Abstract
Magnetic resonance spectroscopy (MRS) is a non-invasive technique used to study metabolites in the brain. MRS findings in traumatic brain injury (TBI) and subconcussive hit literature have been mixed. The most common observation is a decrease in N-acetyl-aspartate (NAA), traditionally considered a marker of neuronal integrity. Other metabolites, however, such as creatine (Cr), choline (Cho), glutamate+glutamine (Glx) and myo-inositol (mI) have shown inconsistent changes in these populations. The objective of this systematic review and meta-analysis was to synthesize MRS literature in brain injury and explore factors (biological factors such as brain region, injury severity, time since injury, demographics and technical methodological factors such as field strength, acquisition parameters, analysis approach) that may contribute to differential findings. One hundred and thirty-eight studies met inclusion criteria for the systematic review and of those, 62 NAA, 24 Cr, 49 Cho, 18 Glx, and 21 mI studies met inclusion criteria for meta-analysis. A random effects model was used for meta-analyses with brain region as a subgroup for each of the five metabolites studied. Meta-regression was used to examine the influence of potential moderators including injury severity, time since injury, age, sex, tissue composition, and methodological factors. In this analysis of 1428 unique brain-injured subjects and 1132 controls, the corpus callosum was identified as a brain region highly susceptible to metabolite alteration. NAA was consistently decreased in TBI of all severities, but not in subconcussive hits. Cho and mI were found to be increased in moderate-to-severe TBI but not in mild TBI. Glx and Cr were largely unaffected, but did show alterations in certain conditions.
Introduction
Traumatic brain injury (TBI) is a heterogeneous injury with both immediate and long-term consequences. 1 TBI severity is often characterized as mild, moderate, or severe. 2 Common symptoms of mild TBI include a combination of headache, dizziness, sleep disturbance, cognitive impairment, fatigue, irritability, anxiety, and depression, 3,4 while more severe injuries typically present with additional complications such as coma, seizures, severe loss of coordination, behavioral changes, disorientation, and long-term physical and cognitive disability. 5 -7 The differentiation of severity is usually based on immediate clinical presentation, 8 duration of loss of consciousness, and post-traumatic amnesia, as well as findings on computed tomography and/or structural magnetic resonance imaging. 2 Although some differentiate concussion as a form of mild TBI, 9 these two terms are often used interchangeably, as will be the convention in this text. A final category of head trauma with increasing recognition is subconcussive hits, with the expectation that repetitive subconcussive hits have an additive effect on brain health, particularly in the long term. 10 –12
Research to best understand the full continuum of brain injury is expanding with increasing use of magnetic resonance imaging to understand brain injury as well as to predict outcomes and inform treatments. 13 –15 Included in this approach is magnetic resonance spectroscopy (MRS), which provides neurochemical information about brain tissue. Briefly, MRS uses a typical magnetic resonance imaging scanner to quantify concentrations of specific metabolites. On the spectrum, each metabolite has a unique chemical signature with peak(s) at specific frequencies used for metabolite identification. The area under the peak(s) is proportional to the concentration of a particular metabolite.
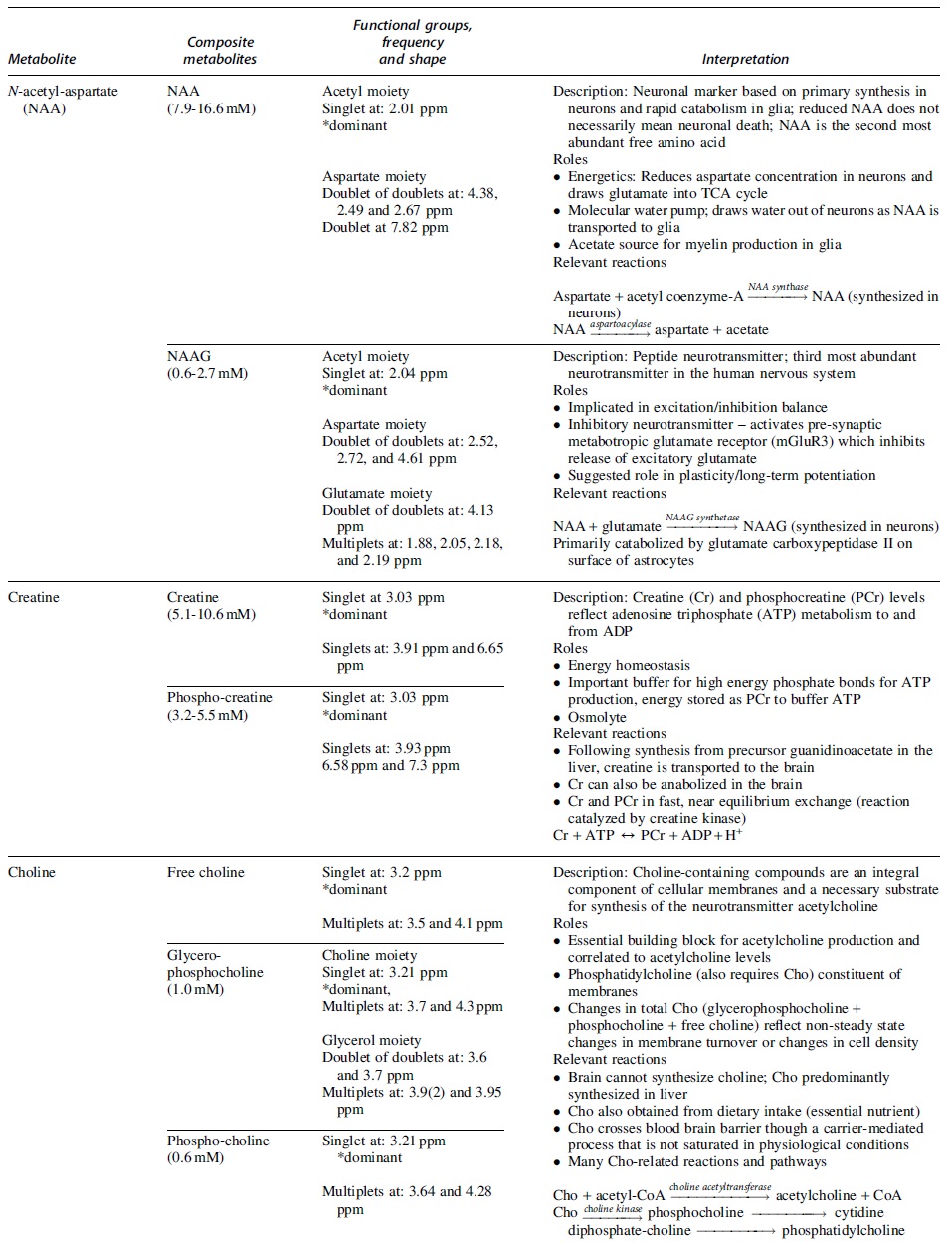
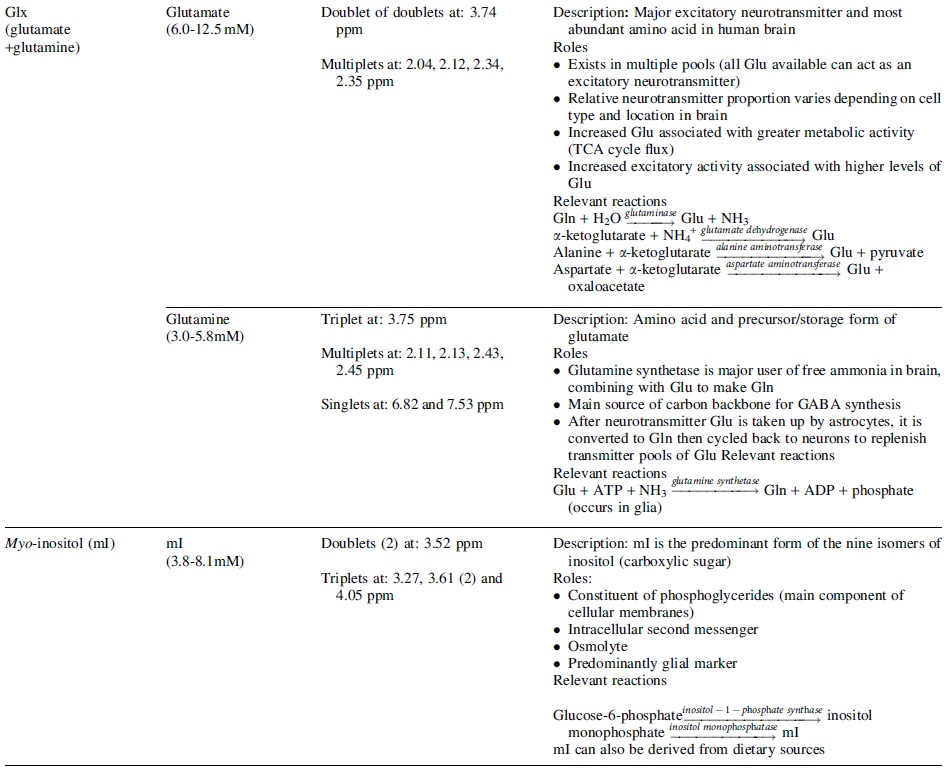
A detailed review of MRS methodology is beyond the scope of this article and readers are directed to several literature resources. 16 –19 More detailed information on terminology can be found in the article by Kreis and coworkers. 20 A number of relevant metabolites can be quantified using MRS. This article will focus on metabolites most commonly studied: N-acetyl aspartate (NAA), creatine (Cr), choline (Cho), glutamate (Glu), and glutamine (Gln), which are often reported as Glx = Glu+Gln, and myo-inositol (mI). In Table 1, we summarize the signal characteristics of these metabolites, including their composite signals and primary reactions.
