Abstract
Traumatic brain injury (TBI) remains a leading cause of death and disability worldwide. Motivations for outcome data collection in TBI are threefold: to improve patient outcomes, to facilitate research, and to provide the means and methods for wider injury surveillance. Such data play a pivotal role in population health, and ways to increase the reliability of data collection following TBI should be pursued. As a result, technology-aided follow-up of patients with neurotrauma is on the rise; there is, therefore, a need to describe how such technologies have been used. A scoping review was conducted and reported using the PRISMA extension (PRISMA-ScR). Five electronic databases (Embase, MEDLINE, Global Health, PsycInfo, and Scopus) were searched systematically using keywords derived from the concepts of “telemedicine,” “TBI,” “outcome assessment,” and “patient-generated health data.” Forty studies described follow-up technologies (FUTs) utilizing telephones (52.5%, n = 21), short message service (SMS; 10%, n = 4), smartphones (22.5%, n = 9), videoconferencing (10%, n = 4), digital assistants (2.5%, n = 1), and custom devices (2.5%, n = 1) among cohorts of patients with TBI of varying injury severity. Where reported, clinical facilitators, remote follow-up timing and intervals between sessions, synchronicity of follow-up instances, proxy involvement, outcome measures utilized, and technology evaluation efforts are described. FUTs can aid more temporally sensitive assessments and capture fluctuating sequelae, a benefit of particular relevance to TBI cohorts. However, the evidence base surrounding FUTs remains in its infancy, particularly with respect to large samples, low- and middle-income patient cohorts, and the validation of outcome measures for deployment via such remote technology.
Introduction
Traumatic brain injury (TBI) is a concern in both public and global health, and is a leading cause of death and disability worldwide. 1,2 Secondary deficits from TBI manifest in multiple ways, often with long-term symptoms in physical, cognitive, and emotional domains, 3 –5 which have a collateral impact, both direct and indirect, on patients, families, and wider society. 2,3,6,7 The purpose of outcome data collection is threefold: to improve patient outcomes, to facilitate research, and to provide the means and methods for wider injury surveillance.
Each purpose may vary in the fidelity of the data sought—injury surveillance efforts may typically aim to garner general morbidity or mortality on a wider scale, whereas research initiatives may use refined, detailed assessment batteries centered upon the phenomena or sequelae in question. In clinical practice, we may seek a balance of both—employing more detailed outcome measures and assessments as required, while also addressing any ongoing sequelae and determining the general status and well-being of the patient.
In 2004, the World Health Organization (WHO) released Guidelines for Essential Trauma Care emphasizing the importance of surveillance data to reduce the global burden of death and disability from injuries. 8 In addition to enabling clinical teams to determine the full extent of physical, mental, and socioeconomic sequalae post-injury, 5,9 –11 accurate data also facilitate the evaluation of systems and services, including: evaluating the efficacy of patient treatment and management decisions 10,11 ; identifying targets of wider systems improvement in injury prevention 12 ; enabling continuous quality improvement projects and trials; enabling the formation of registries that may themselves be incorporated into care pathways, injury prevention strategies, and policies 13,14 ; and lastly, facilitating rehabilitation of an individual and improving rehabilitation pathways and services.
Despite improvements in injury surveillance data, data on disability and long-term functional outcomes remain poorly recorded in both high-income countries (HICs) and low- and middle-income countries (LMICs). 5,10,15,16 However, given the heterogeneity both within and between LMICs, the collection of outcome data is considered more complex 1,10,17 and as such is often limited to collection at hospital discharge only. 10 Despite these limitations very little has been published on the challenges faced in facilitating long-term follow-up and collection of outcome data in LMICs. Of the research that does exists, efforts in data collection at the clinical level were found to be complicated by factors such as weak health care and long-term support infrastructure, resulting in a lack of regular follow-up of patients with trauma. 10
In 2007 the world's population became more urban than rural for the first time. 18 However, in many countries, and in particular LMICs, vast numbers remain in rural settings, and with this, have limited access to general health services. In the context of specialist services such as neurosurgery, which remain heavily centralized to urban settings, clinicians often have limited or no regular access to patients after discharge. 19 –21 Patients who are able to access neurosurgical follow-up often have to travel extensive distances at great personal cost. 22,23 Those unable to access neurosurgical follow-up become lost to follow-up. 24,25
Over the last few decades, there has been rapid advancements in technology, especially regarding telecommunications and its widespread adoption. According to a United Nations (UN) International Telecommunication Union (ITU) 2020 report, 26 47% of households worldwide are estimated to have access to a computer, with 57% of households perceived to have internet access. Mobile phone and cellular network technologies, however, are arguably the “common-denominator” technology worldwide. An estimated 75 per 100 of the world's population are thought to have an active mobile broadband connection, and it is estimated that there are 105 mobile-cellular telephone subscriptions per 100 population.
Similarly, short message service (SMS) technologies, initially made possible by second-generation or “2G” cellular network technology, remains a valuable opportunity for outcome data collection owing to its worldwide penetration and strong coverage, even in an LMIC setting where it is estimated that over 95% of the population have 2G coverage. 27 The adoption of next-generation network architecture, such as fifth generation (5G) cellular technology, can be expected to lead to exciting new possibilities for mobile health assessment owing to its ability to provide accessible, high-speed streaming capabilities 28 for use in high-definition remote video assessments.
Technologies are becoming frequently adapted to harness unique opportunities to connect patients with their health providers upon discharge. These follow-up technologies (FUTs) may provide innovative solutions that mitigate those deemed “lost to follow-up,” fill quantitative gaps in TBI epidemiology, and enable those in the remotest corners of the world to access specialist care.
Prior engagement with the literature on FUTs revealed a complex and diverse evidence base, and no prior attempt to synthesize this substantial body of work. A scoping review was therefore deemed an appropriate methodology to describe the type, characteristics, and size of the evidence in this field. The objective of this review was to identify follow-up technologies, such as telephone- and SMS-based service, videoconferencing, and smartphone applications, implemented across global settings in TBI. In addition, this review aimed to characterize the outcome measures administered and the data collected, among the communication modalities used, and briefly highlight the success of each in context with the patient populations and settings they have been implemented in.
Methods
The reporting of this study is in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for scoping reviews (PRISMA-ScR), 29 with an additional study screening and selection flowchart (Fig. 1) as recommended by the Joanna Briggs Institute (JBI) and PRISMA-ScR reporting guidelines. 29 –31 Unlike systematic reviews, scoping reviews do not require an antecedent protocol registration, 32 notwithstanding, review objectives, eligibility criteria, and preliminary study characteristics to be extracted were determined a priori.
