Abstract
Spinal cord injury (SCI) results in intramedullary microvasculature disruption and blood perfusion deficit at and remote from the injury site. However, the relationship between remote vascular impairment and functional recovery remains understudied. We characterized perfusion impairment in vivo, rostral to the injury, using magnetic resonance imaging (MRI), and investigated its association with lesion extent and impairment following SCI. Twenty-one patients with chronic cervical SCI and 39 healthy controls (HC) underwent a high-resolution MRI protocol, including intravoxel incoherent motion (IVIM) and T2*-weighted MRI covering C1–C3 cervical levels, as well as T2-weighted MRI to determine lesion volumes. IVIM matrices (i.e., blood volume fraction, velocity, flow indices, and diffusion) and cord structural characteristics were calculated to assess perfusion changes and cervical cord atrophy, respectively. Patients with SCI additionally underwent a standard clinical examination protocol to assess functional impairment. Correlation analysis was used to investigate associations between IVIM parameters with lesion volume and sensorimotor dysfunction. Cervical cord white and gray matter were atrophied (27.60% and 21.10%, p < 0.0001, respectively) above the cervical cord injury, accompanied by a lower blood volume fraction (−22.05%, p < 0.001) and a higher blood velocity-related index (+38.72%, p < 0.0001) in patients with SCI compared with HC. Crucially, gray matter remote perfusion deficit correlated with larger lesion volumes and clinical impairment. This study shows clinically eloquent perfusion deficit rostral to a SCI, its magnitude driven by injury severity. These findings indicate trauma-induced widespread microvascular alterations beyond the injury site. Perfusion MRI matrices in the spinal cord hold promise as biomarkers for monitoring treatment effects and dynamic changes in microvasculature integrity following SCI.
Introduction
Traumatic spinal cord injury (SCI) induces secondary injury alterations including microvascular changes at the lesion epicenter. These alterations encompass disruptions of the blood–spinal cord barrier, 1 reorganization of the microvasculature, and a decrease in blood flow, 2 which can contribute to neuronal damage following SCI and limit functional recovery. 3 –5 Preclinical evidence illustrates that spinal cord perfusion impairment progresses dynamically even beyond the injury level, 6,7 affecting mostly the remote gray matter. 2 Understanding perfusion changes after SCI is crucial for developing possible therapeutic approaches targeting the spinal cord microvasculature to facilitate neural repair and regeneration. 5 While microstructural neurodegeneration has been studied in vivo across the spinal cord, 8,9 the spread of intramedullary perfusion impairment and its link with functional impairment remains understudied. The paucity of in vivo perfusion studies in the spinal cord is partly due to the lack of noninvasive tools sensitive to microvascular perfusion changes. Intravoxel incoherent motion (IVIM) is a diffusion-based magnetic resonance imaging (MRI) technique, indirectly sensitive to tissue diffusion and perfusion changes. 10 This method has been successfully applied in a wide range of pathologies across various organs in preclinical and clinical models 11 –14 to study perfusion dysfunction. Recently, IVIM has been also applied in the cervical cord of a cohort of patients with nontraumatic SCI (i.e., degenerative cervical myelopathy [DCM]), 15 providing evidence of microvascular damage in the gray matter rostral to the stenosis. The perfusion impairments were correlated with cervical cord atrophy remote from the level of stenosis. Furthermore, the reliability of the IVIM protocol has been investigated previously in HC. 16 Thus, IVIM holds potential as an MRI method sensitive to perfusion changes in diseases affecting the spinal cord. In this study, we aimed to investigate intramedullary microvascular changes in vivo induced by traumatic injury, in the region rostral to the lesion epicenter (i.e., C1–C3, to avoid image artefacts cause by implants) in chronic SCI. In addition, this study sought to assess the relationship between the magnitude of the blood perfusion changes with lesion volume and functional recovery after injury. We hypothesized that injury-induced intramedullary microvascular and perfusion deficits would be reflected in IVIM matrices remote from the lesion level in chronic cervical SCI and that perfusion changes will correlate with the severity of the injury (lesion volume) and functional recovery in SCI.
Methods
Standard protocol approvals, registrations, and patient consents
The study protocol was conducted in accordance with the Declaration of Helsinki and approved by the local Ethics Committee of Zurich (EK-2018–00937). All participants provided informed written consent before study enrolment.
Participants
Twenty-one patients with chronic (time since injury ≥6 months) 17 traumatic SCI, with a cervical lesion, and 39 healthy controls were recruited at the Balgrist University Hospital (Spinal Cord Injury Center outpatient clinic) between June 2021 and August 2023. Inclusion criteria included an injury at the cervical level (between C4 and C7), chronic stage, no pregnancy, no preexisting neurological condition, and age in the range of 18 and 75 years old. One healthy control was excluded from analysis due to the presence of preexisting spinal cord compression.
Clinical assessment
Patients with SCI underwent a state-of-the-art clinical examination protocol, to relate tissue perfusion integrity in the white and gray matter to clinical impairment. The clinical examination included scoring on the international standards for the neurological classification of spinal cord injury protocol for pinprick and light-touch scores 18 and the American Spinal Injury Association Impairment Scale (AIS) classification (A (i.e., loss of sensory and motor functions—classified as a complete injury), B, C, D (i.e., incomplete), or E (i.e., no functional impairment)). One patient did not complete the clinical protocol.
