Abstract
The benefits of therapeutic hypothermia (TH) in severe traumatic brain injury (sTBI) have been long debated. In 2018, the POLAR study, a high-quality international trial, appeared to end the debate by showing that TH did not improve mortality in sTBI. However, the POLAR-based recommendation to abandon TH was challenged by different investigators. In our recent meta-analysis, we introduced the cooling index (COIN) to assess the extent of cooling and showed that TH is beneficial in sTBI, but only when the COIN is sufficiently high. In the present study, we calculated the COIN for the POLAR study and ran a new meta-analysis, which included the POLAR data and accounted for the cooling extent. The POLAR study targeted a high cooling extent (COIN of 276°C × h; calculated for 72 h), but the achieved cooling was much lower (COIN of 193°C × h)—because of deviations from the protocol. When the POLAR data were included in the COIN-based meta-analysis, TH had an overall effect of reducing death (odds rate of 0.686; p = 0.007). Among the subgroups with different COIN levels, the only significantly decreased odds rate (i.e., beneficial effect of TH) was observed in the subgroup with high COIN (0.470; p = 0.013). We conclude that, because of deviations from the targeted cooling protocol, the overall cooling extent was not sufficiently high in the POLAR study, thus masking the beneficial effects of TH. The current analysis shows that TH is beneficial in sTBI, but only when the COIN is high. Abandoning the use of TH in sTBI may be premature.
Introduction
The usefulness of therapeutic hypothermia (TH) in severe traumatic brain injury (sTBI) has been long debated. In December 2018, the Prophylactic Hypothermia Trial to Lessen Traumatic Brain Injury (POLAR), a multi-center randomized controlled trial, appeared to end the debate by showing that TH did not improve the outcomes, including death rates, in sTBI. 1 However, the POLAR-based recommendation to abandon TH was challenged. 2 –4 It was especially difficult to achieve consensus regarding the use of moderate TH (deep body temperature <35°C), given that 24 of the 35 surveyed experts stated that it had a role in the treatment of sTBI with elevated intracranial pressure (Seattle International sTBI Consensus Conference, Survey 3). 5 After several rounds of voting, the routine use of moderate TH was not recommended, whereas mild TH was recommended, but only as tier-three treatment. 5,6
In our recent meta-analysis, 7 we introduced the cooling index (COIN) to quantify the extent of TH in sTBI and showed that TH was beneficial only when the COIN was high. Our results suggested that the COIN (as a measure of the magnitude of hypothermia) is an important factor in the effectiveness of TH, given that the effect of cooling on neuroprotection can depend on its overall extent determined by depth, duration, and rewarming rate. Could it be that the overall COIN was not sufficiently high in POLAR, thus masking the true potential of TH and limiting its use? To test this hypothesis, we ran a new meta-analysis, which included the POLAR data and accounted for the extent of TH.
Methods
After confirming, through the quality assessment protocol, 7 that the POLAR study met the criteria for high-quality trials (a high level of randomization), we extracted the targeted cooling parameters (viz., cooling temperature, cooling duration, and speed of rewarming), accounted for any deviations from or adjustments to them, and calculated the COIN values (see Table 1 for the formula). Based on the COIN, we included the death rates (assessed as odds ratio [OR]) reported by POLAR in the appropriate subgroup of the COIN-based meta-analysis, as described in details previously, 7 and formally analyzed this new, extended data set, which included data from 13 studies. 1,8 –19
Cooling Parameters Reported in the POLAR Study and the Calculated Cooling Index (COIN)
COIN = ΔT × t + (ΔT × ΔT/R)/2, where ΔT is the difference between normal deep body temperature (36.5°C) and the temperature reached at the end of cooling (in °C); ‘‘t’’ is hypothermia duration (in hours); and ‘‘R’’ is the rate of rewarming (in °C/h).
Publication bias was assessed with funnel plots by using the trim-and-fill method 20 and the test by Egger and colleagues 21 (see Supplementary Fig. S1). Egger's test values <0.1 were considered as indicators of a significant small-study effect. Between-study heterogeneity was tested with the Q homogeneity test (p values <0.05 were considered as indicators of significant heterogeneity) and with the I 2 statistical test, where I 2 is the proportion of total variation attributable to between-study variability (an I 2 value >50% was considered as indicating considerable heterogeneity).
Results
The COIN value would have been high in the POLAR study—if the targeted parameters (Table 1) were met. However, the targeted cooling parameters were reached in less than one half of patients receiving TH (Table 1). In the hypothermia group, 85 patients (33%) were cooled for <48 h; 27 patients (10%) never reached a deep body temperature of 35°C; and 65 patients (27%) never reached 33°C. 1 Hence, many patients in the POLAR study had a low level of COIN, and the overall level achieved in that study was only moderate (Table 1).
