Abstract
After traumatic brain injury (TBI), monocyte/macrophage infiltration is a key early step in the development of an inflammatory cascade that leads to substantial secondary damage. Intravenous (IV) immunomodulatory nanoparticle (IMP) administration after TBI limits inflammatory cell infiltration and reduces both behavioral decline and lesion size without any noticeable toxicity. Here we show that there is a dose-response relationship between the amount of IMP administered and tissue damage which plateaus at a well-tolerated dose. There is a therapeutic window of efficacy for IMP administration of at least 6 h after injury with some benefit observed when treatment was delayed for 12 h after injury. Single cell RNA sequencing demonstrated substantial changes in gene expression after TBI in both neural and non-neural cells in the brain, and IMP administration ameliorated many of the changes. Particularly notable were significant unexpected changes in CCR1, CXCR2, and BDNF expression in vascular smooth muscle cells that may participate in injury responses after TBI. Thus, IMP treatment within 6 h after TBI limits inflammatory responses and gliosis, improves anatomical and behavioral outcomes and prevents detrimental changes in gene expression in both neural and non-neural cellular elements of the brain. IMPs are non-toxic and are made of an FDA-approved material that is stable at room temperature. They could easily be given IV immediately after TBI in the field by emergency medical technicians or in the emergency room to prevent secondary damage, thereby improving outcomes.
Introduction
After traumatic brain injury (TBI), there is a cascade of physiological events over the subsequent hours to days that leads to substantial secondary injury. The secondary damage reflects the effects of infiltrating immune cells, cytokine release, mitochondrial dysfunction, reactive oxygen species, excitotoxicity, and other mechanisms. 1 Despite many preclinical and clinical trials designed to limit such secondary damage, no successful therapies have emerged. However, we recently reported that intravenous (IV) administration of immunomodulatory nanoparticles (IMP) that was initiated 2 h after experimental TBI resulted in remarkable preservation of both tissue and neurological function in both open skull and closed head mouse models. 2
IMP are highly negatively charged, 500 nm-diameter particles composed of the FDA-approved biodegradable biopolymer, carboxylated poly(lactic-co-glycolic) acid (PLGA-COOH). After IV administration, IMP binds to the macrophage receptor with collagenous structure (MARCO) on monocytes (MC), and bound to IMP no longer migrate to sites of inflammation. 3 In our prior study we found that IMP administration at 2, 24, and 48 h after TBI resulted in a profound reduction in the number of infiltrating immune cells in the brain, skewing of the inflammatory cells in the brain towards an anti-inflammatory phenotype, decreased levels of inflammatory cytokines, reduced edema formation assessed by MR-imaging, reduced lesion volumes on anatomical examination, improved electrophysiologic visual function, and improved long-term motor behavior. IV treatment with IMP resulted in anatomical and functional improvement in animal models of tissue injury in other organs as well. The beneficial anatomical, physiological, and behavioral effects of IV IMP administration, the well-defined mechanism of action, 2,3 the lack of any evident deleterious side effects, 2,3 and the ease of administration suggest that IMP treatment may be a clinically translatable acute intervention for TBI.
Our initial study delayed the start of treatment until 2 h after the injury. However, it is possible that the therapeutic window for treatment may be longer which would enhance the potential clinical utility. It also is unclear whether the dose of IMP used in the initial study is the optimal one, and delineation of the dose-response relationship will also be essential. Moreover, although the mechanism of action is known, the profound effects of IMP administration on anatomy, physiology, and behavior suggest that there are far-reaching changes in gene expression and signaling events that remain to be defined. This study established the optimal dosing and therapeutic window for treating TBI with IMP and defined the changes in gene expression in the brain that underlie the beneficial effects of treatment.
Materials and Methods
Animal care and housing
All animal procedures were performed in accordance with the Public Health Service policy on humane care and use of Laboratory Animals. All procedures were approved by the Northwestern University Institutional Animal Care and Use Committee. Male and female 10- to 12-week-old C57BL/6 mice (Charles River Labs, Wilmington, MA) were used for the study and housed at Northwestern University Center for comparative medicine in a 12-h light/dark cycle with a regular chow diet ad libitum.
Animals and surgical preparation
Mice were anesthetized with 2.5% isoflurane in oxygen. The hair atop their skulls was removed using Nair. The surgical site was disinfected with iodine and cleaned with an alcohol wipe. The mice were given a 0.6 mg/kg subcutaneous dose of extended release (ER) buprenorphine and moved to the stereotaxic frame. Their heads were firmly secured using the bite plate and ear bars.
Traumatic Brain Injury
The TBI procedure was performed as previously described. 2 Briefly, the sham operation consisted of surgical preparation, an incision along the midline of their skull, and a 3 mm craniotomy 1 mm caudal and +1.5 mm lateral relative to bregma over the somatosensory cortex. The incision was glued closed using 3M Vetbond. TBI surgery followed the same procedures as described for sham operation through the craniotomy. Once the 3 mm opening was made, a deformation injury was delivered using Leica Biosystems Impact One stereotaxic impactor using a 2 mm tip. The impactor delivered the injury 2 mm deep at 3.00 m/s with a 30 s dwell time. Bleeding was cleaned with sterile cotton swabs, and the incision was glued closed using 3M Vetbond. Enrofloxacin antibiotic (2.5 mg/kg, subcutaneous in 1 mL sterile normal saline) was administered daily for 3 days after injury to reduce the risk of infection.
Immunomodulatory Nanoparticles Injection
The PLGA-COOH nanoparticles (IMP, 500 nanometers in diameter, 100 mg/vial from Cour Pharmaceuticals) were first dissolved in 2500 µL of normal sterile saline to create a stock solution at 40 mg/mL concentration without conjugating to any drug or chemical. Stock IMP solution was further diluted in sterile saline to obtain the 200 µL of injection volume at IMP concentrations of 0.25, 0.5, 1, or 2 mg for the dosage testing experiment, and 2 mg concentration for the rest of the study. IMP was injected intravenously into the lateral tail vein of the mouse. Each animal received three injections. The injections for the effective treatment window study were administered at either 6, 30, and 54 h; 12, 36, and 60 h; or 24, 48, and 72 h post-injury respectively of their assigned cohort. Animals assigned to no-IMP control groups were given 200 µL tail vein injections of normal sterile saline in concert with their respective IMP-treated counterparts.