MRI acquisition
Image acquisition was performed on a clinical 3T MRI scanner (MAGNETOM Prisma, Siemens Healthineers, Erlangen, Germany), equipped with a Siemens Healthineers 64-channel head/neck radiofrequency (RF) coil. All participants were asked to wear a stifneck (Laerdal Medicals, Norway), to prevent undesirable motion in the inferior–superior direction and were instructed to reduce swallowing during the scans to minimize subsequent artefacts. All scans were centered on the C2–C3 disk to cover C1–C3 cervical levels.
Anatomical MRI acquisition
The anatomical MRI protocol included a standard sagittal turbo spin echo (TSE) T2-weighted sequence (MRI parameters: repetition time (TR): 3500 ms, echo time (TE): 84 ms, flip angle: 160°, field of view (FOV): 220 × 220 mm2, in-plane resolution: 0.3 × 0.3 mm2, slice thickness: 2.5 mm, 20 slices) for the localization of cervical levels and assessment of lesion volume, an axial TSE T2-weighted sequence at the injury level (MRI parameters: TR: 5510 ms, TE: 96 ms, flip angle: 150°, FOV: 160 × 160 mm2, in-plane resolution: 0.6 × 0.6 mm2, slice thickness: 3 mm, 23 slices) for visualization of the lesion, and finally an axial 3D multi-echo gradient-echo T2*-weighted sequence (TR: 38 ms, 5 TE [6.85, 10.85, 14.85, 18.85, 22.85] ms, flip angle: 8°, FOV: 192 × 192 mm2, in-plane resolution: 0.5 × 0.5 mm2, slice thickness: 5 mm, 16 slices) for white and gray matter segmentation and calculation of cross-sectional areas.
IVIM MRI acquisition
The IVIM MRI protocol consisted of a cardiac-gated, axial 2D-RF spin-echo echo planar imaging Zonally-magnified Oblique Multislice (ZOOMit) diffusion-weighted sequence with TR: 1800 ms (nominal: 600 ms), TE: 58 ms, FA: 90°, FOV: 101 × 31.8 mm2, in-plane resolution: 0.9 × 0.9 mm2, slice thickness: 5 mm, 9 slices, and with 14 b-values ranging from 0 to 650 s/mm2 with an increment of 50 s/mm2. Twenty repetitions per b-value were acquired in three in-plane diffusion-encoding directions, with acquisitions split into forward and reverse phase-encoding directions for subsequent distortion correction. Due to lack of time, the IVIM protocol was not completed in three patients. The total scan time ranged from 50 to 65 min, depending on the participant’s individual heartbeat.
Image processing
Cross-sectional cord area and lesion volume
Cross-sectional areas of the white and gray matter were determined based on segmentations obtained automatically (followed by manual correction) on the T2*-weighted images using the automatic segmentation algorithm (sct_deepseg_sc and sct_deepseg_gm) 19 of the Spinal Cord Toolbox (SCT, version 5.8). 20 Lesion masks were generated automatically on the sagittal T2-weighted images using sct_deepseg_lesion 19 in SCT (v6.0). The masks were manually corrected, and lesion volumes were extracted using sct_analyze_lesion. Lesions were not visible in three patients due to the presence of metal artefacts. Due to motion artefacts, one SCI patient was excluded from the structural analysis on the T2*-weighted and T2-weighted images.
IVIM Parameters
The IVIM processing pipeline was conducted using the IVIM toolbox
21
to generate IVIM perfusion and diffusion maps. The steps consisted of denoising,
22
artefacts removal using Gibbs
23,24
(DIPY),
25
motion correction (sct_dmri_moco),
20
and distortion correction (FSL topup).
26
Voxel-wise fitting of the MR signal was based on the most used biexponential IVIM model:
10
The IVIM parameters indirectly provide information on microvascular volume fraction (
Due to artefacts and incomplete dataset acquisition, only a subset of the participants was included in the structural analysis (SCI: n = 20/21, HC: n = 38/39) and in the perfusion analysis (SCI: n = 16/21, HC: n = 38/39).
