Abstract
The Glasgow Coma Scale (GCS) and pupillary reactivity are well-known prognostic factors in traumatic brain injury (TBI). The aim of this study was to compare the GCS motor score and pupillary reactivity assessed in the field and at hospital admission and assess their prognostic value for 6-month mortality in patients with moderate or severe TBI. We studied 445 patients with moderate or severe TBI from Austria enrolled to hospital in 2009–2012. The area under the curve (AUC) and Nagelkerke's R 2 were used to evaluate the predictive ability of GCS motor score and pupillary reactivity assessed in the field and at admission. Uni- and multi-variable analyses—adjusting for age, other clinical, and computed tomography findings—were performed using combinations of field and admission GCS motor score and pupillary reactivity. Motor scores generally deteriorated from the field to admission, whereas pupillary reactivity was similar. GCS motor score assessed in field (AUC=0.754; R 2=0.273) and pupillary assessment at admission (AUC=0.662; R 2=0.214) performed best as predictors of 6-month mortality in the univariate analysis. This combination also showed best performance in the adjusted analyses (AUC=0.876; R 2=0.508), but the performance of both predictors assessed at admission was not much worse (AUC=0.857; R 2=0.460). Field GCS motor score and pupillary reactivity at hospital admission, compared to other combinations of these parameters, possess the best prognostic value to predict 6-month mortality in patients with moderate-to-severe TBI. Given that differences in prognostic performance are only small, both the field and admission values of GCS motor score and pupillary reaction may be reasonable to use in multi-variable prediction models to predict 6-month outcome.
Introduction
T
The motor component of the GCS score has been identified as containing virtually all prognostic information in the total GCS score for those with severe TBI (sTBI). 3 Its use was suggested in prognostic models instead of the summary GCS score. This approach has been adopted, for example, in the International Mission for Prognosis and Analysis of Clinical Trials (IMPACT) models. 4 A further study 5 found equal sensitivity and specificity using both measures and advocated the use of the motor score alone in prehospital triage of brain-injured patients.
Besides the GCS, assessment of pupillary reactivity is another standard procedure of neurological evaluation. Acute pupillary dilatation in head-injured patients indicates a neurological emergency. 6 Traditionally, this phenomenon was thought to be caused by uncal herniation resulting from brain edema or a mass lesion, which led to compression of the third cranial nerve, which, in turn, caused the actual pupil dilatation. 7 Another mechanism for pupillary dilatation is decrease of blood flow to the brain stem and the resulting brain-stem ischemia. 6
Both GCS and pupillary reactivity possess good ability to predict outcome in patients with TBI 8 and have been widely used in TBI prognostic models. A review found that, of the identified prognostic models, 50% included GCS as a predictor and 26% included pupillary assessment. 9
An aspect that can influence the predictive value of the GCS is time of assessment. Although admission GCS has been shown to have a stronger association to outcome, compared to assessments at earlier time points, 8 its power as an outcome predictor may have been reduced significantly in more-recent patients as a result of aggressive prehospital treatment causing patients to arrive to hospitals sedated, intubated, or paralyzed. 10 Neurological assessment using any of the GCS components (motor response, eye opening, or verbal response) can become problematic in such cases. 11 Substantial differences exist between assessments in the prehospital phase, compared to in-hospital assessments. This needs to be considered when using GCS or its components and pupillary reactivity in prognostic models. 8
The aim of this study was to compare the GCS motor score and pupillary reactivity assessed in the field and at hospital admission and assess their prognostic value for 6-month mortality in patients with moderate or severe TBI.
Methods
Data
The International Neurotrauma Research Organization in Vienna has been working to improve the outcome of patients with TBI in Austria and the broader European region for over 12 years. One of its most recent studies focused on improvements of outcome of patients with TBI through improved prehospital care. Within this prospective observational study, data on patients with TBI were collected in Austria during the period of 2009–2012 (INRO-PH data set). All patients with GCS score <=12 within 48 h after the accident and/or Abbreviated Injury Scale (AIS) head >2 were included in the study. Data on demographic characteristics, injury type and severity, prehospital treatment, trauma room treatment, surgical procedures, computed tomography (CT) scans, intensive care unit (ICU)-based treatment (first 5 days) and outcome (at ICU discharge, hospital discharge and 6 months after injury) were recorded. The Extended Glasgow Outcome Scale was used to categorize the outcome at hospital discharge and at 6 months after injury.