A Summary of Primary Brain Metabolites Studied in Brain Injury that Can Be Quantified Using 1 H MRS
Typical in vivo concentration in the human brain, peak description, resonant frequencies, and key roles and relevant reactions are described for each metabolite. A comprehensive review of metabolite roles and pathways can be found in Rae. 154 Further details on chemical shift and concentration are described by Govindaraju and coworkers. 89 ADP, adenosine diphosphate; ATP, adenosine triphosphate; Cho, choline; Cr, creatine; GABA, γ-aminobutyric acid; Gln, glutamine; Glu, glutamate; Glx, glutamate + glutamine; NAA, N-acetyl-aspartate; NAAG, N-acetyl-aspartyl-glutamate; PCr, phosphocreatine; ppm, parts per million; TCA cycle, citric acid cycle.
The goal of this article was to comprehensively synthesize MRS research across all severities of brain injury and include consideration of the main factors that may impact MRS findings; specifically, brain region studied, injury severity, time since injury, age, sex and methodological factors (echo time, field strength, quantification approach) associated with acquisition and analysis. We performed independent meta-analyses to investigate NAA, Cr, Cho, Glx, and mI, all of which have been studied in TBI and have provided new insight into the consequences of brain injury. Metabolite levels in brain-injured subjects were compared with controls, and follow-up meta-regression analyses were performed to examine factors that may influence results.
Methods
Literature search and systematic review selection criteria
A comprehensive literature search of five databases (MEDLINE®, Embase, Web of Science, Scopus and SportDiscus) was performed on January 31, 2020, to identify studies that met the following inclusion criteria: Subjects had TBI or subconcussive hits/repetitive head impacts Any type of MRS (single-voxel or magnetic resonance spectroscopic imaging) All ages, males and/or female subjects Any field strength Manuscripts were in the English language
Studies were excluded if subjects were diagnosed with traumatic encephalopathy syndrome, as this is a distinct diagnosis. 121 Additionally, review articles and gray literature (e.g., conference abstracts, government publications, unpublished trial data, dissertations/theses, letters, or editorials) were excluded. For complete search details, please see Supplementary Tables S1–S5.
Two independent reviewers (J.M.J. and P.L.L.) completed all abstract and full-text screening using the Covidence platform.
22
Discrepancies were resolved by a third, independent reviewer (A.D.H.). The protocol for our systematic review was registered in the PROSPERO database (
Meta-analysis selection criteria
The meta-analyses were performed on NAA, Cho, Cr, Glx, and mI, as there is an abundance of 1 H MRS and 1 H magnetic resonance spectroscopic imaging (MRSI) literature for these metabolites in TBI. Studies were included in the meta-analysis if they met inclusion criteria for the systematic review, had a control group, and reported either water- or creatine-referenced (Cr-referenced) metabolite levels or fully quantified metabolite levels. Details on quantification approach criteria can be found in the Data extraction section.
Studies that examined only the less commonly studied nuclei (31P, 13C) and other metabolites available in 1 H spectroscopy were excluded from meta-analyses. Studies that lacked consistent, anatomically defined regions of interest for voxel placement (i.e., were placed in a lesion), case series (n < 5 in control or TBI group) and studies that had metabolite values that were either not reported or unextractable, were also excluded. For studies that were identifiably from the same study sample, only the study with the largest sample size was included in the meta-analysis.
Data extraction
Reviewers J.M.J. and P.L.L. independently extracted all data from the included studies using a customized data extraction form (see Supplementary Table S6). If studies reported metabolite values in a table or in the text, values were extracted directly. For studies that did not report metabolite values but included bar graphs that displayed the metabolite mean and standard deviation or scatter plots, WebPlotDigitizer 23 was used to manually extract data. Some studies subdivided TBI groups by good and poor health outcomes or other clinical measures. In such cases, we combined the mean and standard deviation of TBI subgroups for comparison with controls using the formula described in the most recent edition of the Cochrane Handbook for Systematic Reviews of Interventions. 24 A comprehensive list of all variables extracted is reported in Table S6. Often, Glu and Gln are reported as a combined Glx signal because the signals are highly overlapped, although it is known that Glu is more concentrated and dominates the Glx signal (see Table 1). The majority of studies reported Glx; however, if data were reported for Glu alone, this was included in the Glx meta-analysis.
