Abstract
It is important for patients and clinicians to know the potential for recovery from concussion as soon as possible after injury, especially in patients who do not recover completely in the first month and have concussion with persisting concussion symptoms (C+PCS). We assessed the association between the causes of concussion and recovery from C+PCS in a consecutive retrospective and prospective cohort of 600 patients referred to the Canadian Concussion Center (CCC) at Toronto Western Hospital. Data were obtained from clinical records and follow-up questionnaires and not from a standardized database. A novel method was used to assess long-term recovery, and multi-variable Cox proportional hazards models were used to assess relationships between cause of concussion and time to recovery. We examined the subsequent recovery of patients who had not recovered after at least one month from the time of concussion. Patients were grouped into the following four causes: sports and recreation (S&R, n = 312, 52%); motor vehicle collisions (MVC, n = 103, 17%); falls (n = 100, 17%); and being struck by an object including violence (SBOV, n = 85, 14%). The MVC group had the highest percentage of females (75.7%), the oldest participants (median: 40.0 [interquartile range (IQR):30.5–49.0] years), the most symptoms (median:11.0 [IQR:8.5–15.0]), and the longest symptom duration (median: 28.0 [IQR:12.0–56.00] months). In contrast, the S&R group had the highest percentage of males (58.1%), the youngest participants (median:20.0 [IQR:17.0–30.0] years), the best recovery outcome, and shortest symptom duration (median:22.0 [IQR:8.0–49.5] months). Significant differences among the four causes included age (p < 0.001), sex (p < 0.001), number of previous concussions (p < 0.001), history of psychiatric disorders (p = 0.002), and migraine (p = 0.001). Recovery from concussion was categorized into three groups: (1) Complete Recovery occurred in only 60 (10%) patients with median time 8.0 (IQR:3.5–18.0) months and included 42 S&R, 7 MVC, 8 falls, and 3 SBOV; (2) Incomplete Recovery occurred in 408 (68.0%) patients with persisting median symptom time of 5.0 (IQR:2.0–12.0) months; and (3) Unknown Recovery occurred in 132 (22.0%) patients and was because of lack of follow-up. In summary, the cause of C+PCS was associated with the type, number, and duration of symptoms and time required for recovery, although all causes of C+PCS produced prolonged symptoms in a large percentage of patients, which emphasizes the importance of concussions as a public health concern necessitating improved prevention and treatment strategies.
Introduction
Concussion is an important public health, economic, and social concern because of its high incidence, 1 frequent failure of complete recovery, prolonged duration of incomplete recovery, 2 and potential for catastrophic results from the consequences of repetitive concussion including second impact syndrome and cognitive decline. 3,4 A recent report, 5 found that “concussion patients, caregivers and clinician priorities for concussion researchers were early and accurate concussion diagnosis, effective symptom management and prediction of poor outcomes.” The present report addresses these priorities.
Concussion is also known as mild traumatic brain injury (mTBI), although these diagnoses are not identical. For example, mTBI includes patients with Glasgow Coma Scale (GCS) score of 13–15, whereas all patients with concussion are GCS score of 15. Currently, acute concussion is considered to last about 30 days as we defined previously, 6 similar to the that of the Concussion in Sport Group, and if symptoms persist beyond that time, the condition is termed persisting concussion symptoms (PCS), 7 a condition previously termed post-concussion syndrome. 2 The definitions used in this report are also in keeping with recent recommendations. 8,9
Concussion is induced by mechanical forces to the brain such as linear and rotational acceleration and deceleration that produce incompletely understood pathophysiological processes. 10 –12 The diagnosis of concussion depends on the clinical signs and symptoms because imaging 10 –12 and biofluid biomarkers lack sufficient sensitivity and specificity. 13,14 Computed tomography (CT) or magnetic resonance imaging (MRI), however, are often required to rule out brain injuries more severe than a concussion. We reported that brain imaging is normal in C+PCS in which all patients have a GCS score of 15, 15 but may be abnormal in mTBI, which includes patients with a GCS score of 13–15. 16 –19
Previously, we reported that the duration of symptoms after C+PCS increases with the number of signs and symptoms 2 , but we did not assess the association between the cause of concussion and the likelihood of complete recovery or the duration of symptoms in incomplete recovery. In the present article, we used a novel method for assessing recovery after C+PCS based on available follow-up information as described in the Methods Section.
Mainly because of poor compliance with follow-up, it remains uncertain what proportion of patients with an acute concussion have complete recovery in one month, with estimates ranging widely. 20 –22 Thus, despite the high prevalence of concussion, poor compliance has resulted in a small number of longitudinal studies. Indeed, we have reported previously poor compliance in the S&R group. 23 We are aware of one longitudinal study that showed significant variation in the time taken to achieve complete recovery 24 and other studies of PCS, including ours, that found that many patients did not achieve complete recovery even after several years. 2,25,26 The association between the cause of concussion and recovery, however, has been assessed in only a small number of studies.
The percent of concussed patients in whom PCS develops ranges from 5% 27 to 58%, 28 with the most reported incidence being 10–15%, 16 –18,29,30 although we and others have found it to be 25–30%. 31,32 Patients with PCS experience a myriad of at least 65 symptoms, including headache, dizziness, fatigue, irritability, cognitive difficulty, depression, anxiety, and sleep disturbance. Indeed, when these symptoms persist for months to years after injury, the long-term disability is substantial, with many unable to return to learn, sports, or work. 33 –35 We have shown previously that there is a strong relationship between the number of symptoms and the speed and completeness of recovery from C+PCS. 2
The following risk factors have been identified for increasing the risk of concussion or the risk of PCS or both: children and adolescents, 24,36 older adults, 25,30,37,38 female sex, 18,39 –44 history of previous concussion, 16,24,29,45 –48 pre-existing learning disabilities and attention deficit disorder/attention-deficit hyperactivity disorder (ADD/ADHD), 49 history of migraines, 50,51 history of psychiatric conditions, 25,30,43,47,52 –58 and type of sport and position played. 59 –61 The cause of injury as a risk factor, however, has not been studied extensively.
The four most common causes of concussion are sports and recreation (S&R), motor vehicle collisions (MVC), falls especially in the elderly, and being struck by an object (SBOV), including partner violence, other types of assault, and falling objects. Seiger and associates 61 examined whether the cause of injury affected recovery from concussion, but the study was limited to sports and MVC and only in a high-school population.
Our previous report 62 examined the relationship between cause and PCS in 437 patients and found significant associations between PCS and age, sex, and cause of injury. For example, older patients were concussed by falls and MVC while younger participants were concussed by S&R (p < 0.01), and patients with a history of only one concussion sustained it in an MVC while those with multiple concussions sustained them in sports. The relationships between the causes of concussion and long-term recovery were not examined, however.
The main purpose of the present study of 600 patients with C+PCS was to examine the association between the four leading causes of concussion described above and the likelihood of recovery from concussion including the speed and completeness of long-term recovery. With this method, we examined the association between the following: (1) the cause of C+PCS and the time required to achieve complete recovery and (2) the cause of C+PCS and the duration of persisting symptoms in those with known incomplete recovery. This is the first study to examine the association between long-term recovery and cause of concussion in a longitudinal cohort of C+PCS.