Statistical analyses
Differences in age- and sex distributions between patients with SCI and healthy controls were tested with the Mann–Whitney U test and Fisher’s exact test, respectively. Structural MRI-derived cross-sectional areas were compared using two-sample Wilcoxon rank sum exact tests, to account for the non-normal data distribution and inequality of variances (tested with the Shapiro–Wilk test and Levene’s test, respectively) in the white and gray matter. IVIM parameters were compared between patients with SCI and healthy controls with two-sample Wilcoxon rank sum exact tests. In addition, influence of sex on perfusion was investigated by testing for potential differences in the IVIM parameters between males and females, in both cohorts separately, using two-sample Wilcoxon rank rum exact tests. Furthermore, differences in terms of IVIM parameters across the different AIS grades in patients with SCI were explored using a Kruskal–Wallis test. Finally, relationships between IVIM parameters and lesion volume, adjusted for lesion level, between IVIM parameters and lesion level, between IVIM parameters and time since injury, as well as between IVIM parameters and sensorimotor clinical scores (including upper extremity motor score (UEMS), cervical light-touch (CLT) and cervical pinprick (CPP)), were explored in patients with SCI using Kendall rank correlation coefficients, due to the non-normal distribution of IVIM data, with a statistical significance level set to 0.05. Regression models were corrected for the time between the clinical examination and the MRI scan to account for variability among patients. All statistical analyses were conducted in R statistical software (version 4.3.1). 30
Data availability
Anonymized datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
Results
Clinical, radiological, and neurological characteristics
MRI data were acquired on 21 patients with chronic cervical SCI (age [mean ± SD]: 48.10 ± 15.09 years, 6 females, months since injury [mean ± SD]: 106.52 ± 113.54) and 39 healthy controls (age [mean ± SD]: 49.29 ± 15.54 years, 20 females). There was no statistically significant difference in terms of distribution of age and sex between the two cohorts, as tested with the Mann–Whitney U test (W = 441, p = 0.42) and Fisher’s exact test (p = 0.27), respectively. Five patients with SCI were classified as functionally complete (AIS A) and 16 as functionally incomplete (AIS B-D). The lesion was located at level C4 in 7 patients, at C5 in 6 patients, at C6 in 6 patients, and at C7 in 2 patients. The mean spinal cord lesion volumes, averaged across patients with SCI, was 199.74 ± 252.94 mm3, ranging between 5.20 and 906.94 mm3. Patients with AIS A showed a mean lesion volume of 616.76 ± 410.38 mm3 (n = 2), had a mean lesion volume of 394.06 ± 299.53 mm3 (n = 3), and of 116.11 ± 98.78 mm3 (n = 12). Clinical, radiological, and neurological characteristics are reported at the group level for the SCI cohort in Table 1.
AIS, American Spinal Injury Association Impairment Scale; LEMS, Lower Extremity Motor Score; CLT, Cervical Light Touch; CPP, Cervical PinPrick; SCI, spinal cord injury; SD, standard deviation; UEMS, Upper Extremity Motor Score.
Remote tissue-specific neurodegeneration
Mean cross-sectional area (averaged across C1–C3 levels) was lower in the SCI group compared with HC in the white matter (SCI: 47.22 ± 10.53 mm2, HC: 65.22 ± 8.59 mm2, Δ = −27.60%, p < 0.0001) and in the gray matter (SCI: 11.45 ± 1.83 mm2, HC: 14.51 ± 1.05 mm2, Δ = −21.10%, p < 0.0001) (Fig. 1).
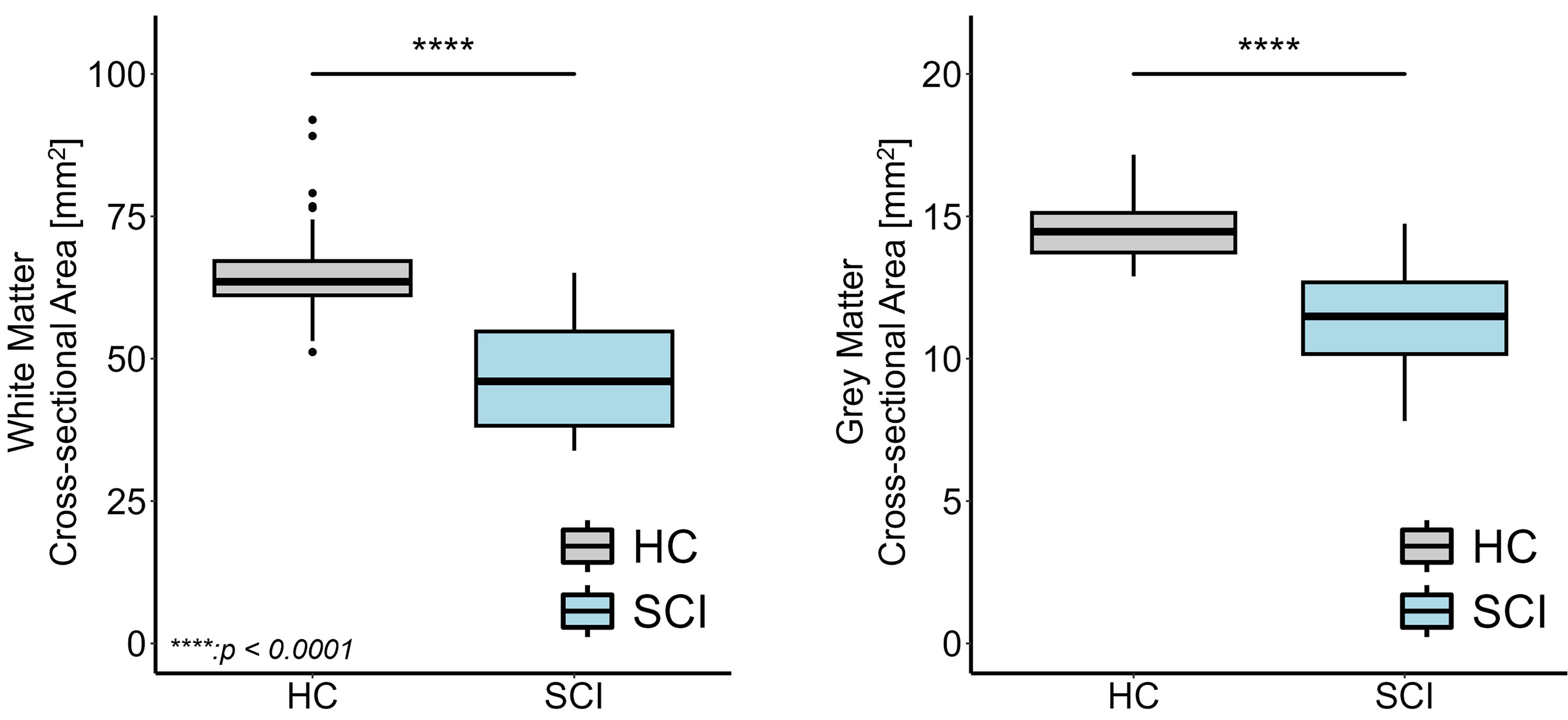
Comparing cervical cross-sectional areas in SCI and healthy controls (HC) across C1-C3 levels. Box and whisker plots of the cross-sectional areas of white matter and gray matter (in mm2) in the cervical cord, averaged across C1-C3 levels, in healthy controls (HC) and patients with SCI. ****p < 0.0001. The box extends from the 1st to the 3rd quartiles, whiskers extend to the smallest and largest value no further than 1.5 times the interquartile range, while dots represent outlying values beyond the end of the whiskers. SCI, spinal cord injury.