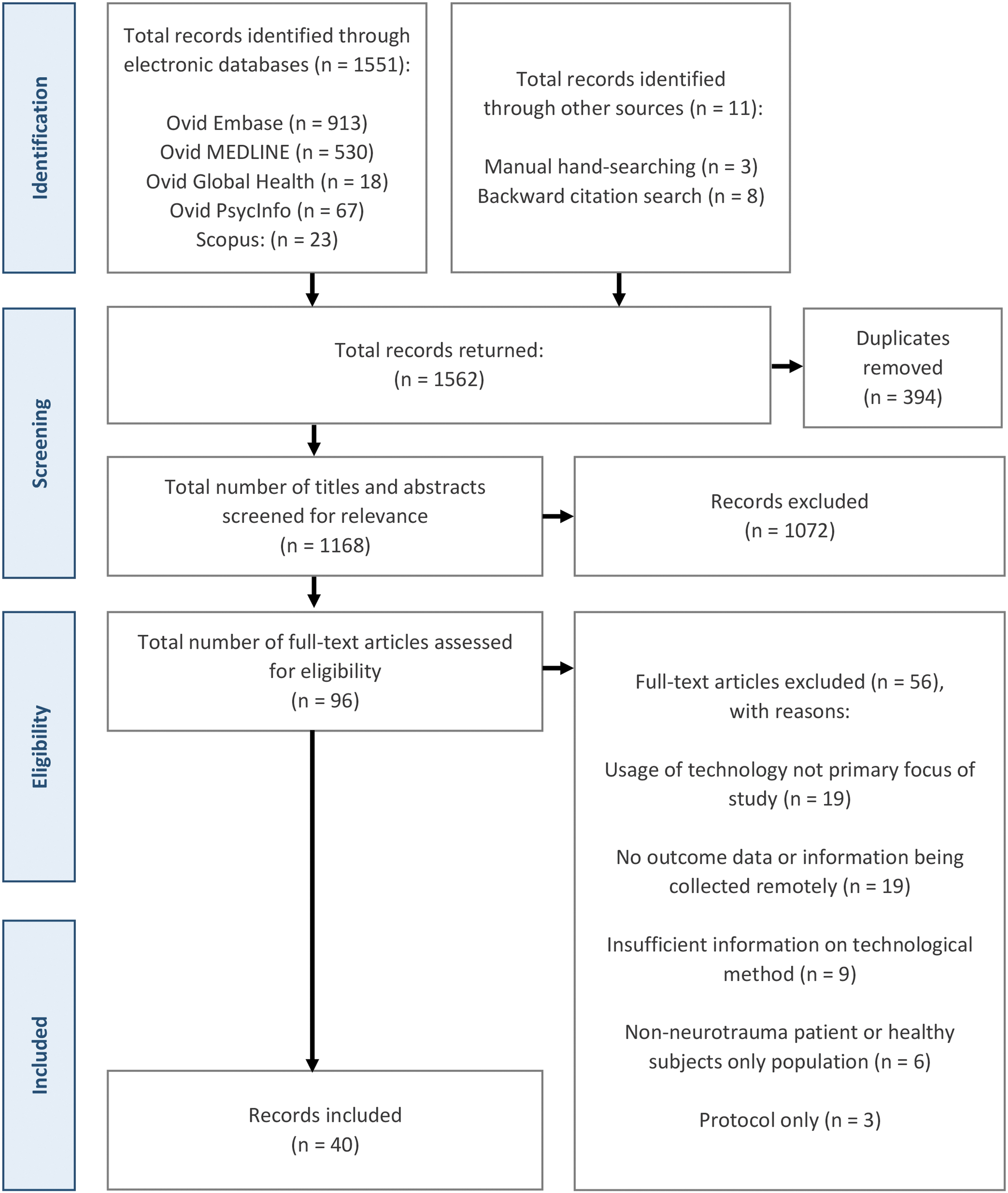
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flowchart detailing the study selection process.
Our scoping review was guided by the comprehensive scoping review framework by Arksey and O'Malley, 33 with minor refinements, including those suggested by Levac and colleagues 34 and the JBI. 30 This framework includes: (1) identifying the research question; (2) identifying relevant studies; (3) study selection; (4) charting the data; (5) collating, summarizing, and reporting the results; and an optional (6) consultation exercise. This review serves as part of a wider program of work in which stakeholders (neurotrauma physicians) are formally engaged in a separate qualitative research process, examining their views, experiences, and opinions of FUTs as applied to post-TBI care provision. Prior to commencing the review, we shared similar perceptions to those of Levac and colleagues 34 regarding the challenges of conducting and integrating the results of stakeholder consultation within a review's findings. In light of this, and given the potential of qualitative inquiry to empower rich and in-depth investigations of the human experience, 35 stage 6 was not carried out within this review. Notwithstanding, the lead author (BGS) discussed findings with senior co-authors, who by their nature as LMIC neurosurgeons are stakeholders within this context.
Research question
What research has been conducted to describe, examine, or assess the use of follow-up technologies in traumatic brain injury cohorts worldwide?
To answer this question, the following sub-questions were posed: What technologies are being used as FUTs in a global setting? In what settings is FUT research being conducted? What patient cohorts (demographics, injury severity) have been included in FUT research? What factors may constitute successful implementation of FUTs? What, if any, validated outcome measures are being deployed via FUTs?
Search strategy
The final search strategy was determined with the assistance of an academic medical librarian following a consultation to derive keywords based on the review objective and concepts of “telemedicine,” “traumatic brain injury,” “outcome assessment,” and “patient-generated health data.” The authors selected a number of “indicator papers”—predefined articles that one would expect to appear in their final search results—to test the quality and robustness of the search strategy. Several pilot searches were attempted before a final strategy was established and translated across a number of databases. The search strategy was executed on the October 1, 2021 on the following electronic databases: OVID Embase, OVID MEDLINE, OVID Global Health, OVID PsycInfo, and Scopus. An example search strategy for OVID MEDLINE can be found in Supplementary Table S2. These databases were selected owing to their sufficient coverage given the multi-disciplinary nature of TBI, outcomes, and their assessment. A limited manual search was conducted on Google Scholar and a number of technology- and head-injury focused journals (Journal of Neurotrauma, The Journal of Head Trauma Rehabilitation, World Neurosurgery, NEUROSURGERY, Journal of Medical Internet Research, Journal of Telemedicine and Telecare).
Following a pre-protocol pilot gray literature search, it became apparent that, of the limited materials retrieved, most were unsuitable for a number of reasons, including: insufficient indication of external peer review (of particular importance in commercial reports); a lack of FUT description or elucidation of the methods of their use; and description or evaluation of the FUT was not perceived to be the primary focus of the resource. Compounded by the resource- and time-intensive nature of conducting a gray literature search in this context, gray literature sources were omitted in the final search strategy. Finally, backward citation searching was undertaken, whereby the reference lists of articles deemed eligible for inclusion, and review articles that were not eligible for inclusion, were screened for relevant studies. Searching for additional sources was completed on April 25, 2022.
Eligibility criteria
Scientific articles reporting original research of the application of technology written in English were included. All databases were searched from inception to achieve the largest scope possible and to detail early innovations in this field.
The Population/Participants, Concept & Context (PCC) framework 36,37 was used to inform our inclusion criteria, search, and data charting strategies.
Eligibility criteria for articles to be included in this review were: (1) any published original research, including: primary studies, reports, editorials, opinion articles, letters, conference abstracts, theses, and book chapters; (2) reports with a primary aim to describe, assess, or examine the use of FUTs to facilitate remote collection of patient outcome data; and (3) adult and pediatric all-severity cohorts of TBI patients. Articles were excluded if they were (1) study protocols or secondary research (reviews); (2) reports describing the collection of family or caregiver outcomes only; or (3) no TBI population or involved healthy volunteers only.
Population/Participants
Studies were only eligible for inclusion in this review if their primary aim related to the development, implementation, or validation of technologies contributing to the provision of follow-up care of discharged patients following TBI of any severity, whether directly or via proxy (family members/relatives, caregivers, and guardians). Studies of mixed-pathology cohorts were included.