We hypothesized that there are important relationships between the causes of C+PCS and recovery from C+PCS, including the completeness of recovery and the time taken to recover, and that these outcomes will differ depending on age and gender. We also hypothesized that the causes of C+PCS will be a determinant of the symptoms of concussion. Our specific objective was to improve the precision with which patients are treated for this condition by determining the risk factors.
Methods
Study design and subjects
We assessed long-term recovery in a large, consecutive retrospective and prospective cohort (N = 600) with C+PCS all referred by other physicians to the CCC at Toronto Western Hospital. All patients had an initial clinic visit between 1990 and 2018, and all initial visits were accomplished pre-COVID pandemic. The treatment offered for PCS was multi-disciplinary, individualized, and symptom-oriented for managing headaches, mental health issues, dizziness, sleep disorder, and other symptoms with an emphasis on early return to physical and cognitive activity and respect for thresholds at which symptom exacerbation occurred.
The CCC focuses on long-term management of C+PCS through a policy of providing long-term follow-up by repeated visits and periodic questionnaires to assess the longitudinal effects of concussion. To accomplish this, all patients were specifically directed to return for at least one or more follow-up visits, and after completion of follow-up office visits, patients were requested to complete follow-up questionnaires mailed to them approximately every 18–24 months until they were fully recovered from C+PCS. The study was approved by the University Health Network Research Ethics Board.
As noted above, there is still no uniform definition of PCS in the literature. For the present study, we used a definition similar to the that of the Concussion in Sport Group that we used previously 6 : three or more persisting concussion symptoms lasting for at least one month after the concussion. Of the 815 consecutive patients with concussion seen by the first author (CHT) at the CCC from 1990–2018, 600 with C+PCS were included in the present study (Fig. 1).
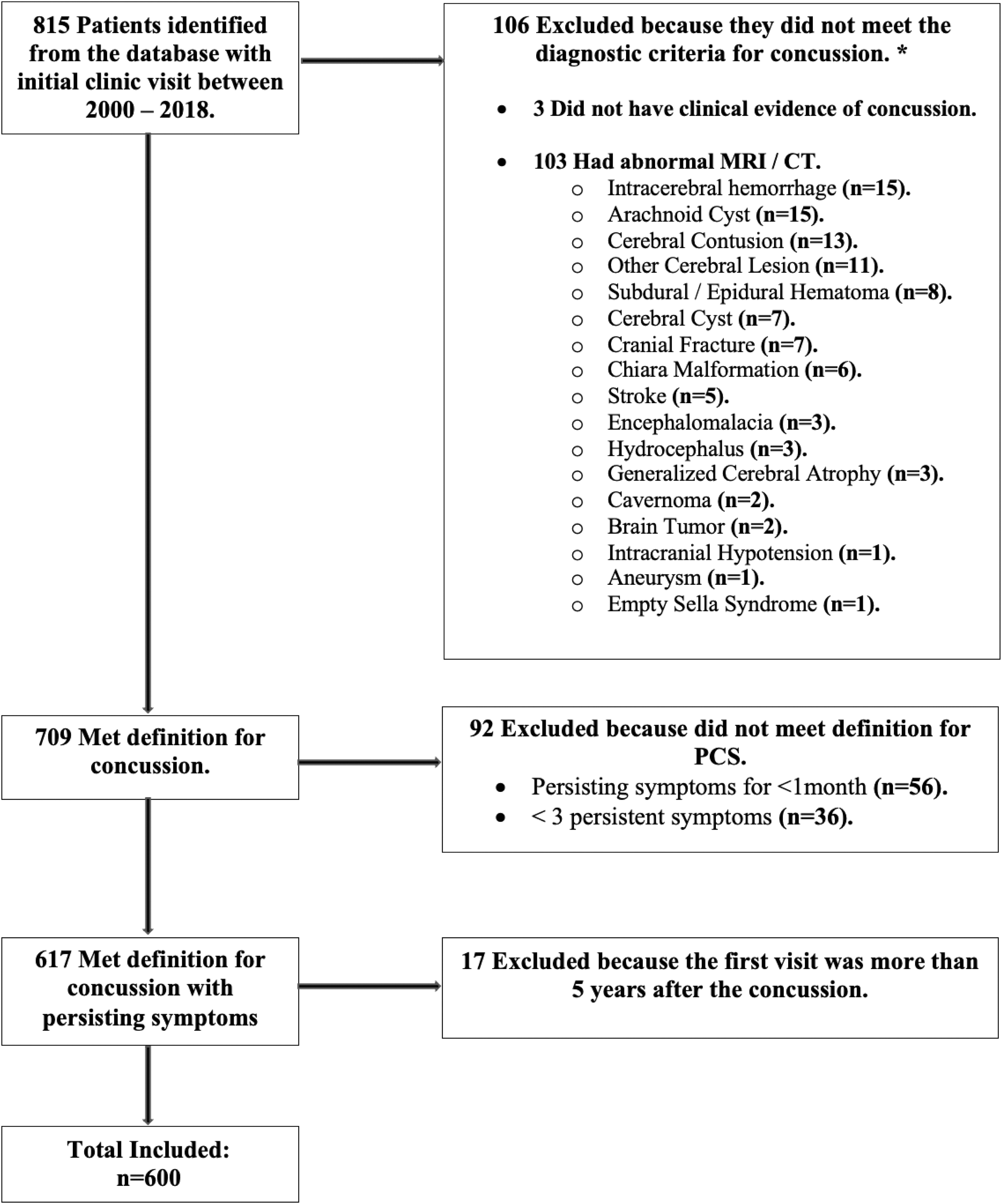
Patient inclusion and exclusion criteria for study (N = 600). *Concussion definition as defined in the Methods section.
The reasons for the 215 exclusions were as follows: three did not have clinical evidence of a concussion; 106 had abnormal MRI/CT imaging results indicative of a brain injury of greater severity than concussion including intracerebral hemorrhage and cerebral contusion, or other intracranial abnormalities such as arachnoid cysts; 92 were excluded because of failure to meet the clinical criteria for PCS in this study (36 had fewer than three persisting symptoms, and 56 had symptoms lasting less than one month). Last, 17 patients were excluded to eliminate recall bias because their first CCC clinic visit was more than five years after their concussion.
We recently reported the first imaging criteria for PCS, 44 and all patients in the present series who underwent imaging (MRI + CT: n = 190, MRI alone: n = 299, CT alone: n = 50), conformed with these criteria.
Definitions
Concussion was defined according to the 5th International Conference on Concussion in Sport, 7 and the index concussion was the concussion that brought the patient to the CCC for consultation. Our Centre has used a definition of PCS described previously 6,41 based on the following Berlin guidelines of the Concussion in Sport Group: (1) any three symptoms of concussion; (2) persistence of symptoms for at least one month; and (3) absence of clinical or imaging evidence of a more severe TBI such as cerebral contusion, subdural hematoma, subarachnoid hemorrhage, or intracerebral hemorrhage identified by the presence of focal neurological defects such as limb weakness or by lesions seen on MRI or CT scans.
Thus, the diagnosis of PCS was made clinically with exclusions based on clinical and any available imaging findings. All measures of duration of symptoms and time to recovery were in relation to the date of the index concussion.
The definition of complete recovery was complete absence of any persisting concussion symptoms as reported by the patient, and the definition of incomplete recovery was continuing presence of one or more persisting symptoms of concussion reported by the patient. Although information about recovery was obtained during clinic visits or via questionnaire, wherever possible, the actual time of recovery was based on the patient's stated date of recovery rather than the time of the visit or completion of a questionnaire.
