Abstract
While chronic neurological effects from concussion have been studied widely, little is known about possible links between concussion and long-term medical and behavioral comorbidities. We performed a retrospective cohort study of 9205 adult patients with concussion, matched to non-concussion controls from a hospital-based electronic medical registry. Patients with comorbidities before the index visit were excluded. Behavioral and medical comorbidities were defined by International Classification of Diseases, Ninth and Tenth Revision codes. Groups were followed for up to 10 years to identify comorbidity incidence after a concussion. Cox proportional hazards models were used to calculate associations between concussion and comorbidities after multi-variable adjustment. Patients with concussion were 57% male (median age: 31; interquartile range [IQR] = 23–48 years) at enrollment with a median follow-up time of 6.1 years (IQR = 4.2–9.1) and well-matched to healthy controls. Most (83%) concussions were evaluated in outpatient settings (5% inpatient). During follow-up, we found significantly higher risks of cardiovascular risks developing including hypertension (hazard ratio [HR] = 1.7, 95% confidence interval [CI]: 1.5–1.9), obesity (HR = 1.7, 95% CI: 1.3–2.0), and diabetes mellitus (HR = 1.8, 95% CI: 1.4–2.3) in the concussion group compared with controls. Similarly, psychiatric and neurological disorders such as depression (HR = 3.0, 95% CI: 2.6–3.5), psychosis (HR = 6.0, 95% CI: 4.2–8.6), stroke (HR = 2.1 95% CI: 1.5–2.9), and epilepsy (HR = 4.4, 95% CI: 3.2–5.9) were higher in the concussion group. Most comorbidities developed less than five years post-concussion. The risks for post-concussion comorbidities were also higher in patients under 40 years old compared with controls. Patients with concussion demonstrated an increased risk of development of medical and behavioral health comorbidities. Prospective studies are warranted to better describe the burden of long-term comorbidities in patients with concussion.
Introduction
Concussion, or mild traumatic brain injury (TBI), is a major public health problem with high rates in contact sports and after falls, particularly in the elderly. 1 Concussions may have a wide range of signs and symptoms, including sleep disorders, memory impairment, headaches, dizziness, impulsivity, and depression. Additional accumulating evidence indicates that brain injury may trigger progressive degenerative processes affecting brain health and other organ systems. 2,3
The long-term health outcomes of repetitive mild TBI have gained extensive public attention recently, with epidemiological and pathological studies of military veterans and professional American-style football players reporting chronic multi-system affliction and increased mortality after injury. 4 –6 In addition to the consequences of repetitive head injury, psychiatric and medical comorbidities have been self-reported by patients after a single moderate or severe TBI. 7,8 Among those with severe brain injury, chronic comorbidities can increase healthcare costs, complicate the course of recovery, and increase mortality. 7 Despite the high prevalence of concussion (mild TBI), little is known about long-term medical and behavioral sequelae post-injury.
While the acute neurological effects of concussion often resolve spontaneously over relatively short periods, long-term effects of concussion on psychiatric, cerebrovascular, and cardiovascular comorbidities are uncertain. Lack of long-term tracking and follow-up after TBI could be contributing to this uncertainty. 9 Studying the timing of the onset of comorbidities, relevant risk factors, and underlying mechanisms are important to identify at-risk patients and understand the late life decline and earlier death after mild and severe TBI. 10,11
We present results from a large observational age-, race-, and sex-matched cohort study up to a 10-year period to (1) assess the risk and time to cardiovascular, psychiatric, endocrine, and cerebrovascular diagnoses in patients with concussion compared with non-concussed healthy controls, and (2) evaluate the association between age and loss of consciousness on development of comorbidities.
Methods
Study design
In this retrospective cohort study, data were derived from the Mass General Brigham Research Patient Data Registry (RPDR) that captures all inpatient, outpatient, and emergency department (ED) diagnoses. The Mass General Brigham Institutional Review Board approved this study.
Patient selection
Patients aged 18 years or older who had at least one inpatient or outpatient visit for concussion between 2000 and 2015 and subsequently at least one inpatient or outpatient follow-up visit between 2000–2019 were included. Controls were identified using RPDR by selecting patients with the same age, sex, and race distribution as the concussion cohort. Controls with a history of TBI were excluded. The index date for concussion patients was concussion diagnosis date. For controls, index date was a random hospital or outpatient clinic encounter in the system. Age was calculated at time of first concussion diagnosis or index date for controls. All concussion patients and controls with any comorbidities of interest before the TBI or index date were excluded. The remaining patients and controls were followed for up to 10 years to assess the risk of comorbidities over time.
