Abstract
The effect of decompressive craniectomy (DC) on functional outcomes and mortality in children after severe head trauma is strongly debated. The lack of high-quality evidence poses a serious challenge to neurosurgeons' and pediatric intensive care physicians' decision making in critically ill children after head trauma. This study was conducted to compare DC and medical management in severely head-injured children with respect to short-term outcomes and mortality. Data on patients <18 years of age treated in Germany, Austria, and Switzerland during a 10-year period were extracted from TraumaRegister DGU®, forming a retrospective multi-center cohort study. Descriptive and multi-variable analyses were performed to compare outcomes and mortality after DC and medical management. Of 2507 patients, 402 (16.0%) received DC. Mortality was 20.6% after DC and 13.7% after medical management. Poor outcome (death or vegetative state) occurred in 27.6% after DC and in 16.1% after medical management. After risk adjustment by logistic regression modeling, the odds ratio was 1.56 (95% confidence interval 1.01-2.40) for poor outcome at intensive care unit discharge and 1.20 (0.74-1.95) for mortality after DC. In summary, DC was associated with increased odds for poor short-term outcomes in children with severe head trauma. This finding should temper enthusiasm for DC in children until a large randomized controlled trial has answered more precisely if DC in children is beneficial or increases rates of vegetative state.
Introduction
Severe traumatic brain injury (TBI) is a relevant cause of morbidity and mortality in children around the globe. 1 Research interest in TBI has grown in recent years and progress has been made in gathering evidence on the best management of pediatric TBI. 2 The first tier of post-TBI treatment is designed to avoid exacerbation of cerebral injury by prevention or treatment of intracranial hypertension by optimizing cerebral perfusion pressure and brain tissue oxygenation. 3 The second tier of therapy has a low evidence level and consists of rescue measures to control intracranial hypertension by application of hyperosmolar solutions, barbiturate infusion, hypothermia treatment, hyperventilation, and decompressive craniectomy. 2,3
The potential of decompressive craniectomy (DC) to improve mortality and functional outcomes is highly debated. DC lowers elevated intracranial pressure (ICP) in children, 4 –11 but one in 10 patients undergoing DC suffers from a complication requiring additional interventions. 12 In adults, two randomized, controlled trials (RCTs) investigated the effect of DC on overall mortality and outcome. 13,14 The studies differed in design, with the Decompressive Craniectomy (DECRA) trial investigating the effect of DC on early and the RescueICP on late refractory ICP elevations. The findings supported DC for late rather than for early refractory ICP elevation with more survivors but more patients with low Glasgow Outcome Scores. 13,14 The effect of DC on outcomes and mortality in children has not been studied in high quality randomized controlled trials (RCTs).
Only one RCT including 27 patients is available on DC in children, 6 along with numerous small studies, case series, and one larger retrospective study including 150 children with DC. 4 –11,15 –19 Studies on functional outcome reported better outcomes in children after DC compared with conservative treatment 4,6,20 or no difference. 5 Two studies found lower mortality in the DC groups compared with medical management. 4,20 However, almost all reports are limited by small case numbers, retrospective and heterogenous study designs, and different outcome measures. 3,16 Urgent questions concerning the timing and location of surgery, cut-off values for ICP, and the effect of DC on mortality and outcomes remain unanswered.
The aim of our study was to compare functional outcomes and mortality at discharge from the intensive care unit (ICU) after DC versus medical management in children with severe head trauma in a large dataset. By extracting data from the TraumaRegister DGU® (TR-DGU) from Germany, Austria, and Switzerland, we conducted a retrospective international multi-center cohort study.
Methods
Data collection
The TraumaRegister DGU of the German Trauma Society (Deutsche Gesellschaft für Unfallchirurgie, DGU) was founded in 1993. The aim of this multi-center database is a pseudonymized and standardized documentation of severely injured patients.
Data are collected prospectively in four consecutive time phases from the site of the accident until discharge from hospital: 1) pre-hospital phase; 2) emergency room and initial surgery; 3) intensive care unit; and 4) discharge. The documentation includes detailed information on demographics, injury pattern, comorbidities, pre- and in-hospital management, course on intensive care unit, relevant laboratory findings including data on transfusion, and outcome of each individual. The inclusion criterion is admission to hospital via emergency room with subsequent ICU/intermediate care or reach the hospital with vital signs and die before admission to ICU.
The infrastructure for documentation, data management, and data analysis is provided by AUC–Academy for Trauma Surgery (AUC–Akademie der Unfallchirurgie GmbH), a company affiliated to the German Trauma Society. The scientific leadership is provided by the Committee on Emergency Medicine, Intensive Care and Trauma Management (Notfall-, Intensivmedizin und Schwerverletztenversorgung, Sektion NIS) of the German Trauma Society. The participating hospitals submit their data pseudonymized into a central database via a web-based application. Scientific data analysis is approved according to a peer review procedure laid down in the publication guideline of TraumaRegister DGU.
The participating hospitals are primarily located in Germany (90%), but a rising number of hospitals of other countries contribute, data as well (at the moment from Austria, Belgium, Finland, Luxembourg, Slovenia, Switzerland, The Netherlands, and the United Arab Emirates). Currently, almost 30,000 cases from more than 650 hospitals are entered into the database per year. For this study, 2507 pediatric cases of severe TBI were analyzed.