To assess recovery and duration of symptoms, patients were placed in one of three recovery categories based on the patient's own reporting: (1) Known Complete Recovery: the patient had CCC follow-up via clinic visit or returned questionnaire and reported complete recovery, the duration of which was based on the time elapsed between the date of the index concussion and the date of recovery provided by the latest follow-up visit or questionnaire; (2) Known Incomplete Recovery: the patient had at least one follow-up visit or questionnaire, did not report complete recovery, but reported either no change or improving but incomplete recovery. In this category, the known duration of symptoms is the time elapsed between the date of the index concussion and the date provided at the latest follow up visit or questionnaire, and thus, denotes the patients' known total symptomatic time. It should be noted that in this category, the subsequent duration of symptoms is unknown because of the termination of follow-up; (3) Unknown Recovery: the patient attended only one clinic visit at which time he or she reported incomplete recovery and did not return any questionnaires. Questionnaires were sent every 1-2 years to patients who had not recovered, with the last batch of questionnaires distributed in 2021–2022.
Data collection
Data collected from the patient's clinical records were from many sources, including CCC narratives from the examining physicians, MRI and CT imaging reports, information from the referring MD, and from other physicians and healthcare professionals to whom patients had been referred, before or after the initial CCC visit. Data were also collected from the questionnaires sent every 1–2 years to discharged patients continuing to be symptomatic and non-compliant patients who did not return for visits. The CHT office of the CCC to which all 600 patients were referred did not use a standardized concussion symptom checklist. The patient's narrative of symptoms and responses to questions were thoroughly documented, however, including any accompanying persons' comments.
All interviews were conducted by an experienced physician specializing in concussions, or by supervised concussion fellows who were physicians. The Concussion Clinic is an ambulatory care clinic in the Toronto Western Hospital and accepts physician referrals of concussed patients from age 11 upward with no upper age limit.
The Concussion Clinic was Toronto's first such clinic and was initially specializing in sports concussions, but since 2011, patients with concussion of all causes were welcomed. The information collected included patient characteristics such as sex, age, previous concussions, and pre-existing condition, radiographic findings, concussion cause, symptoms, recovery, and extracranial injuries. Data were treated confidentially and de-identified to protect patient identity. Trained Research Assistants and Graduate Students extracted the data from patient charts.
Independent variables were patient characteristics and the four causes of concussion (S&R, MVC, falls, and SBOV), and dependent variables were recovery (known complete, known incomplete, or unknown), and the number and type of persisting symptoms. Fifty-six different symptoms were recorded from these sources (Supplementary Table 3). Only the index concussion, which was the concussion that resulted in clinic referral, was used for analysis of the causes and symptoms of patients with multiple concussions. Patients with intracranial injuries more severe than concussion were excluded, but patients with major extracranial injuries such as limb fractures, internal organ injuries, chest injuries, and whiplash were included and tabulated, although minor injuries such as lacerations and bruising were not tabulated.
Statistical analysis
Continuous variables were summarized using Median (IQR defined as 1st quartile–3rd quartile), and the Kruskal-Wallis test was used to compare groups. Percentages were reported for categorical variables and the Chi-square test was used to compare groups. Fisher exact test was used to assess differences in learning disability and some symptom prevalence. Because of the unequal follow-up time between subjects, we used rates of recovery (per person-year), as well as numbers (percentages) within different fixed lengths of follow-up.
Multi-variable Poisson models were used to assess the association of cause of injury with the number of symptoms, adjusted for age and length of follow-up. We used Kaplan-Meier curves with Log-Rank tests, as well as univariable and multi-variable Cox PH models for evaluating the association between cause of injury and time to recovery. Patients who did not come for a follow-up visit and did not return any questionnaires were censored at the time of their first visit, and patients without a documented date of complete recovery were considered censored at the date of the last known visit or returned questionnaire or indicated recovery date.
Multi-variable models were adjusted for potential confounders selected based on their significant associations with the outcomes and significant differences between the four cause groups, as well as clinical importance.
All hypothesis tests were conducted against a two-sided alternative. The p-values were considered statistically significant when less than 0.05. All analysis was performed in R.4.2 (R Foundation for Statistical Computing, Vienna, Austria.)
Results
Causes of concussion
Supplementary Table 1 shows the complete list of the causes of C+PCS in the 600 patients: 312 (52%) in S&R, 103 (17%) in MVC, 100 (17%) in falls, and 85 (14%) in SBOV. The S&R group was further categorized into winter sports (n = 160), field sports (n = 61), floor sports (n = 29), bicycling and other street sports (n = 29), water sports (n = 13), other sports (n = 18), and unknown sports (n = 2). The MVC included automobile or truck (n = 98), motorcycle (n = 3), and pedestrian-motor vehicle (n = 2).
Falls included tripped/slipped (n = 24), slipped on ice (n = 15), fell downstairs (n = 12), fainted (n = 10), and other falls/unspecified (n = 39). The SBOV category included struck by a stationary object (n = 35), struck by a moving object (n = 19), struck by a falling object (n = 13), assault (n = 11), and unspecified (n = 7).
Patient characteristics
Table 1 shows the demographic features of the patients included in the study. There was no missingness in any of the demographic characteristics. There was a significant sex difference (p < 0.001) among the four causes of concussion. The total cohort consisted of 329 (54.8%) females. In the S&R group (n = 312), there were 130 (41.7%) females, MVC (n = 103) comprised 78 (75.7%) females, falls (n = 100) comprised 68 (68.0%) females, and SBOV (n = 85) comprised 53 (62.4%) females.
The median age for the total cohort was 29 (IQR:18–45) years, and there was a significant difference in median age at the time of the index concussion p < 0.001) among the different causes of concussion: S&R: 20 (IQR:17–30); MVC: 40 (IQR:30.5–49); falls: 46 (IQR:29–56); and SBOV: 39([IQR:23–53).
Demographic Features of Persisting Concussion Symptom Patients in the Four Cause of Injury Groups (N = 600)
MVC, motor vehicle collision; SBOV, struck by object including violence; IQR, interquartile range; ADD, attention deficit disorder; ADHD, attention deficit hyperactivity disorder.
There was a significant difference in the median number of previous concussions (p < 0.001) for the four causes of concussion. The median number of previous concussions for the total population was 1.00 (IQR:0.00–3.00, Table 1). Previous concussions were highest in the S&R group with a median of 2.00 (IQR:1.00–3.00), and lowest in the MVC and SBOV group, which both had a median of 0.00 (IQR:0.00–2.00). In addition, there were significant differences in the prevalence of previously diagnosed psychiatric conditions (p = 0.002) and migraine headaches (p = 0.001) among the four causes of C+PCS. As well, the incidence of extracranial injuries differed significantly among the four causes of injury with MVC having the greatest incidence of extracranial injuries (Table 1).
Patient compliance for follow-up visits and questionnaires returned in relation to recovery and duration of symptoms
Supplementary Table 2 shows the compliance of the 600 patients based on follow-up clinic visits and returned questionnaires. If a patient did not come for a follow-up visit and did not return any questionnaires, recovery was designated as unknown (n = 135, 22.5%). During the first and only clinic visit, however, three patients reported that they had already completely recovered, which explains why the unknown recovery group consisted of 132 patients (Supplementary Table 2).