Concept
In this review, we defined follow-up technology as any system, device, equipment, component, or machinery used to both transmit and receive digital information electronically between a remote outpatient or their proxy and a member of their clinical team. The focus of this exchange was to attain data from the patient as to their current welfare status in the form of either patient-generated health data (PGHD), that is, self-reported, or clinician-derived health data (CDHD), that is, garnered through clinician-led assessment or interview, either from the patient or their nominated proxy. In addition, we defined “remote” as, at the time of information exchange, the patient was in a community-based setting (e.g., home, regional care provider such as a general practitioner or regional hospital, or other public settings) at a distance from the clinical team managing their follow-up.
Herein, we refer to synchronicity as the temporal aspect of the encounter between clinician and patient. Synchronous FUTs function to collect data in real time, often allowing direct contact between clinician and patient through sensor-, text-, voice-, or video-based technology. Asynchronous FUTs collect data by store-and-forward techniques—data are gathered, stored, and transmitted for later review by the clinician at two independent time-points; that is, they do not interact in real time. 38
Finally, we define “follow-up” as any attempt to monitor, assess, communicate, or liaise with a patient, or their proxy, from the point of hospital discharge, for the benefit of furthering their health and well-being, research, or injury surveillance.
Context
FUTs utilized in any global health setting
We utilized HIC and LMIC classifications as defined by the 2021–2022 World Bank list of economies. 39
Study selection
A two-stage screening process was followed. All search results were initially imported into the Zotero (Corporation for Digital Scholarship, Virginia, USA) reference manager for title and abstract review. Where necessary, duplicates were removed manually. Two researchers (BGS and ST) independently screened all titles and abstracts, and potentially eligible studies were identified for full-text review.
Disagreements arising from the selection process were either resolved by consensus, or where this was not possible, a third reviewer (OM) was consulted for resolution. Following preliminary screening, the remaining articles were exported to Microsoft® Excel (Microsoft, Redmond, Washington, USA), where full texts were independently screened by two researchers (BGS and ST) for final eligibility; a third researcher (OM) was consulted for disagreements as required. Selected studies formed the final repository of evidence for subsequent data extraction (charting) and collation.
Data charting and synthesis of results
Owing to the exploratory nature of a scoping review, a precursor proforma was developed to facilitate data extraction. Within this document, initial elements of interest that sought to answer our research question were informed by our PCC framework and agreed upon by researcher consensus (BGS, ST, CJW, AGK). The proforma was subdivided into five key sections, including: authorship and study characteristics, description of neurotrauma, characteristics of FUT(s) utilized, and major findings and challenges. This was iteratively updated and refined as the charting process progressed, adding to pre-identified elements of interest (see Supplementary Table S1). Data charting was conducted independently for all articles by two researchers (BGS and ST). Following the charting of the first five studies, in line with recommendations by Levac and colleagues, 34 the authors reconvened to ensure proforma suitability in addressing the research question, and to advance the proforma following familiarization with this initial subset of studies. Final completed proformas were cross-checked for conformity, and a third researcher (OM) was consulted as necessary in cases of a dispute. No formal critical appraisal, or quality of evidence assessment, was conducted as it would fall beyond the remit of a scoping review. 40 Following data charting, a narrative summary of included articles was constructed in relation to the review's overarching question and sub-questions.
Results
Characteristics of included studies
Executing the search strategy across the five electronic databases yielded a total of 1562 potentially eligible citations. Following de-duplication, 1168 unique articles remained, with a subsequent title and abstract review delineating a pool of 96 citations for further full-text review. This final stage of screening concluded with 40 articles for inclusion; inclusive of 11 citations discovered through manual and citation searches. A full PRISMA-ScR flowchart for the study search, selection, and exclusion process is depicted in Figure 1.
Of the 40 articles retrieved, the plurality (n = 15) were reported or deemed to be descriptive in design, 19,41 –54 and included small, non-randomized pilot studies and secondary analyses of data, or in two studies, were conducted in or described the retrospective analysis of a quality improvement initiative format. 52,53 Citations with an observational design formed the second most common type (n = 10), 10,55 –63 encompassing prospective 55,56,58 and retrospective 10 cohort studies and cross-sectional studies. 57,60,61 Research of experimental (n = 9) 64 –72 and quasi-experimental (n = 6) 73 –78 design was similar in frequency. Experimental designs included single-center 66,68 –71 and international multi-center 65,67 randomized trial designs, among non-randomized, open-label trials. 64,72 Results of these studies were published primarily as original research articles (n = 31), 10,19,41 –44,48 –53,55,56,59,61 –75,78 with a lesser number as conference abstracts or research posters (n = 8), 45 –47,57,58,60,76,77 and research summary letters (n = 1). 54
International context
The adaption of technologies for follow-up delivery has been used in multiple settings throughout the world. The majority of articles reported on FUTs in HICs (n = 34, 85%), including the United States (n = 24), 41 –43,51 –61,63,64,66,67,72 –74,76 –78 Australia (n = 3), 70,71,75 Canada (n = 3), 45,47,68 the Netherlands (n = 1), 62 Ireland (n = 2) 46,50 and a joint endeavor between Italy, Spain, and Belgium (n = 1). 65 Whereas only six (15%) reported findings from studies in LMICs including Uganda (n = 2), 19,49 Ethiopia (n = 1), 10 India (n = 1), 44 Iran (n = 1), 69 and Indonesia (n = 1). 48,79
Patient population demographics and TBI characteristics
The majority of articles (n = 27, 67.5%) described civilian adult population cohorts (>18 years of age), 10,41,42,44 –48,50 –52,55,57 –60,62,65,66,69 –71,73 –75,77,79 from sample statistics reported representing 3442 patients. A further five studies described military or veteran cohorts 43,61,64,67,76 representing an additional 207 adult patients. Five studies reported exclusively pediatric patient cohorts, 19,54,63,68,72 representing 287 patients. Three studies investigated mixed adult and pediatric cohorts, 49,56,78 adding a further 774 patients to previous approximations. Two studies did not report the demographics of the cohorts investigated. 44,53
With respect to TBI severity, FUTs were most frequently implemented in cohorts of patients with TBI of undefined severity (n = 14) 10,44,45,49,52,53,55,58,62,65,67,70,71,77 —in some of these cases, patient cohorts were pooled with other diagnoses (trauma, spinal cord injury, stroke, orthopedic, acquired brain injury, among other neurological conditions). Where TBI severity was defined, seven studies (n = 7) explored the implementation of FUTs in all-severity TBI patient cohorts. 19,41,48,51,73,74,76 In studies recruiting patients with particular injury severities, concussion or mild TBI formed the majority (n = 12), 43,46,50,54,56,59,61,63,66,68,72,78 whereas only one study investigated FUTs as applied to a cohort of patients with severe TBI exclusively. 75 No studies reported moderate TBI cohorts exclusively. Of the remaining studies, two reported FUTs for mild to moderate TBI cohorts, 47,69 and four for moderate to severe TBI cohorts. 42,57,60,64 Further, two studies described their TBI cohort as chronic. 51,58
Almost all (n = 37) studies used follow-up technologies while the patient was at home or in another non-health setting in the community. The remaining studies (n = 3) used technology while the patient was at other clinical settings away from the team responsible for follow-up assessment, including a polytrauma rehabilitation center in one study, 76 and a hospital research laboratory 15 km from the assessing clinician 70,71 in two linked studies by the same author team.