Participation in TraumaRegister DGU is voluntary. For hospitals associated with TraumaNetzwerk DGU; however, the entry of at least a basic data set is obligatory for reasons of quality assurance.
Patients aged <18 years from Germany, Austria and Switzerland with Abbreviated Injury Scores (AIS) for the head ≥3 who were admitted between 2010 and 2019 (10 years) were included. Patients transferred to another hospital within the first 48 h after admission were excluded due to missing data on final outcomes. Patients transferred in from another hospital were excluded, as well. Only patients with complete standard documentation including surgical interventions were included.
Predicted mortality, injury severity, and outcome measures
Predicted mortality was calculated using the Revised Injury Severity Classification, version II (RISC II) score, which was developed and validated by the TR-DGU to predict survival of trauma patients based on 13 predictive pre-clinical factors. 21 The included predictors are the worst and second worst injuries based on the AIS, head injury based on the AIS, age, sex, pre-trauma American Society of Anesthesiologists Score (ASA score), pupil reactivity, pupil size, motor function, injury mechanism, blood pressure, coagulation measured by international normalized ratio, base deficit, hemoglobin concentration, and cardiopulmonary resuscitation performed before hospital admission.
Injury severity was assessed using the Injury Severity Score (ISS) and AIS for the head and body. Outcome was measured unblinded by the physician in charge at ICU discharge. The TraumaRegister gives concise instructions on how to complete each item in the guide to completion that can be downloaded publicly on their website. Outcome categories were defined as good recovery, moderate disability (handicapped but independent), severe disability (conscious but handicapped and dependent on help), vegetative state (permanent vegetative state) or death. We defined poor outcome as death or vegetative state. Good outcome was defined as severe disability, moderate disability, and good recovery.
Guidelines and ethics approval
The present study is in line with the publication guidelines of the TR-DGU and is registered as project ID 2020-033. All methods concur with relevant guidelines and regulations and the study was approved by the ethics committee of the Medical Faculty of the University of Duisburg-Essen (21-10116-BO).
Statistical analysis
Quantitative variables are presented as mean and standard deviation (SD) or median and interquartile range (IQR) in case of skewed data. For qualitative factors, absolute and relative frequencies are given. For mortality rates, 95% confidence intervals (CIs) were calculated for comparison of observed and predicted mortality by the RISC II score.
Adjusted odds ratios with 95% CIs were calculated using logistic regression models with in-hospital poor outcomes and mortality as dependent variables. Covariates were selected based on previous knowledge on predictors of outcome in trauma patients: Because the RISC II score sums up pre-clinical factors, we included additional, mainly in-hospital, predictive factors in the model. These factors were helicopter transport, blood transfusion in the shock room, computed tomography performed, treatment in a Level 2 or 3 trauma center, placement of cerebrospinal fluid drain or hematoma evacuation, cervical spine injury, cerebral contusion, epidural hematoma, subdural hematoma, subarachnoidal hemorrhage, intracerebral hemorrhage, brain edema, brain stem injury, skull fractures, and age (stratified into groups from 0-5, 6-11, and 12-17 years). The severity of head injury (assessed by the AIS head) was not entered into the model separately, because it forms part of the RISC II score. Cases with missing data on covariates were excluded from logistic regression.
Statistical analyses were performed with SPSS Version 26 (IBM Inc., Armonk, NY) and SAS Enterprise Guide 7.1 (SAS Institute Inc., Cary, NC). Figures were produced with SAS Enterprise Guide 7.1.
Results
Description of the cohort
Out of 352,899 patients reported to the TraumaRegister DGU during the study period, 11,000 were children, of which 2507 met the inclusion criteria (Fig. 1). More than 90% of patients were treated in a Level 1 trauma center and male sex prevailed (Table 1). DC was performed in 402 (16.0%) patients. Patients undergoing DC did not differ with regard to age (Fig. 2), but suffered more severe head injury, had higher ISS, lower initial Glasgow Coma Scale (GCS) values, and longer ICU and hospital stays compared with medical management (Table 1). In patients with AIS head 5, the findings on ISS and GCS were inverted, with lower ISS and higher GCS values in patients receiving DC (Table 2).