Brain injury severity was classified based on the severity description reported in the original research articles; subconcussive hits (subconcussive hits + repetitive head impacts), mild TBI, moderate/severe TBI, and mixed TBI (mild + moderate/severe TBI). As MRS studies often combined moderate and severe TBI data, these categories were merged into a single moderate/severe TBI group. If severity was not specifically indicated, it was defined using Glasgow Coma Scale (GCS) scores (mild = GCS 13–15, moderate = GCS 9–12, severe TBI = GCS <9). 8 Time since injury was defined as the mean time since injury to MRS scan as reported by the study. We further categorized data into hyperacute (within 72 h), acute (> 72 h–2 weeks), subacute (> 2 weeks–3 months) and chronic (> 3 months) phases. Age was defined as the mean age of the sample. Sex was defined as the percentage of males within the study. Brain regions and tissue composition (white matter dominant, gray matter dominant, or mixed white+gray matter) were defined by the description provided in the original research articles. If no description was explicitly provided, brain regions and tissue composition were categorized by visually inspecting figures with voxel placements and comparison with similar voxel placements from other articles. Generally, one of three quantification approaches were used across the studies included in the meta-analysis including: (1) Cr-referencing, (2) water-referencing assuming constant parameters in the voxel, thus reporting metabolite levels in institutional units (i.u.), in this article, which will be referred to as relative water-referenced data, and (3) complete tissue correction, which includes accounting for differential T1, T2, and water density in white matter, gray matter, and cerebrospinal fluid (CSF), here referred to as absolute (Abs) quantification. Another quantification method is to use an external solution reference, in which a phantom solution that contains metabolites with known concentration is scanned and used as a reference for metabolite quantification. Because of the challenges associated with this approach, 17 only one study 54 included in this review had used this approach. See the consensus paper by Near and coworkers for additional details on complete tissue correction and external solution references. 17
Statistical analysis
Our primary analyses were quantitative comparisons of metabolite levels in brain-injured subjects relative to controls, with brain region as a subgroup. Nine brain regions were defined: frontal, frontoparietal, parietal, parietooccipital, occipital, temporal, subcortical, corpus callosum, and cerebellum. We used five distinct random-effects models (restricted maximum likelihood method) to generate the mean effect size (Hedges' g and 95% confidence interval [CI]) of brain injury on NAA, Cho, Cr, Glx, and mI levels in each brain region of interest and across all brain regions. Whole-brain MRSI metabolite measures were not evaluated as a subgroup in our regional analysis, as our primary interest was to explore whether metabolite alterations in brain injury differ by brain region.
Between-study heterogeneity was quantified using the I2 statistic. Sensitivity analyses were conducted for each metabolite using the leave-one-out method. Small study effects were examined using Egger's test and through visual inspection of funnel plots.
Univariate meta-regression was used to assess other potential moderators of metabolite levels including injury severity, time since injury, age, sex, tissue type (white matter dominant, gray matter dominant, mixed tissue), field strength, echo time (TE), quantification approach (absolute compared with relative water-referencing or Cr-referencing), and year of publication. Studies that did not report a moderator of interest were excluded from that specific moderator analysis. In the moderator analyses, we tested whether a moderator impacted the detection of group differences. The exception was for the quantification approach. As absolute quantification is generally recommended, 17 this moderator analysis compared whether relative water- and Cr-referencing results showed different effects than absolute quantification (basis of comparison). A common study design included multiple time point measurements for the brain-injured group (i.e., longitudinal) and a single measurement for the controls. When this study design was used, we used only the time point of the brain-injured group with the largest sample size to avoid repeated sampling of the control group in moderator analyses. However, for the time since injury meta-regression, the concern of repeated sampling was outweighed by the bias toward larger sample sizes at more acute visits and the desire to have as much data across time points as possible. Therefore, we included all time points to avoid undue sampling bias toward more acute visits that were less likely to have dropouts. Following standard practice for both the primary and moderator analyses, subgroups with fewer than two metabolite measurements were excluded. All statistical analyses were performed in STATA/BE 17.0 (Stata Corp., College Station, TX).
Risk of bias assessment
The National Institutes of Health (NIH) quality assessment tool for observational cohort and cross-sectional studies was used to assess the risk of bias of all studies included in meta-analyses. This tool is composed of 14 yes/no questions designed to assess the risk of bias. The quality rating was assigned as good/fair/poor and was completed by two independent reviewers (J.M.J. and P.L.L.) and conflicts were resolved by a third individual (A.D.H). Assessment of the quality of data was based on current standards in the field. 17,18
Results
One hundred and thirty-eight studies met inclusion criteria for the systematic review 25 –163 and of those, 62 NAA, 24 Cr, 49 Cho, 18 Glx, and 21 mI studies met inclusion criteria for meta–analysis (see Fig. 1). Most studies included in the systematic review used a conventional PRESS acquisition sequence (83 studies) of which 54 were single voxel studies. The remaining single-voxel studies used STEAM (25 studies), MEGA-PRESS (7 studies), and other less frequently used sequences including EPSI, L-COSY, DRESS, SPECIAL, and TSI (28 studies total). Eighty studies examined special populations: 34 studies examined exclusively pediatric cohorts defined as participants <18 years of age, 24 studies were of athletes (either varsity or professional), 16 studies were of sports-related concussion (specifically non-varsity, or professional athletes), and 6 studies were of military cohorts. Of all brain injury severities, mild TBI was the most studied (59 studies). Many studies have been conducted on moderate/severe TBI subjects (47 studies), with fewer studies of subconcussive hits and mixed severities (13 and 26 respectively). Fifty-five studies examined chronic TBI (> 3 months post-injury), 46 studies were of acute TBI (72 h-2 weeks post-injury) and 40 were of subacute TBI (2 weeks-3 months post-injury), while only 25 studies examined the hyperacute time point (< 72 h post-injury).
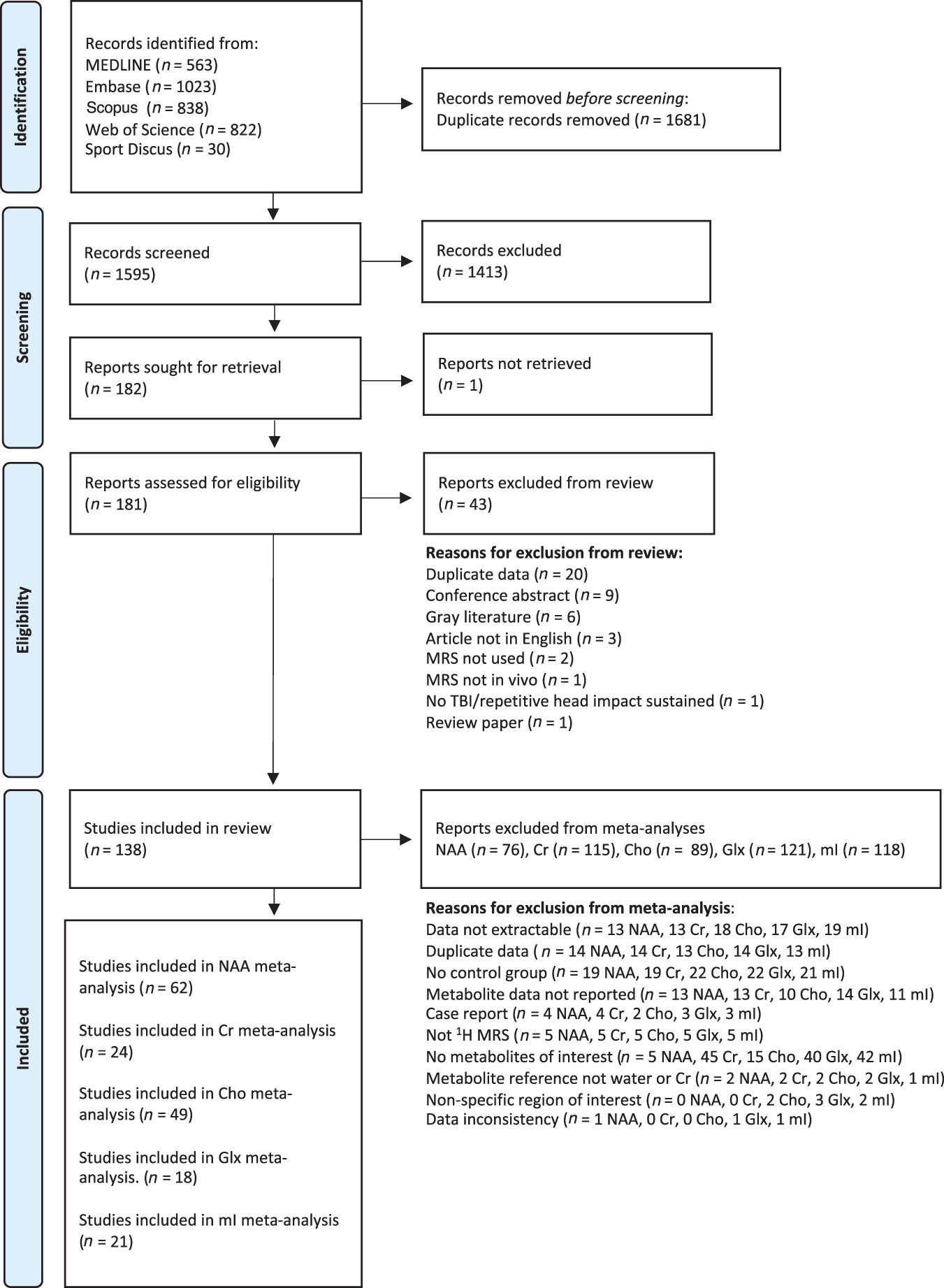
An overview of study selection and reasons for exclusion.