Based on the above, we included the POLAR data in the “Moderate” COIN subgroup of our meta-analysis (Fig. 1). For all data, including POLAR, the OR for death was 0.686 (p = 0.007), indicating that, overall, TH significantly decreased mortality in sTBI. However, a significant decrease in OR (indicating a beneficial effect of TH) was observed only in the “High” COIN subgroup (0.470; p = 0.013). ORs in subgroups with “Low” or “Moderate” cooling intensity were 0.718 (p = 0.081) and 0.846 (p = 0.533), respectively.
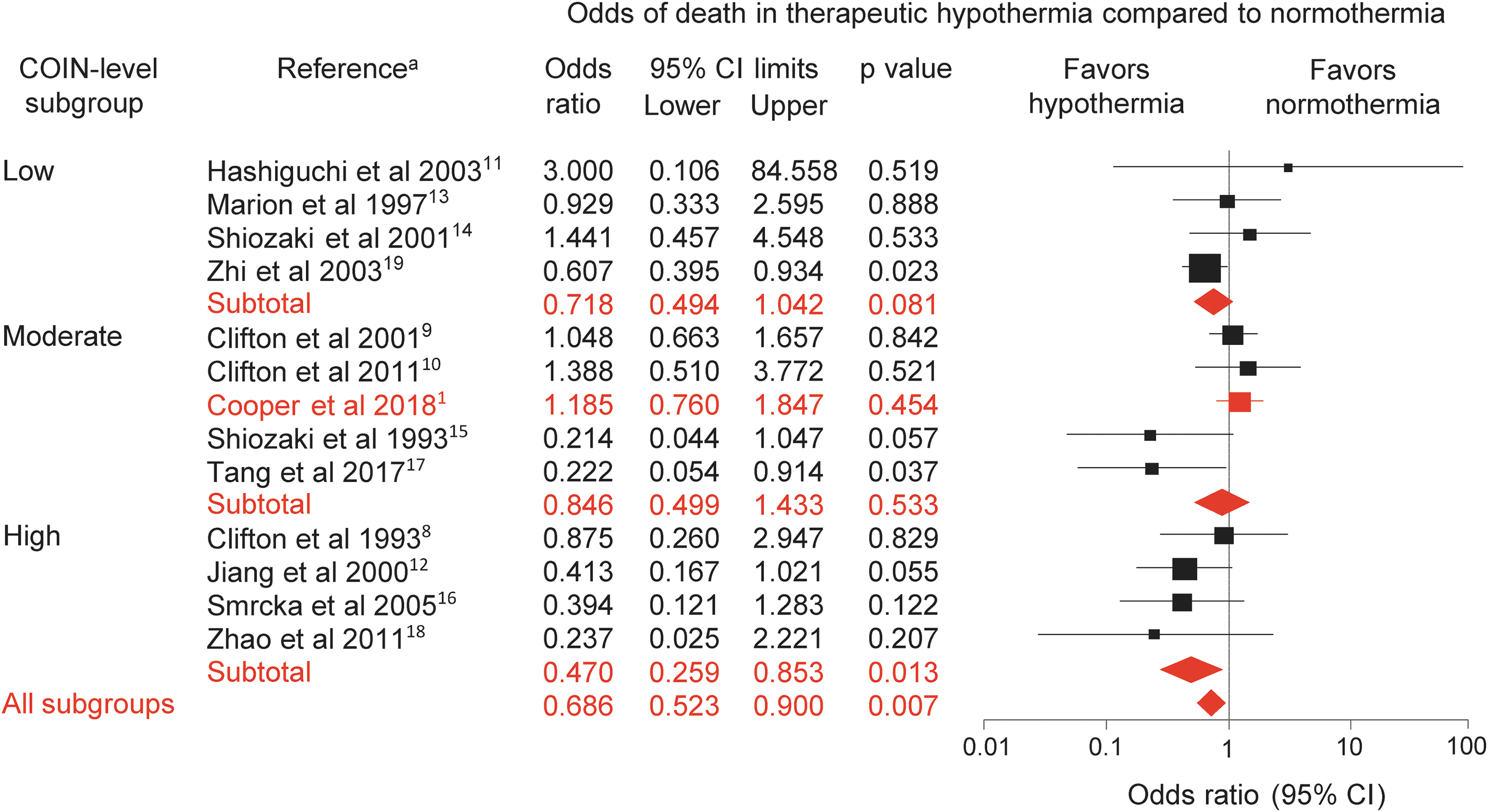
Forest plot of the effects of therapeutic hypothermia on mortality in patients with severe traumatic brain injury. The odds ratio was calculated by dividing the odds of death to survival in the therapeutic hypothermia group with the odds of death to survival in the normothermia group. A ratio <1 indicates that therapeutic hypothermia reduced the odds of death, whereas a ratio >1 indicates increased odds of death in therapeutic hypothermia. The odds ratios were compared by using a random-effects model in high-quality, randomized controlled trials divided into “Low” (<160°C × h), “Moderate” (160°C–200°C × h), and “High” (>200°C × h) subgroups based on the cooling index (COIN). All new data compared to our previous analysis 7 are highlighted in red. Note that the POLAR study (Cooper and colleagues 2018) 1 is included in the “Moderate” COIN subgroup (for details, see Table 1). aFull references to the analyzed studies can be found in the list of references. CI, confidence interval. Color image is available online.