Ladder Rung Walking Test
All cages were blinded by a lab member not associated with the analysis of the behavioral tests. The researcher analyzing the behavioral data remained blinded until all behavioral tests were completed and analyzed. The tests were performed as previously described. 2,4 Briefly, mice were habituated on the custom-made ladder rung apparatus by allowing each mouse to walk across the ladder rung once daily for three days prior to the experimental day. The ladder was elevated using a box of equal height on both sides and a mouse cage on each side. The starting side used a clean cage while the mice’s original home cage was used to elevate the ladder at the opposite side. Mice were only allowed to cross the ladder one way, start to finish, and their path was blocked with sterile cotton swabs in the instances the mice tried to reverse direction. Videos of the mice crossing were captured with a GoPro10 camera situated below the center of the bridge, with illumination from below provided by LED light strips to improve the image quality. Each day the rungs were changed to avoid the bias of learning the walking pattern. On the experimental test day, the camera recorded each animal crossing in ultra slow-motion mode (240 frames per second) and the videos were played back in slow motion to count the number of total steps taken and the number of times the mice’s feet fell below the ladder. The ladder rung walking test and habituation were performed at 1- and 3-week post-injury between 10 am and Noon.
5-Ethynyl-2′-Deoxyuridine (EdU) Injections
A 2.5 mg/mL stock of EdU was made from 50 mg EdU from invitrogen (A10044) in sterile phosphate-buffered saline (PBS). Mice received 2 doses of 100 mg/kg EdU via intraperitoneal injection (IP) 6 and 3 h before euthanasia.
Whole Brain Collection for Immunohistochemistry
Mice were euthanized with CO2 and perfused with 10 mL ice-cold PBS followed by 10 mL ice-cold freshly made 4% paraformaldehyde (PFA) in PBS with a pH of 7.4. The whole brain was then removed and placed in 9 mL of ice-cold freshly made 4% PFA in PBS and incubated at 4°C for 2 h. Brains were then washed two times with cold PBS and placed in 9 mL cold 30% sucrose in PBS overnight. Brains were then embedded in optimal cutting temperature ([OCT], Sakura) and sectioned to 10 µm thickness on a cryostat (Leica CM1850) and collected on glass slides that were stored at −80°C until use.
Cortical Tissue Collection for Western Blot Analysis
Mice were euthanized with CO2 and perfused with 10 mL ice-cold PBS. The whole brain was then removed and a 3 mm3 perilesional sample was collected and stored at −80°C until protein extraction. Samples were later thawed on ice and placed in 200 µL of T-Per (Thermo Fisher #78510), followed by homogenization with an electric pestle in 1.5 mL tubes and spun in a tabletop centrifuge at 4°C for 5 min. The supernatant was transferred to new 1.5 mL tubes and stored at −80°C until use.
Western Blot Analysis
Protein concentration from tissue lysates where first measured by Bicinchoninic Acid (BCA) protein Assay (Pierce, #23225) before proceeding to Western blot (WB) analyses. Using standard Western blot techniques, 8 µg of protein was run in a 26-well 4–20% gel. After protein transfer, polyvinylidene fluoride (PVDF) membranes were blocked in 5% milk in Tris Buffer Saline with 0.1% Tween 20 (TBS-T) or 5% bovine serum albumin in TBS-T depending upon the primary antibody’s specifications (see section Antibodies), followed by incubation in horse radish peroxidase (HRP)-conjugated secondary antibodies and detection with Radiant Q ECL reagents (Azure Biosystems, #AC2101). A C600 imager (Azure Biosystems) was used to image Western blots. ImageJ software was used to measure optical density and normalized against glyceraldehyde 3-phosphate dehydrogenase (GAPDH).
Immunohistochemistry
For immunohistochemical staining (IHC), slides containing sectioned brain tissue were stored at −80°C after sectioning and brought to room temp before beginning staining. Phosphate Buffer Saline with 0.05% Triton X-100 (PBS-T) was used to wash slides before blocking in blocking media for 1 h (blocking media: 5% Normal Donkey Serum or Normal Goat Serum in PBS-T). After blocking, slides were washed three times with PBS-T. The primary antibodies (see section Antibodies) were diluted in the blocking solution and incubated at 4°C overnight. On the following day, the slides were washed six times in PBS-T for 1 h before incubating in fluorescently labeled secondary antibodies diluted in blocking buffer (see section Antibodies). Following secondary antibody incubations, slides were washed six times for 1 h before mounting in ProLong Gold mounting media (Thermo Fisher) under glass coverslips and allowed to cure overnight at room temperature.
Antibodies
Microscopy
Slides with immunofluorescent staining of astrocyte and microglia (MG) markers were imaged on an SP5 confocal microscope (Leica Biosystems) as previously described. 2 Briefly, acellular lesion centers were identified by paucity of Hoechst stain in the dorsal cortex. Glial fibrillary acidic protein (GFAP) immunoreactivity was used to identify the border of lesion, and five randomly selected regions of interest within 200 µm from the lesion border were imaged at 400 × magnification using optical thickness of 1 µm z-steps at the resolution of 1024 by 1024. Whole brain images used for lesion volume measurements were acquired at 10 × and auto-stitched using a Keyence BZ-X810 fluorescent microscopy.
Lesion Volume Measurement and Reactive Gliosis Quantification
Multichannel immunofluorescent images were assembled in Adobe Photoshop. The lesion volume was calculated by using ImageJ to measure the lesion area of cryostat sections taken approximately every 300 µm. The areas between sections were averaged and multiplied by the distance to calculate the volume between sections. The total volume was the sum of the volumes between sections. For quantifying reactive astrocytes (AC) and MG in the injury site, GFAP +EdU+ and ionized calcium-binding adapter molecule-1 (IBA-1)+EdU+ cells within 200 µm of lesion border were quantified manually with number tracking assist using ImageJ.