Remote perfusion compromise
The mean perfusion maps visually revealed lower microvascular volume fraction (
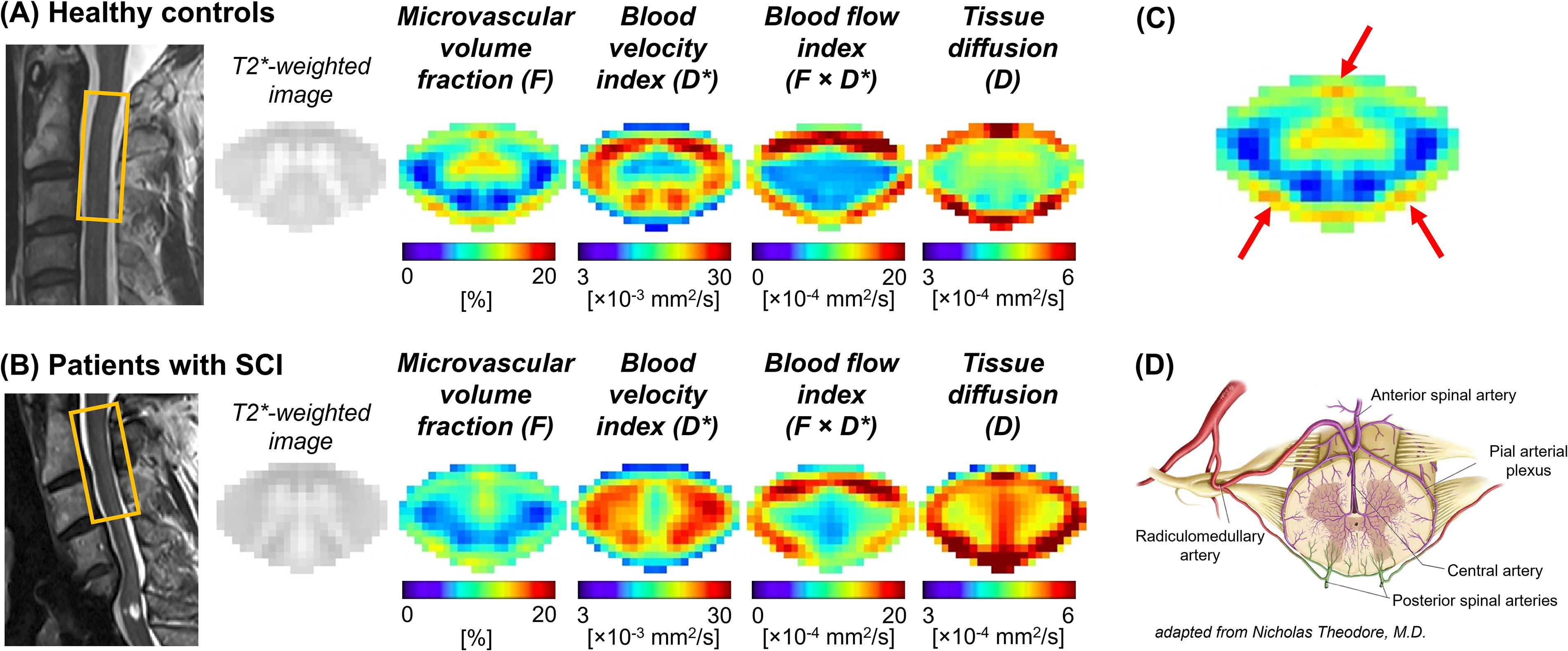
Illustration of intravoxel incoherent motion (IVIM) maps in healthy controls and patients with SCI.
A reduction in microvascular volume fraction (
Spinal Cord Tissue-Specific IVIM Parameters
Intravoxel incoherent motion (IVIM) parameters (mean and standard deviations [SD] across participants) in the white matter (WM) and gray matter (GM) of patients with spinal cord injury (SCI, n = 16) and healthy controls (HC, n = 38), along with the relative differences between the two groups and p-value of the statistical t-tests.
IVIM parameters were extracted in subject space level-wise and averaged across C1–C3 levels. Significant p-values are highlighted in bold (significance level set to 0.05).