Exposure
Concussion was identified by International Classification of Diseases, Ninth and Tenth Revision (ICD-9/10) codes according to the Centers for Disease Control and Prevention (CDC) criteria. 12 The cause or injury resulting in the TBI was derived from the ICD external cause of injury codes (ICD-E), 13 including falls, motor vehicle accident (MVA), struck by/against, assault, and other/unspecified.
Comorbidities
Comorbidities included: psychiatric disorders (depression, anxiety, psychosis, bipolar disorder, sleep disorder, suicide ideation/intent/attempt); substance abuse (drug abuse, opioid abuse, alcoholism); neurological disorders (ischemic stroke, Alzheimer disease, vascular dementia, epilepsy/post-traumatic seizures); cardiovascular disease (CVD) and risk factors (hypertension, hyperlipidemia, obesity, coronary artery disease); and endocrine disorders (thyroid, pituitary, adrenal, diabetes mellitus); erectile dysfunction.
Comorbidity diagnoses were made during subsequent outpatient or inpatient encounters. Comorbidities were identified based on ICD9/10 codes on the first date of diagnosis (Supplementary Table 1). 14 We assessed the risk and determined the time to development of each comorbidity after concussion and index date as the date when each comorbidity first appeared in the medical record. We also evaluated the impact of age and documented loss of consciousness (LOC; as a surrogate for injury severity or vulnerability 15 ) on the incidence of medical and mental health comorbidities post-concussion.
Baseline Characteristics of Concussion Patients and Age, Sex, and Race-Matched Controls
MVA, motor vehicle accident; IQR, interquartile range.
To ensure concordance between electronic records and physician or specialist diagnosis, a trained abstractor performed manual chart review on a randomly selected 5% of the ICD codes of all comorbidities obtained from electronic medical records (EMR).
Statistical analysis
As appropriate, data were summarized by count and proportion, or median and interquartile range (IQR). We used Cox proportional hazards models to examine associations between concussion and diagnosis of mental health or medical conditions in a cohort with uneven follow-up. Patients with concussion and controls were censored at the time of comorbidity diagnosis, death, or last encounter in RPDR, whichever came first. We used Schoenfeld residuals to test the proportional hazards assumption. Interaction terms that included a time variable were added when the model did not initially meet the proportional hazards assumption.
To explore encounter bias and to reduce the likelihood that increased medical surveillance accounted for the increased incidence in patients with concussion, we excluded those with only a single encounter in the EMR system. Using this restricted dataset, we evaluated associations between all studied comorbidities in patients with concussion and controls. 16 To examine bias introduced by differences in the number of encounters before the comorbidity diagnosis in controls versus patients with concussion, we also compared the median number of encounters before diagnosis in both the concussion and control groups. We also separately investigated whether patients with concussion evaluated in the inpatient-only or outpatient-only setting for their first concussion encounter carried a higher risk for comorbidities that could produce bias.
We examined incident comorbidities in patients with concussion compared with controls after stratifying by age and LOC status. Because of loss of effective matching in these analyses, the age-stratified analyses were adjusted for sex and race, and LOC-stratified analysis was adjusted for age, sex, and race. Kaplan-Meier plots were used to describe the cumulative incidence rates of each comorbidity with log-rank test to compare risk difference between concussion and control groups. Differences in associations between concussion and comorbidities by age were examined by assessing multiplicative interaction terms in Cox models. Statistical analyses were performed using R v.3.6.1. 17
Results
We identified a total of 22,855 patients with concussion between 2000 and 2015. Of these, 4440 patients under 18 years of age and 9210 patients with histories of previous comorbidities were excluded. The remaining 9205 patients with concussion without a history of comorbidities at the time of concussion (median age = 31, IQR: 23–48 years, 57% male, 72% White) were matched with 9188 age-, sex-, and race-matched controls (median age = 33, IQR: 25-48 years, 57% male, 73% White). The majority (64%) of the patients were in the 18-40–year age group. Falls were the most common cause of concussion, accounting for 27% of cases, followed by MVA (21%). Most (83%) concussions were evaluated in outpatient settings, and 5% were inpatient. Controls were well-matched to concussion cases on age, sex, and race, but had a smaller number of healthcare visits over the follow-up period (Table 1).