Data were prospectively collected in 16 centers, and a total of 778 patients were enrolled. Of these, a subset of 445 patients was selected where data both on prehospital and admission assessments of the GCS motor score and pupillary reactivity was available. These patients were used for the analyses in this study.
Assessment of neurological status
Prehospital treatment within the studies was provided by emergency physicians or paramedics. The treatment protocol included rapid examination with documentation of vital signs (GCS score, pupillary status, blood pressure, heart rate, and oxygen saturation). Rapid-sequence intubation, facilitated by hypnotics and relaxants, ventilation, treatment of hemorrhage, and fluid resuscitation, were done as appropriate. After admission, each patient was examined by a trauma team (anesthesiologists, trauma surgeons, and/or neurosurgeons, radiologists, and nurses), clinical status (GCS score, pupillary status, heart rate, blood pressure, and oxygen saturation) was documented again, and a CT scan was done. Both the GCS and pupillary reaction at admission were assessed by the anesthesiologist, who was primarily responsible for the monitoring of vital signs. Patients then underwent surgery as appropriate and/or were admitted to the ICU. Given that GCS score and pupillary reactivity were assessed in all patients twice (in the field, and at admission to the hospital), it was feasible to make comparisons of neurological status assessed at both time points.
Statistical analyses
The GCS motor score and pupillary reactivity in field and at admission were compared using cross-tabulations. The differences in the distributions were tested with McNemar's test.
To evaluate the prognostic value, logistic regression models were fit using GCS motor score and pupillary reactivity assessed in field and at admission as single predictors and in different combinations, with 6-month mortality as the response variable.
The IMPACT core and IMPACT extended models 4 for prediction of 6-month mortality were refit on data to further evaluate the predictive performance of both measures. GCS motor score and pupillary reactivity assessed in field and at admission were used in various combinations (along other predictors in the IMPACT core and extended models 4 that remained unchanged) and performances were compared (see Supplementary Table 1). The area under the receiver operating characteristic curve (AUC) was calculated to evaluate the discriminative ability, and Nagelkerke's R 2 was used as a measure of predictive ability. Bootstrapping with 500 repetitions was used to correct for optimism of both these measures when refitted using the IMPACT core and extended models. Ninety-five percent confidence intervals were calculated for optimism-corrected performance measures using the standard deviation of bootstrapped measures as an indication of uncertainty.
Results