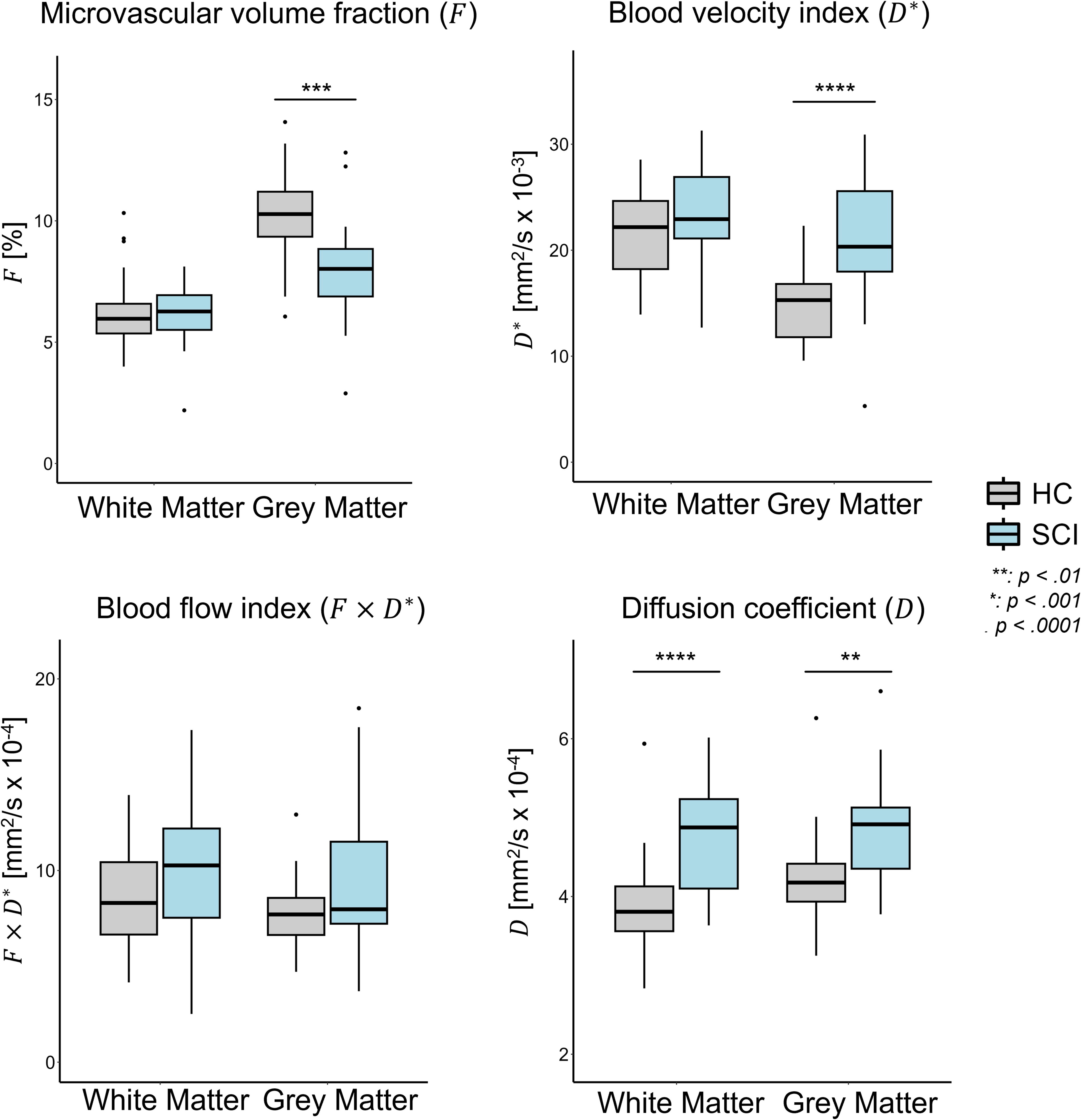
Comparing intravoxel incoherent motion (IVIM) parameters in patients with SCI and healthy controls (HC). Box and whisker plots of IVIM parameters in subject space, averaged across C1-C3 levels, in HC and SCI groups. **p < 0.01; ***p < 0.001; ****p < 0.0001. The box extends from the 1st to the 3rd quartiles, whiskers extend to the smallest and largest value no further than 1.5 times the inter-quartile range, while dots represent outlying values beyond the end of the whiskers. SCI, spinal cord injury.
Perfusion impairment correlated with lesion characteristics
In patients with SCI, a greater lesion volume was associated with a smaller microvascular volume fraction (τ = −0.52, p = 0.01) and associated with a higher blood velocity index (τ = 0.58, p = 0.003) in the gray matter (Fig. 4A). A greater lesion volume was also associated with a higher diffusion coefficient in both the white (τ = 0.52, p = 0.01) and gray matter (τ = 0.41, p = 0.047) (Fig. 4B). No significant relationship was observed between perfusion readouts and the level of injury, nor between perfusion readouts and time since injury.