Risk of multi-system comorbidities post-concussion
Compared with controls, patients with concussion had significantly higher risk of psychiatric disorders including depression (hazard ratio [HR] = 3.0, 95% confidence interval [CI]: 2.6–3.5), anxiety (HR = 2.8, 95% CI: 2.5–3.3), and psychosis (HR = 6.0, 95% CI: 4.2–8.6; Table 2). Risk of sleep disorders (HR = 2.8, 95% CI: 2.3–3.4), substance use disorder (HR = 3.5, 95% CI: 2.5–4.9), opioid abuse (HR = 3.1, 95% CI: 2.1–4.4), and alcoholism (HR = 3.4, 95% CI: 2.6–4.6) were significantly higher after concussion compared with controls within up to 10 years of follow-up.
Hazard Ratios for Multi-System Comorbidities Post-Concussion Compared with Control, Matched on Age, Race, and Sex
HR, hazard ratio; CI, confidence interval; TIA, transient ischemic attack.
Significance denoted by * < 0.05, ** <0.01, *** <0.001
Reference = Control group
The incidence of CVD risk factors including hypertension (HR = 1.7, 95% CI: 1.5–1.9), hyperlipidemia (HR = 1.7, 95% CI: 1.4–1.9), obesity (HR = 1.7, 95% CI: 1.3–2.0), and coronary artery disease (HR = 1.9, 95% CI: 1.4–2.4) were significantly higher in the concussion group compared with controls. Among endocrine disorders, diabetes mellitus risk was higher in the concussion cohort compared with controls (HR = 1.8, 95% CI: 1.4–2.3).
Because there was a significant difference in the total number of encounters between the concussion and control groups (Table 1), we explored the potential effects of encounter bias. We calculated the median number of encounters for concussion and control groups before the diagnosis of each comorbidity. There were no significant differences between the median number of medical encounters before diagnosis for all but one comorbidity (thyroid disorders; p = 0.013). The difference between encounter frequency was almost entirely because of more frequent visits for the concussion group than controls after comorbidity diagnosis (Supplementary Table 2).
Sensitivity analyses restricted to all patients with greater than two follow-up visits and to all patients seen in the outpatient setting for their index visit demonstrated the same statistically significant associations between concussion and comorbidities as seen in the full dataset (Supplementary Tables 3 and 4). We manually reviewed 5% of the ICD codes to ensure concordance between EMR and physician diagnosis and found that greater than 95% were concordant (data not shown).
Stratification by Loss of Consciousness and Risk of Medical and Mental Health Comorbidities Post-Concussion, Adjusted for Age, Sex, and Race
LOC, loss of consciousness; HR, hazard ratio; CI, confidence interval; TIA, transient ischemic attack.
Significance denoted by * < 0.05, ** <0.01, *** <0.001
Reference = Control group
Age stratification and risk of multi-system comorbidities post-concussion
We found significantly higher risks for psychiatric disorders such as depression and anxiety post-concussion compared with controls in all age groups (p < 0.001), except for bipolar disorder, which was not significant in older patients with concussion (age >60 years) compared with controls. Risk for psychosis was also significantly higher especially in young (age 18–40 years, HR = 4.1, 95% CI: 2.7–6.2) and middle-aged patients with concussion (age 41–60 years, HR = 15, 95% CI: 5.4-40.4) compared with controls. The notable increase in psychosis for the middle-aged group compared with younger patients was significant (interaction p value <0.001; data not shown) but not for older ages.
Middle-aged patients with concussion were also at much higher risk for substance use disorders (age 41–60 years, HR = 8.7, 95% CI: 3.5–21.8) compared with similarly aged controls (Supplementary Table 5). The HR for this group was significantly different than that of younger (interaction p value = 0.009) but not older age groups. It is important to note, however, that relatively small numbers in the control groups for these comorbidities may have led to artificially wide CIs.
Differences in the effects of concussion on the HR of many comorbidities by age group were significant for hyperlipidemia, depression, bipolar disorder, schizophrenia/psychosis, anxiety disorder, sleep disorder, suicidality, substance use disorder, and Alzheimer disease (interaction p value range: <0.001 to 0.05; data not shown).