Technology modality
Remote FUTs were grouped by their underlying modality: telephone, SMS, smartphone (e.g., mobile application), videoconference, and “miscellaneous”—namely a personal digital assistant (PDA) and custom touchscreen device—each demonstrated in one study respectively. Telephone-based FUTs (52.5%) were the first to appear for use with cohorts of patients with TBI in 1997, 41 and remained the modal technology utilized at the time of search strategy execution. Videoconference-based technologies (10%) were next to appear a decade later in 2008 as part of a multi-center randomized trial. 65 A single study exploring PDA (2.5%) as a remote FUT for ecological momentary assessment (EMA) in a pediatric concussion cohort followed shortly after in 2009. 63 SMS-based FUTs (10%) followed in 2012, forming the asynchronous communication element of a pilot EMA study in the United States. 43 Smartphone-based FUTs (22.5%) were introduced in 2015. Lastly, a single study 62 explored the use of a custom electronic touchscreen device, the PsyMate, in 2019 to investigate the feasibility of EMA to explore the interactions between person, environment, and effect in an acquired brain injury (ABI) cohort. Figure 2 demonstrates the technology modalities implemented over time.
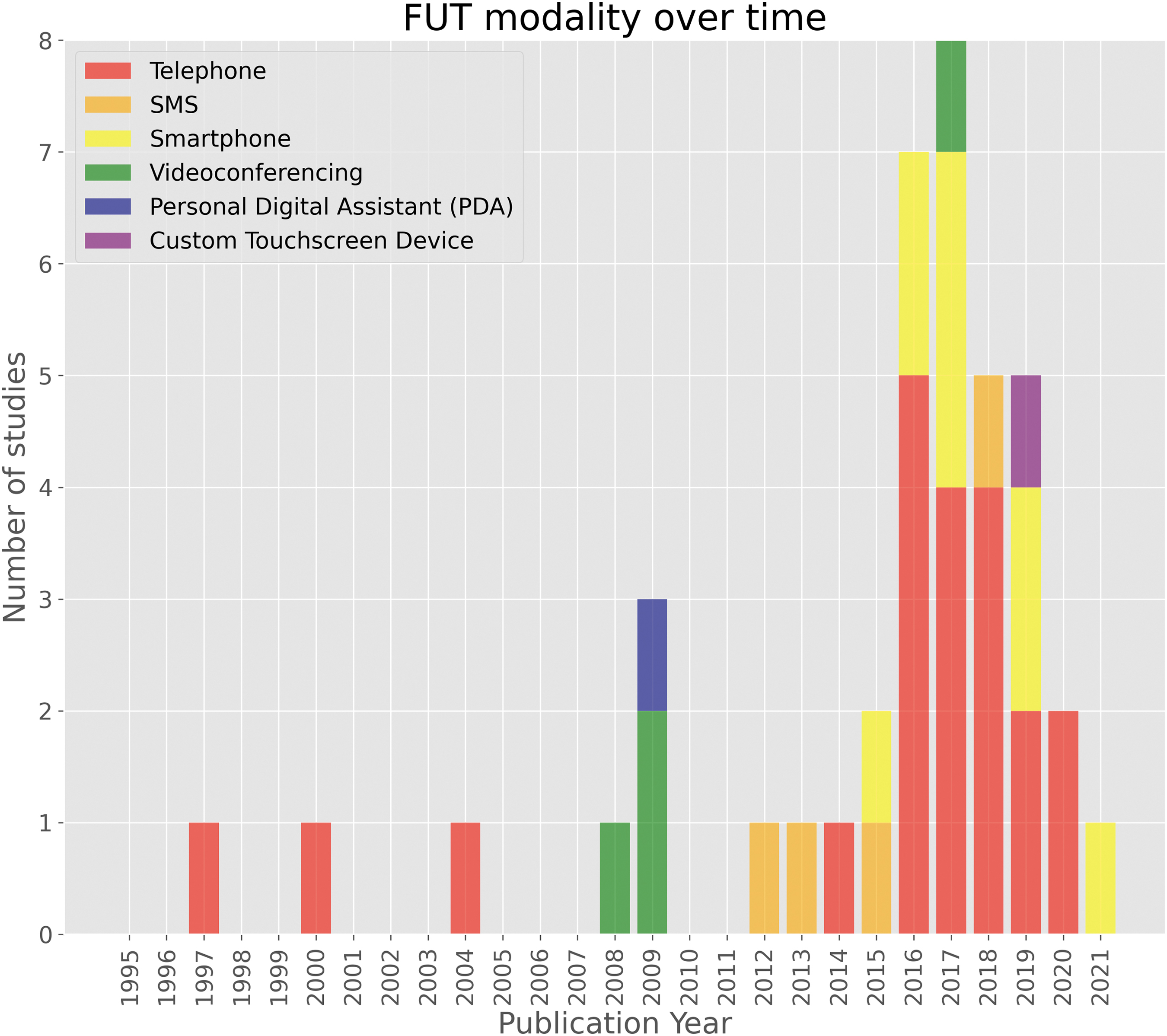
Follow-up technology modality over time. PDA, personal digital assistant; SMS, short message service.
Telephone-based follow-up
Telephone-based follow-up technology (Table 1) was used in 21 studies (52.5%). 10,19,41,42,44 –50,52,53,55,57,60,64,68,69,74,76 Telephones were primarily used as a synchronous means of two-way communication to conduct structured interviews with patients and their proxies. Although almost all studies, where defined, used clinical or research staff to conduct follow-up, one used an external call center that had integrated its systems with the hospital's electronic medical records 44 to complete the follow-up interview. Another study 55 had no human facilitator in the administration of telephone-based follow-up, and instead used an asynchronous and interactive voice response system (IVR), whereby pre-recorded questions were played to the patient, and either a voice or keypad could be used to respond. One study that used scripted, structured telephonic follow-up described the additional use of a secure web-based data capture platform (REDCap) with branching logic to conduct the interview. 52 Lastly, one study reported the use of a computer-assisted telephone interviewing system, whereby the assessor could follow a script in the collection of data, enabling an assessor without familiarity with TBI to conduct the interview. 74
Citations Reporting the Use of Telephone-Based Follow-Up
Denotes research published in two parts.
[C], control; CVA, cerebrovascular accident; ED, emergency department; F, female; h, hours; min, minutes; GCS, Glasgow Coma Score; [I], intervention; LoC, loss of consciousness; RCT, random controlled trial; SD, standard deviation; TSI/D, time since injury or diagnosis; w/o, without; y, years.
SMS-based follow-up
The studies included demonstrated a multi-modal use of SMS-based technology (Table 2). Four studies demonstrated SMS exclusively as the means of data collection, often in an asynchronous and automated manner, through the delivery of timed symptom assessments to patients in the community, 43,56,59,66 whereby responses were often logged in a database for later review. Two studies, despite not using SMS directly in the collection of outcome data, utilized SMS to deliver prompts or updates. These text messages prompted the patient to log their current status and well-being on other systems such as a secure website or app-based portal. 67 In another study, SMS was used as a reminder system, informing patients to expect to receive shortly a telephone call, with a focus on improving telephonic response and attrition rates. 45
Citations Reporting the Use of SMS-Based Follow-Up
ED, emergency department; F, female; h, hours; min, minutes; PTSD, post-traumatic stress disorder; SD, standard deviation; TSI/D, time since injury or diagnosis; y, years.