Single Cell RNA Sequencing and Analyses
For transcriptomic profiling of perilesional cellular responses to IMP treatment, two months old C57/Bl6 male mice that received either TBI or sham injury were injected with IMP or saline at 2 h post-injury and sacrificed 24 h post-injury. After perfusing with PBS, 3 mm3 of the perilesional tissue from the dissected brains of 2 mice per experimental condition were removed and dissociated following the protocol outlined in the Papain Dissociate System (Worthington Biochemical), dissociated single cells were verified for greater than 90% viability and submitted for single cell RNA sequencing with the Chromium Single Cell 3′ Solution V3.1 (10× Genomics) with assistance from the University of Chicago genomics core. Ninety thousand cells from two independent samples were combined, captured, and barcoded on a microfluidic chip, and subsequently used for cDNA library construction before sequencing on the Illumina Hiseq 6000. Cell Ranger (version 7) was used to align the FASTQ files for each sample to the mouse reference genome and the number of reads from each cell that aligned to each gene were quantified. The matrix files, which summarized the alignment results, were then imported into Seurat (v4, Satija Lab, NYGC) for further analysis. The Seurat object data was first filtered by total count and mitochondrial DNA content to eliminate doublets and poor-quality cells respectively. Data from all samples then underwent normalization and scaling using the R package SCTransform, 5 followed by integration via the CCA integration method. 6 Cell clusters were identified using SNN resolution of 0.2, followed by the identification of cluster specific differentially expressed genes (DEG) using Seurat function FindAllMarkers, followed by cell type identification using cell type-specific marker expression via the scType package. Cell cluster distribution in two dimensions were visualized using uniform manifold approximation and projection (UMAP) dimensional reduction, and the expression of genes of interest on the UMAP was presented using the FeaturePlot function in Seurat. Pathway analysis was performed on the NCATS BioPlanet (2019) platform, 7 using Enrichr. 8 The R package CellChat (v1) 9 was used to identify differentially regulated ligand-receptor pairs in each experimental condition based on a curated receptor-ligand pairs database in CellChatDB (cellchat.org/cellchatdb) using standard author suggested parameters. For the circle plot representation of significant interactions, only the top 10% of significant interactions were graphed. Matrix data from the single cell RNA-sequencing (scRNA-seq) is deposited at NCBI Gene Expression Omnibus repository at accession number GSE266001.
Statistics
Data were analyzed using GraphPad Prism software (v10), and statistical significance was assigned at a predetermined cutoff of p < 0.05. Comparisons were conducted using one-way analysis of variance (ANOVA) with the Tukey multiple comparison post hoc test. All data are presented as means ± standard error of the mean (SEM) unless otherwise noted.
Results
A dose of at least 1 mg per injection was required for maximal protection in mice after TBI
Previously, we demonstrated the efficacy of IV IMP administration in reducing secondary damage in both controlled cortical impact (CCI) and closed head injury (CHI) models of TBI. 2 In that study mice were treated with 1 mg of IMP IV at 2, 24, and 48 h after injury. To define the dose-response relationship for IMP treatment and to determine the optimal dosage, both male and female mice were treated after CCI with different doses of IMP using the same schedule for dosing that was used in the initial study (Fig. 1A). The ladder rung test was used to assess motor function 2 and 4 weeks post CCI (wpi), and the animals were sacrificed at 5 wpi for analysis of lesion volume (Fig. 1).
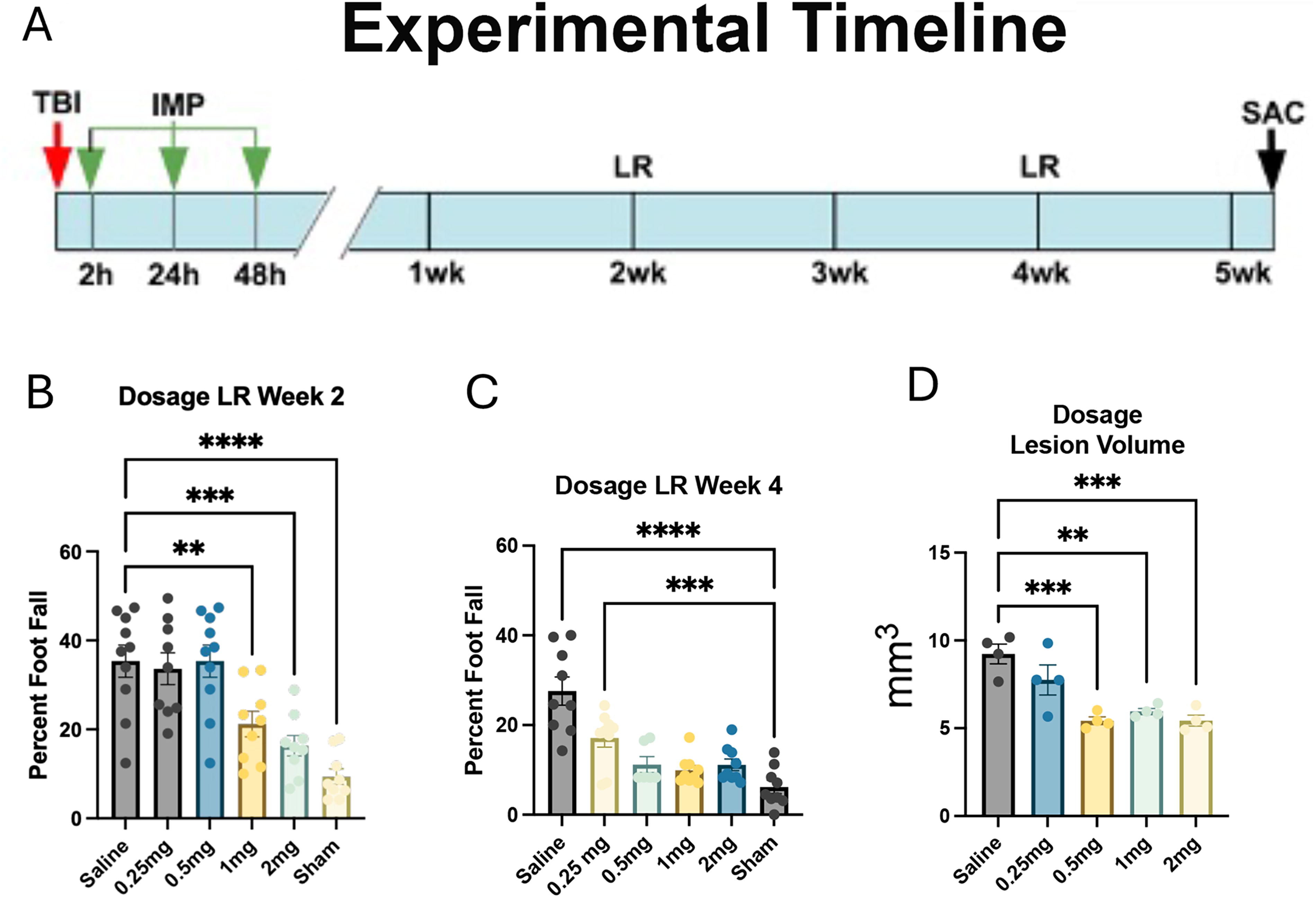
Determination of minimum effective dose of IMP treatment.