The largest group of patients in the table comprised 149 patients, 25% of the total 600 patients, and they had 1–2 visits but returned no questionnaires. The MVC group was the most compliant group with the highest number of visits and returned questionnaires, although the number of compliant patients was still very small. The least compliant group of 132 patients had no follow-up visits, returned no questionnaires, and consisted of 74 S&R (23.1%), 16 MVC (15.5%), 27 falls (27.0%), and 18 SBOV (21.1%). Thus, the falls group had the highest percent of non-compliant patients, defined as no follow-up visit and no questionnaires. It is likely that that there were multiple reasons for non-compliance including socioeconomic factors, transfer to other healthcare professionals, and movement of subjects to other locations.
Recovery from PCS based on Known Complete Recovery, Known Incomplete Recovery, and Unknown Recovery
The basis for the categorization of recovery was given in the Methods section. Table 2 shows that 60 (10.0%) patients had known complete recovery, and 408 (68.0%) had known incomplete recovery as determined by visit or returned questionnaire during follow-up. In contrast, in 132 (22.0%) patients, recovery was unknown because of lack of compliance with follow-up.
Recovery from Concussion With Persisting Concussion Symptoms (N = 600): Known Complete Recovery, Known Incomplete Recovery, and Unknown Recovery *
C+PCS, concussion with persisting concussion symptoms; IQR, interquartile range.
See text for description of the three categories of recovery.
In three patients, the exact time to complete recovery is unknown.
It is of interest that patients with complete recovery came for their first clinic visit in the shortest time after the concussion with a median of 3.00 (IQR:1.00–7.25) months. Those with known incomplete recovery had a median of 5.00 (IQR:2.00–12.00) months, and those with unknown recovery had a median of 8 (IQR:4.00–18.00) months. Only 60 (10%) patients had complete recovery, 57 of whom with known duration of symptoms recovered in a median of 8.00 (IQR:3.50–18.00) months, whereas those with known incomplete recovery (n = 408) were symptomatic for at least a median of 36 (IQR:18.0 − 64.00) months.
Relationships between the cause of C+PCS and the total number, duration, and prevalence of symptoms
There were significant differences in the total number of symptoms for each cause of concussion (p < 0.001) (Table 3). In S&R, the median number was 8.00 (IQR:6.00–12.00), MVC 11.00 (IQR:8.50–15.00), falls 8.00 (IQR:6.00–12.00), and SBOV 10.00 (IQR:6.00–15.00). After MVC, PCS symptoms lasted longer than the other causes with a median of 28 (IQR:12.00–56.00) months, and in contrast, after S&R, the median was 22 (
Association of Cause of Concussion with Symptoms and Recovery (N = 600)
S&R, sports and recreation; MVC, motor vehicle collision; SBOV, struck by object including violence; PCS, persisting concussion symptoms; IQR, interquartile range.
Known symptom duration is defined as follow-up duration for those who did not recover, or time to recovery for those who had complete recovery.
Of the 60 patients with known complete recovery, 42 (13.46%) recovered in the S&R group (n = 312), 7 (6.79%) in the MVC group (n = 103), 8 (8%) in the falls group (n = 100), and 3 (3.52%) after SBOV (n = 85). The S&R group had the fastest rate of recovery (S&R = 0.038 cases/person years), compared with the other causes (MVC = 0.02, falls = 0.025, and SBOV = 0.012).
In a multi-variable model adjusted for age and time from index concussion to latest follow-up visit or questionnaire, we found MVC and SBOV were associated with a higher expected number of symptoms, when compared with the S&R group (RR = 1.17, 95% CI (1.09,1.26), p < 0.0001; and RR = 1.11, 95% CI (1.02,1.2), p = 0.014, respectively.), while no association was present for falls (RR = 0.99, 95% CI (0.91,1.07), p = 0.74) (Table 4).
Association of Cause of Concussion With the Number of Symptoms and Adjusted for Length of Follow-Up and Age
RR, risk ratio; CI, confidence interval; S&R, sports and recreation; Ref., reference group; MVC, motor vehicle collision; SBOV, struck by object including violence; V, visit; Q, questionnaire.
Multi-variable Poisson Model adjusted for length of follow-up and age.
Table 5 shows the length of time patients experienced symptoms for each cause of concussion and the percentage of patients who recovered completely within a given interval. We used bins of unequal size because in the earlier months after the concussion, patients had more frequent visits. Also, after 20 months they became more spread out, while after 20 months, full recovery was uncommon.
Timing of Known Complete Recovery from Concussion With Persisting Concussion Symptoms in 57 * Patients at Increasing Time Intervals from the Index Concussion to Latest Follow-Up Visit or Questionnaire (N = 600)
C+PCS, concussion with persisting concussion symptoms; S&R, sports and recreation; MVC, motor vehicle collision; SBOV, struck by object including violence.
All 600 patients are included in this table. There is no 0–1 month column because all patients in the study had PCS lasting at least one month. Although 60 of the 600 patients reported complete recovery, data on time to recovery was available for 57 patients. Known Complete Recovery was rare after 29 months of C+PCS, with only seven patients achieving complete recovery after that time.
Notably, patients with the shortest symptom duration and fastest complete recovery were from the S&R group, which had the largest percentage of patients (13 patients, 2.3%) who experienced symptoms within 1–3 months and recovered completely. No one from the other causes recovered completely within 1–3 months except 1 (0.2%) participant from the MVC group. After 30 months, very few patients recovered completely. In the range of 30–39 months, 1 S&R (0.4%) patient, and 4 falls (1.7%) patients experienced symptoms for 30 months and then recovered completely by 39 months. After 40 months, only 1 S&R patient and 1 falls patient recovered completely.
The type and prevalence of persisting symptoms are displayed in Supplementary Table 3. In total, 56 symptoms were recorded and categorized as peri-injury (3 symptoms), somatic (23 symptoms), cognitive (14 symptoms), emotional (13 symptoms), and sleep (3 symptoms) based on the Centers for Disease Control and Prevention's (CDC) classification of concussion symptoms. 43
For all symptom categories and all causes of concussion, the most prevalent symptoms were headache (n = 529, 88%, p = 0.609), memory difficulties (395 patients, 65%, p = 0.092), and balance impairment (329 patients, 54.8%, p < 0.001). When evaluating symptom prevalence for each concussion cause individually, the first and second most prevalent symptoms for each cause was also headaches (S&R: n = 279, 89%; MVC: n = 87, 84%; falls: n = 88, 88%; SBOV: n = 7 5, 88%), and memory problems (S&R: n = 195, 62%; MVC: n = 78, 76%; falls: n = 68, 68%; SBOV: n = 54, 63%), respectively. The third most common symptom varied among the four causes: concentration difficulties in S&R (166 patients, 53%), balance problems in MVC and falls (n = 69, 66%, and n = 66, 66%, respectively), and sensitivity to light in SBOV (n = 52, 61%).
Of the 56 recorded symptoms, we were able to calculate p values for 51, and there was a significant difference in symptom prevalence with respect to the causes of concussion for nine symptoms (Supplementary Table 3). Symptoms were significantly different if the p value was less than or equal to the adjusted p value (0.05/51 = 0.001) and included balance (p < 0.001), irritability (p = 0.001), noise sensitivity (p < 0.001), vertigo (p < 0.001), anxiety (p < 0.001), depression (p = 0.001), post-traumatic stress disorder (PTSD) (p < 0.001), sleeping less (p < 0.001), and speech problems (p < 0.001).