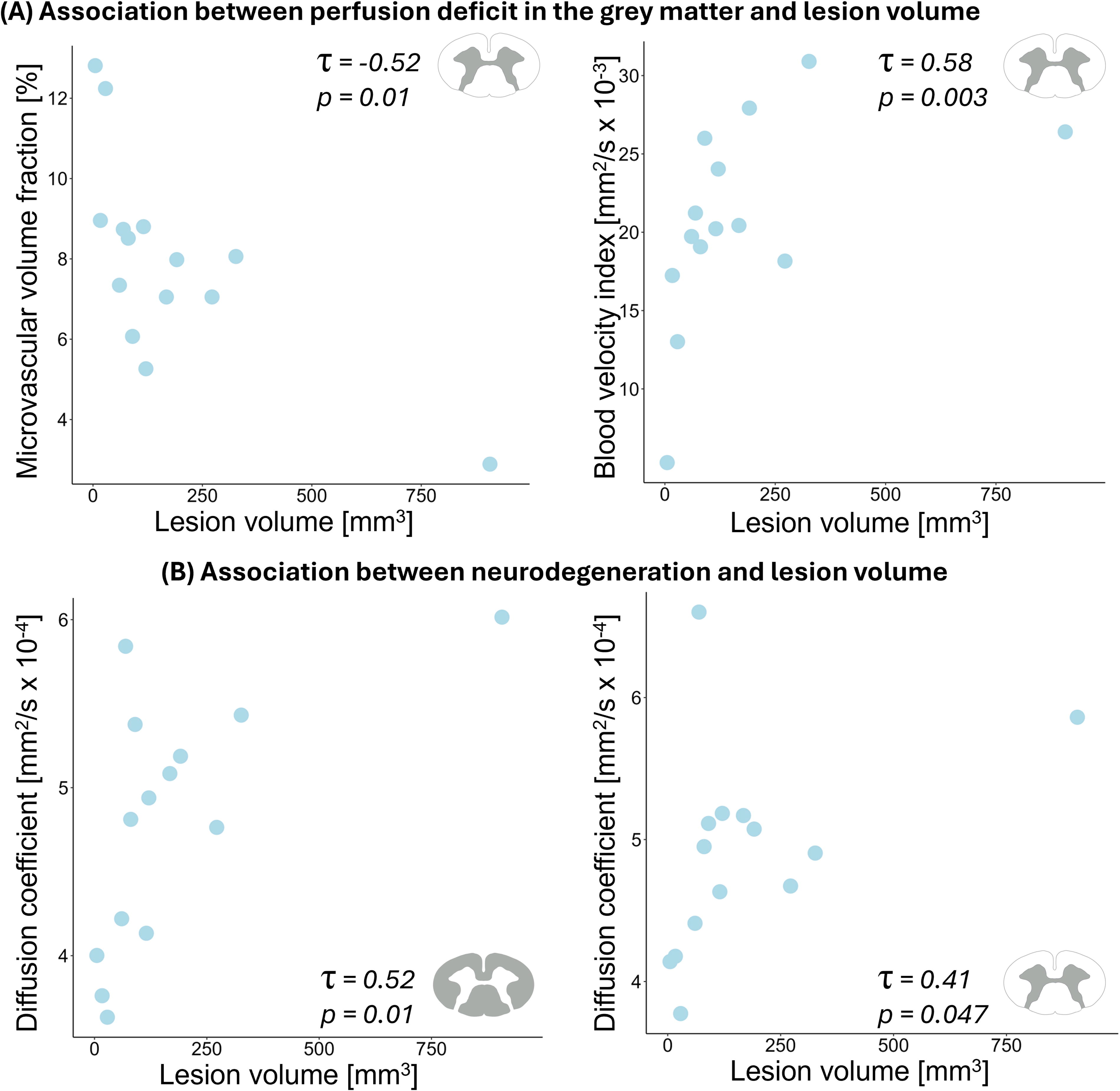
Association between lesion volume and intravoxel incoherent motion (IVIM) parameters in patients with SCI.