At 2 wpi the animals that received 1 or 2 mg doses of IMP had significant reductions in foot falls on the ladder rung test (Fig. 1B). There was no significant difference between the 1 and 2 mg groups. However, the 0.5 and 0.25 mg doses were ineffective in reducing motor deficits at 2 wpi. At 4 wpi there was a generalized recovery with reduced foot falls in all injured mice (Fig. 1C). The protective effects of the 1 and 2 mg doses persisted, and the mice performed identically to sham-injured mice. Interestingly, mice that received only 0.5 mg also performed identically to sham-injured mice. Mice treated with 0.25 mg performed significantly worse than sham-injured mice but still better than saline-treated CCI mice. Thus, a dose of at least 1 mg was required for maximal behavioral protection after CCI, but some beneficial effects were observed even with a dose as low as 0.25 mg.
To assess the relative efficacy of different doses of IMP in reducing secondary damage after CCI, we measured the lesion volumes in injured brains at 5 wpi. There were significant reductions in lesion volume with all doses except the 0.25 mg dose. Doses at 0.5 mg and above reduced lesion volume by almost 50%. No differences in dosage responses were observed between male and female mice. In toto, the behavioral and anatomical observations indicated that a dose of at least 1 mg was required for maximal protection in mice.
IV IMP administration within 6 h post-injury reduces the severity of damage after CCI
In our previous studies, IMP administration began 2 hpi as we considered it a clinically relevant time point for a TBI patient to receive emergency care. However, to determine whether more delayed treatment was still effective, we delayed the onset of IMP administration to either 6 h, 12 h, or 24 hpi. Two mg of IMP were used for each dose. The second and third doses were given 24 and 48 h after the first dose. One week post-injury, IMP administration starting at 6 h and 12 hpi significantly reduced the number of foot falls compared with injured animals that received only saline. There was no significant reduction when IMP was delivered 24 h after injury (Fig. 2A). Similar to the results seen in the dose response ladder rung test, there was a generalized improvement across all injured groups over time. At 3 wpi the IMP-treated injured animals no longer showed statistically significant decreases in foot falls compared with the TBI+saline animals. However, the TBI+saline animals continued to perform significantly worse compared with the sham operated animals at 3 wpi (Fig. 2B).
We then assessed the protective effects of delayed treatment on the size of the lesion and the inflammatory response 5 wpi. Mice received 2 doses of 100 mg/kg of EdU via IP 6 and 3 h before euthanasia. A significant reduction in lesion volume was observed in animals when they received IMP 6 hpi (Fig. 2C). There was a trend towards improvement in the animals that began treatment 12 h after injury, but there was no reduction in cortical tissue loss when IMP treatment was delayed to 24 h after insult (Fig. 2D).
In addition, there was evidence of decreased immune reactivity in animals that were treated within 6 h. We examined the lesion site by western analysis for levels of the inflammatory β-galactoside-binding lectin, galectin-3, and for the marker of astrocytic responses, glial fibrillary acidic acid (GFAP) (Fig. 3). Treatment after 6 h but not 12 h significantly reduced galectin-3 to the same level found in sham mice (Fig. 3A, B). There was a trend towards reduction in GFAP when treatment was initiated at 6 h (Fig. 3C, D); levels of GFAP in the 6 hpi group did not differ significantly from sham. The lesion sites then were examined immunocytochemically for levels of the activated microglial marker, IBA-1, and for EdU incorporation (Fig. 3E, F, and G). Both IBA-1 and EdU immunoreactivity were significantly increased in saline-treated injured mice compared with sham. There was no difference in IBA-1 between sham and injured mice that received IMP 6 hpi, and both differed significantly from the saline group. However, IBA-1 in mice treated with 12 hpi did not differ from the saline group. There was a similar trend in IBA-1+EdU+ cells that did not reach statistical significance. No differences in delayed treatment window responses were observed between male and female mice. Overall, these observations indicate that the therapeutic window for IMP after TBI is at least 6 h after injury.
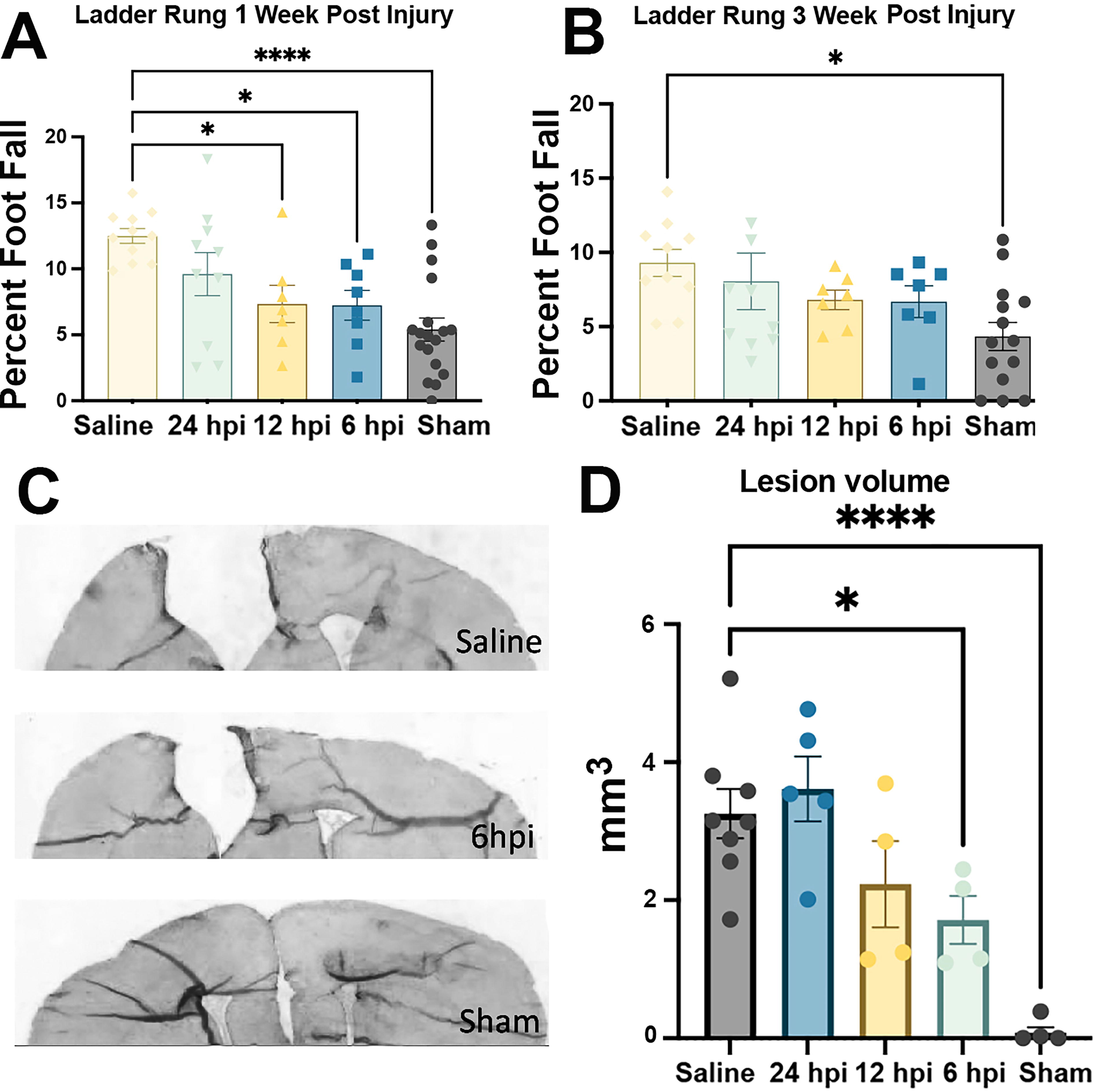
Determination of the longest effective time of IMP treatment post-injury.