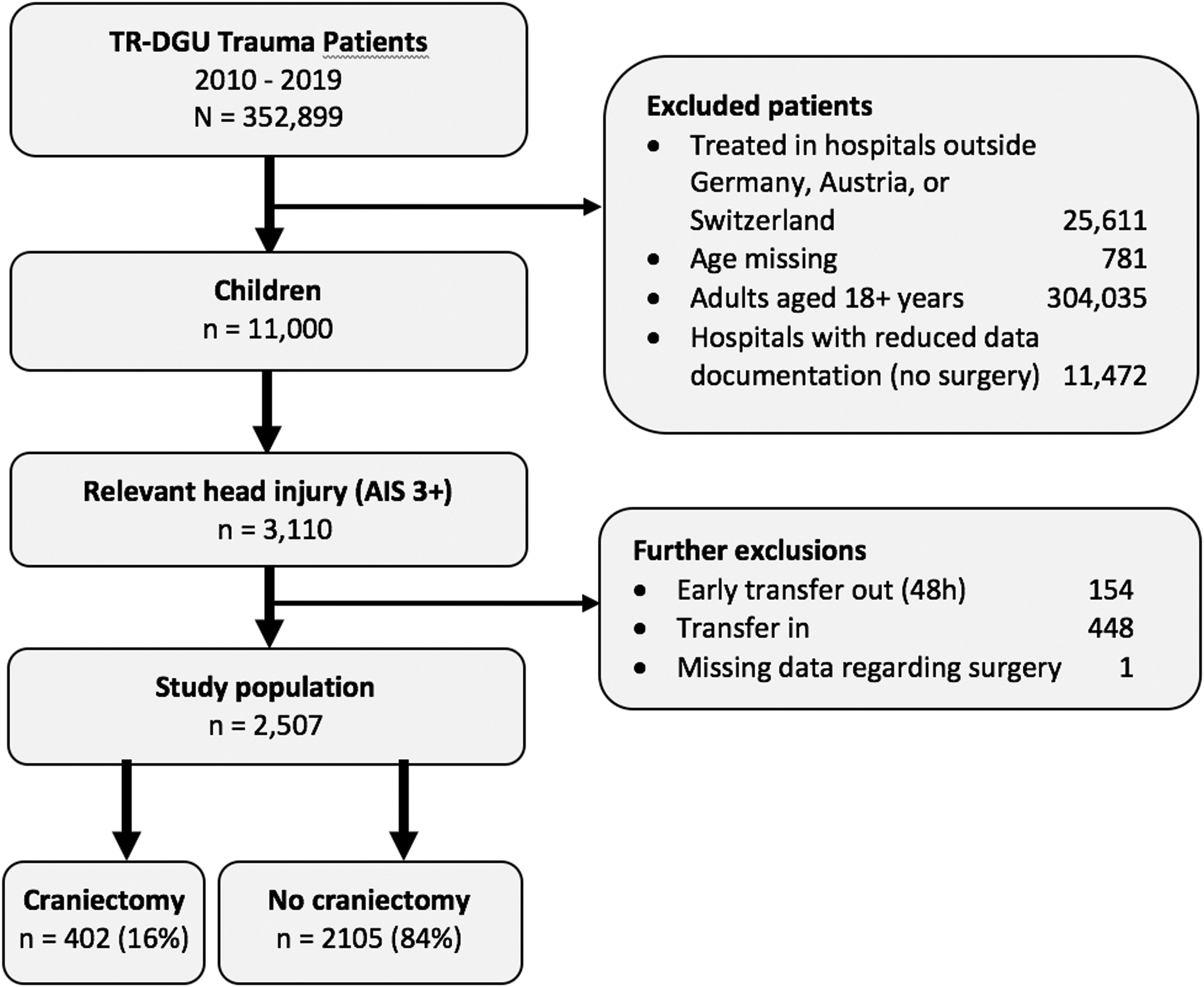
Flow chart of included and excluded patients
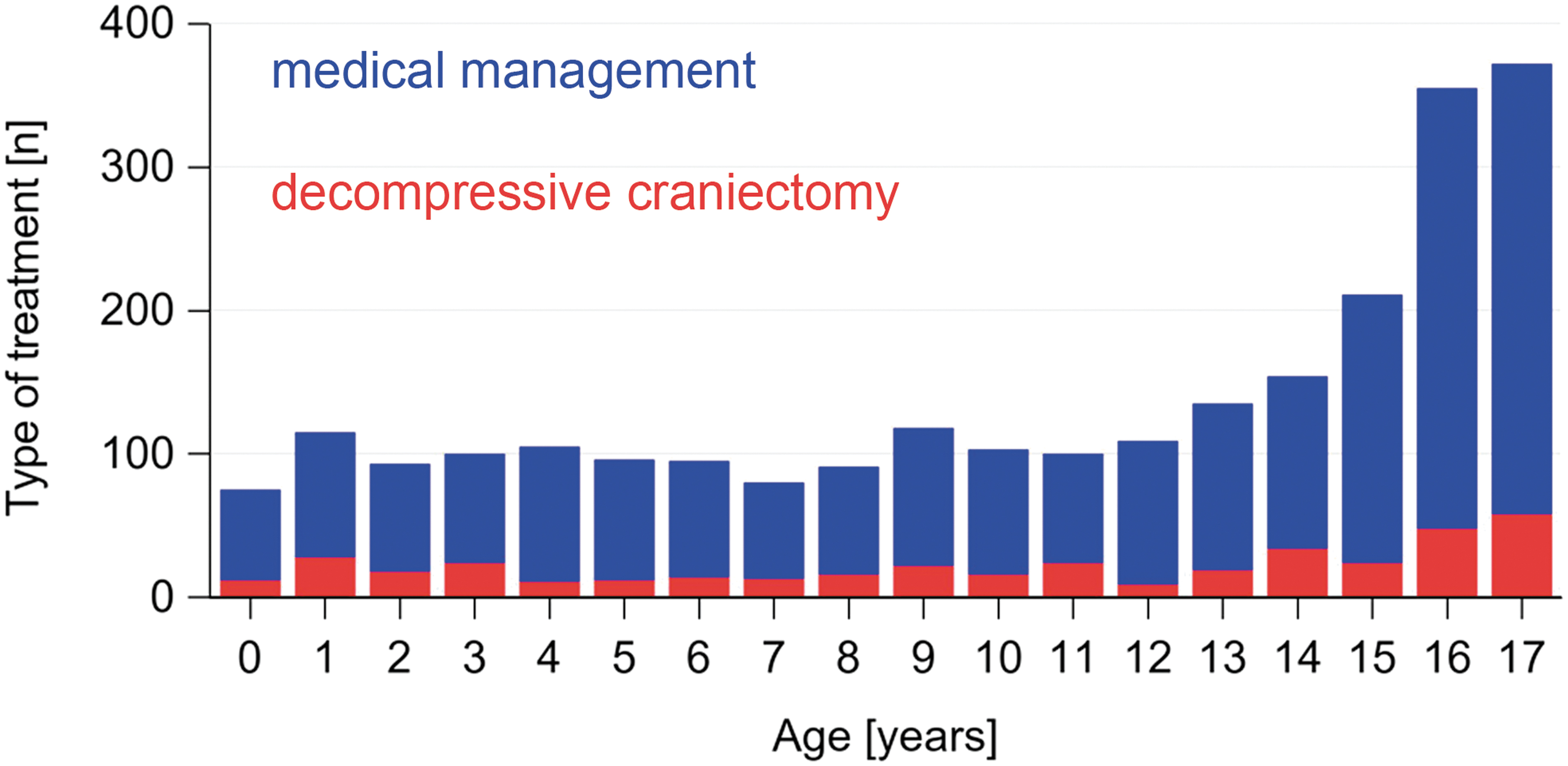
Frequencies of medical management and decompressive craniectomy by age. The proportions of decompressive craniectomy versus medical management are constant, but absolute frequencies increase with age due to motor vehicle accidents. Color image is available online.
Patient Characteristics
Missing data on outcome: n = 67 (2.7 %), n = 56 (2.7 %) for medical management, n = 11 (2.7 %) for DC.
SD, standard deviation; ISS, Injury Severity Score; GCS, Glasgow coma scale; ICU, intensive care unit; IQR, interquartile range; RISC, Revised Injury Severity Classification; SR, shock room.
Injury Severity and Combined Outcomes by AIS Head and Treatment Group