The following sections report results from the five meta-analyses; abbreviated forest plots of all meta-analyses are in Figure 2. Metabolite results across different brain regions can be found in Figure 3. Detailed forest plots for each metabolite are reported in Supplementary Figure S1a–e, which depict the classification of injury severity, time since injury, and brain region for each study included.
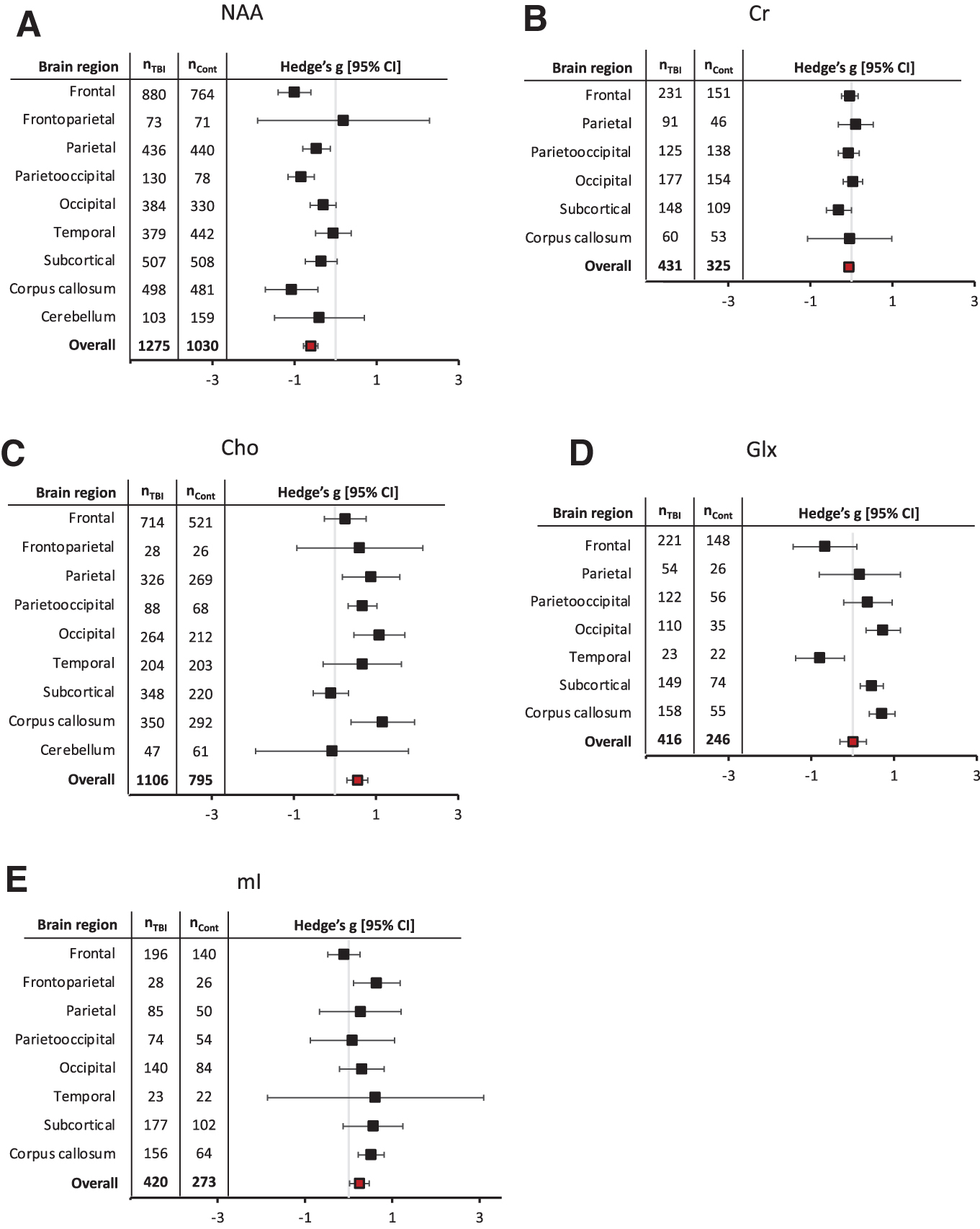
Metabolite effect sizes (Hedge's g) and 95% confidence intervals (CI) stratified by brain region studied for
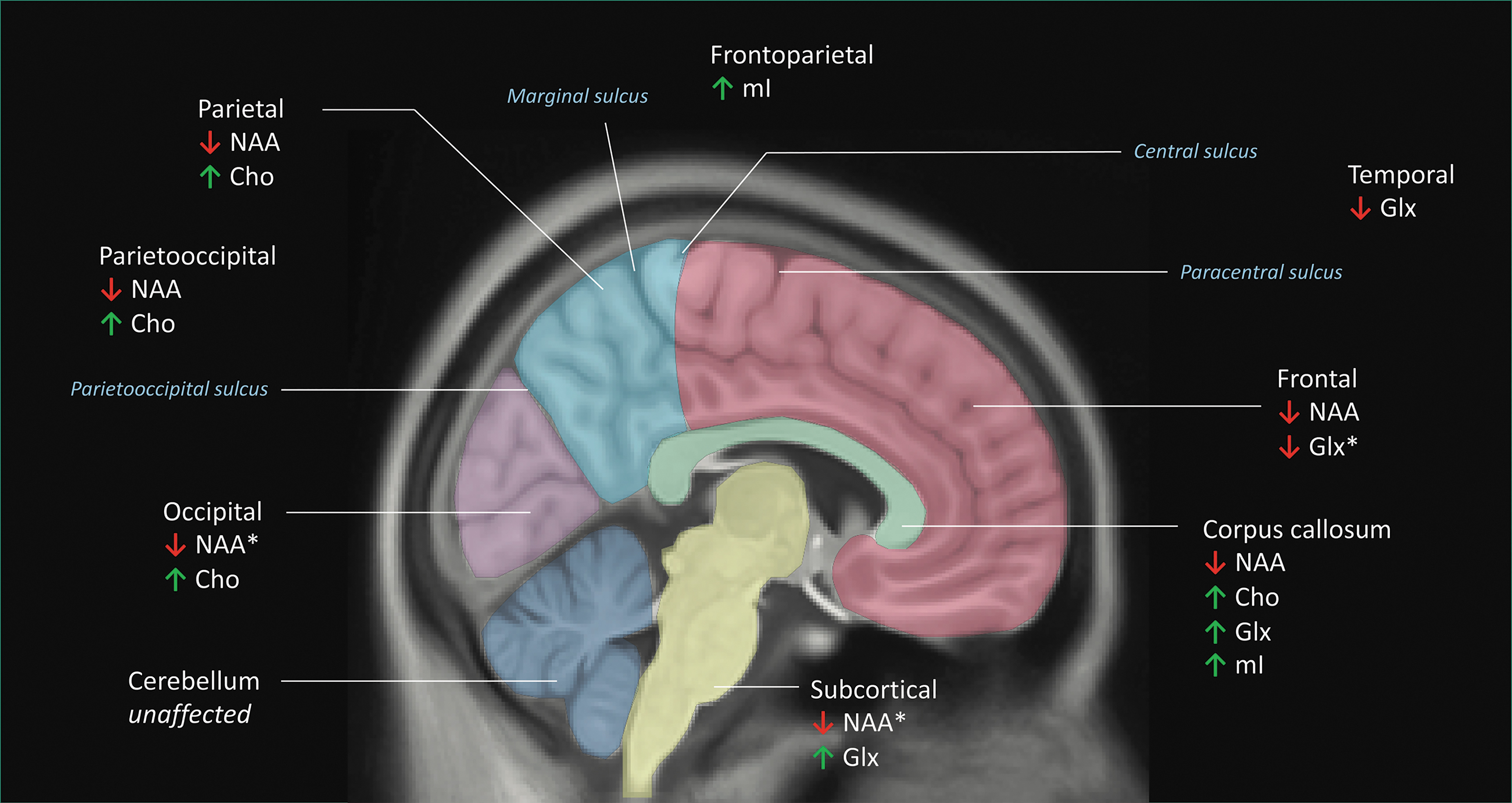
Schematic of the regional metabolite meta-analyses (nine regions defined: frontal [pink], frontoparietal, parietal [light blue], parieto-occipital, occipital [violet], temporal, subcortical [yellow], corpus callosum [green], and cerebellar regions [dark blue]). Brain regions were defined by the description provided in the original research articles. If no description was explicitly provided, brain regions were categorized by visually inspecting figures depicting voxel placement and comparison with similar voxel placements from other articles. Regional delineations can be seen for the frontal, parietal, occipital, subcortical, corpus callosum, and cerebellar regions. The frontoparietal cortex (not explicitly shown in the figure) was defined as a region transecting the central sulcus that was composed of both the frontal and parietal lobes. Similarly, the parieto-occipital cortex was defined as a region transecting the parieto-occipital sulcus, and was composed of both the parietal and occipital lobes. Although not shown here, the temporal lobe was delineated by the Sylvian fissure and the pre-occipital notch. Metabolites that were found to be significantly altered in brain-injured subjects relative to controls are indicated. ↑ = Metabolite significantly increased in brain-injured subjects. ↓ = Metabolite significantly decreased in brain-injured subjects. * = Metabolite alterations that approached statistical significance.
NAA
Sixty-two studies (nBrain-Injured = 1275; nControl = 1030) met inclusion criteria for the NAA meta-analysis. NAA was lower in the brain injury group, with an overall medium effect size (g = -0.60, CI: -0.78, -0.43). Brain region was a statistically significant factor (Qb[8] = 19.50, p = 0.01). The following brain regions had significantly lower NAA: frontal (g = -1.00, CI: -1.40, -0.60), parietal (g = -0.47, CI: -0.80, -0.13), parietooccipital (g = -0.84, CI: -1.16, -0.52), and the corpus callosum (g = -1.07, CI: -1.71, -0.43). No region showed increased NAA with brain injury. The detailed NAA forest plot can be found in Figure S1a.
Age was a statistically significant moderator of NAA effects, with more pronounced NAA reductions in older cohorts. Mild, moderate/severe, and mixed TBI groups (but not subconcussive hits) showed decreased NAA with comparable effect sizes. NAA was decreased at all time points studied, and reductions were greatest at the hyperacute time point. Reductions in NAA were found in gray matter dominant, white matter dominant, and mixed tissue brain regions. Statistically significant reductions in NAA were seen at both 1.5T and 3T, with a greater decrease in NAA at the lower field strength; no NAA reductions were seen at 7T. Larger NAA decreases were seen with relative water-referencing and Cr-referencing than with absolute quantification. TE was a statistically significant moderator of NAA effect sizes with studies of TE = 30 ms, 60 ms, and ≥100 ms having statistically significant effects. NAA effect sizes had a statistically significant relationship with publication year, with smaller effect sizes found in more recent studies. Lastly, sex was not a statistically significant moderator. NAA moderator analyses are summarized in Table 2.