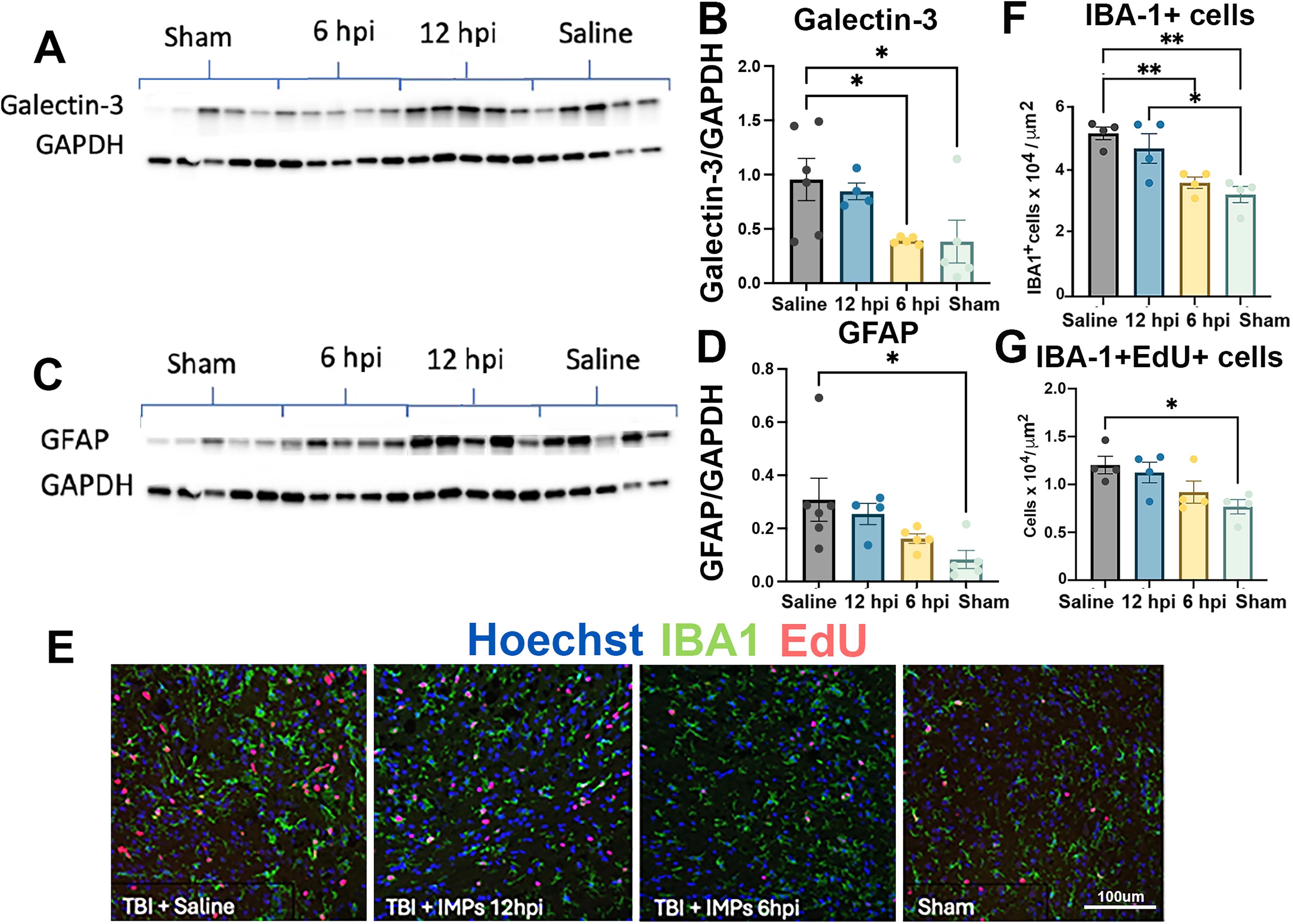
Molecular and cellular phenotypes of perilesional glial responses to IMP treatments initiated at 6 h post-injury.