Sums of absolute frequencies for combined outcomes can differ from the number n given in the headers due to missing data on Outcome.
AIS, abbreviated injury score; MM, medical management; DC, decompressive craniotomy; ISS, injury severity score; GCS, Glasgow coma scale; ICU, intensive care unit; SD, standard deviation.
Blunt trauma was the leading mechanism of injury and no differences were observed in the type of accident between groups (Table 2). Intracranial hemorrhage and brain edema were more prevalent in children undergoing decompressive craniectomy, while skull fractures and cerebral contusions occurred similarly often (Table 3).
Mechanism and Type of Injury
TBI, traumatic brain injury; AIS, abbreviated injury score.
Mortality
Predicted and observed mortality was higher in children undergoing DC (Table 1) and with more severe head injury (Fig. 3A, 3B). Mean time to death was 2.6 h (± 3.1) after the accident for patients receiving medical management and 5.4 h (± 11.4) for patients undergoing DC. Mortality was higher in patients receiving medical management during the first 6 h after the accident, but at 24 h this finding was inverted (Fig. 4).
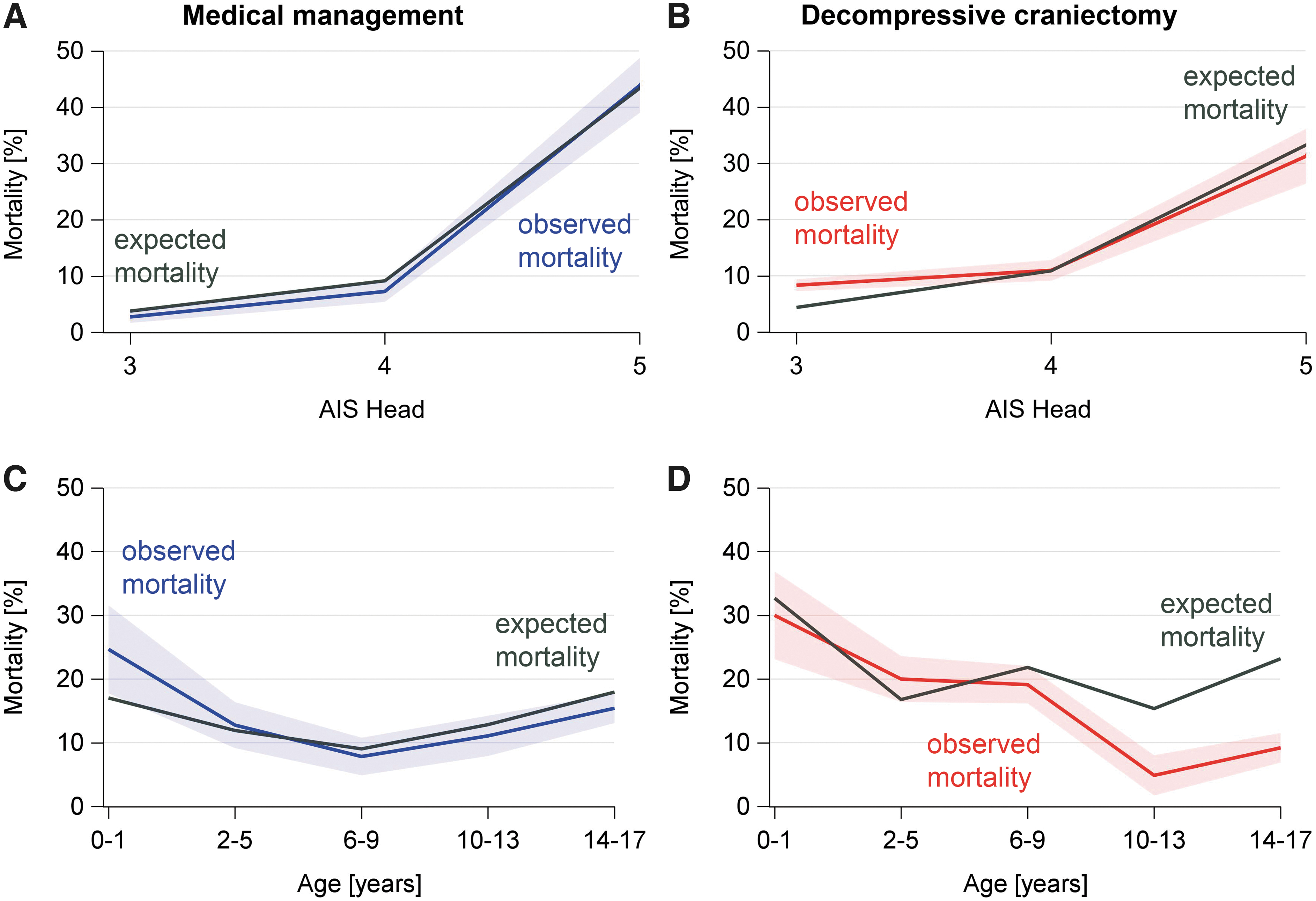
Predicted and observed mortality for medical management and decompressive craniectomy. The predicted mortality was calculated based on the revised injury severity classification (RISC II Score) as the risk of mortality for each individual. Then the group mean was calculated for each subgroup to be presented in this graph (gray line).
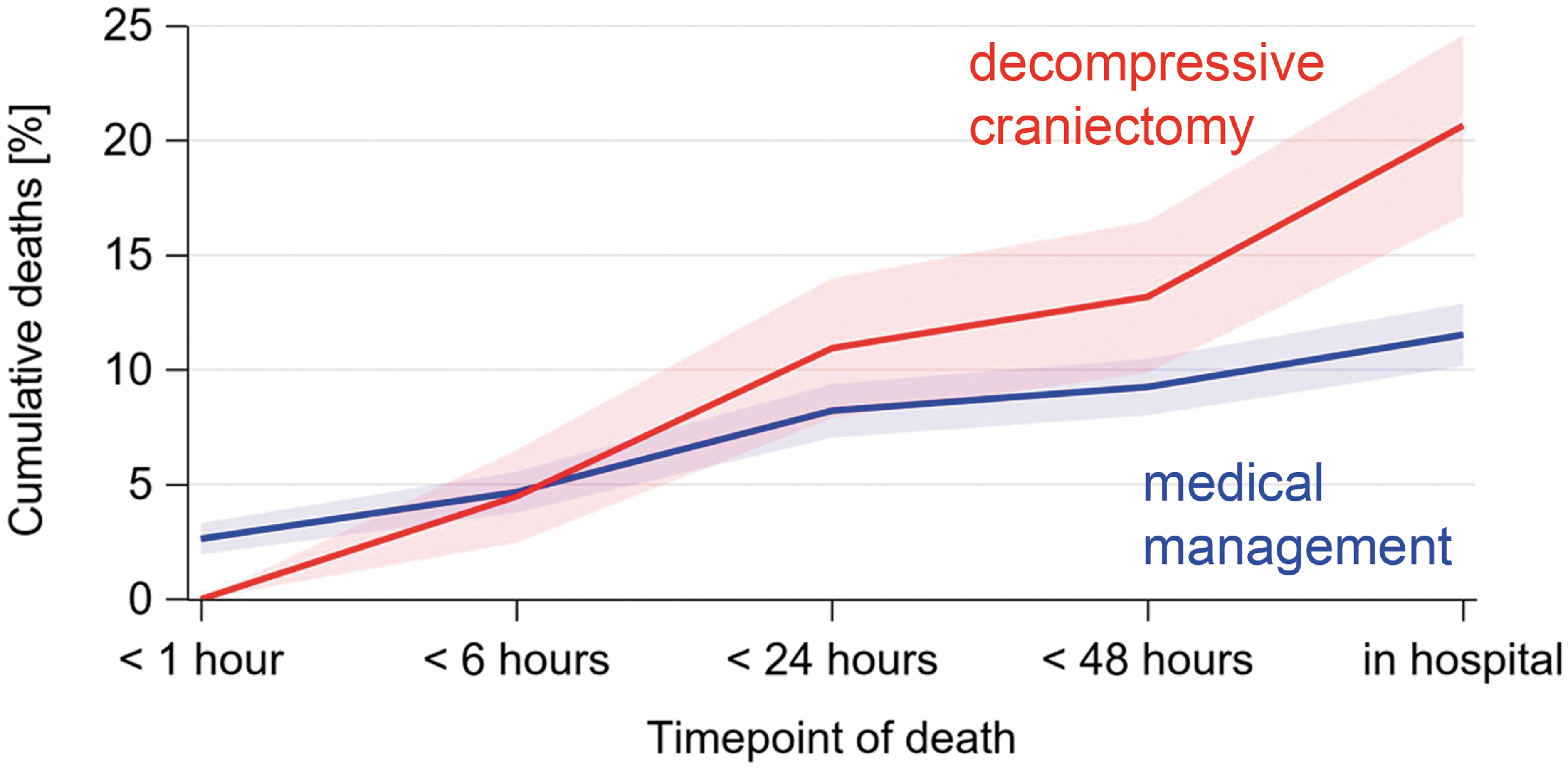
Cumulative deaths by time after the accident. Colored bands: 95% confidence intervals. Color image is available online.