NAA Moderator Analysis Summary
For quantification approach, the analysis used absolute units (Abs) as the basis of comparison for the other metabolite approaches (i.e., relative water-referencing and Cr-referencing). n = number of included studies (in all cases where n = 1 study, > 1 regional measurement was included in the analysis, conforming to inclusion methods). p-values of statistically significant results are bolded. NAA, N-acetyl-aspartate; SE, standard error; CI, confidence interval; TBI, traumatic brain injury.
Cr
Twenty-four studies (nBrain-Injured = 431; nControl = 325) met inclusion criteria for the Cr meta-analysis. There was no overall effect (g = -0.06, CI: -0.18, 0.06) of brain injury on Cr, nor did effect sizes differ across brain regions (Qb[5] = 3.78, p = 0.58). The detailed Cr forest plot is in Figure S1b.
For the Cr moderator analyses, time since injury was a statistically significant factor. Acutely post-injury Cr was increased while in the chronic phase, Cr was decreased relative to controls. No Cr differences were found at the hyperacute or subacute time points. TE was a statistically significant moderator, with studies of TE = 20 ms, 40 ms, and 135 ms having statistically significant effects. Cr was not moderated by field strength, injury severity, quantification approach, age, sex, tissue type, or publication year. Cr moderator analyses are summarized in Table 3.
Cr Moderator Analysis Summary
For quantification approach, the analysis used absolute units (Abs) as the basis of comparison for the other metabolite quantification approach (i.e., relative water-referencing). n = number of included studies (in all cases where n = 1 study, >1 regional measurement was included in the analysis, conforming to inclusion methods). p-values of statistically significant results are bolded. Cr, Creatine; SE, standard error; CI, confidence interval; TBI, traumatic brain injury.
Cho
Forty-nine studies (nBrain-injured = 1106; nControl = 795) met inclusion criteria for the Cho meta-analysis. Compared with controls, brain-injured subjects showed an overall increase in Cho with a medium effect size (g = 0.55; CI: 0.29, 0.80), and brain region was a statistically significant factor (Qb[8] = 16.93, p = 0.03). The corpus callosum (g = 1.16; CI: 0.39, 1.94), parietal (g = 0.88; CI: 0.18, 1.58), parietooccipital (g = 0.67; CI: 0.32, 1.02), and occipital (g = 1.08; CI: 0.46, 1.70) regions showed statistically significant increases in Cho in brain-injured individuals compared with controls. The detailed Cho forest plot can be found in Figure S1c.
Injury severity was a statistically significant moderator of effect size, with only the moderate-to-severe TBI group showing statistically significant increases in Cho. TBI subjects in the subacute and chronic phase post-injury, but not the hyperacute or acute phase, were found to have elevated Cho. Age had an influence on Cho effect sizes, with younger age groups showing larger Cho differences. TE was a statistically significant moderator of Cho effect sizes with studies of TE = 30 ms and 70 ms having statistically significant effects. Relative water-referenced, but not Cr-referenced, data differed significantly from absolute quantification. Only mixed tissue regions (not white matter dominant or gray matter dominant tissue regions) and field strength of 3T showed statistically significant Cho increases. Sex and publication year did not significantly impact Cho effect sizes. Cho moderator analyses are summarized in Table 4.