Between-study heterogeneity was relatively small, as indicated by the Q homogeneity test (Q = 19.1; p = 0.085) and the I 2 statistical test (I 2 = 37.3%). Neither of the used assessment methods indicated the presence of publication bias (Supplementary Fig. S1).
Discussion
Including the POLAR results in our COIN-based meta-analysis strengthened our former conclusion that TH has a significant beneficial effect on death rate in sTBI, but only when COIN is sufficiently high. The benefits of TH were also shown in the recent review by Moore and colleagues. 22 The authors applied the umbrella review methodology to several potentially low-value clinical practices and found that TH was the only one with evidence of benefit. However, the POLAR study was not included in any of the systematic reviews analyzed in the umbrella review by Moore and colleagues. 22
When the results of POLAR were translated into treatment guidelines preceding the present work, 5,6 the absence of an overall beneficial effect led to the recommendation to reduce the use of TH in sTBI. However, some deviations from the cooling protocol occurred at different POLAR-participating centers and decreased the overall extent of cooling from “High” (targeted) to “Moderate” (overall achieved) and even “Low” (observed in many patients). This decrease in the COIN was likely to mask the benefits of TH in the overall cohort. It would be desirable to analyze separately the outcomes in those patients who fully met the targeted cooling criteria and those who did not. Until such results are available from the POLAR group or obtained in other high-quality trial(s), it may be premature to abandon the use of TH in sTBI.
Our meta-analysis included four multi-center randomized controlled trials, 1,9,10,14 whereas the remaining nine studies were single-center randomized controlled trials. 8,11 –13,15 –19 It should be mentioned that intervention effects for binary outcomes (such as mortality) were shown to be, on average, larger in single-center randomized controlled trials than in multi-center trials. 23 With regard to TH, some differences between results from single-center versus multi-center trials were noticed previously. 24 The differences were explained by the difficulty of keeping all parameters that influence the outcome constant across the centers in multi-center trials. Indeed, several significant intercenter differences (e.g., in disease severity, drug selection, drug doses, and personnel experience) were found in a careful analysis of a multi-center trial. 24
In our earlier report, 7 we noted that large multi-center trials are often considered to have higher quality than single-center trials when pharmacological treatments are investigated. When a complex intervention is involved, and the precise execution of this intervention is crucial (as in the case of TH), then different protocols used in different centers can lead to heterogeneous results, which can mask the differences between the treated and control groups. It should be also mentioned that the adherence to the TH protocol, including all the surrounding management, can be more closely monitored and controlled in single-center studies than in large multi-center randomized trials.
Given that positive results of single-center trials were occasionally contradicted when tested in multi-center settings, some authors concluded that physicians should apply the findings of single-center trials only after careful evaluation of their methodology. 25 It should be also noted, however, that the only way to avoid publication bias is to base meta-analyses on as complete collections of studies as possible. 26 Publication bias (i.e., the possibility of missing studies) must be always addressed according to the PRISMA guidelines, 27 which were followed in our study. In our original review protocol (registration no.: CRD42017056535), we aimed at including all available studies in the meta-analysis, without limitations to the study type—in order to achieve the most comprehensive review of the topic and avoid publication bias. This approach produced a heterogeneous set of trials with regard to both clinical and statistical designs. To reduce the heterogeneity, we used a novel approach: We extended the conventional study selection protocols by the detailed evaluation of statistical, clinical, and methodological design aspects. 7
As a result, we identified a group of 12 studies that were homogenous with regard to all three design aspects. Importantly, the POLAR study also fulfilled the inclusion criteria and did not increase heterogeneity in the present analysis. Moreover, the presence of any sizable publication bias was successfully avoided (Supplementary Fig. S1). Based on the above, we believe that the inclusion of both single-center and multi-center studies in our analysis is justified: This is the only way to conduct the most extensive analysis of the available data while minimizing the risk of publication bias.
Conclusion
In conclusion, including the POLAR study in our COIN-based meta-analysis suggests that the COIN should be flipped again to settle the dispute on the use of TH in sTBI.
Footnotes
Funding Information
This work was supported by the Hungarian National Research, Development and Innovation Office (grant FK 124483), the New National Excellence Program of the Hungarian Ministry for Innovation and Technology (grants UNKP-20-3-II-PTE-877 and UNKP-20-5-PTE-736), and the Janos Bolyai Research Scholarship of the Hungarian Academy of Sciences.
Author Disclosure Statement
No competing financial interests exist.
Supplementary Material
Supplementary Figure S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