Transcriptomic profiling via scRNA-seq revealed reduced microglial activation at 24 h post-TBI with IMP treatment
To determine how IMP treatment influences gene expression in different cell types around the site of injury after TBI with and without IMP treatment, we next performed scRNA-seq of cells collected from perilesional cortical tissue harvested from mice that received sham injury with saline injection (sham+saline), mice that received TBI with saline injection (TBI+saline), and mice that received TBI with IMP treatment (TBI+IMP, Fig. 4A). After the removal of cells with poor quality based on gene number and mitochondrial DNA level, expression data from 24,000 cells from all experimental groups were integrated for subsequent hierarchical clustering and cell type identity assignment via the Seurat R package. Based on cell type specific marker expression, 13 molecularly distinct cell clusters were identified in all experimental groups (Fig. 4B, C). This included cells of the neural lineage such as neurons (Ex.N and In.N), AC, oligodendrocytes (OL), and oligodendrocyte progenitors (OPCs) as well as non-neural cell types including vascular cells like endothelial cells (EC), pericytes (PC), and smooth muscle cells (SMCs). Cells of hematogenous origin, including MG MC, neutrophils (NT), and T-cells, also were identified. As previously demonstrated by FACS analysis, we found a significant reduction in the number of MC in mice that received TBI+IMP treatment compared with TBI+saline, consistent with the known effects of IMP treatment in sequestering circulating MC and preventing their infiltration into injury sites. 2,3
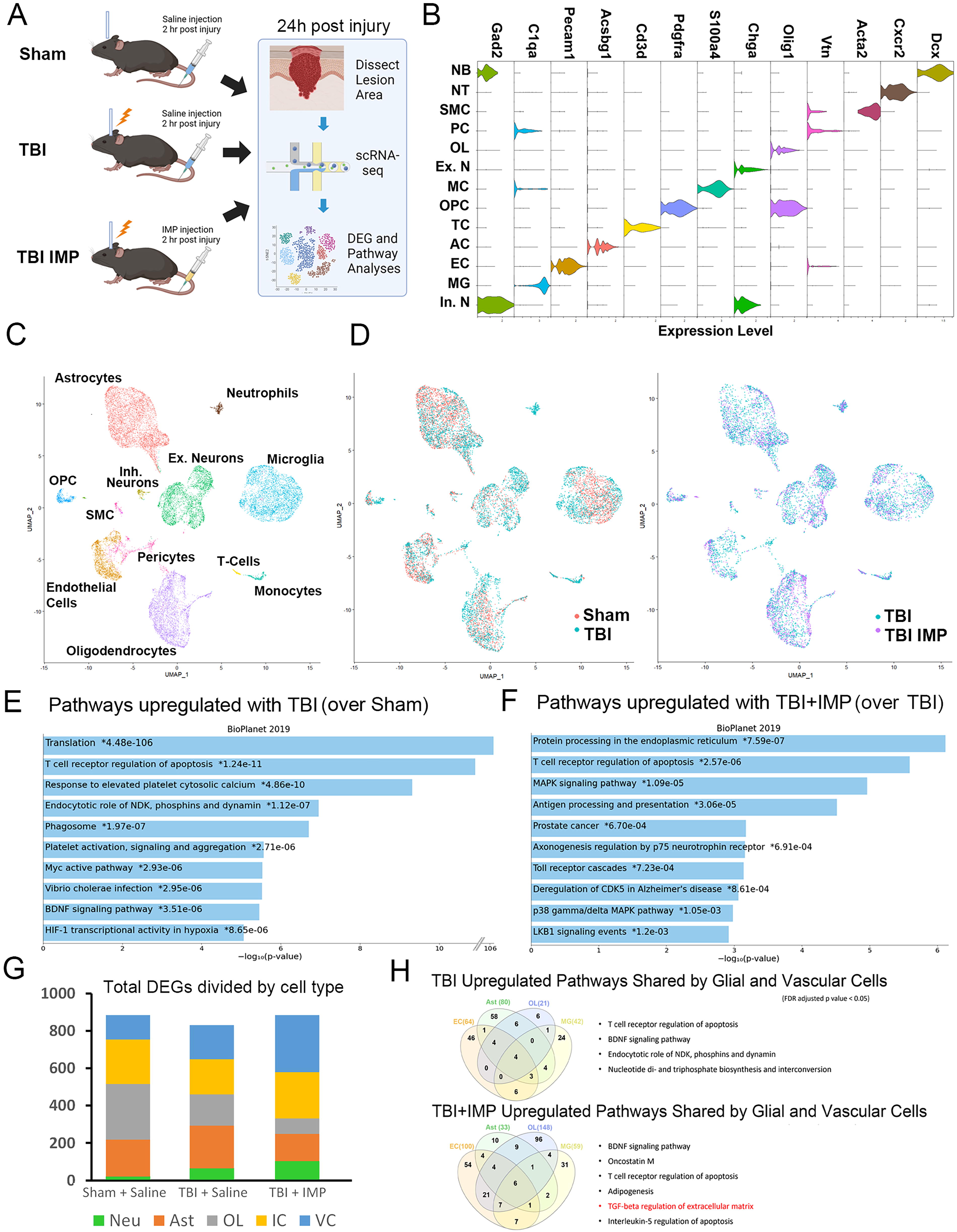
Single cell RNA sequencing analysis revealed differentially regulated pathways by IMP after TBI.
Comparison of overall UMAP spatial distribution between cell clusters in the sham+saline and the TBI+saline groups, as well as between TBI+saline and TBI+IMP groups, revealed no significant overall shifts in gene expression profiles between integrated experimental datasets (Fig. 4D). Before investigating the cell type-specific changes in gene expression, we first used the overall DEGs between experimental conditions to identify molecular pathways that are enriched in the TBI and the TBI+IMP brains using the aggregated BioPlanet as well as the KEGG pathway analysis databases. 7,10 We found that when compared with the sham+saline controls, TBI+saline animals showed increased expression of ribosomal protein genes associated with translation, apoptosis, and calcium signaling (Fig. 4E). This expression pattern is consistent with the injury-induced stress/regenerative response, which has been observed in multiple cell types. 11 –13 In contrast, while similar pathways are also observed in IMP treatment TBI brains, heat shock proteins are among the top DEGs in the TBI+IMP group and were not detected in the TBI only group, suggesting that IMP treatment promotes pathways associated with the protective unfolded protein responses regulating protein folding, homeostasis, and degradation (Fig. 4F).
We next performed differential gene expression analysis in a cell type-specific manner to determine the transcriptomic level responses to TBI and TBI with IMP treatment (Fig. 4G). To simplify the analysis, the major cell types were grouped into five categories that include the three neural lineage cells (neurons, AC, and OL) plus immune cells (MG, MC, and T-cells), and vascular cells (pericytes, endothelial, and SMCs). Interestingly, we found that vascular and immune cells both show increases in DEGs with IMP treatment when compared with TBI alone. In contrast, OL and AC show reductions in the number of DEGs in the TBI+IMP group compared with the TBI group. This finding led us to hypothesize that IMP treatment modulates changes in reactive gliosis associated gene expression in neuroglia post-injury. To validate this hypothesis, we examined the expression of reactive astrocyte enriched genes and found IMP treatment indeed reduces levels of these transcripts in one of the four subclusters of AC that are spatially shifted in the UMAP in the TBI+saline and TBI+IMP groups (cluster A4, supplementary Fig. 1A–C). IMP treatment also appears to promote opposing expression profiles in many anti-inflammatory DAM-suppressed and M2 MG markers when compared with the TBI+saline group (Supplementary Fig. S1). These findings support results from our protein expression analyses via Western blots and immunohistochemistry and provide further insights into how IMP treatment modifies post-injury glial responses.