Remote perfusion impairment was associated with sensorimotor impairment
Microvascular volume fraction (
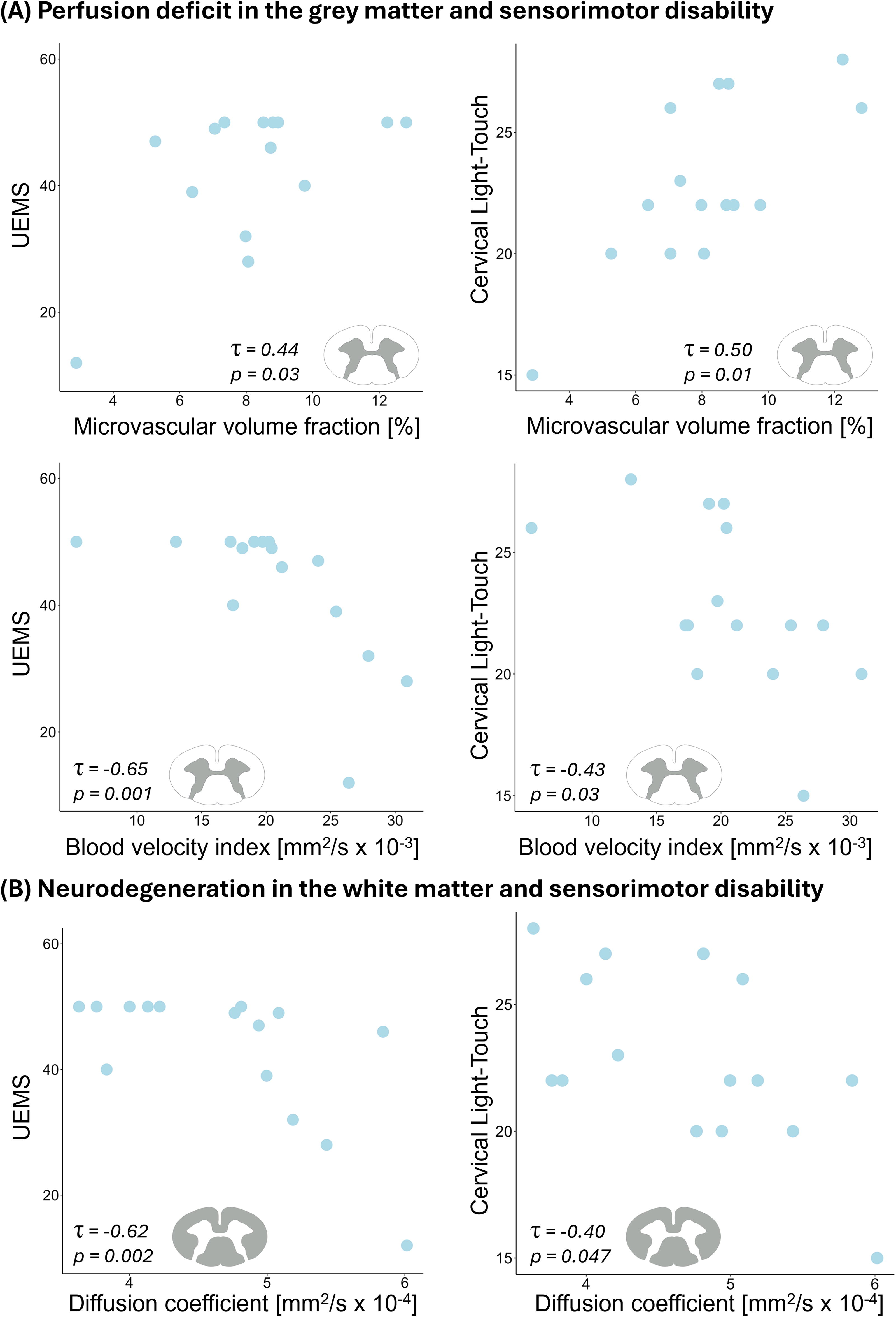
Associations between intravoxel incoherent motion (IVIM) parameters and sensorimotor impairment in SCI.
Discussion
This study detected intramedullary microvascular and perfusion deficit, extending into the region rostral to the lesion epicenter in the cervical cord of patients with chronic tetraplegic SCI. Tissue-specific perfusion deficit was associated with the extent of lesion and clinical impairment in SCI. Specifically, a larger lesion volume was associated with a smaller remote microvascular volume fraction and a higher blood velocity index in the gray matter. Microvascular changes indicated extensive perfusion compromise rostral to the injury. IVIM matrices sensitive to perfusion changes can therefore enable us to investigate the underlying mechanisms of microvascular deficit driving SCI-induced pathophysiological changes in vivo.
Remote perfusion deficit and spinal cord neurodegeneration
The pathophysiological signature of the secondary injury consists of dynamic rearrangement of the microvascular architecture, blood spinal cord barrier alteration, and disruption in macro- and microstructural composition of spinal cord tissue.
31
In this study, the microvascular volume fraction (
At the macro and microstructural level, there was higher atrophy and diffusion coefficient (
Relationship between perfusion impairment and lesion volume
The degree of perfusion and diffusion compromise was directly associated with the extent of the lesion volume. Specifically, a greater lesion volume was associated with a smaller microvascular volume fraction and with a higher blood velocity index. Those findings indicate that the extent of perfusion disturbance may be tied to the severity of injury, 4,39 reflected by the lesion volume. Of note, one patient had an extended lesion volume (906.94 mm3), which was still in the range of previously reported values, 40,41 underlining the heterogeneity in cord damage in the SCI population. The degree of perfusion and diffusion compromise did not significantly correlate with the injury level.
Relationship between perfusion impairment and sensorimotor dysfunction
The current findings show that the magnitude of remote perfusion impairment in the gray matter rostral to a traumatic injury in vivo relates to impairment in sensorimotor functions and may thereby contribute to the severity of clinical disability. This is in line with previous studies reporting perfusion and demyelination readouts in the spinal cord associated with clinical deficit and locomotor improvement. 6,37 Our results may be explained due to the higher density of neuronal cell bodies located in the gray matter and its greater metabolic requirement, highlighting this region’s microvasculature as possibly crucial for the preservation of sensorimotor functions.
Sensorimotor impairment most likely results from a combined effect of lesion severity and neurodegenerative changes at both microstructural and microvascular levels. Assessing the individual impact of those factors on clinical disability is beyond the scope of the current study; however, the current results are crucial to highlight the clinical significance of perfusion readouts at C1–C3 levels, underlining their relevance for longitudinal studies.
