality of care and/or patient outcomes, and (8) quality of evidence supporting guideline recommendation. These criteria are based on the ADAPTE Manual and Resource Toolkit (Version 2.0, 2019, accessed from www.g-i-n.net June 18, 2023) and tailored for the present study. The expert survey was conducted from October to November 2023.
Statistical analysis
Rankings were reverse coded so that the highest rank (1) equals the highest score (10). Unranked topics were assigned a score of 0. We calculated an average of the reverse-coded rankings within each group. Higher mean item scores (maximum = 10) reflect topics that were ranked higher by more people. As a complementary metric of perceived importance, we also report the proportion of respondents in each group who included a given topic in their top 10, regardless of rank within top 10. Data are reported descriptively, except for a supplementary analysis in which we used independent samples t-tests to compare priority ratings in PWLE with lengthy versus no hospitalization for TBI.
Results
We received a total of 288 responses to the public prioritization survey, including 157 from PLWE (patients and caregivers) and 131 from clinicians. Of this response pool, 112 (71.3%) of PWLE provided valid data (i.e., selected at least one topic as important) and 99 (75.6%) clinicians provided valid data. Only 11 (3.8%) of the total responses and 6 (2.8%) of the valid responses were completed in Spanish. Participant characteristics are reported in Table 1. The majority of PWLE identified as women and White, reported having a college degree and an annual household income of at least $50,000, and were from the United States. The sample of clinicians had a similar demographic profile but was more balanced on gender.
Demographics of Survey Respondents
CPG, clinical practice guideline; PWLE, people with lived experience; TBI, traumatic brain injury.
The majority of PWLE respondents (84 of 101; 83.2% of valid responses) reported on a TBI that occurred more than a year ago. Nearly a third of PWLE (30 of 99; 30.3% of valid responses) reported that the most recent TBI involved a hospital admission of more than seven nights (see Table 1). PWLE who reported on a TBI requiring hospitalization more than seven nights (n = 30) had similar ratings to those reporting on nonhospitalized TBIs (n = 53) for their highest priority topics, including computed tomography (Q1), magnetic resonance imaging (MRI) (Q2), neuropsychological assessment (Q5), and computerized cognitive testing (Q6), all p < 0.05. However, PWLE respondents who had experience with a TBI involving lengthy hospitalization rated early education (Q12) as significantly more important than those who had experience with a nonhospitalized TBI [M = 4.20, SD = 3.7 vs. M = 1.7, SD = 2.9; t(81) = 3.43, p = 0.0009].
Neurologists, occupational therapists, and speech-language pathologists were the most represented disciplines. The majority of clinicians were senior (independent practice >15 years) and worked at an academic medical center or outpatient rehabilitation clinic. Virtually all clinicians reported caring for adults in the general population and a substantial portion reported providing care to athletes (n = 45; 46%) and/or military service members or veterans (n = 29; 30%). The results of the public prioritization survey are presented in Table 2, specifically, respondents’ rankings for topics that the expert group ranked within the top 10, and their own top 10 ranked topics.
Prioritization Survey Results
PWLE, people with lived experience; Green, topics rated in the top 15 by all three groups; Orange, topics rated in the top 15 by two groups; Red, topics rated in the top 15 by one group.
Rankings were reverse coded so that 1 (highest ranking) =10 (highest score), unranked topics were assigned a score of 0, and an average was calculated across respondents in each group. Higher scores (maximum = 10) reflect topics that were ranker higher by more people.
The proportion of respondents who included this topic in their top 10 (regardless of rank within top 10).
All 17 CPG Working Group members were invited to complete the expert survey, and 14 did. Their characteristics are also reported in Table 1 and results in Table 2. There were seven topics rated in the top 15 by all three groups (rows colored green) and an additional six topics rated in the top 15 by two groups (rows colored orange). Within the latter category, there were some marked discrepancies between groups. For example, PWLE and clinicians both rated neuropsychological assessment and computerized cognitive testing as very high priorities, whereas experts ranked these topics as 24th and 42nd out of 42. Only two topics were rated in the top 15 by one group and neither of the other two groups (rows colored red): screening for health condition that could complicate recovery, and when to order a brain scan.
Finally, the CPG Working Group integrated the survey results from the PWLE and clinicians with their own to create a “top 10” list of priority topics. They started by including all topics ranked highly by at least two groups (n = 14; green and yellow rows of Table 2) and those topics deemed essential by the CPG Working Group before the prioritization surveys (n = 5), leading to a list of 19 topics (Table 3—left column). The CPG Working Group then consolidated topics that could be covered in a single recommendation. For example, three topics were assessments/diagnostic tests/investigations intended to clarify how TBI affected brain structure and function, and so were condensed into a single topic. After seven iterations, the CPG Working Group reached unanimous consensus on a final “top 10” list (Table 3—right column).