Cho Moderator Analysis Summary
For quantification approach, the analysis used absolute units (Abs) as the basis of comparison for the other metabolite approaches (i.e., relative water-referencing and Cr-referencing). n = number of included studies (in all cases where n = 1 study, > 1 regional measurement was included in the analysis, conforming to inclusion methods). p-values of statistically significant results are bolded. Cho, Choline; SE, standard error; CI, confidence interval; TBI, traumatic brain injury.
Glu + Gln, Glx
Eighteen studies (nBrain-injured = 416; nControl = 246) met inclusion criteria for the Glx meta-analysis. There was no overall effect of brain injury on Glx (g = 0.01; CI: -0.31, 0.33). However, effect sizes differed significantly across brain regions (Qb[6] = 29.91, p < 0.01). Relative to controls, Glx was significantly higher in the corpus callosum (g = 0.71; CI: 0.40, 1.02), occipital (g = 0.73; CI: 0.32, 1.15), and subcortical regions (g = 0.46; CI: 0.18, 0.74) in brain-injured individuals. In contrast, Glx was significantly lower in the temporal lobe (g = -0.79; CI: -1.38, -0.20) and approached statistical significance in the frontal region (g = -0.67; CI: -1.44, 0.10). The detailed Glx forest plot can be found in Figure S1d.
Age and tissue type were statistically significant moderators of effect size. Adolescent and young adult brain-injured groups had lower Glx than controls, whereas adults (> 25 years) tended to show higher Glx. Increased Glx was found in white matter dominant, but not gray matter dominant or mixed tissue. Glx was increased at 1.5T but not at 3T. Sex, publication year, and TE were not statistically significant moderators of Glx. Glx moderator analyses are summarized in Table 5.
Glx Moderator Analysis Summary
For quantification approach, the analysis used absolute units (Abs) as the basis of comparison for the other metabolite approaches (i.e., relative water-referencing and Cr-referencing). n = number of included studies (in all cases where n = 1 study, > 1 regional measurement was included in the analysis, conforming to inclusion methods). p-values of statistically significant results are bolded. Glx, glutamate + glutamine; SE, standard error; CI, confidence interval; TBI, traumatic brain injury.
mI
Twenty-one studies (nBrain-Injured = 420; nControl = 273) met inclusion criteria for the mI meta-analysis. There was an overall positive effect (g = 0.25, CI: 0.02, 0.47), indicating that when pooling data across all regions, mI is increased with brain injury. mI effects were not significantly different across brain regions (Qb[7] = 9.01, p = 0.25); that is, in no case did one region show a significantly different effect from another region. The detailed mI forest plot can be found in Figure S1e.
Age, time point, severity, field strength, TE, and publication year were all statistically significant moderators of mI effect size. Age had a positive relationship with mI, indicating a larger mI increase in older subjects with brain injury. mI was unaffected in the hyperacute and acute phase post-injury but was significantly increased at subacute and chronic time points. Severity was a moderator of effect size, with only the moderate/severe TBI group exhibiting statistically significant increases in mI. mI was significantly increased at 1.5T but not at 3T, and mI effect sizes were smaller in studies that were published more recently. TE was a statistically significant moderator of mI effect sizes, with studies of TE = 35 ms data showing statistically significant mI increases. Sex and tissue type did not moderate effect sizes. mI moderator analyses are summarized in Table 6.
mI Moderator Analysis Summary
For quantification approach, the analysis used absolute units (Abs) as the basis of comparison for the other metabolite approaches (i.e., relative water-referencing and Cr-referencing). n = number of included studies (in all cases where n = 1 study, > 1 regional measurement was included in the analysis, conforming to inclusion methods). p-values of statistically significant results are bolded. mI, myo-inositol; SE, standard error; CI, confidence interval; TBI, traumatic brain injury.
Risk of bias assessment
Overall, studies clearly stated their research question and goals. Inclusion/exclusion criteria was specified for a large majority of studies, but were often broad and captured heterogeneous cohorts. Only ∼30% of studies clearly defined their study population and controlled for confounding variables. Studies rarely justified their sample size and did not report the participation rate of eligible participants. Typically, assessors were not blinded to participant group. As there were few longitudinal studies, loss to follow-up after baseline was largely not applicable. Results from the risk of bias assessment are summarized in Supplementary Figure S2.
Sensitivity analyses
Visual inspection of funnel plots revealed evidence of asymmetry in some metabolites included in meta-analyses. Funnel plots of Cho and Cr were visually symmetric, but Glx, NAA and mI had a skew of varying magnitude. Funnel plots can be found in Supplementary Figure S3.
Egger's test indicated that NAA and Glx showed larger negative effect sizes in studies with smaller sample sizes. Egger's test was repeated while accounting for our primary moderator of interest (brain region). When the variance caused by brain region was included in Egger's test, NAA and Glx continued to be biased by small studies. Cho and mI also showed small-study effects; however, for these metabolites, larger positive effect sizes were associated with smaller sample sizes when accounting for brain region. Cr did not have small-study effects regardless of whether brain region was included in the analysis. Egger's test results are reported in Supplementary Table S7.
The results of the leave-one-out sensitivity analysis are reported in Supplementary Figure S4a–e and show that no single study had a disproportionate effect on the overall effect size for NAA, Cho, Cr, or Glx. mI had differing results depending on the study removed in the leave-one-out analysis; several studies changed the mI result from being statistically significant to a non-statistically significant trend.
Discussion
The purpose of this review was to synthesize the literature on MRS findings in brain-injured subjects to better understand the effect of brain injury on brain metabolites. To date, this is the largest meta-analysis synthesizing the effect of brain injury on metabolites with 1428 unique brain-injured subjects. We examined the effect of brain injury on five frequently studied metabolites using MRS (NAA, Cr, Cho, Glx, and mI). NAA was consistently decreased across different brain regions, TBI of all severities, and time since injury (early after injury and in the subsequent weeks and months). Cho and mI were increased with TBI, although alterations were less consistent than NAA and were moderated by various injury characteristics and demographic factors. Glx and Cr were largely unchanged, but did exhibit some alterations.
Brain region and tissue composition
Certain brain regions appear to be more prone to metabolite changes, and this vulnerability varied by metabolite. Interestingly, although the frontal lobe was the most studied region, only NAA showed a statistically significant alteration (reduction) in this region. The frontal lobe has functional relevance to brain injury given its important roles in cognition 164 and emotional regulation, 165 likely contributing to the frequent selection of this region of interest, particularly for studies of mild TBI. As Cho and mI were not altered in mild TBI but overall were changed in moderate/severe TBI, the lack of statistically significant alterations of these metabolites may be related to injury severity rather than the measurement region.
The high incidence of metabolite differences in the corpus callosum was common across metabolites. Diffuse axonal injuries are often considered a result of shear-strain deformation of the brain at gray and white matter interfaces. 166 Local metabolic disturbance is also thought to contribute to axonal injury, particularly with respect to delayed axonal swelling. 167 Because the corpus callosum is a critical bridge between cerebral hemispheres, it has been proposed that damage to it may contribute to interhemispheric disconnection and circuit disruption seen in functional MRI studies after TBI. 168 Consistent with the reported vulnerability of the corpus callosum to microstructural damage in brain injury, 169 our data show that this area is also susceptible to metabolic alteration.