The risk of many common chronic diseases usually associated with aging was increased even in the youngest group (age 18–40 years) post-concussion. Hypertension (HR = 1.3, 95% CI: 1.1–1.8) and obesity (HR = 1.6, 95% CI: 1.2–2.1) were significantly associated with concussion in the 18–40 years age group and in older age groups (Fig. 1a). We found similar patterns of increased risk of epilepsy (HR = 3.8, 95% CI: 2.5–5.9) and substance use disorder (HR = 2.9, 95% CI: 2.0–4.2) post-concussion in the younger age group (18–40 years, Fig. 1 b,c).
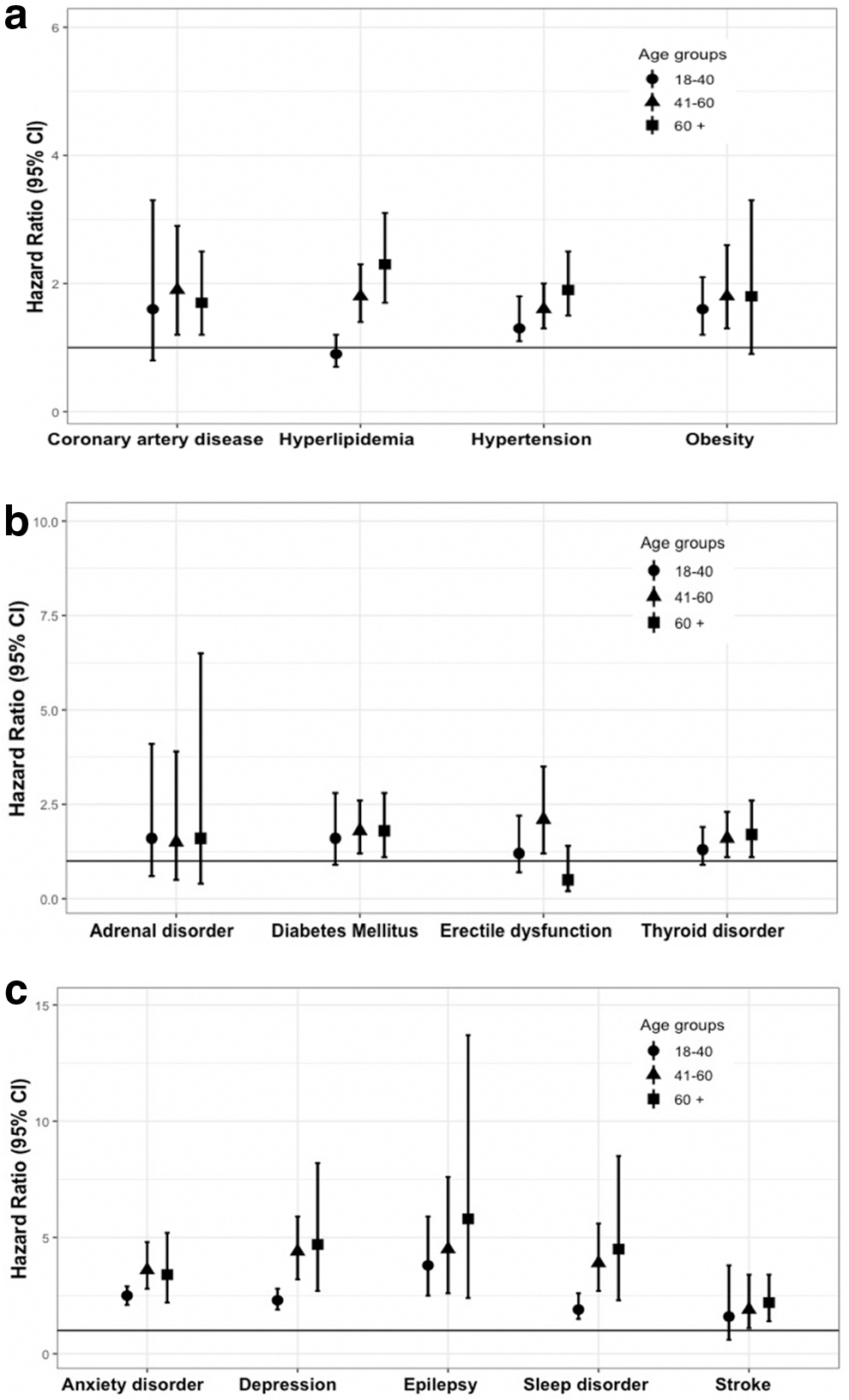
Risk trends of multi-system comorbidities after concussion by age. Figure shows (
Stratification by LOC and risk of multi-system comorbidities post-concussion
Forty-one percent of the concussion group had concussion with LOC. Compared with controls, both patients with concussion with and without LOC had more post-concussion psychiatric and cardiovascular comorbidities than controls (p < 0.001). Patients with concussion who experienced LOC, however, had increased risk of psychosis (HR = 7.8, 95% CI: 5.3–11.5), suicidal ideation/attempts (HR = 5.6, 95% CI: 3.3–9.3), and substance use disorder (HR = 4.6, 95% CI: 3.2–6.6) compared with controls. Neurodegenerative diseases such as Alzheimer disease (HR = 2.7, 95% CI: 1.1–6.8) and vascular dementia (HR = 4.1, 95%CI: 1.2–14.0) were also more common post-LOC compared with controls (Table 3).