Association of cause of concussion with time to complete recovery from PCS
Overall, there was a significant difference in time to complete recovery between the four causes of concussion (p = 0.018) (Fig. 2). Because of the small sample size of completely recovered patients in the falls, MVC, and SBOV groups, we combined the falls, MVC, and SBOV groups into an “Other” category and compared complete recovery with the S&R group and found a significant difference in time to complete recovery between the two groups (p = 0.0029) (Fig. 3).
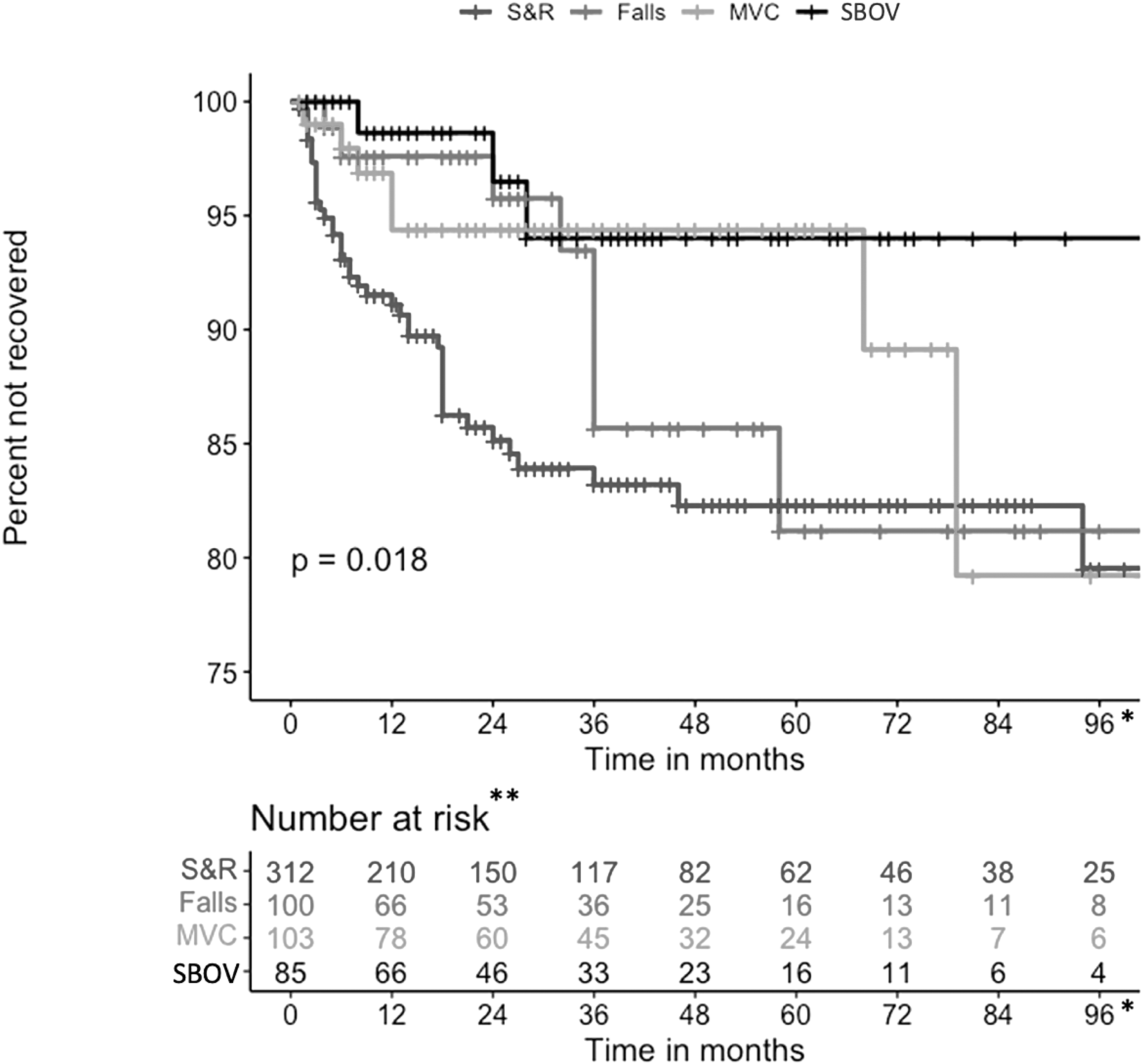
Kaplan-Meier curve for Complete Recovery among all four causes over 96-month follow-up (N = 600). S&R, sports and recreation; MVC, motor vehicle collision; SBOV, struck by object including violence. *Maximum length of follow-up was 250 months; all events happened before 96 months. **Number at risk refers to patients not yet recovered or not yet censored.
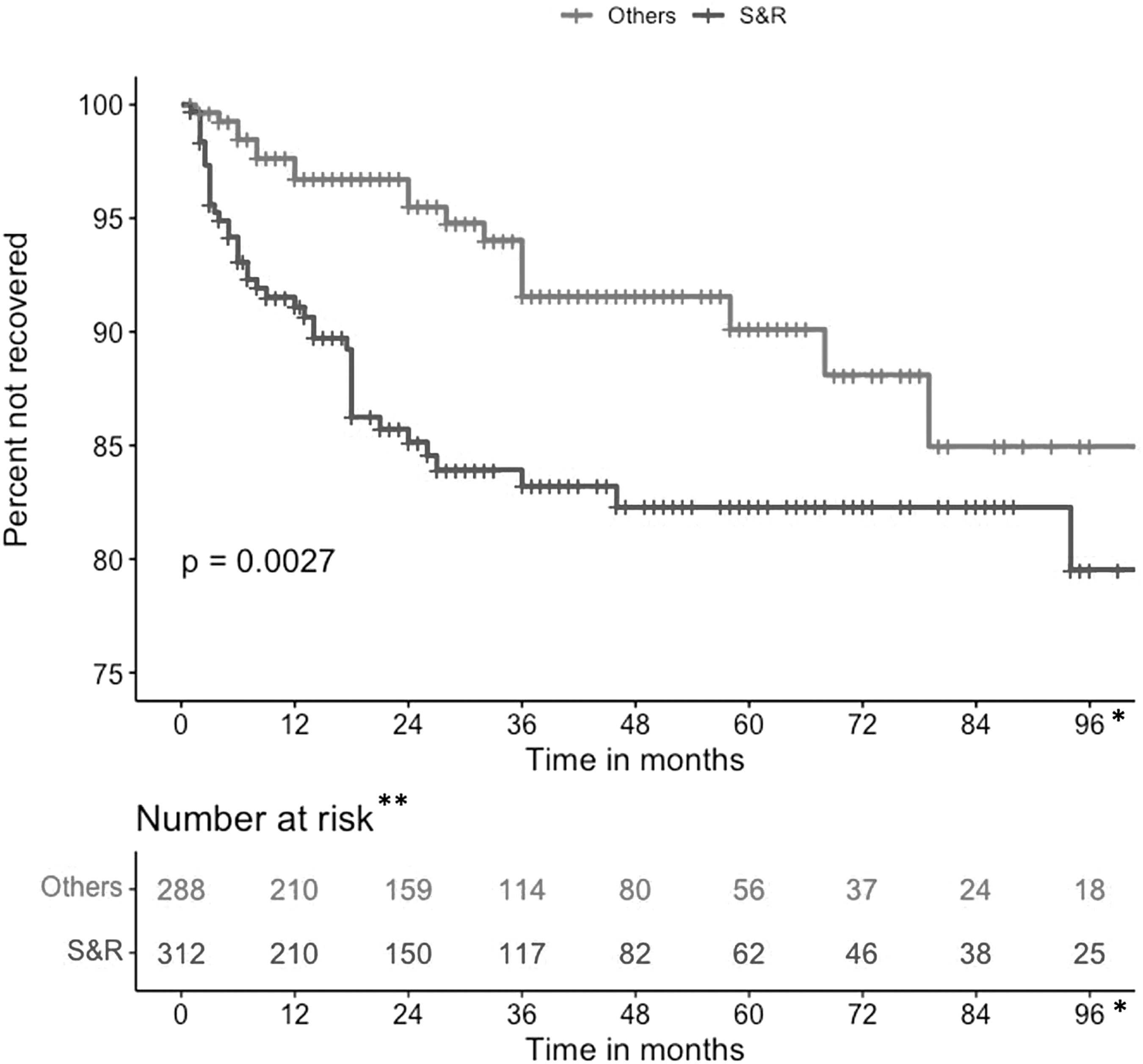
Kaplan-Meier curves for Complete Recovery between S&R and the other three causes over 96-month follow-up (N = 600). S&R, sports and recreation; Others defined as: MVC, motor vehicle collision; SBOV, struck by object including violence; or falls. *Maximum length of follow-up was 250 months; all events happened before 96 months. **Number at risk refers to patients not yet recovered or not yet censored.