Smartphone application-based follow-up
The second technology most frequently used in the remote collection of outcome data were mobile applications, or “apps,” installed on Apple and Android devices such as smartphones and tablets (Table 3) (n = 9). 51,54,58,61,67,72,73,77,78 Such implementations of mobile applications ranged from gamified symptom journals and social networking 72 to delivering questionnaires for EMA. 51,58,73 Three studies described the further use of the device's onboard sensors and additional functionality in the delivery and collection of data, including native “push notifications” to prompt patients to input data, 78 the GPS tracking function in the collection of activity and community participation data, 61 or in one study, the Apple iPod Touch's in-built accelerometer to capture objective measurements of physical activity. 54
Citations Reporting the Use of Smartphone-Based Folllow-Up
This was a secondary analysis of a previous pilot feasibility study, a73, and is included as a separate entry for totality.
CI, confidence interval; F, female; h, hours; min, minutes; OR, odds ratio; SD, standard deviation; TSI/D, time since injury or diagnosis; y, years.
Videoconferencing-based follow-up
Three studies utilized videoconferencing exclusively for remote assessment (Table 4). 70,71,75 Two studies, by the same authorship group, described the use by the assessing speech-language pathologist of two remote-controlled robotic web cameras. 70,71 Additionally, in their second article with a system re-design, the authors describe concurrent automatic store-and-forward facilities integrated into their system, enabling video and audio data of higher quality than that streamed over the 128kbit/sec videoconference connection to be sent to the assessing clinician for later review. 71 Further, one study reported the use of a novel, custom, portable home care activity desk (HCAD) installed in the patient's home. Each unit consisted of sensorized tools and videoconferencing facilities, providing store-and-forward capability between the patient's home and hospital servers. 65 One study additionally described the use of telephone as a backup option utilized in cases of videoconferencing technical difficulty. 75
Citations Reporting the Use of Videoconference-Based Follow-Up
Studies conducted by the same authors; bstudy included separately due to revised methods and a novel patient cohort.
[C], control; CVA, cerebrovascular accident; F, female; h, hours; [I], intervention; min, minutes; MS, multiple sclerosis; SD, standard deviation; TSI/D, time since injury or diagnosis; y, years.
Miscellaneous technologies for follow-up
Two studies, pooled together as “miscellaneous” (Table 5), described the use of portable touchscreen electronic devices, one proprietary device (PsyMate) 62 and one commercially available PDA device. 63 Both studies described auditory prompt capabilities with their devices, enabling semi-randomly scheduled EMA throughout the day. 62,63 One study previously noted described allowing the patient to choose between a secure Internet website portal or telephonic IVR to submit self-reported outcome data. 55
Citations Reporting the Use of Miscellaneous Technology-Based Follow-Up
F, female; SD, standard deviation.
Clinical facilitators
Clinical facilitators were most often explicitly described in the article as the research staff (i.e., research coordinators and care managers, research assistants, and clinical researchers) 19,42,49,52,59,61,72,73,75 or hospital outcome management staff 55 and blinded examiners. 66 Nursing personnel 41,52,69 formed the next largest pool of facilitators, and included specialist psychiatric nurses, 64 rehab-based nurse practitioners, 47 or neurosurgical nurse personnel experienced in TBI and/or neurotrauma outcome measure administration. 48,53,80 Undefined clinical staff, 43 speech-language pathologists, 70,71 physicians, 52 general therapists, 65 occupational therapists, 68 and care team members 67 were described as facilitators in a smaller subset of studies. In three studies, facilitators were primarily external non-clinical staff without familiarity of TBI, such as data clerks, 10 call center personnel, 44 or interviewers otherwise undefined. 74 However, 15 studies did not explicitly report on who facilitated the follow-up reported in the article. 45,46,50,51,54,56 –58,60,62,63,76 –78,81 Although it may be assumed that the facilitators of technology in these studies were the author teams themselves, this cannot be confirmed.
Despite not being described as active facilitators, it is of note that in two studies, family members, friends, and relative facilitators were incorporated in the delivery of the follow-up technology, 43,72 and instead may be designated as “passive facilitators.” One study with a pediatric population describes a “social networking” function built in to the smartphone application, allowing friends and family to connect with and receive notifications of the patient's activities and progress. 72 An additional study describes how designated friends or relatives could opt in to receive notifications should their injured relative fail to maintain contact with the FUT services, or if they returned a score below a pre-set threshold that warranted further one-to-one contact outside of the FUT. 43
Timing of follow-up and time since injury, diagnosis, or discharge
The timing of remote follow-up delivery with respect to the patient's time since injury/diagnosis (TSI/D), or time since hospital discharge, could be established directly or approximated in 77.5% (n = 31) of articles retrieved. Studies were broadly grouped together by respective timeframes of: less than 1 month, 1–3 months, 3–6 months, 6–12 months, and 12 months or more. Eight (20%) studies reported remote assessment within a month of discharge or injury; 42,45,47,54,59,66,68,78 within this group, five studies 54,59,66,68,78 described recruitment and assessment of participants from discharge up to 2 weeks post-discharge for the assessment of concussion or mild TBI. Three (7.5%) studies 41,48,64 assessed patients between 1- and 3-months post-injury or discharge, and all utilized telephone as the modality of choice. Five (12.5%) studies 10,52,63,69,76 conducted remote assessment beyond 3 months and within 6 months of discharge or injury. Four studies (10%) described assessment between 6 and 12 months. 46,58,70,72 Lastly, 27.5% (n = 11) of studies 19,49 –51,57,60,65,71,73 –75 depicted an average time-point of remote assessment of 1 year and beyond hospital discharge or injury. Nine studies (22.5%) did not formally define the time-point at which remote assessment was attempted. 43,44,53,55,56,61,62,67,77
Intervals between sessions
The most frequently reported (n = 14) timeframe of outcome data collection by FUT was at one single time-point following injury or discharge. 10,19,44,46,47,49,50,52,55,60,70,71,74,75 Of these studies, 11 (n = 11, 78.6%) utilized telephone-based technology. 10,19,44,46,47,49,50,52,55,60,74 One study used two modalities in a single instance of follow-up, namely a secure web-portal and telephone-based IVR system. 55 Three studies used videoconferencing in a single instance. 70,71,75
The remaining studies (n = 26, 65%) reported more than one outcome data collection point. One study reported follow-up at yearly intervals for 2 years post-injury. 57 Another study reported two follow-up points at quarterly intervals of 3- and 6-months post-injury. 41 One study described data collection on a monthly basis up to 3 months following discharge. 48 Similarly, a further study described fortnightly instances of follow-up by telephone over the course of 4 weeks. 45 Three studies reported collecting outcomes on a weekly basis 43,64,69 —two of which were by telephone 64,69 —and the first use of SMS is seen at this weekly interval. 43 In those studies using follow-up technology on a daily basis, eight studies utilized smartphone applications 51,54,58,61,67,72,73 and one employed a videoconference-based, sensorized HCAD previously described. 65
Seven studies reported collecting data multiple times throughout the day 56,59,62,63,66,77,78 ; this session interval also used the most diverse range of technologies of the intervals described thus far. Three studies explored the use of SMS up to five times daily, 56,59,66 and two studies demonstrated the use of smartphone applications up to four times daily. 77,78 The remaining studies used a PDA 63 and custom touchscreen device 62 to examine outcomes up to 10 times daily. Lastly, three studies employing telephone as an FUT collected data at steadily increasing intervals up to 9 months post-injury. 42,68,76 One additional study, owing to the nature of the telephone-based service (a neurotrauma hotline), was not able to define an interval between sessions; however, it reported an average of 3.3 calls per day over a 12-month period. 53 A visual summary of the intervals between FUT sessions with respect to technology modality can be found in Figure 3.