Top 10 Priority Topics for a Clinical Practice Guideline on Management of Nonhospitalized Traumatic Brain Injury
TBI, traumatic brain injury.
Discussion
Many people with TBI do not receive follow-up care, 16,17 or receive inadequate or inappropriate care, predisposing them to suboptimal recovery and chronic disability. 3 This may be especially true in marginalized and minoritized groups. 18,19 CPGs can reduce the variability in care by synthesizing empirical evidence and translating them into actionable recommendations. Quality CPGs for TBI exist, but most target subpopulations (e.g., athletes with sport-related concussion 13 ) or specific providers (e.g., physical therapists 16 ) or the emergency department/acute care hospital setting. 14,20,21 None are ideally suited for clinicians in the general community, where the greatest variability in TBI care practices likely exists. 3 The comprehensiveness (voluminousness) of prior CPGs may also limit their implementation in primary care. 22 The CPG Working Group of the TBI Forum’s Action Collaborative on TBI care therefore sought to identify the highest priority clinical actions in the days to months following nonhospitalized TBI, with input from PWLE and clinicians in the community.
Several prioritization survey findings were noteworthy. There was fair agreement between PWLE, clinicians, and the CPG Working Group that follow-up care should include early education, guidance on return to activity, and assessing risk for prolonged recovery and the need for return visits and referral to specialty TBI services. Interestingly, the three surveyed groups also agreed that screening and initiating treatment for mental health complications were a priority, which aligns with evidence on the high incidence and impact of psychiatric comorbidity on TBI recovery. 23 –27 There were also areas of disagreement. Of the five most highly ranked topics by PWLE, four concerned assessments/diagnostic tests/investigations to characterize the effects of TBI on brain structure and function, or in other words, to help answer the question “how has my brain been injured?” Clinicians similarly valued these kinds of assessments. The large representation of occupational therapists and speech-language therapists in this sample, who are often providers of cognitive assessment, may partly explain this finding. However, the CPG Working Group did not rank assessments among their highest priorities. Debriefing in the CPG Working Group meetings suggested a primary reason—a low likelihood of results that would change clinical management. For example, MRI in patients with persisting symptoms following TBI not requiring hospitalization will usually show no traumatic intracranial abnormalities 28,29 or abnormalities that do not alter the treatment plan. That said, confirming an absence or presence of TBI-related pathology on MRI may be helpful in some clinical contexts.
The top 10 topics listed in Table 3 are not implementation-ready. While they highlight important content areas for TBI outpatient care, they do not provide specific guidance on what actions clinicians should take, when to take them, and with whom. Development of these topics into a new CPG could achieve that necessary specificity. We only included prior CPGs that were published in English, which is a limitation. Another limitation of the present study is underrepresentation of certain regions (outside of the United States) and demographic groups (e.g., Black and Latino/Hispanic respondents) in the public survey. The public survey also did not specify a time frame after TBI to consider when identifying their priorities. The priorities in the acute stage post-injury may not have been captured well, as the vast majority of PWLE group referred to the injury sustained >1 year ago. Moreover, despite our attempts to advertise the public survey as for PWLE with “mild TBI/concussion,” a proportion of respondents likely experienced more severe TBIs, given that 30% were hospitalized for more than 7 days and 15% required neurosurgery. This may have contributed to differences in priorities between the surveyed groups, although we found evidence of this for early education only. Primary care physicians were not well-represented in the clinician survey sample. They provide the majority of post-acute TBI care and so should be engaged in future CPG development and implementation efforts.
The CPG Working Group is now in the process of writing a new CPG by translating each of the 10 priority topics identified in the present study into actionable recommendations based on syntheses of existing CPGs. We acknowledge that merely publishing a new CPG will not markedly enhance TBI care without theory and evidence-informed implementation efforts. 22,30 Moreover, widespread adoption of a new CPG could potentially exacerbate rather than reduce care variability, particularly by recommending interventions that are costly or otherwise inaccessible in some communities. Attention to health inequity considerations in the CPG design will be essential to avoid that unintended outcome. 31,32
The top 10 priority clinical actions identified here overlap with the top 10 research priorities identified in the recent James-Lind Alliance Priority-Setting Partnership on concussion. 15 For example, early referral to TBI specialty care and treatments for post-traumatic headache appear in both lists. That PWLE and clinicians in both priority setting efforts identified similar areas of importance provides reassuring convergence. The fact that the James-Lind Alliance Priority-Setting Partnership aimed to identify the most important unanswered research questions, however, raises a concern that there may be insufficient evidence to write strong recommendations on these topics.