Infants and adolescents had the highest predicted and observed mortality across groups and when receiving medical management (Fig. 3C). In patients between 6 and 17 years of age undergoing DC, the observed mortality was lower than predicted (Fig. 3D).
Combined outcomes
The percentage of favorable outcomes was higher in children with lower AIS head scores (Table 3; Fig. 5). For AIS head 3 and 4, poor outcomes and mortality were more frequent in children undergoing DC compared with medical management, whereas in children with AIS head 5, better outcomes and lower mortality were observed after DC (Fig. 3 and Fig. 5; Table 3).
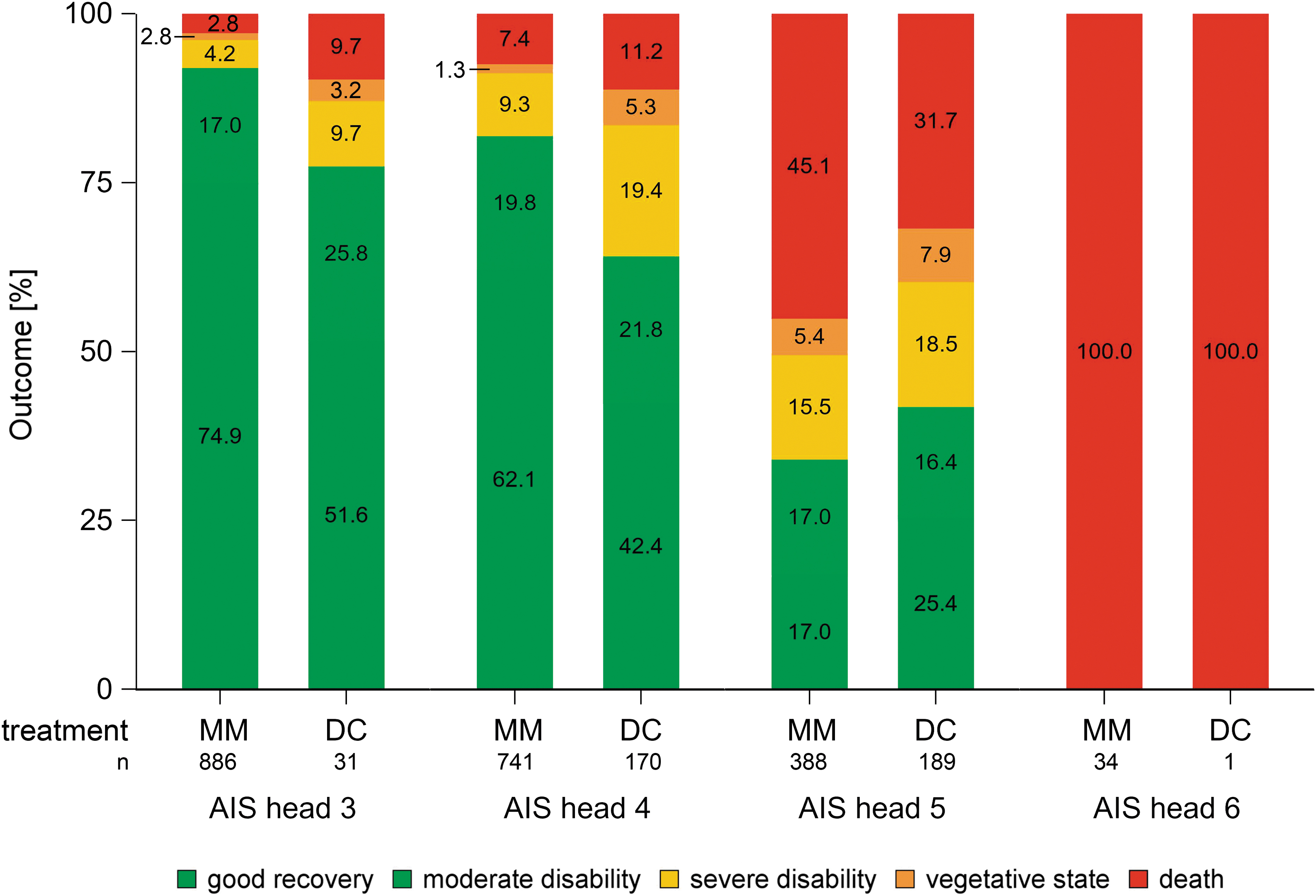
Mortality and outcomes by severity of head injury. MM, medical management; DC, decompressive craniectomy; AIS, Abbreviated Injury Score. Color image is available online.