Time to development of comorbidities after concussion
Comorbidities across all systems developed within a median time of five years after concussion (Supplementary Table 6). Cumulative incidence risk for psychiatric, sleep, and substance use disorders, post-traumatic epilepsy, and stroke were significant post-concussion compared with controls (p < 0.005 for all comparisons, Fig. 2c–e), but not for Alzheimer disease or vascular dementia (Supplementary Fig. 1). Cumulative incidence risk for all studied CVD risk factors including hypertension, hyperlipidemia, obesity, and coronary artery disease showed significant differences compared with controls (p < 0.01 for all comparisons; Fig. 2a,b). Similarly, the trend for diabetes mellitus (p = 0.0013) was significant, but not for adrenal, thyroid, and pituitary diseases or for erectile dysfunction, compared with controls.
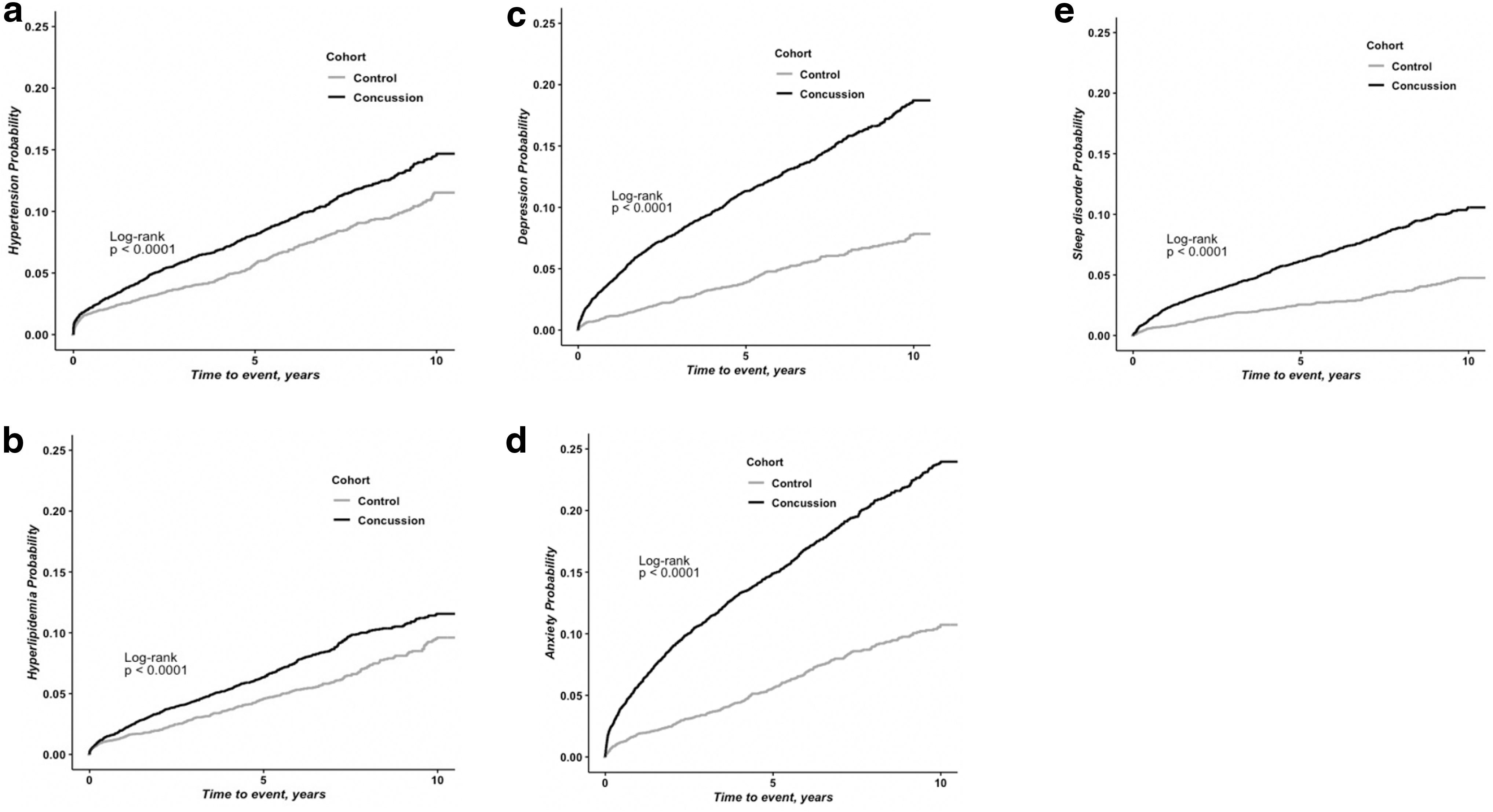
Kaplan-Meier graphs for risk of multi-system comorbidities post-concussion. Kaplan-Meier graphs show the cumulative incidence rate of multi-system comorbidities post-concussion including (
Discussion
To our knowledge, this is the first large study to examine the association between concussion and risk of chronic medical and behavioral health conditions with up to 10 years of follow-up. Compared with controls, patients experiencing concussion without pre-existing comorbidities were at increased risk of numerous chronic conditions developing including CVD, endocrine diseases, and neurological and mental health comorbidities. Higher post-concussion risks were not confined to older ages and were observed among patients as young as 18–40 years old. Most of the comorbidities developed within a median of less than five years after concussion.
Emotional disturbances, sleep and behavioral problems are common after concussion and are typically transient, but in some cases may last for months to years. 18 Similarly, we found a significant increase in reported sleep disorders and neuropsychiatric comorbidities such as anxiety and depression in all age groups after concussion. 