In a univariable Cox PH model, S&R was significantly associated with a 2.5 times increase in the “hazard” of full recovery compared with the other three mechanisms of injury (hazard ratio [HR] = 2.52, 95% CI (1.42, 4.49), p < 0.01). After adjusting for age, sex, and history of previous psychiatric disorders, however, the association became both weaker in magnitude and no longer significant (HR = 1.42, 95% CI (0.73,2.76), p = 0.31), suggesting at least a partial confounding effect. Similarly, while the MVC group was associated with worse recovery in the univariable model compared with the other three mechanisms of injury (HR = 0.43, 95% CI (0.17, 1.08), p = 0.07), the association is no longer present after adjusting for the above characteristics (HR = 0.71, 95% CI (0.28, 1.87), p = 0.497) (Table 6). All Cox PH models satisfied the PH assumption.
Association of Cause of Concussion with Time to Recovery
HR, hazard ratio; CI, confidence interval; Ref., reference group.
Others defined as: S&R, sports and recreation; MVC, motor vehicle collision; SBOV, struck by object including violence; or falls.
Univariable and multi-variable Cox PH model adjusted for age, sex, and previous history of psychiatric disorders.
Discussion
The present study confirmed several findings we reported earlier about the epidemiology of C+PCS 62 and also identified several novel findings with respect to the associations between the causes of concussion, the demographic features of patients with C+PCS including the type, number, and prevalence of PCS symptoms, and the recovery from concussion. Similar to our previous report, we found several relationships between age, gender, and cause of injury, but in addition, we identified several other demographic findings related to the specific cause of injury, including significant differences in the number of previous concussions, history of psychiatric disorders, and migraine. We also identified a greater percentage of women than men who sustained C+PCS caused by MVC, falls, and SBOV (Table 2).
The present study focused on long-term recovery from C+PCS, which very few previous studies have examined. Figures 2 and 3 show the association between long-term recovery and the four causes of concussion with a follow-up duration of 96 months. Although our clinic has follow-up data of up to 250 months, we show the first 96 months because all the curves showed a continuous flat line after that indicating that no patient made a complete recovery after 96 months among the many cases followed for longer durations after that.
The S&R group had a higher likelihood of complete recovery from PCS than the three other mechanisms of injury combined (Fig. 3). The S&R group was also the largest group, had the highest percent of males (p < 0.001), and was significantly younger (p < 0.001) at the time of their index concussion compared with the other causes. When the possible confounders were assessed, the highest complete recovery in the S&R group was likely because of the significantly younger age (Table 5). We assessed the relationship between complete recovery among the different age groups, and Figure 4 shows that individuals who were younger recovered better than older individuals: for example, the less than 20-year-old group had the best recovery, ages 21–30 years had the second-best recovery, and individuals aged 31–50 years or older than 50 years had less recovery.
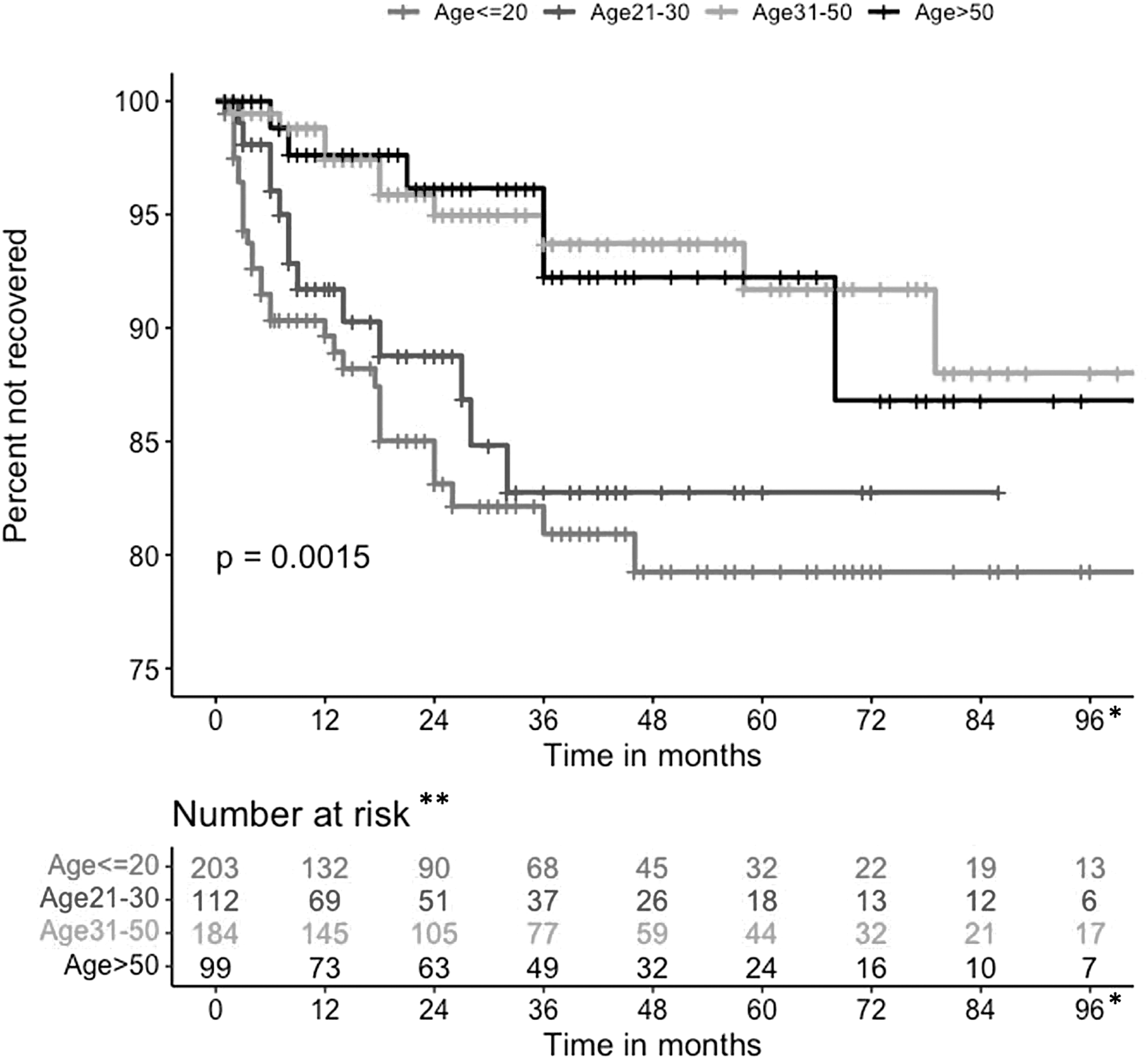
Kaplan-Meier curves for Complete Recovery between different age groups over 96-month follow-up (N = 600). *Although the maximum length of follow-up was 250 months in one patient, all Complete Recovery events happened before 96 months. **Number at risk refers to patients not yet recovered or not yet censored.