To determine what molecular pathways in glial and vascular cells are differentially regulated by TBI and IMP treatment, we cross referenced differentially regulated pathways identified in AC, OL, immune, and vascular cell types. Although the TBI+saline and TBI+IMP groups shared many of the same enriched pathways, the TBI+IMP group uniquely showed differential regulation of Transforming Growth Factor beta (TGF-β) signaling and adipogenesis pathways (Fig. 4F). Taken together, these findings indicate that IMP treatment after TBI leads to transcript level changes in both neural and non-neural cell types, with the largest effects observed on gene expression in immune and vascular cells.
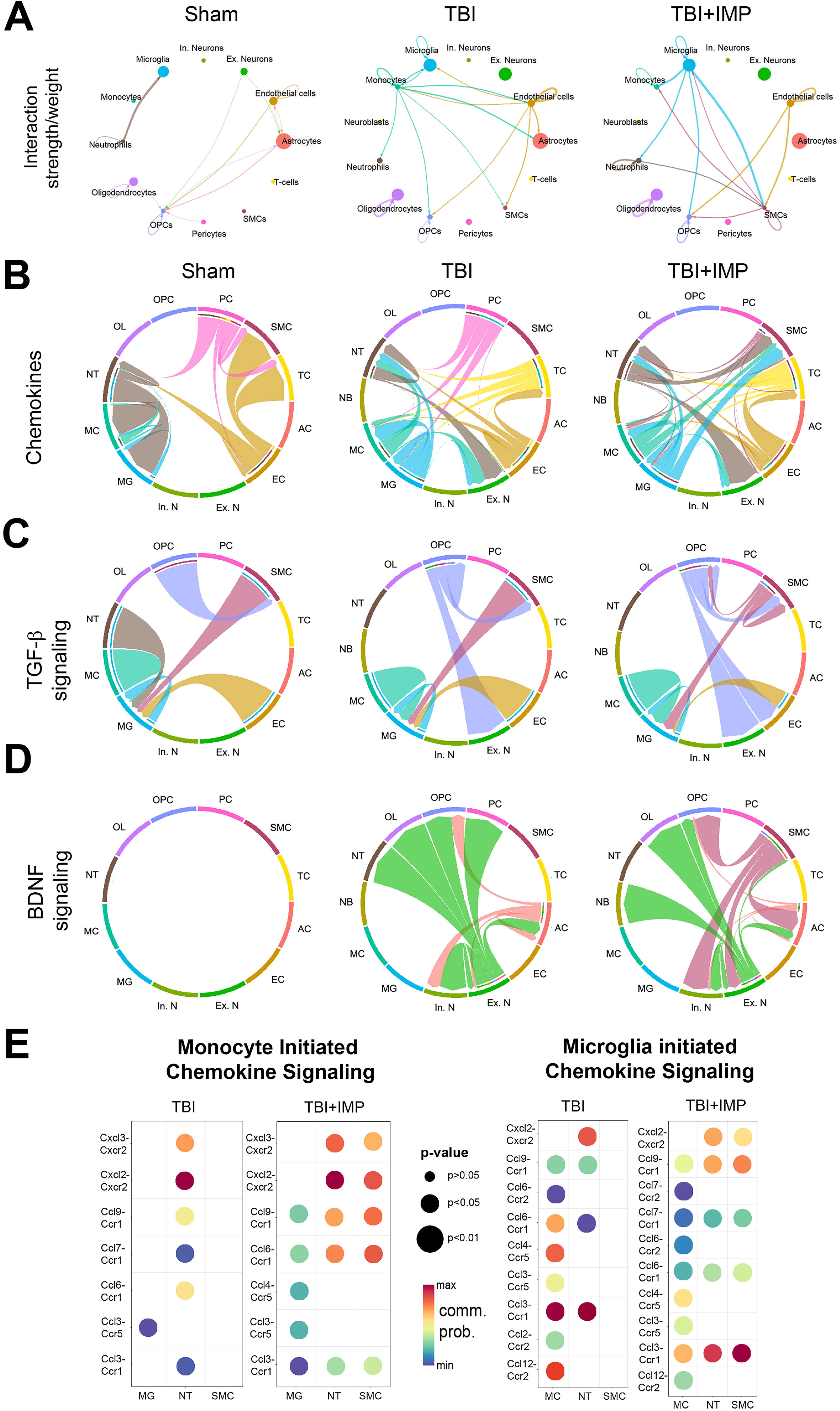
CellChat analyses identify vascular smooth muscle cells as mediators of IMP-modulated signaling interactions.
We next sought to determine which inter-cell communications correlate with specific TBI+IMP-enriched signaling pathways. Since chemokine signaling is critical for post-injury immune responses, we examined how IMP treatment acutely alters chemokine ligand and receptor interactions among all identified cell types (Fig. 5B). We found that in addition to interactions observed in the TBI+saline group, the TBI+IMP group shows monocyte, MG, and neutrophil-initiated chemokine signaling to SMCs and vice versa. Furthermore, pericyte-initiated chemokine signaling, which is present in both the sham+saline and TBI+saline groups, is not detected in the TBI+IMP group. We next analyzed ligand-receptor interactions in the TGFβ signaling pathway, which was identified to be enriched in the TBI+IMP group. In comparison to the TBI+saline group, we found that IMP treatment led to a gain of SMCs to OPCs signaling and a loss of neutrophil to MG signaling. Finally, while Brain-Derived Neurotrophic Factor (BDNF) signaling is enriched in both TBI and TBI+IMP groups, signals initiated from SMCs to OPC, AC, and neurons are present only in the TBI+IMP group.
To investigate specific ligand-receptor pairs that mediate the IMP treatment-induced changes in chemokine signaling, we compiled all monocyte-SMCs and MG-SMCs chemokine ligand-receptor pairs and examined their communication probabilities based on transcript expression level and prevalence (Fig. 5E). We found that the increases in interaction probability between MC-SMCs and MG-SMCs result from increased chemokine receptors CXC-motif chemokine receptor 2 (CXCR2) and Chemokine receptor 1 (CCR1) in the SMCs, which coincides with the TBI+IMP group-specific increase in BDNF transcripts in the SMCs. These findings are consistent with prior observations that modulating chemokine signaling may be beneficial after TBI. 14 –16
Discussion
After TBI, there is substantial secondary injury attributable to infiltrating immune cells, cytokine release, reactive oxygen species, excitotoxicity, and other mechanisms. 1 At present there are no effective therapies for preventing secondary damage or for preventing the anatomical and functional consequences of the injury. 17,18 Monocyte/macrophage infiltration is a key early step in the development of the inflammatory cascade, with subsequent elaboration of cytokines and infiltration of additional immune components. 19 –21 After IV administration, IMP binds to the MARCO on MC, and MC bound to IMP no longer migrates to sites of inflammation. 2,3 IV IMP administration prevented secondary damage and improved outcomes after a variety of injuries to different organs including the brain and spinal cord. 2,3,22 There were no apparent side effects or tissue damage associated with IMP treatment, and IMP was well tolerated in humans when used to treat Gluten intolerance. 23 IMP are made of an FDA-approved material that is stable at room temperature and could easily be given intravenously immediately after TBI in the field by emergency medical technicians or in the emergency room to prevent secondary damage, thereby improving outcomes. However, the development of IMP as a therapy for TBI requires knowledge of the dose-response relationship, the therapeutic time window for treatment after injury, and the overall consequences of treatment on different cellular elements of the brain.