19 Significantly different HRs found between different age groups for a number of comorbidities may be an effect of aging or may be because of underlying differences in the population strata because recruitment required having no comorbidity diagnoses at baseline. Risk of psychosis post-concussion was significantly higher in middle-aged (15-fold) and older patients (9-fold) and remained high even in patients younger than 40 (4-fold) compared with controls, although the number of events was relatively low.
Previous studies suggested a possible link between moderate and severe TBI and delayed onset psychosis, 20 but a similar association with mild TBI has not yet been reported. The impact of other factors known to increase the risk for psychosis and other neuropsychiatric comorbidities such as pre-morbid biological risk, family history, and history of previous neurodevelopmental conditions still require further investigation.
The few studies that have focused on LOC in civilian populations showed that LOC at the time of injury is predictive for depression and post-concussion symptoms in the few months post-TBI. 21,22
In our study, we observed a higher incidence of psychosis, suicidality, and substance use disorder among patients who experienced LOC compared with controls. The overall high incidence of neuropsychiatric disorders may play a role in patients' decision making and potentially increase the risk of suicide attempts, chronic substance use disorder, and even chronic comorbidities in some patients. 18,23,24 The directionality of these behavioral patterns is still not clear.
Compared with controls, we found a significant increase in CVD and CVD risk factors including hypertension, sleep disorders, and obesity after concussion even in the younger age group (18–40 years). Patients with concussion are at risk for weight gain, sleep disturbances, chronic fatigue, and reduction in functional health post-injury, which could all play a role in increasing CVD and its risk factors. 25 –28 Overall, it is still not clear whether the heightened CVD risk post-concussion is from mechanistic causes, other medical comorbidities, subsequent lifestyle changes that put patients at higher risk, or other factors.