Additional confounding variables also influenced recovery, such as sex (Fig. 5). Although, the S&R group comprised the youngest participants, it also had the highest percentage of males. In contrast, the MVC group comprised the oldest patients, had the longest duration of symptoms, and had the highest proportion of females at 75.7% (p < 0.001) (Table 1). There is other evidence that women have C+PCS for longer durations than men. 63
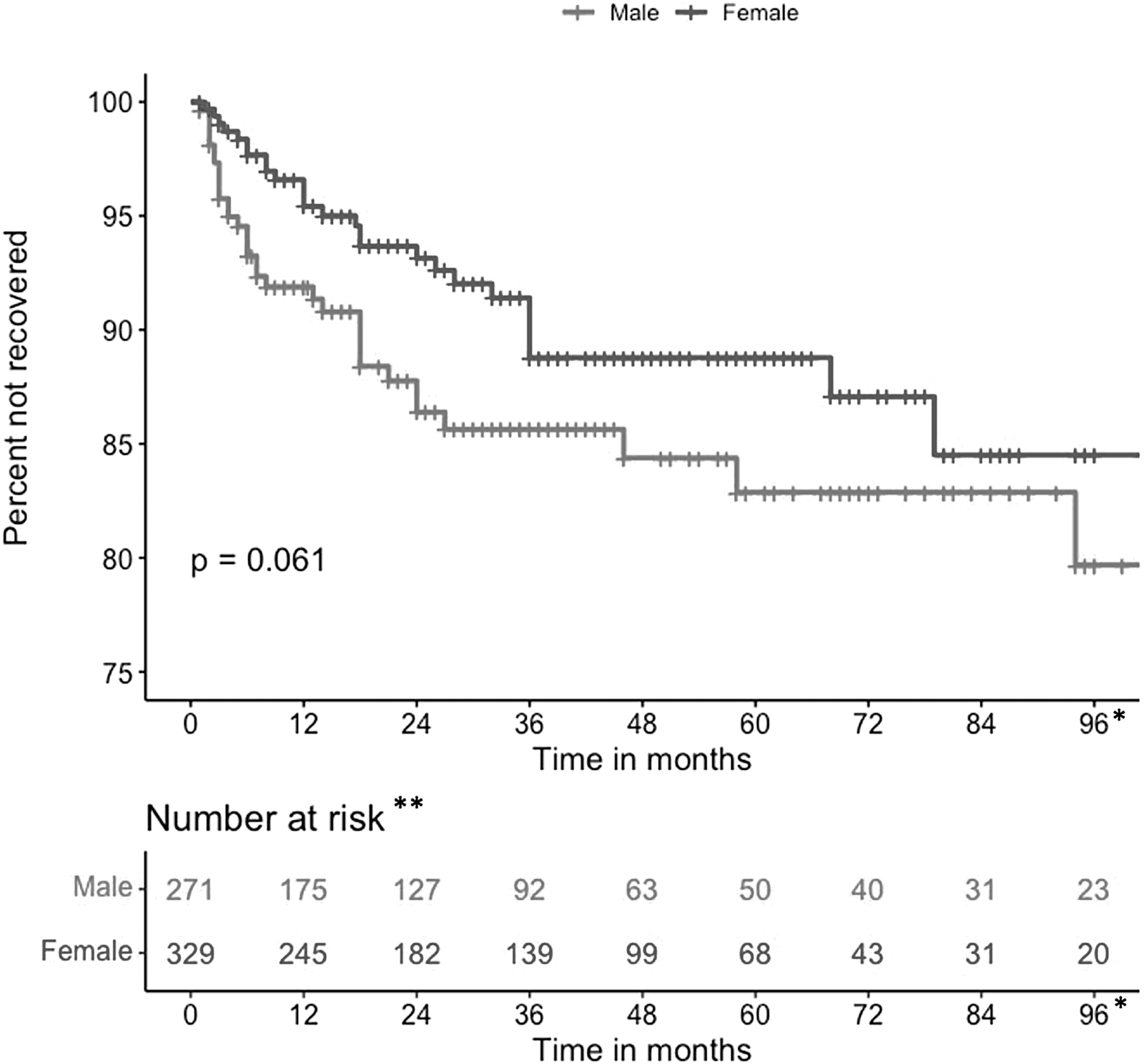
Kaplan-Meier curves for Complete Recovery between males and females over 96-month follow-up (N = 600). *Maximum length of follow-up was 250 months; all events happened before 96 months. **Number at risk refers to patients not yet recovered or not yet censored.
Males and females differ in brain anatomy and in neck muscle strength, 64 with females often having smaller and weaker neck muscles, which may make them more susceptible to concussion. It has also been hypothesized that hormonal differences and the female menstrual cycle may influence recovery from C+PCS. 63 There was also a significant difference in concomitant extracranial injuries among the four cause groups (Table 1), and the greater incidence of extracranial injuries in the MVC group likely contributed to the lower recovery rates in that group. Other factors such as litigation, insurance claims, and socio-economic differences that we could not measure may also have influenced recovery.
We identified that poor patient compliance was a major reason for the significant number of patients in all four cause groups with missing long-term follow-up information, a problem we had identified previously in the S&R group. 23 Because follow-up was incomplete in many patients, we created a novel way to assess recovery based on three patient groups: (1) Known Complete Recovery and Known Total Duration of Symptoms; (2) Known Incomplete Recovery in which the duration of symptoms was calculated based on the time elapsed from the date of the index concussion to the latest visit or questionnaire; and (3) Unknown Recovery because patients did not return for a follow-up visit or return a questionnaire (Table 2).
Median recovery time for patients with Known Complete Recovery was 8 [3.5–18.0] months (Table 2). Those with Known Incomplete Recovery were symptomatic for a median (IQR) of 36 (18–64) months. Our previous study found that patients who were still symptomatic after 3 years did not make a full recovery from PCS. 2 In the current study, however, 2 patients in the total cohort (N = 600) did achieve complete recovery after 3 years of PCS, with complete recovery at 46 months and 58 months, respectively.
In addition, we determined that those who made a complete recovery came for their first initial clinic visit sooner than those with known incomplete recovery. This may suggest that these patients were more proactive about seeking medical attention for their injury and were thus more compliant in informing us of their recovery. It may also suggest that seeking medical attention earlier after concussion promotes recovery.