There was a clear dose-response relationship both in the improvement in motor function and in limiting the size of the lesion. Maximal effects were observed at a dose of 1 mg (40 mg/kg) given three times. Doubling the dose to 2 mg did not result in any significant improvement compared with the 1 mg dose. Reduction in the dose to 0.5 mg marginally reduced behavioral improvement, and reduction in the dose to 0.25 mg significantly reduced both anatomical and behavioral improvement. In view of the apparent lack of toxicity, a higher dose of 2 mg was used for subsequent studies.
Establishing a therapeutic window after TBI when IMP treatment is effective is particularly important since not all patients can reach a medical facility, be evaluated, and receive treatment within the 2-h window established in our original study. 2 Here we demonstrated that significant behavioral improvement effects were still observed when the initial treatment was delayed until 6 or 12 h after the injury. Delay until 24 h resulted in almost complete loss of efficacy. For all behavioral assessments, we did not observe any behavioral improvements beyond the 1-week time point. This is likely due to the natural resiliency of mice in recovery from the injury, as well as the result of limited sensitivity of the ladder rung test. Using long-term cellular phenotype as a recovery measure, treatments initiated at 6 h post-injury led to reduced lesion volume as well as MG activation that were not observed in mice that were treated at 12 or 24 h post-injury. These observations suggest that treatment should be initiated as soon as possible after injury, but that treatment remains effective even with a delay of up to 6 h after injury.
We then sought to define the molecular mechanisms underlying the therapeutic benefits of IMP treatment. Our prior study demonstrated that IMP treatment after TBI reduces infiltration of inflammatory cells, levels of inflammatory chemokines and cytokines, and reactive gliosis, and improves neuronal health in the perilesional zone with a reduction in tissue loss and improved behavior. 2 However, it remained unclear which perilesional cell types respond to IMP treatment, and how different cell types contribute to the therapeutic effects. Our scRNA-seq studies first confirmed the reduction in monocyte infiltration to the brain parenchyma and the decrease in molecular markers of reactive astrogliosis in the perilesional zone. In addition, our analysis revealed several novel insights regarding the cellular and molecular mechanisms of IMP-mediated therapeutic effects of TBI. First, despite limited changes in the UMAP spatial distribution when compared across TBI groups with and without IMP treatment, immune cells including MC and MG showed increases in the number of DEGs in the TBI+IMP group, consistent with the hypothesized immune modulating function of the particles. Importantly, we find that IMP treatment led to altered chemokine signaling from immune cells to vascular SMCs, primarily as the result of IMP-induced increases in CXCR2 and CCR1 expression in SMCs. These IMP-associated changes coincide with the initiation of BDNF signaling from SMCs to both excitatory and inhibitory neurons. Since BDNF is well characterized as a trophic factor for neuronal survival, this immune-SMCs-neurons signaling cascade may in part mediate the protective effects of IMP treatment. While direct regulation of BDNF transcript by CCR1 or CXCR2 has not been reported, CCR1 and BDNF are both known to be induced in SMCs by vascular injury or inflammatory stimuli in non-neural systems. 24,25 Signaling by other chemokine receptors has been shown to mediate detrimental sequela of TBI, 11,15,26 and CXCR2 signaling has been implicated in nociceptive sensitization after TBI. 27 Future studies that perturb CXCR2 and CCR1 expression in SMCs will allow us to determine whether BDNF expression is dependent on chemokine induced activation and whether it contributes to the therapeutic effects of IMP treatment after TBI.
In summary, IMP treatment within 6 h after TBI limits inflammatory responses and gliosis and improves anatomical and behavioral outcomes. At the molecular level, IMP treatment prevents detrimental changes in gene expression in both neural and non-neural cellular elements of the brain. In the pursuit of higher treatment efficacy, future studies will require refining the PLGA molecular properties that improve uptake, permeability, and retention in rodent and primate animal models. Cellular and molecular studies that further our understanding of the mechanisms of action may also lead to molecular targets for the functionalization of PLGA nanoparticles with pathway-specific conjugates. IMPs are made of an FDA-approved material that is stable at room temperature and could easily be given immediately intravenously after TBI in the field by EMTs or in the emergency room to prevent secondary damage, thereby improving outcomes.
Transparency, Rigor, and Reproducibility
In all experiments, the investigators who performed all procedures and analyses were blinded to the identity of the animals to ensure a lack of bias and to enhance the rigor and reproducibility of the studies. The number of animals in each group necessary for each measurement was predetermined by a power analysis done with G*Power 3.1.9.2 using the standard deviations and effect sizes from our preliminary studies, an alpha of 0.05, and a power (1-β) of 0.80. 122 mice were subjected to experimental injury in toto in the reported studies. Six mice died before randomization to study groups and 5 mice died after randomization. No mice were excluded from data analysis in any experiment. Every experiment was repeated twice more except the transcriptome studies were only done for 2 mice in each group. Data were analyzed using GraphPad Prism software (v5.04), and statistical significance was assigned at a predetermined cutoff of p < 0.05. Comparisons between 3 or more groups were conducted using ANOVA with the Tukey multiple comparison post hoc test. Data from the scRNA-seq are deposited at the NCBI Gene Expression Omnibus repository at accession number GSE266610.
Footnotes
Acknowledgment
IMP was generously provided by Cour Pharmaceuticals Inc.
Authors’ Contributions
R.B.: investigation, formal analysis, data curation, writing original draft, visualization. J.E.K.: investigation, formal analysis, data curation, editing. T.M.: investigation, project administration. C.Y.P.: investigation, validation, formal analysis, data curation, writing and editing, visualization. J. A.K.: conceptualization, validation, formal analysis, writing and editing, visualization, supervision, funding acquisition.
Funding Information
This work was funded by NIH grant
Author Disclosure Statement
No competing financial interests exist.
Supplementary Material
Supplementary Figure S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