Multi-variable analyses
Logistic regression was performed only for patients with complete data sets (n = 2389 [95.3%] for mortality and n = 2332 [93.0%] for poor outcome). There were no missing data on mortality, while information on poor outcome was missing in 67 (2.7%) datasets. The remaining exclusions were due to missing data on transport and blood transfusions. We found increased odds ratios for poor outcome (1.56, 95%, CI 1.01-2.40) and mortality (1.20, 95% CI 0.74-1.95, not significant) in children undergoing DC (Fig. 6). Children who received a cerebrospinal fluid (CSF) drain or evacuation of hemorrhage had lower odds for poor outcome (OR 0.57, 95% CI 0.37-0.86) and mortality (OR 0.39, 95% CI 0.25-0.63). Brain edema and age younger than 6 years were independently associated with higher odds for poor outcome and mortality. Intracerebral hemorrhage increased odds for poor outcomes. Epidural hematoma was independently associated with both lowered ORs for poor outcomes and mortality. Comprehensive results of the logistic regression are presented in Figure 6.
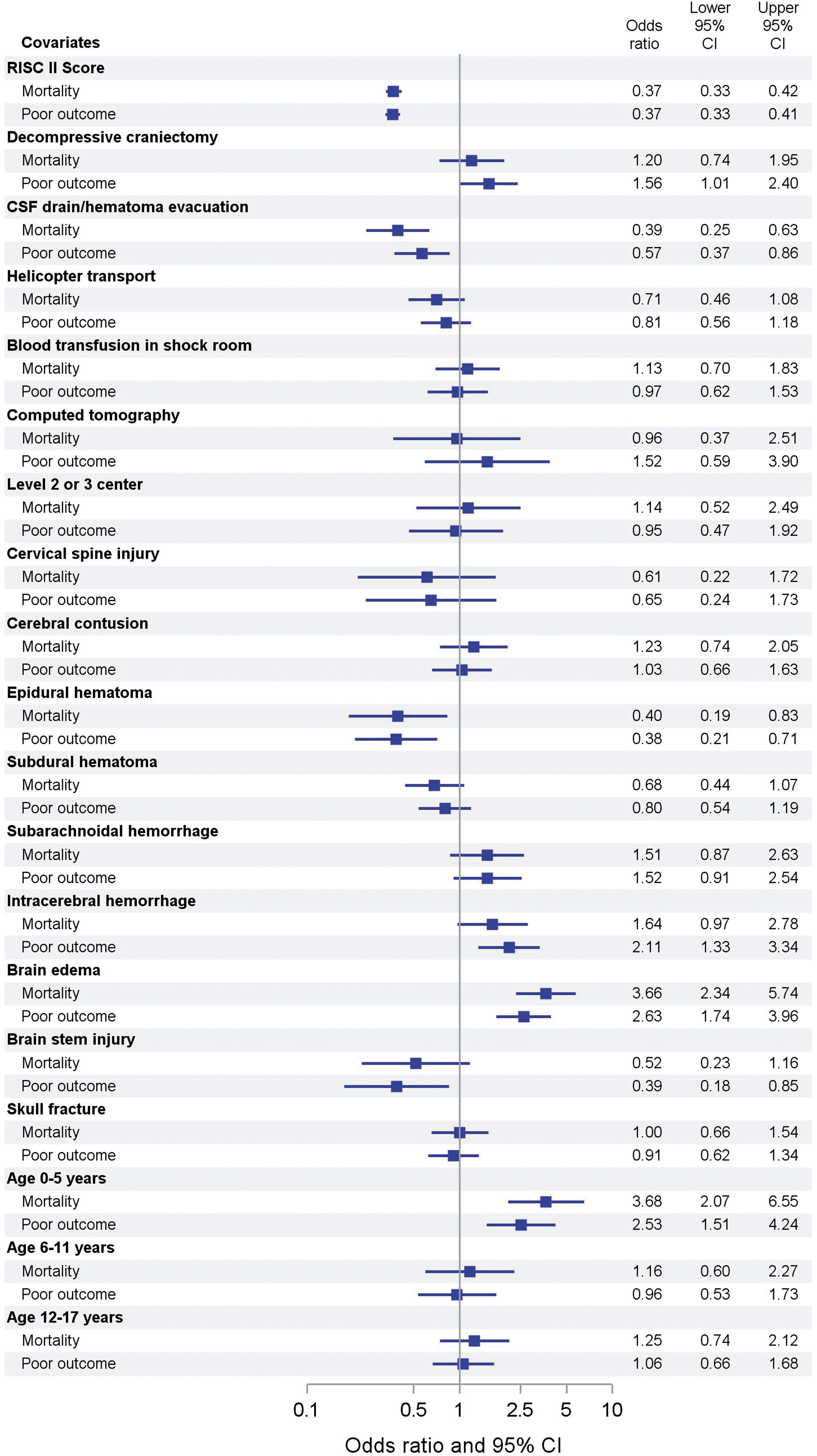
Adjusted odds ratios for mortality and poor outcomes. RISC, Revised Injury Severity Classification; CSF, cerebrospinal fluid drain. Color image is available online.
Discussion
This large retrospective study on decompressive craniectomy in children with severe TBI found increased adjusted odds for death and poor outcome in children undergoing DC compared with medical management. Children receiving decompression by CSF drain or evacuation of hematoma had lower odds for death or poor outcome.