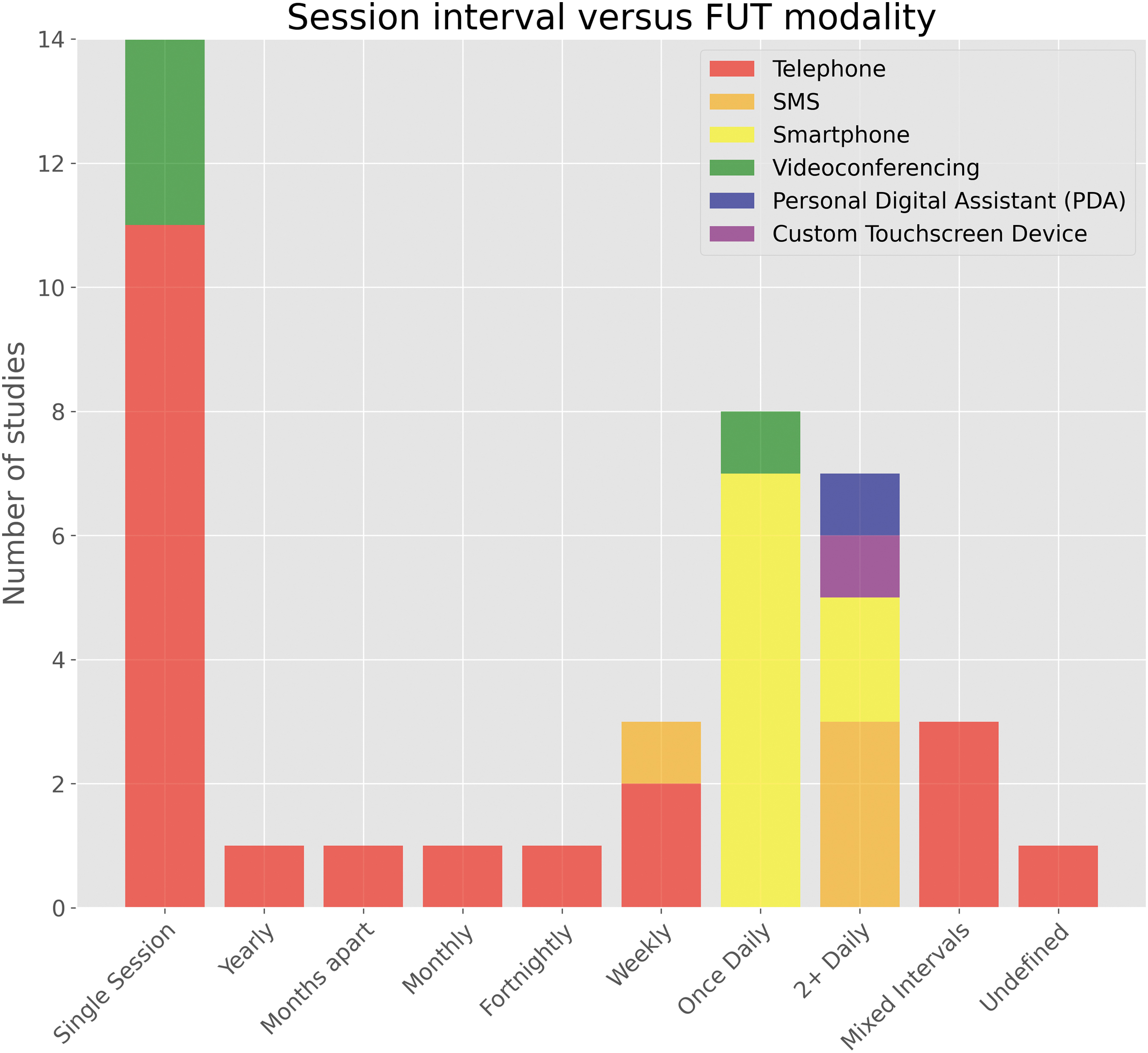
Intervals between remote follow-up sessions with respect to follow-up technology modality. SMS, short message service.
Synchronicity
Half of the studies utilized synchronous or real-time assessment in the collection of outcome data (n = 20, 50%). 10,19,41,42,44 –50,52,57,60,64,68,69,74 –76 Sixteen (40%) used an asynchronous or store-and-forward approach. 43,51,54 –56,58,59,61 –63,66,67,72,73,77,78 Four studies (10%) demonstrated the use of mixed synchronicity, 53,65,70,71 often utilizing different capabilities of the technology in its service delivery, such as voicemail (asynchronous) services built in to a neurotrauma hotline (synchronous). 53
Use of proxies
In 29 studies, there was no reported use of proxies (72.5%). 41,43 –47,50,51,54 –63,65 –67,70 –74,76 –78 Eight studies (20%) were extended to both patients or their proxies in remote follow-up. 10,42,48,49,52,53,64,75 Of these studies, two (5%) used a mixture of both patient and proxy data, usually collected in the same sitting, to assess the patient's well-being. 42,75 A further two studies described the assistive role of proxies where the patient was unable to directly report their well-being themselves. 10,52
Two studies (5%) described the role of proxies in assisting initiating contact with the patient if they were initially unable to be reached, although did not further disclose whether the proxies were used to seek information on behalf of the patient. 48,49 One study, in addition to assisting the patient to report their well-being, used proxies to report data pertaining to mortality as necessary. 52 One study, 43 despite not using a proxy to ascertain data regarding the status of health of the patient on their behalf, utilized a clinical staff or “buddy” contact system of friends and family to facilitate further check-in with the patient, should they not respond to the asynchronous technology-based well-being report, or not meet a threshold score indicating they were otherwise well.
Three studies (7.5%) 53,64,69 described telephone-based support made available for patients and their proxies, 53,64 and their proxies exclusively, 69 to share symptoms or well-being status (on behalf of the patient where required), or to seek support at their own convenience, in between scheduled requests or prompts for patient status reports. 64,69
Three studies (7.5%) described the use of proxies exclusively to assess patients in their FUT deployment, without directly communicating with the patients themselves. 19,68,69 Pediatric patient cohorts formed the basis of two of these studies, 19,68 whereby follow-up and outcome data collection was solely provided by the patient's caregivers owing to the patient's age.
Deployment of outcome measures
Several studies utilized one or more validated outcome measures in their technologies, whereas a small number of studies used internally developed measures, scores, and scales in the remote assessment of patients. The Glasgow Outcome Scale-Extended (GOS-E) was the most frequently used in the studies included (n = 6), followed by the Rivermead Post-Concussion Questionnaire or its derivatives (n = 5), and the 5-Level EuroQol 5-Dimension instrument (n = 4). A full list of outcome measures deployed in FUTs for TBI, ordered by frequency, can be found in Table 6.