Conclusions
The present study scoped existing CPGs that covered, at least in part, outpatient care for nonhospitalized TBI, compiled a comprehensive list of topics covered in those CPGs, and then gathered input from experts, PWLE, and clinicians to refine the top 10 priorities for implementation. Another key contribution of the present study was to show how perceptions of importance aligned and differed between these groups. Including perspectives from PWLE in the CPG development process is critical to ensure that their needs are effectively addressed in outpatient TBI care.
Transparency, Rigor, and Reproducibility
The study protocol was not preregistered. The analyses were descriptive. No null hypothesis testing was planned or performed, and accordingly, the sample size was pragmatic rather than based on power calculations. Data were acquired from October 2023 to March 2024. The surveys are made available as supplementary material, to support reproducibility. De-identified data from this study will be made available (as allowable according to IRB standards) by emailing the corresponding author.
Footnotes
Acknowledgments
The authors wish to acknowledge Moriah Cogan for operational support for the Action Collaborative, Scott Hamilton for providing input on the public survey, and Eloisa Martinez for translating the public survey into Spanish.
Authors’ Contributions
N.D.S. and K.L. co-led the Working Group, liaising with G.M., leader of the Action Collaborative. N.D.S. prepared the first draft of the article. A.M., M.T.B., E.W.E., J.T.G., C.H., F.M.H., D.A.I., C.M., J.V.D.N., M.R.P., O.T., V.W., and M.V.G. participated in Working Group activities, including the study design and data collection and interpretation. All authors critically reviewed and approved the article.
Members of the Action Collaborative on TBI Care:
Amy Markowitz, University of California-San Francisco; Caterina Mosti, University of California-San Francisco; Cathra Halabi, University of California-San Francisco; Christopher Giza, University of California-Los Angeles; Daniel Ignacio, St. Jude Brain Injury Network; David Brody, Uniformed Services University of Health Sciences; David Okonkwo, University of Pittsburgh; Flora Hammond, Indiana University; Gavin Attwood, Brain Alliance of Colorado; Geoffrey Manley, University of California-San Francisco; Javier Cardenas, University of West Virginia; Jeff Bazarian, University of Rochester; Joel Scholten, US Department of Vetran Affairs; John Corrigan, Ohio State University; Joseph Giacino, Spaulding Rehabilitation Hospital and Harvard Medical School; Joukje van der Naalt, University of Groningen; Katherine Lee, MS, Secretary of Defense for Health Readiness Policy and Oversight; Kelly Sarmiento, MPH, Centers of Disease Control and Prevention; Laura B. Ngwenya, University of Cincinnati; Mark Bayley, University of Toronto; Mayur Patel, American College of Surgeons Committee on Trauma and Vanderbilt University Medical Center; Michael Huang, University of California-San Francisco; Michael McCrea, Medical College of Wisconsin; Monica Verduzco-Gutierrez, University of Texas Health Science Center at San Antonio; Monique Pappadis, University of Texas Medical Branch; Moriah Cogan, University of California-San Francisco; Noah Silverberg, University of British Columbia; Odette Harris, Stanford University; Olli Tenovuo, University of Turku; Rachel Lazarus, AARP; Raymond Raven, OSS Health Network; Rebeccah Wolfkiel, National Association of State Head Injury Administrators; Scott Hamilton; Steven Flanagan, NYU Langone; Christina L. Master, Children's Hospital of Philadelphia and University of Pennsylvania Perelman School of Medicine; Vincent Wang, University of Texas-Austin; Wesley Ely, Vanderbilt University Medical Center.