The age distribution in our study concurs with the typically described incidence peaks during infancy and adolescence for the occurrence of and mortality from TBI. 1,22 The initial GCS values are in line with findings from other reports on children undergoing DC for severe TBI. 23,24 Age younger than 6 years was an independent risk factor for poor outcomes and death in the logistic regression. Manfiotto and colleagues reported that of 150 children undergoing DC, those with poor outcome were significantly younger at the time of the accident. 25 Age-specific metabolic demands and susceptibility to tissue hypoxemia may contribute to the differences in outcomes.
Several studies reported favorable outcomes several months or years after DC, with percentages varying between 50 and 100%. 4,7,20,24,25 In contrast, one study found that 38% of patients undergoing DC died and cognitive abnormalities were present in more than 50% of survivors. 23 The only pediatric RCT reported better outcomes after DC within 6 h of ICP rise compared with medical management (54 vs. 14% good outcomes).
Two other studies point in a different direction: Thomale and colleagues found no differences in functional outcomes between DC and medical management, and Kan and colleagues observed increased mortality when DC was performed due to raised intracranial pressure only. 5,9 Further risk factors, such as generalized brain edema, the site of the lesion, and timepoint of DC, likely play an important role in identifying patients who might benefit from this intervention. However, this relationship has not yet been systematically investigated due to small case numbers in the present studies.
There are several limitations to our study. No data were available on the decision-making triage leading to DC, the timing of DC, and ICP values. Possibly, there were three subgroups in our dataset: catastrophic injuries where no surgical intervention was contemplated, very severe TBI with evident brain swelling and consecutive DC, and less severe TBI where caregivers felt no need for decompression. Some of these aspects are accounted for by the RISC II score (e.g., pupil reactivity and severity of head injury according to the AIS). However, the RISC II score was developed to predict mortality after trauma and not specifically for TBI. It primarily aims to predict the risk of mortality prior to hospital admission and does not take into account consecutive clinical management or secondary insults, which play an important role in the context of TBI. For these reasons, risk adjustment was not optimal for the TBI population. A further limitation is the fact that outcomes were not assessed blindly along with the lack of information on long-term outcome. Outcomes after TBI remain dynamic during the first year after the accident. We accounted for that by adding the outcome category “severe disability” to good outcome group, but follow-up data until at least 12 months after the accident would have strengthened the study.
Of note, at the time-point of outcome assessment, no DC patient could have had their cranioplasty performed. Yet, the strength of our study is the large case number derived from an international multi-center cohort that is continuously collected, updated, documented in a standardized way, and subject to quality control. The large size of the cohort enabled stratified analyses and correcting for numerous confounding factors.
Although the overall level of evidence for practice recommendations remains low in children, DC is currently suggested to treat neurological deterioration, herniation or refractory intracranial hypertension (level III recommendation). 2,3 The aims are to control ICP, improve outcomes and reduce mortality. 2,3 After risk adjustment, DC was associated with increases in poor outcomes and mortality, especially when compared with CSF drain or evacuation of hematoma. From our data, it remains a matter of debate whether DC directly increases rates of poor outcomes or whether it is a surrogate marker of clinical deterioration. Larger prospective trials are needed to determine tolerable ICP values, subgroups of children who benefit from DC, the optimal timing, and best surgery technique. DC needs to be evaluated not only in comparison with medical management, but also with surgical alternatives such as CSF drain or evacuation of hematoma. CSF drains are relatively simple to place and offer the possibility to reduce elevated intracranial pressure to some degree. This finding deserves closer investigation, as intracranial pressure monitoring is inconsistently used and its importance has not been finally clarified. 26,27
Conclusion
High-quality evidence on the effect of DC in children is extremely difficult to obtain. In this retrospective analysis, we found increased odds ratios for poor outcome at ICU discharge (1.56, 95% CI 1.01-2.40) in children undergoing DC after severe head trauma. A large randomized controlled trial to determine long-term outcomes after DC and identify patients that should be offered DC is urgently needed. Until then, the findings of this study call for very careful decision making on DC in children.
Footnotes
Acknowledgments
The current study extracted data from the TraumaRegister DGU® according to ethical guidelines. Ethics approval was obtained from the Ethics Committee of the Medical Faculty of the University of Duisburg-Essen (21-10116-BO). A consent to participate was not necessary according to local legislation.
The original data are the property of the AUC Akademie der Unfallchirurgie and were accessed via the TraumaRegister DGU. Only aggregated data are available from the TraumaRegister DGU® for researchers who meet the criteria for access.
Authors' Contributions
Conceptualization: NB, CDS, OK. Methodology: RL, OK, MD. Statistical analyses: RL. Interpretation of data: NB, CDS, RL, UFM. Writing – original draft preparation: NB. Writing – review and editing: CDS, RL, OK, UFM, MD.
Funding Information
NB received funding from the Medical Faculty of the University of Duisburg-Essen (IFORES program) and the Stiftung Universitätsmedizin Essen.
Author Disclosure Statement
RL declares that his institution receives an ongoing support from AUC Akademie der Unfallchirurgie, the data holder of TR-DGU, which includes statistical support of data analysis from registry data.
For the other authors, no competing financial interests exist.