Related to the regional analysis is the effect of tissue type. Whereas many studies included in our meta-analyses examined mixed white and gray matter regions, some studies targeted white matter dominant or gray matter dominant regions specifically. Given the differential distributions of some metabolites between white and gray matter, 170,171,174 we aimed to determine whether metabolite alterations were impacted by tissue type. Metabolite effect sizes in white matter dominant and gray matter dominant tissue were largely similar for all metabolites except for Glx. Relative to controls, Glx was higher following brain injury only in white matter dominant tissue, and Glx was the only metabolite that was increased or decreased depending on brain region (increased in corpus callosum, occipital, and subcortical regions; decreased in temporal lobe; trend of decrease in frontal lobe). Glu is reportedly higher in gray matter than in white matter, 120,170 –173 whereas Gln is higher in white matter than in gray matter. 174 As Glu and Gln have distinct roles (see Table 1), they may respond differently to brain injury, which may contribute to varied Glx results across regions. It is well known that Glu is more abundant than Gln, and that therefore Glx is often interpreted as Glu. The convention of reporting Glx is also a result of the difficulty in reliably separating Glu from Gln, as they are highly overlapped on the 1 H MRS spectrum. 175 As the number of studies that examined Glu alone are limited, we did not compare Glu and Glx results in this meta-analysis. However, the possibility of differing Glu and Gln effects raises an important question to be explored in future work, possibly with customized approaches or ultra-high field. 175 Another limitation, discussed in detail subsequently, is the inability to specifically differentiate the effects of region compared with tissue type with the univariate approaches used here.
Injury severity
NAA was consistently decreased across TBI of all severities but did not exhibit differences in subconcussive hits. Compared with the other metabolites, NAA appears to be the most sensitive to brain injury. Cho and mI were elevated only in moderate-to-severe TBI, whereas Glx and Cr did not appear to be mediated by injury severity. Interestingly, NAA reductions were not larger with more severe TBI.
NAA has been traditionally considered as a marker of neuronal integrity 117 ; however, NAA has many roles as described in Table 1. Decreased NAA in moderate-to-severe TBI has been suggested to be related to neuronal loss along with tissue loss. However, it must be noted that Cho and mI are increased in moderate-to-severe TBI, suggesting that tissue loss alone does not explain alterations in brain metabolites. Decreased NAA in moderate-to-severe TBI may instead reflect a combination of neuronal dysfunction and tissue loss. Similarly, reductions in NAA following mild TBI may reflect a transient state of poor neuronal health or energy impairment, as NAA levels have been seen to increase with recovery. 176,177 The lack of statistically significant NAA reductions in subconcussive hits may indicate that NAA alterations may be too subtle to detect, or it may be that the injury is not severe enough to elicit a lasting change in NAA. Currently there is limited MRS literature on subconcussive hits; therefore, the topic warrants further study to obtain a better understanding of the cumulative effect of head impacts on brain metabolites.
Time since injury
Immediately post-TBI, the brain is in a state of ionic and energetic flux, 178 and long-term consequences are less well known. The largest NAA reductions were detected during the hyperacute phase post-injury, which is widely acknowledged as a period of metabolic disturbance 179 with high risk of re-injury. 166 NAA alterations lasting beyond the hyperacute period may be related to severity of initial injury or the capacity to recover from injury. Cho and mI alterations were only found in the subacute and chronic phases post-injury. Clinical studies and histopathological work have provided evidence of prolonged neuroinflammation following TBI, 181,182 particularly in moderate-to-severe TBI, which may explain Cho and mI increases. Alternatively, Cho and mI increases may reflect membrane remodeling during recovery.
Age and sex
Clinically, the relationship between age and TBI is complicated. Central nervous system anatomy and physiology differ between children and adults, which results in different outcomes following initial head impact as well as in response to treatment interventions. 183 Differences between pediatric and adult TBI are reviewed in detail by Figaji and coworkers. 183 In the present meta-analysis, weaker NAA effects were found in younger populations, suggesting that older individuals are more susceptible to damage resulting in lower NAA following injury. It may be that the more plastic pediatric brain is more adaptable and/or resilient, resulting in a smaller change in NAA. This interpretation, however, is complicated by the fact that NAA increases with development. 184 In contrast, larger Cho effects were seen in younger cohorts. Typically, Cho concentrations are higher in white matter than in gray matter, 174,185 –187 and white matter proliferation and maturation are characteristic of early brain development. 188,189 As previously discussed, white matter is also highly susceptible to brain injury. 190 Brain injury during this crucial developmental period may result in larger Cho increases than in adulthood. 184 Alternatively, because the brain is highly plastic in early life, 191 more pronounced elevations in Cho seen in younger ages may reflect a greater ability of the brain to respond and recover from damage to neural membranes. Overall, the results of our meta-analyses support the proposal that adult MRS TBI results do not extend to the pediatric population.
Sex differences in TBI pathophysiology and symptomology are not clearly defined. 192 It is widely reported that women have worse outcomes than men following TBI. 193 –196 However, there is also research to suggest that these outcomes differ by symptom 197 and may be influenced by TBI severity. 192 Here, we did not find sex to be a statistically significant moderator of metabolite effect sizes. Sex was operationally defined as the percentage of males of each study. Although this approach is common in meta-analyses, 198 –203 it does not capture differences in sex distributions between groups, and it is possible that the influence of sex on metabolite alterations is masked in this analysis. Although we did not find a relationship between sex and metabolite effect sizes, we acknowledge that this area of research merits further investigation.
Control groups
A source of variability across studies was the definition of controls used for comparison with brain-injured subjects. In some studies, broad inclusion criteria were used for the control group with the only specification being that controls were “healthy.” 67,78,92 Other studies excluded controls with any previous brain injury history. 55,56,84 Studies of contact sport athletes often recruited a non-contact athlete control group to account for the biological and psychosocial effects of participation in sports. 36,48,63,90,95,118,163 Similarly, orthopedic injury controls are becoming increasingly common, as they share a common trauma experience to brain-injured subjects. 97,113,151,204 The selection of controls is an important consideration, as a recent imaging study comparing children and adolescents with mild TBI, those with orthopedic injury, and uninjured controls found that the structural connectome of the mild TBI and orthopedic injury groups did not significantly differ, but that both groups were significantly different from the uninjured group. 199 This finding suggests that differences in brain imaging are related to injury experience, rather than specifically to brain injury. Therefore, the selection of controls for MRS studies may also have statistically significant effects on outcomes.
Technical moderators
Early MRI systems were typically field strengths of 1.5T, whereas increasing adoption and widespread use of 3T and higher field strengths are now more common. Acquisition and analysis methods have also improved with the development of more advanced hardware and software, including but not limited to: water suppression, shimming, and post-processing (coil combination and retrospective frequency correction). 206 Our analyses found that NAA and mI effect sizes were larger in earlier studies, with weaker effects found in more recent years. This may reflect an insensitivity of measurement methods to detect subtle changes in metabolites in earlier work and a lack of publication of null results. Alternatively, although still meeting the criteria for mild, moderate, or severe TBI, the injuries themselves may have been more severe in older literature. Indeed, there is some evidence that as information and knowledge about concussion are more widespread, there is increased presentation of mild TBI to clinicians. 207 Interestingly, Cho effects were statistically significant at 3T but not 1.5T, which may be related to an increased ability to detect changes with improved methods. As stated earlier with the statistically significant effects of moderate/severe TBI in Cho, it would be interesting to explore how severity, field strength, and publication year impact these results, as there appears to be sampling bias toward more severe TBI being studied earlier and at 1.5T, compared with more recent work at 3T, with a larger focus on mild TBI.