Author Disclosure Statement
N.D.S. has received research operating funds from multiple granting agencies (Canada Foundation for Innovation, Canadian Institutes of Health Research, Mitacs, Ontario Brain Institute, U.S. Department of Defense, WorkSafeBC, VGH+UBC Hospital Foundation) for research related to TBI diagnosis, prognosis, and treatment. He has received speaker fees for providing continuing medical education on these topics. He serves as chair of the American Congress of Rehabilitation Medicine’s Brain Injury Special Interest Group Task Force on mild TBI (unpaid). He has served as an expert panel member for the Living Concussion Guidelines and as an external reviewer for other clinical practice guidelines on concussion/TBI (unpaid). He has provided expert testimony and medical–legal consulting in the past 5 years (<10% of total income). K.L. reports no conflicts of interest. A.M. has received a fellowship from the University of British Columbia (UBC) Institute of Mental Health (IMH) Marshall Fellows Program. M.T.B. reports no conflicts of interest. D.L.B. is an employee of the U.S. Dept. of Defense. Research currently funded by the Department of Defense. Equity in Inner Cosmos LLC. Royalties from Sales of Concussion Care Manual (Oxford University Press). Honoraria from Mary Ann Liebert, Inc. Publisher of Journal of Neurotrauma for services as Editor-in-Chief. Consulting for Pfizer Inc, Intellectual Ventures, Signum Nutralogix, Kypha Inc, Sage Therapeutics, iPerian Inc, Navigant, Avid Radiopharmaceuticals (Eli Lilly & Co), the St Louis County Public Defender, the U.S. Attorney’s Office, the St Louis County Medical Examiner, GLG, and Stemedica. He has provided expert testimony and medical–legal consulting in over 50 cases (<1% of total income). None of these entities was directly involved in the present work. E.W.E. discloses that he receives ongoing funding from the Veterans Affairs and the National Institutes of Health. He received honoraria within the past 3 years from Pfizer for continuing medical education lectures he gave at international medical conferences that received Pfizer’s sponsorship. He does not have any formal consultancies or stock investment to disclose with any industry groups or companies. J.T.G. reports grant funding from the National Institute of Neurological Disorders and Stroke, Department of Defense, National Institute on Disability, Independent Living, and Rehabilitation Research for research related to TBI diagnosis, prognosis, treatment, and outcome. He also receives research support from the Barbara Epstein Foundation and SameYou Foundation. He occasionally receives honoraria from academic institutions and medical centers for training seminars concerning the Coma Recovery Scale—Revised. He serves on the Scientific Advisory Boards of the Traumatic Brain Injury Model System National Data and Statistical Center and the Neurocritical Care Society’s Curing Coma Campaign. He has provided medicolegal consultation within the last 3 years (<10% of total income). C.H. has received research support from the Pac12 Student Health and Wellbeing Initiative for concussion recovery, the Weill Institute for Neurosciences Neurohub for stroke recovery, and UC Noyce Initiative for stroke rehabilitation, unrelated to the current work. Dr. Halabi has also received research support from the National Institute on Deafness and Other Communication Disorders for aphasia recovery after stroke, also unrelated to the current work. F.M.H. has served on scientific advisory boards for Avanir and Otsuka, and received funding to her academic institution from the National Institute on Disability Independent Living and Rehabilitation Research, National Institutes of Health, Department of Defense, Patient Centered Outcomes Research Institute. D.A.I. reports no conflicts of interest. C.B.M. reports no conflicts of interest and no financial disclosures. J.V.D.N. reports no conflicts of interest. M.R.P. has received ongoing research funding to her institution from the National Institutes of Health/National Institute on Aging and the National Institute on Disability, Independent Living, and Rehabilitation Research. She serves as a consultant for TIRR Memorial Hermann, Craig Hospital, and Indiana University. O.T. has received research funding for TBI research from the EU Commission, Business Finland, and several Finnish foundations supporting scientific research. He has received speaker fees for providing continuing medical education on these topics from the Brain Injury Society of Finland and several Finnish universities and hospitals. He has served as chair of Finnish Traumatic Brain Injury Guidelines (unpaid) and participated in the development of Scandinavian guidelines on TBI as a member of the Scandinavian Neurotrauma Committee (unpaid). He has been a member of the expert panel preparing an European Consensus Panel Recommendation for patients with TBI of GCS13-15, work supported by Abbott. He has provided expert testimony and medicolegal consulting for the past 20 years (<10% of total income). He has received consultation fees from NeuroTrauma Sciences, LLC. V.W. has no financial disclosure and no conflict of interest. M.V.G. discloses she has received ongoing AHRQ grant funding for Long COVID. She has received honoraria from AbbVie, Merz, Revance, and Ipsen for speaking or consulting work on spasticity. She is on the board of the Association of Academic Physiatrists (unpaid). She has provided expert testimony and medical–legal consulting in the past 5 years (<10% of total income). K.B. reports no conflicts of interest. G.T.M. discloses research grants from the U.S. Department of Defense, the National Institutes of Health/National Institute of Neurological Disorders and Stroke, U.S. Department of Energy, the National Football League (NFL) Scientific Advisory Board, NeuroTrauma Sciences, and Abbott Laboratories. Dr. Manley is currently a member of the steering committee for an NIH-NINDS initiative for improved characterization/nomenclature of TBI and a member of the NASEM TBI Forum.
Funding Information
The Action Collaborative on TBI Care is funded by generous donations from an anonymous donor, the Weill Family Foundation, and the Maurice Marciano Family Foundation.
Supplementary Material
Supplementary Data S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