It is important to note that the perfusion readouts were measured at C1–C3 levels, while clinical scores reported information from all cervical levels (C2–C8). This approach was chosen because obtaining perfusion data at lower cervical levels poses greater technical challenges due to degraded image quality near the lesion, which can be caused by implant artefacts. 42 However, we hypothesize that perfusion impairment likely extends beyond C1–C3 at lower levels and that the severity of the vascular impairment increases towards the vicinity of the injury epicenter, as shown in preclinical models. 2,7 Of note, the absence of significant differences in IVIM parameters across AIS grades may be partially attributed to the small sample size of patients with AIS A (n = 4) and AIS C (n = 1), compared with the larger proportion of patients classified as AIS D (n = 11). While this may reduce the statistical power to detect differences between AIS grades, it contributes to a more homogeneous SCI cohort, which is beneficial for the rest of the study.
The present observations therefore suggest that gray matter vascular capabilities may be necessary for the maintenance of long-term sensorimotor functions. Our findings demonstrate spinal cord vasculature impairments in vivo and their interplay with lesion severity and clinical dysfunction in tetraplegic SCI, long after injury.
Limitations
This study presents several limitations. Despite the clinical relevance of perfusion readouts as possible MRI markers, the long acquisition time of the MRI protocol currently prevents its adoption in clinical setting and further research focusing on acquisition optimization is warranted. Moreover, the cross-sectional aspect of the current work inherently limits information that can be gained on the temporal evolution of the perfusion compromise. Future longitudinal studies have been planned and can provide further insights on the time course of perfusion changes following a traumatic injury and the potential detrimental effect they may elicit on regeneration processes. Furthermore, due to the lower number of severely affected patients, natural variability in patients’ characteristics and heterogeneous underlying pathophysiology of SCI could not be controlled for, but such recruitment would be more logistically demanding and is a common drawback in SCI studies. The definition of chronic stage can vary among studies, ranging from 6 months to 12 or 24 months post-SCI. In this study, the inclusion criterion for the chronic phase was established as being more than 6 months post-injury, as previously outlined. 17 Among the patients included in the perfusion analysis, three patients had sustained the injury 6 months before the examination, one was assessed 10 months post-injury, while the remaining patients had experienced their injuries over 24 months before the assessment. Finally, perfusion readouts at the injury level could bring additional insights; however, due to the challenge of obtaining noise-free and high-quality MRI readouts at the injury site due to implants artefacts, those were not acquired in this study and would necessitate further technical developments.
Conclusion
This study provides insights into intramedullary microvascular impairment in vivo and shows that IVIM parameters, sensitive to perfusion, can reliably capture perfusion compromise in the region rostral to cervical SCI. Microvascular damages were associated with the lesion volume and sensorimotor dysfunction in SCI. Characterization of injury-induced perfusion impairment is of great importance in SCI as it may hinder recovery and render the injured spinal cord more vulnerable. It therefore appeared promising to provide potential biomarkers in therapeutic strategies targeting microvasculature repair, such as angiogenic factors administration, cell transplantation, or biomaterial implantation-based therapies. 5 Future research will focus on characterizing perfusion longitudinally starting at the acute phase following a traumatic injury to explore the temporal profile of perfusion changes. This will shed light on the complexity of the progressive microvascular reorganization occurring in the acute and subacute SCI.
Transparency, rigor, and reproducibility
This cross-sectional study is not a clinical trial and was not formally registered. Images were collected in 39 healthy controls (HC) and 21 patients with SCI between June 2021 and August 2023. Imaging data were collected using a 3T MRI scanner (MAGNETOM Prisma, Siemens Healthineers, Erlangen, Germany). All equipment and software used to perform imaging and image analysis are widely available from commercial sources. The key inclusion criteria for this study are established standards in the field. Due to artefacts and incomplete dataset acquisition, only a subset of participants’ data was included in the structural analysis (SCI: n = 20/21, HC: n = 38/39) and in the perfusion analysis (SCI: n = 16/21, HC: n = 38/39). Small sample size and non-normal distribution of data have been addressed by using two-sample Wilcoxon rank sum exact tests and Kendall rank correlations. Test-retest repeatability of the imaging protocol has been conducted. 16 De-identified data from this study will be made available upon reasonable request by emailing the corresponding author. The processing pipeline code used to conduct the image processing is available in a public repository (https://github.com/slevyrosetti/ivim-toolbox). The authors agree to publish the article using the Mary Ann Liebert Inc. “Open Access” option under appropriate license.
Footnotes
Acknowledgments
The authors would like to thank all the participants for their time. This work is based on experiments performed at the Swiss Center for Musculoskeletal Imaging, SCMI, Balgrist Campus AG, Zurich, with the support of Prof. Daniel Nanz, and methodological developments performed at the Centre de Résonance Magnétique Biologique et Médicale, CRMBM-CEMEREM, Aix-Marseille University/CNRS, Marseille, France.
Authors’ Contributions
A.L.: formal analysis, investigation, visualization, and writing—original draft preparation. S.F.: investigation and writing—review and editing. S.L.: software and writing—review and editing. A.C.: writing—review and editing. V.C.: writing—review and editing. P.F.: writing—review and editing. M.S.: conceptualization, funding acquisition, project administration, supervision, investigation, and writing—original draft preparation.
Author Disclosure Statement
S.L. is an employee of Siemens Healthcare Pty Ltd. The other authors have no competing interest to disclose.
Funding Information
This project has received funding from the