Outcome Measures Deployed via Follow-Up Technologies in TBI Populations
In addition, many studies used generalized questionnaires briefly ascertaining overall well-being 43,52,62,67 and fatigue, 51,58,62,73 presence of pain, 67 past and current medical concerns, 42 physical deficit or symptom checklists, 19,45,49,64 both generalized and specific (e.g., headache, irritability, depression, memory problems, medication compliance, and other miscellaneous complications and symptoms). A handful of studies deployed similar generalized questionnaires, yet with a focus on reporting-by-proxy through the patient's caregiver. 69
Where reported, these broader questions often existed either alongside or exclusive to generic questions encompassing: employment or return to work, 41,42,49,60 household and leisure activities, or activities of daily living, 19,41,49,54,64,68 mood, 42,67 energy and sleep status, 67 personal finance management, 41 relationship status, 67 subjective return to pre-injury or baseline status, 49 travel and location, 41,62 and lastly social context, and social activities and community participation. 41,62 –64 In pediatric populations, some generalized questionnaires sought information regarding the impact of injury or its sequelae on schooling and education, 49,68 and play activities. 68 One study with pediatric participants 54 described measurement of daily cognitive rest and exertion by calculating a composite score of number of text messages sent, minutes of screen time and gaming, and minutes of reading and schoolwork.
Other domains, inclusive of those aforementioned, were assessed by one study as part of a structured interview addressing 17 broad domains including personal care, ambulation, home management, leisure, alcohol and drugs use, legal issues, and spirituality. 42 One study 76 differed from those previously reported in deploying a neuropsychological assessment battery (including standard verbally administered tests of attention, memory, working memory, processing speed, language, executive skills, and auditory-verbal adaptions of trail-making). Another study 71 utilized informal oromotor and perpetual speech assessments as part of a wider speech-language battery. Weight status was included in one study with a longer duration of 36 weeks. 67
One study, instead of directly asking for a subjective measure of travel, activities, or social and community participation, collected GPS-based activity data (MOVES Storylines) to quantify this measure objectively and indirectly. 61 Similarly, another study 54 utilized the device's onboard accelerometer to quantify step count as part of activity monitoring. A minority of studies sought to assess the access of further care as part of their technology-based follow-up assessment 19,49,69 —studies seeking this information were conducted solely in LMICs. One article describing the use of a telephone hotline for neurotrauma, by the nature of the technology differed greatly from others included by not reporting use of any outcome measures or other proforma for data collection. 53
Technology evaluation
Where technology was evaluated, most studies employed generalized questionnaires developed internally, and often used visual analog or 5- or 7-point Likert scales to gauge overall user satisfaction 61,62,65,69 –73,77 asking questions about acceptability, user friendliness, aesthetic, task difficulty, task appropriateness, and general impression of the technology. A smaller set of studies assessed technology feasibility 48,72,79 (such as retrospectively analyzing those eligible for FUT enrollment, those consented, and those who completed all instances of assessment, among reasons for loss of contact where realized) and compliance, 72,73,77 often which was calculated retrospectively rather than evaluated by the patient cohorts themselves, such as the use of a technology with respect to the investigator's target dose. 72 Further, two studies reported anecdotal or qualitative feedback pertaining to the patient's experience in using the technology. 48,79 One study asked participants to log daily unexpected, technology-related events such as errors as part of the evaluation process, and their perceived accuracy of the GPS-based activity data with respect to their actual activities. 61 Only one study utilized an externally validated assessment of telehealth services, the Telehealth Usability Questionnaire (TUQ). 73
Discussion
The purpose of this review was to describe the breadth of technologies implemented for follow-up, and highlight the instruments deployed with respect to their successes for all-severity TBI in a global setting. Forty-two studies were retrieved that utilized FUTs for symptom surveillance and outcome data collection and described technologies that fell under broad categories of telephone-, SMS-, smartphone-, videoconference-based technologies among a small number of miscellaneous devices that may fit under a number of these categories.
Smartphones are widely recognized to be both well-positioned and well-suited for emotional, 73 behavioral, 82 and physical monitoring, 83 particularly when applied to an EMA or experience sampling method (ESM) methodology. Traditional, face-to-face assessments conducted at infrequent intervals along the patient's journey of recovery rely upon retrospective self-reports that themselves are predisposed to recall bias, 84 to which patients with TBI are believed to be more susceptible. 85 This is further compounded by the numerous challenges patients with TBI face with cognitive impairment, 86 poor memory, 87 and impaired self-awareness. 88,89 Smartphone and other FUT-based remote assessment, as mirrored in this review, show promise to quantify symptoms more accurately, and with respect to their temporal variability, otherwise uncaptured at a single time-point, and further facilitated in one's own natural environment.
The United Nations General Assembly in 2015 highlighted the impact technology-enabled breakthroughs have had in the health care sector, enabling greater numbers of people to have access to services otherwise out of reach or unaffordable. 90 Although the implementation of technology to deliver follow-up is better than a complete absence of services, an informed understanding of the capabilities and technological fluency of the target population will be imperative for comprehensive and proper integration within standard practice. Several barriers to successful access of remote telehealth assessments and consultations have been identified in the literature and in recent WHO 2019 guidelines, 91 including: disability such as hearing or cognitive impairment, lack of equipment, poor networking access and speeds, lack of organizational support, difficulty using the systems, security or privacy concerns, and unfamiliarity with technology. 91 –94 One qualitative study exploring non-participant views of a wider telehealth and telecare trial depicted that some patients were hesitant to adopt new approaches where existing face-to-face services were often highly valued. 93 Patients with stigmatized health conditions may also possess additional concerns about the privacy of their information 91 when handled digitally. Developing an awareness of the challenges faced by populations, particularly those most vulnerable such as the elderly and those recovering from the sequelae of injury, will ensure progress toward digital equality of service access, and mitigate the risk of bias or inaccurate data being introduced into technology-enhanced trials, registries, and injury surveillance campaigns.
Although the studies included often quantify their successes by compliance, adherence, and response rates, among broad and crude evaluations of these novel services, it is worth highlighting that any communication fostered by these technologies outside of predefined (and often sparsely scheduled) outpatient clinic meetings was appreciated and welcomed by both pediatric and adult patients and their caregivers, reflected by satisfaction rates reported in the studies included, and in part by strong compliance and return rates in the majority of FUT implementations in TBI. To our knowledge, no studies reported the use of a requirements elicitation survey or equivalent exercise in the design and development of FUTs, and thus this would be welcomed in future research in this area when examining what factors should be addressed and built into these services to improve patient compliance and satisfaction.
Further, the “successes” of technology-based follow-up delivery (of which one may consider to be derived from compliance, attrition, or response metrics) do not appear to be reported in a standardized format across the studies retrieved. As a result, conducting systematic comparisons between technology modalities, and across cohorts of varying severity and demographics, remains a challenge. Although the majority of studies attempted to quantify the success of their technology (Tables 1–5), such as comparing response rates versus prompts delivered, or the number of patients reached after an arbitrary number of contact attempts, further research or initiatives addressing this gap would serve favorably.
The compliance and satisfaction of clinicians remains key in developing technologies for integration into standard practice. It is not a new notion that neurosurgical services across the globe encounter heavy workloads and large patient numbers, especially in LMIC settings, where a disproportionate volume of cases must be handled by smaller specialist workforces when compared with their HIC colleagues. 95 The benefits of asynchronous technologies thus become clear: enabling the clinician to review and act upon the data of their own accord, along with providing patients with the ability to self-report at their discretion, and at more frequent instances otherwise impossible to fulfill synchronously by clinical staff.