Quantification approaches have also been refined over time. Specifically investigated here was the contrast between Cr-referencing (often used in earlier literature), relative water-referencing, and absolute quantification. Our findings suggest that Cr-referenced and relative water-referenced data had larger metabolite effect sizes than absolute quantification (relative water-referenced and Cr-referenced data showed larger NAA deficits than absolute quantification; relative water-referenced data detected larger increases in Cho than absolute quantification). Generally, absolute quantification is now the recommended approach, 17 however, Cr-referencing may be preferred in some cases where the water signal differs. For example, Cr-referencing may reduce inter-site variability. 206,208
Sensitivity analyses
Our sensitivity analyses found that there are small study effects present in the included data, which suggests the presence of publication bias in the field. All the metabolites included in our meta-analyses can be acquired in a single conventional spectroscopy acquisition, yet we noted a much higher prevalence of studies that examined NAA than that examined Cho, Cr, Glx, or mI. This observation may reflect a reporting bias as well as technical specifications of MRS sequences. Early MRS work favored long TEs, which minimizes the spectral baseline and macromolecule contamination. However, metabolites such as Glx and mI have a short T2 relaxation time limiting their quantification at long TEs, 209 which likely contributes to the relative paucity of studies on Glx and mI. As Cr is commonly used as a quantification reference, Cr data are also limited.
Limitations
High heterogeneity was found in studies included in our review and was expected given the nature of brain injury. We explored this heterogeneity through univariate moderator analyses and have identified specific factors (brain region, tissue type, age, injury severity, time since injury, field strength, TE, quantification approach, and publication year) that contribute to variability in metabolite results. However, we also found that individual studies often included cohorts with highly diverse demographic and injury characteristics, which added a degree of uncertainty to the relationships found with different moderators. For example, studies often did not target specific TBI severities, 40,43,65,82 age ranges, 125,155,156 or time periods post-injury. 40,43,82,125 Future work can help limit this within-study heterogeneity by implementing clear, specific inclusion and exclusion criteria for participant recruitment.
Even with clearly defined cohorts, there are limitations to univariate approaches. In particular, univariate approaches do not explore the relationships between moderators. The importance of such interactions is illustrated by considering the influence of age on metabolite alteration following brain injury. For example, NAA and N-acetyl-aspartyl-glutamate (NAAG) are distinct with respect to their regional developmental changes. 210 Specifically, NAA in gray matter increases in child development; whereas NAA levels in white matter remain constant. In contrast, the opposite has been reported for NAAG. 210 Therefore, whereas total NAA (NAA + NAAG) increases in childhood and early adolescence, the underlying metabolites contributing to this change differ by tissue. Further, structural development is not uniform across the brain and has been shown to vary with tissue and region. 211 As such, the age and tissue relationship becomes important to consider when interpreting whether children or adults are more susceptible to metabolite alteration following brain injury. Another example of interacting relationships between moderators is the tissue type and regional analyses. Some regions are dominated by white matter or gray matter, whereas other regions are more mixed, and, specifically, differentiating differences that are driven by location and those driven by tissue type are beyond the scope of the univariate approaches used in this meta-analysis. Further, only the frontal lobe has sufficient data across all metabolites for performing this type of analysis, but this would impart a different type of regional bias to the analysis. Here, interpretation of the tissue-type and regional results must consider these inter-related effects. Multivariate approaches for moderator analyses can help capture interactions among different biological and technical factors to further tease apart their influence on metabolism following brain injury.
Consistent with other MRS systematic reviews, 212 –214 we have used an inclusive requirement for subgroup definitions (a minimum of two MRS measurements per subgroup) rather than selectively omitting studies or features less reported in the literature. This results in the consensus of these smaller subgroup analyses being less compelling than those with multiple studies and measures included; however, given that a goal of this review was to provide an overview and synthesis of all the literature as well as to identify future opportunities, this approach seemed most appropriate here. The limitation resulting from this definition is an imbalance in the number or studies, measures, and participants in different moderator analyses and subgroups. For example, in our regional analysis, some brain regions were less frequently studied than others. Only two studies that included a total of 55 participants (23 brain injured and 22 controls) were included in the Glx temporal lobe analysis, whereas nine studies that included a total of 369 participants (221 brain injured and 148 controls) were included in the frontal lobe analysis. Analyses of smaller subgroups carry a different level of confidence than larger samples and the leave-one-out sensitivity analysis does not enable comparing the levels of confidence between the different moderator analyses. Readers are therefore encouraged to consider the number of studies and study sample size included in each subgroup (see Fig. 2) when interpreting each analysis.
Areas for future research
We have identified several areas of research that were beyond the scope of this review, but that offer opportunities for future syntheses. First, we observed that studies frequently examined metabolite levels in relation to TBI symptom and neuropsychological outcomes. 27,38 –40,53,55,61,64,68,69,71,79,82,86,90,95,97,116,117,151,152,154,159,160 For example, many studies reported a relationship between low NAA levels and worse symptom or neuropsychological outcome. 39,40,55,61,62,64,79,82,97,117,151,152,160 Although symptom associations with other metabolites were identified, findings were less consistent. 27,38,40,53,55,62,64,68,69,75,79,82,90,95,116,117,151,159,160 Determining the consistency of metabolite-symptom relationships across the whole brain and across TBI severities is an opportunity. It is also ambiguous whether symptom resolution precedes or follows metabolic recovery. A clearer picture of the relationship between symptoms and metabolites may serve to inform treatment and hasten recovery.
Second, although less often examined, other metabolites such as lactate, 31,50,78,92,100,104,114,121,140,143,153 γ-aminobutyric acid (GABA), 3,86,95,143,154,158 glutathione, 27,64,90 and aspartate 109,130,143 have been measured using 1 H MRS in brain injury. As many of these metabolites are present in low concentrations and some have complex signal patterns, reliable quantification requires advanced MRS methods, typically spectral editing. Similarly, TBI studies using 31 P MRS 67,122,134,138,139 are rare, but some studies are present in the literature. Although the technical requirements for acquisition of these metabolites are more complex, they offer important insights into cortical metabolism. As such, it would be valuable to synthesize and explore the existing literature on these less frequently studied metabolites either alone or in combination with other metabolite results.
Conclusion
This review identified 138 studies that have used MRS in brain-injured individuals. Our meta-analyses of 1428 unique brain-injured subjects show that TBI affects brain metabolites across widespread regions, with the corpus callosum showing high susceptibility to metabolite alteration. NAA was the most consistent marker of brain injury and was found to be decreased in TBI of all severities, but not in subconcussive hits. Cho and mI were altered only in moderate-to-severe TBI. Glx and Cr were less commonly affected but did exhibit alterations under specific conditions. Moderator variables, including time since injury, age, tissue type, and metabolite referencing method, were all found to influence effect sizes and should be considered in analyses. Collectively, this review synthesizes the large body of MRS work in brain injury and raises questions to help tease apart some of the finer aspects of injury sequelae in future studies.
Footnotes
Acknowledgments
We thank the University of Calgary Health Sciences Librarians, including Nicole Dunnewold, for guidance and assistance with the literature search strategies.
Authors' Contributions
Julie M. Joyce was responsible for the conceptualization, methodology, formal analysis, investigation, writing – original draft, writing – review and editing, and visualization; Parker L. La was responsible for the conceptualization, methodology, formal analysis, investigation, writing – original draft, writing – review and editing, and visualization; Robyn Walker was responsible for investigation, formal analysis, writing – review & editing, visualization; Ashley D. Harris was responsible for conceptualization, methodology, investigation, resources, writing – original draft, writing – review and editing, supervision, project administration, and funding acquisition.
Funding Information
Support was provided by the Hotchkiss Brain Institute and the Alberta Children's Hospital Research Institute. A.D.H. holds a Canada Research Chair in Magnetic Resonance Spectroscopy in Brain Injury.
Author Disclosure Statement
No competing financial interests exist.
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
Supplementary Table S5
Supplementary Table S6
Supplementary Table S7
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.