We did not find a noticeably high risk of Alzheimer disease and other types of cognitive impairment in our cohort, even in the older age group (>60 years), possibly because our patients with concussion had a healthy baseline before concussion, having excluded patients with pre-existing medical and mental health comorbidities that may increase risk of Alzheimer disease. We found an increased risk of stroke subsequent to concussion comparable to that reported in recently published studies. 29 The exact mechanisms are not well studied, but this finding could be related to underlying worsening medical comorbidities post-injury, 20,21 as well as reported post-concussion mechanisms such as neuroinflammation, vascular dysfunction, and increased risk of thrombosis, which could also contribute. 30
Our study has several limitations. First, this is a registry database study from a single academic health system providing primary and tertiary care using structured data captured in the EMR using ICD 9/10 codes. The diagnoses relied on the accuracy and completeness of ICD diagnosis codes, which may have led to some misclassification. The manual review of hospital records, however, showed a high degree of concordance with the ICD coding. Second, we were not able to identify and exclude patients with previous exposure to sport or repetitive head injuries, nor could we identify patients with concussion who did not seek medical attention or whose medical data were stored at other facilities. This weakness is counterbalanced by the large number of patients and long follow-up time line of the study.
Third, we were unable to distinguish between the effect of single and repetitive head injuries on the risks of comorbidities given the limitations of ICD 9/10 codes and EMR structure. In addition, it is uncertain whether findings are generalizable to other cohorts including sport and military personnel. Fourth, we did not quantify concussion severity or general trauma burden, opting instead to explore LOC as a severity index in sensitivity analyses. Fifth, there may be underlying differences that place some persons at higher risk for concussion as well as at higher risk for some of the neuropsychiatric conditions studied here, which could confound data analysis and introduce bias to risk estimates.
Sixth, results could also be influenced by possible ascertainment bias given less frequent follow-up visits for controls compared with patients with concussion. In this setting, simulations have shown that simple adjustments for frequency of visits can also introduce bias. 16 We calculated the median number of encounters for concussion and control groups before the comorbidity diagnosis. While we found no significant differences between the median number of medical encounters before diagnosis for all but one comorbidity, there may be unmeasured encounter bias contributing to results. Seventh, we did not adjust for multiple comparisons despite the inclusion of many comorbidities because this was an exploratory study, and we did not want statistical penalties to inhibit discovery.
Conclusion
Although many post-concussive symptoms may resolve without treatment, concussion may have substantial, long-term consequences at any age. We have identified associations between concussion and the risk of chronic behavioral health and medical comorbidities, largely within five years after a documented concussion. Even young adults who sustain concussions are at relatively high risk for comorbid events compared with controls. Future prospective health and mechanistic studies that account for all of the above limitations are warranted. Such studies are needed to understand the biopsychosocial links and investigate the causality or directionality of these comorbidities and assess the role of medical surveillance and preventive protocols.
Footnotes
Authors' Contributions
S. Izzy, Z. Tahir, R. Grashow, D. Cote, D. Nathan, K. Miller, A. Baggish, M. Weisskopf, and R. Zafonte contributed to study concept and design. S. Izzy, Z. Tahir, R. Grashow, D. Cote, A. Al Jarrah, M. Weisskopf contributed to data acquisition and analysis. S. Izzy, Z. Tahir, R. Grashow, D. Cote, A. Al Jarrah, A. Dhand, H. Taylor, M. Whalen, D. Nathan, K. Miller, F. Speizer, A. Baggish, M. Weisskopf, and R. Zafonte contributed to interpretation of data, drafting and reviewing the manuscript. All authors provided final approval for publication.
Funding Information
A. Baggish, H. Taylor, R. Grashow, R. Zafonte, F. Speizer, and M. Weisskopf are either partially or fully supported by the Football Players Health Study at Harvard University, which is in turn sponsored by the NFLPA. The NFLPA had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication. Other authors report no potential or actual conflicts of interest.
Author Disclosure Statement
For S. Izzy, Z. Tahir, A. Al Jarrah, A. Dhaud, M. Whelan, D.M. Nathan, and K.K. Miller, no competing financial interests exist.
Supplementary Material
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
Supplementary Table S5
Supplementary Table S6
Supplementary Figure S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