Similar to our previous study, 2 we identified a relationship between the number of symptoms reported and recovery time. In the present study, however, we were also able to associate recovery with the cause of injury. To our knowledge, this is the first study to examine the relative frequency of each persisting concussion symptom (n = 51) among the four main causes of concussion.
As in our previous study, 2 patients who reported fewer symptoms such as the S&R group tended to recover faster than those who reported more symptoms, such as the MVC group (Figs. 2 and 3). The MVC patients reported the most symptoms and had the longest symptom duration compared with all other injury causes (Table 3). As indicated above, the greater incidence of concomitant injuries may account for the prolonged recovery time.
In addition, in the present study, we were also able to assess possible relationships between symptom type and cause of concussion and found significant differences in incidence for 9 reported symptoms; all 9 symptoms were highest in MVC. The symptoms and percentage of MVC patients who experienced these 9 symptoms were as follows: imbalance (67%), irritability (59.2%), noise sensitivity (58.3%), vertigo (30%), anxiety (67.0%), depression (67.0%), PTSD (10.7%), too little sleep (35.9%), and speech problems (13.6%). Individually and collectively, these are important and disabling symptoms that can interfere significantly with return to work or school and family life.
Thus, the present study provides evidence that the cause of injury plays an important role in both the duration and type of PCS symptoms. Tarkenton and colleagues 65 used the Sports Concussion Assessment Tool (SCAT3) and its Graded Symptoms Checklist (GSC) to assess the severity of patient symptoms and found patients who sustained a concussion in an MVC had a higher symptom severity score when compared with S&R— which is similar to that in our previous study. 62
The MVC may cause the most symptoms because of higher forces of energy to the brain such as in high-speed crashes compared with the other causes, as has been suggested. 61 The additional psychological stress associated with MVCs and the overlap between PTSD and PSC may also contribute to this finding. 65 Indeed, we are currently analyzing the factors that may be involved, especially in concussed female occupants of motor vehicles involved in MVC, because the majority of patients in our MVC group were female occupants.
Our study has several methodological limitations. All the C+PCS patients were seen by physicians at one tertiary hospital concussion clinic under the supervision of one of the authors (CHT). Further, even though it is a consecutive cohort followed prospectively, the population of patients is not representative of the entire spectrum of concussion patients. For example, by policy, the clinic sees only youths at least 11 years of age and, importantly, the CCC is a referral center specializing in PCS and in those with cognitive decline suspected of chronic traumatic encephalopathy. In general, the CCC did not see acutely concussed patients, especially during the first month after injury during which time about 75% of concussed patients recover, as discussed above.
Another limitation is related to the potential secondary gain for underreporting symptoms by athletes wishing to return to play or overreporting by MVC crash victims for insurance claims and litigation. Another potential limitation relates to the narrative system of symptom recording by physicians specializing in concussion versus the use of a symptom checklist such as the SCAT3 or other patient self-report checklist. There is limited evidence for which system is more accurate.
Meier and coworkers 66 conducted a study on self-reporting of concussion symptoms and found that athletes reported significantly fewer symptoms using standardized self-report symptom measurements compared with reporting during confidential in-person consultations. 66 The study of consistency of concussion reporting by Wojtowicz and associates 67 found good consistency of self-reporting by high school athletes on two occasions that varied from 3 months to 3 years apart using the same questionnaire about the number of concussions they had sustained, but symptom reporting was not assessed. They also found more inconsistent reporting of concussion history in athletes with ADHD.
Another reason for bias in our patient sample is the long interval between the index concussion and the first visit in many patients in this series. During this interval, less severely symptomatic patients may have recovered and cancelled their appointments. Thus, the results may not represent all patients with C+PCS because our clinic's patients may have been more symptomatic than those reported by other authors. In an attempt to minimize this possible effect, we eliminated patients in whom the interval between the index concussion and the first visit was greater than 5 years.
Another possible limitation is the changing referral pattern in our region during the study period. In the earlier years, S&R was the predominant cause of concussion in the referred patients reflecting community concern and our research interest in sports concussion, which explains why this cause comprises 52% of the cases in the present report. In contrast, during the more recent years, there was broadening of both community concern and our concussion research interests to include all causes of concussions.
Conclusion
For the first time, in a longitudinal cohort we have provided evidence that the cause of injury is associated with the number, type, and duration of the symptoms reported and the recovery from C+PCS. We found that all causes of C+PCS can lead to significant symptoms and prolonged recovery time, although MVC caused the greatest number and longest duration of PCS symptoms.
We recommend further development of targeted prevention strategies for all causes of concussion and further research into more effective treatments for all patients with C+PCS because of their long-lasting effects. Further research is also necessary into methods for improving compliance with follow-up in patients with C+PCS.
Transparency, Rigor and Reproducibility
The study was approved by the University Health Network Research Ethics Board (13-6167). The study and analysis plan were not formally pre-registered because we did not decide to do this study at the beginning of patient accrual. Also, this was not a control trial, but rather a longitudinal, retrospective study, and we felt that the Research Ethics Board approval was sufficient. No power analysis was performed for this study. A total of 815 patient charts were reviewed to determine eligibility for the study, and a total of 600 patients met the inclusion criteria. Figure 1 shows how the participants were recruited and screened from our databank; at least one patient died after suspected suicide during the study period. Recovery was assessed based on clinic visits and returned questionnaires. Patients with known incomplete recovery attended at least one clinic visit or returned at least one questionnaire, in which they did not report complete recovery, and thus they were not available for further follow-up. Patients with unknown recovery had incomplete assessments because they failed to attend a follow-up clinic visit or return a questionnaire and did not report complete recovery at their only visit. Data was collected by trained research assistants through chart review of patients who came for a first initial clinic visit between 1990 and 2018, and from follow-up clinic visits or returned questionnaires. Research assistants could not be blinded because they needed to extract all the information from patient records. All questionnaires used in the study are available if requested from the authors. All information collected was confidential, and patients had a unique study ID, so that patients' names were not associated with any information collected. The methods section explains the nature of the statistical tests used. Two of the co-authors are statisticians and performed all the analyses using R.4.2 [R Foundation for Statistical Computing, Vienna, Austria]. All authors have agreed to publish the manuscript using the Mary Ann Liebert. Inc “Open Access” option, and with the appropriate license it will be freely available upon publication.
Footnotes
Acknowledgment
An abstract of this work was published previously in Abstracts from The 40th Annual Symposium of the National Neurotrauma Society held June 25–28, 2023 in Austin, Texas, USA
Author Contributions
Charles Tator: Conceptualization, Methodology, Writing, Supervision; Connor Moore Methodology, Data acquisition, Writing and Data Curation; Chloé Buso: Methodology, Data Acquisition, Writing; Ella Huszti: Formal Analysis, Statistics; Qixuan Li: Formal Analysis, Statistics; Emma Prentice: Methodology, Data acquisition and Data Curation; Mozhgan Khodadadi: Project administration; Olivia Scott: Data Acquisition; Carmela M. Tartaglia: Conceptualization, Methodology, and Writing.
Funding Information
This study was funded by specific donations to the CCC via the University Health Network Foundation.
Author Disclosure Statement
No competing financial interests exist.
Supplementary Material
Supplementary Table 1
Supplementary Table 2
Supplementary Table 3
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