Wearable technologies such as smart watches, biometric monitors, and smart clothing may offer additional asynchronous sources of data and are slowly being introduced as medical technologies, although these were not identified in our review. Across medical disciplines, wearables have been demonstrated to enable real-time monitoring of vital signs, physical status, and physiological parameters as patients go about their daily lives. 96 Although the literature describing wearables for TBI-afflicted individuals remains sparse, 97 we clearly envision the role these technologies may play in enabling additional remote, data-driven approaches for post-TBI monitoring and early sequelae management. 98
Lastly, it is of note that due to the global prevalence and availability of mobile phone or telephone services, many studies screened often briefly stated in their reports the use of a telephone for follow-up practice; however, they excluded a deeper insight, evaluation, or formal assessment of the utility of the remote technology for conducting the act of said follow-up itself. In this respect, due diligence was exercised by the authors across the screening stages.
This scoping review serves as a foundation for the application of technology in follow-up and outcome data collection. Herein, we propose a number of recommendations for future research and practice.
First, from this review, there appears to be grounds for further research exploring, and perhaps refining, what outcome measures are deployed, and at what intervals assessments are conducted. Table 6 reveals a broad range of outcome measures used in the assessment of patients with TBI, some measures of which were deployed in multiples. To ensure technology functions optimally and simultaneously for both patients and clinicians, a balance must be struck between the richness of assessments and the efforts required on the patient's behalf in quantifying their health and well-being at a distance. Further, it would be beneficial for future research to assess the validity of outcome measures when delivered sequentially in a single instance, and namely whether delivering multiple outcome measures introduces confounding effects, including when delivered remotely and without the option of clinical assistance or clarification. Similarly, when they have been applied to FUT-led research it would be beneficial to explore and address the validities of outcome measures that were initially designed to be deployed on a face-to-face basis. We believe that there may be assumptions to challenge regarding whether a party perceived an outcome measure could be successfully delivered by a technological medium with ease, that the validities of such a measure must transfer simultaneously. As such, we encourage further efforts into FUT-led research practices themselves.
Second, a wide variety of outcome measures and instruments have been deployed by technology for remote assessment. Although beyond the scope of this review, it may be useful for further research to retrospectively address the validity of measures deployed, often designed for in-person use, with respect to their adaption for FUT deployment. Lastly, further investigation may be warranted to assess the effects on outcome measure validity in studies deploying multiple patient-reported outcome measures at single or close-together time-points. In addition, for this research investigators may wish to examine whether these instruments when delivered individually or as sets remotely can continue to accurately quantify outcomes from the acute to long-term stages of all-severity TBI and where these technologies may assist in facilitating data collection for common data elements as part of large-scale research efforts.
Strengths and limitations
To our knowledge, this review is among the first to map the current global evidence base of technologies deployed to augment traditional modes of in-person follow-up. A broad and comprehensive search across five electronic databases was conducted, and as such this review serves as a strong foundation for understanding the use-cases of technology-based follow-up for TBI in a global setting. Although the number of studies retrieved is relatively small considering our eligibility criteria (FUTs employed in a global context for all-severity TBI in pediatric and adult cohorts), we present a particular slice of the evidence base in which included articles have a primary focus of exploring FUT deployment.
In the context of future research, we trust this review will serve further as a comprehensive frame of reference for those wishing to apply FUTs in clinical practice or research. However, owing to a sheer number of terms describing global health, technologies, and their implementation, outcome measures, and follow-up, some published articles that may have met the predefined inclusion criteria could have been inadvertently excluded from this review. Further, we acknowledge limitations of our retrieved articles with respect to our eligibility criteria; we are aware of a number of large, high-profile injury surveillance studies, such as those utilizing the U.S. TBI Model Systems (TBIMS) database, 99 –102 that were not included, despite using FUTs in their research, as a description or assessment of FUTs was not the primary focus of such works. Similarly, by the nature of global health and technological reports, which may not always be confined to health journals and/or databases, or always depicted in the English language, we are aware of the limitations in the literature retrieved, and of the literature that may exist in alternate academic or commercial domains and mediums. Considering these limitations, and with an understanding of the parameters of FUTs afforded by this review, a further systematic review as the evidence base evolves, inclusive of gray literature, non-English publications, and articles utilizing FUTs but not necessarily as their primary aim, is recommended.
Conclusions
Our review has demonstrated that the evidence base surrounding FUTs remains in its infancy, particularly with respect to recruiting large patient cohorts, conducting a formal technology assessment, and the representation of research outside high-income settings, respectively. Of the use-cases described, incorporating technologies, both asynchronous and synchronous in nature, may leverage a clinician's abilities in gaining insights of the patient's well-being from discharge and beyond between traditional and often sparsely scheduled face-to-face appointments. FUTs may additionally serve to provide a more precise picture of the status of the patient with TBI through their ability to collect data at time-points closer in proximity to in-person follow-up, harmonious with the WHO's adjuration and promotion of outcome data collection and injury surveillance in the reduction of global TBI burden. Further systematic analyses may prove useful in empirically quantifying the utility, acceptability, feasibility, and costs of each technology modality in neurotrauma practice. Future research may wish to characterize the challenges of implementing, sustaining, and adhering to these novel systems from the perspectives of patients, their proxies, and physicians.
Footnotes
Acknowledgments
The views expressed are those of the author(s) and not necessarily those of the U.K. National Health Service (NHS), the National Institute for Health Research (NIHR), or the Department of Health and Social Care.
Authors' Contributions
The authors contributed as follows: Brandon G. Smith: conceptualization, methodology, formal analysis, data curation, writing—original draft; Stasa Tumpa: investigation, writing—review and editing; Orla Mantle: investigation; Charlotte J. Whiffin: methodology, writing—review and editing, supervision; Harry Mee: writing—review and editing; Davi J. Fontoura Solla: writing—review and editing; Wellingson Silva Paiva: writing—review and editing; Virginia F.J. Newcombe: writing—review and editing; Angelos G. Kolias: conceptualization, methodology, writing—review and editing, supervision; Peter J. Hutchinson: conceptualization, writing—review and editing, supervision.
Funding Information
The research was supported by the NIHR Brain Injury MedTech Co-Operative based at Cambridge University Hospitals NHS Foundation Trust and the University of Cambridge, and by the NIHR Global Health Research Group on Neurotrauma (grant number 16/137/105) using U.K. aid from the U.K. government.
Author Disclosure Statement
AGK and PJH are supported by the NIHR Cambridge Biomedical Research Center, the NIHR Brain Injury MedTech Co-Operative, and the NIHR Global Health Research Group on Neurotrauma. BGS and CJW are supported by the NIHR Global Health Research Group on Neurotrauma. PJH is also supported by an NIHR Research Professorship, and by the Royal College of Surgeons of England. The NIHR Global Health Research Group on Neurotrauma was commissioned by the U.K. NIHR using Official Development Assistance funding (project no. 16/137/105). VFJN is funded by an Academy of Medical Sciences/The Health Foundation Clinician Scientist Fellowship.
Supplementary Material
Supplementary Table S1
Supplementary Table S2
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