In total, 445 patients were included, with a median age of 50 years (Table 1). The CT scan showed a mass lesion in over 1 of 3 of patients, which indicates severe injuries to the brain.
IQR, interquartile range; GCS, Glasgow Coma Scale; ISS, Injury Severity Score; AIS, Abbreviated Injury Scale.
Field and admission assessments
A shift toward more-severe GCS motor score was apparent from prehospital stage to admission, whereas the pupillary reactivity remained similar at both assessments (Tables 2 and 3). A large number of patients showed relatively good motoric responses in the field, but were classified as more severe at admission assessments (Table 2 and Supplementary Table 2) (see online supplementary material at
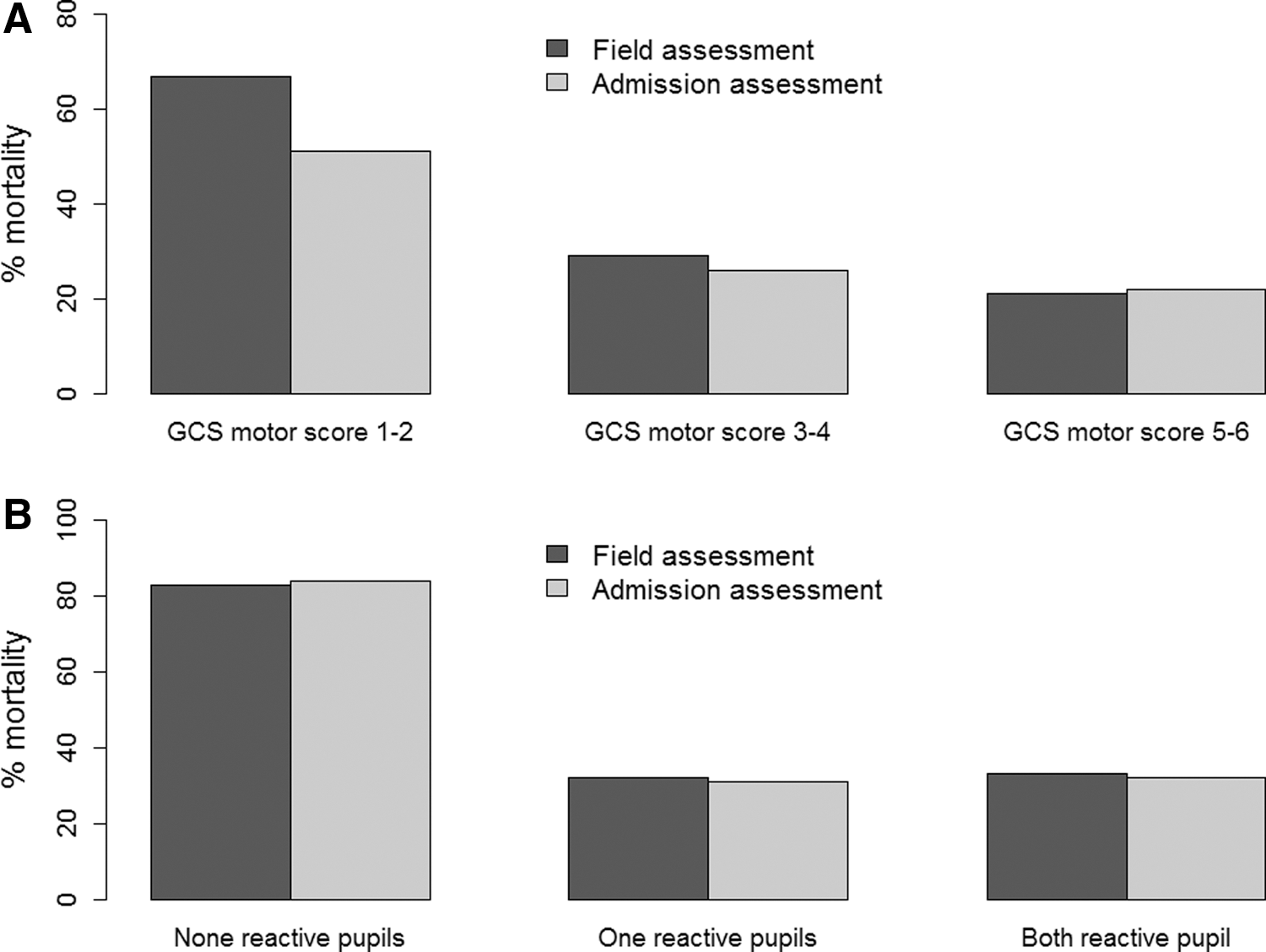
Six-month mortality in subsets of patients created based on their GCS motor score (
p<0.001 (McNemar's test).
GCS, Glasgow Coma Scale.
p=0.395 (McNemar's test).
Prognostic value
The prognostic performance of field and admission GCS motor score and pupillary reactivity in predicting 6-month mortality was assessed as a univariate model, in different combinations and using the IMPACT core and extended models. The size of effects of both GCS motor score and pupillary reactivity on the mortality decreased as more covariates were included. The odds ratios were smallest in the IMPACT extended model (Table 4). The effect of GCS motor score assessed in the field was stronger, compared to the admission assessment in the univariate analysis, but the differences were smaller after adjustment. The effect of pupillary reactivity was rather similar at both assessments in all scenarios.
Data are shown as odds ratios with 95% confidence interval.
The IMPACT core model uses age, pupillary reaction, and GCS motor score as predictors. Thus, the odds ratios are adjusted for age and pupillary reaction (when referring to GCS motor score) and for age and GCS motor score (when referring to GCS pupillary reaction).
The IMPACT extended model uses age, CT classification, hypoxia, hypotension, traumatic subarachnoid hemorrhage, and epidural hematoma as additional predictors besides pupillary reaction and GCS motor score. Thus, the odds ratios are adjusted for these parameters and pupillary reaction (when referring to GCS motor score) and for these parameters and GCS motor score (when referring to GCS pupillary reaction).
When used in IMPACT models, sets of GCS motor score and pupillary reaction values assessed at the same time point are used (e.g., field GCS motor score with field pupillary reaction, admission pupillary reaction with admission GCS motor score, and so on.).
Odds ratios were not possible to obtain because of singularity.
GCS, Glasgow Coma Scale; IMPACT, the International Mission for Prognosis and Analysis of Clinical Trials; CT, computed tomography.
The patterns of predictor effects are in line with patterns of overall prognostic performance (Figs. 2 and 3; Supplementary Table 1) (see online supplementary material at
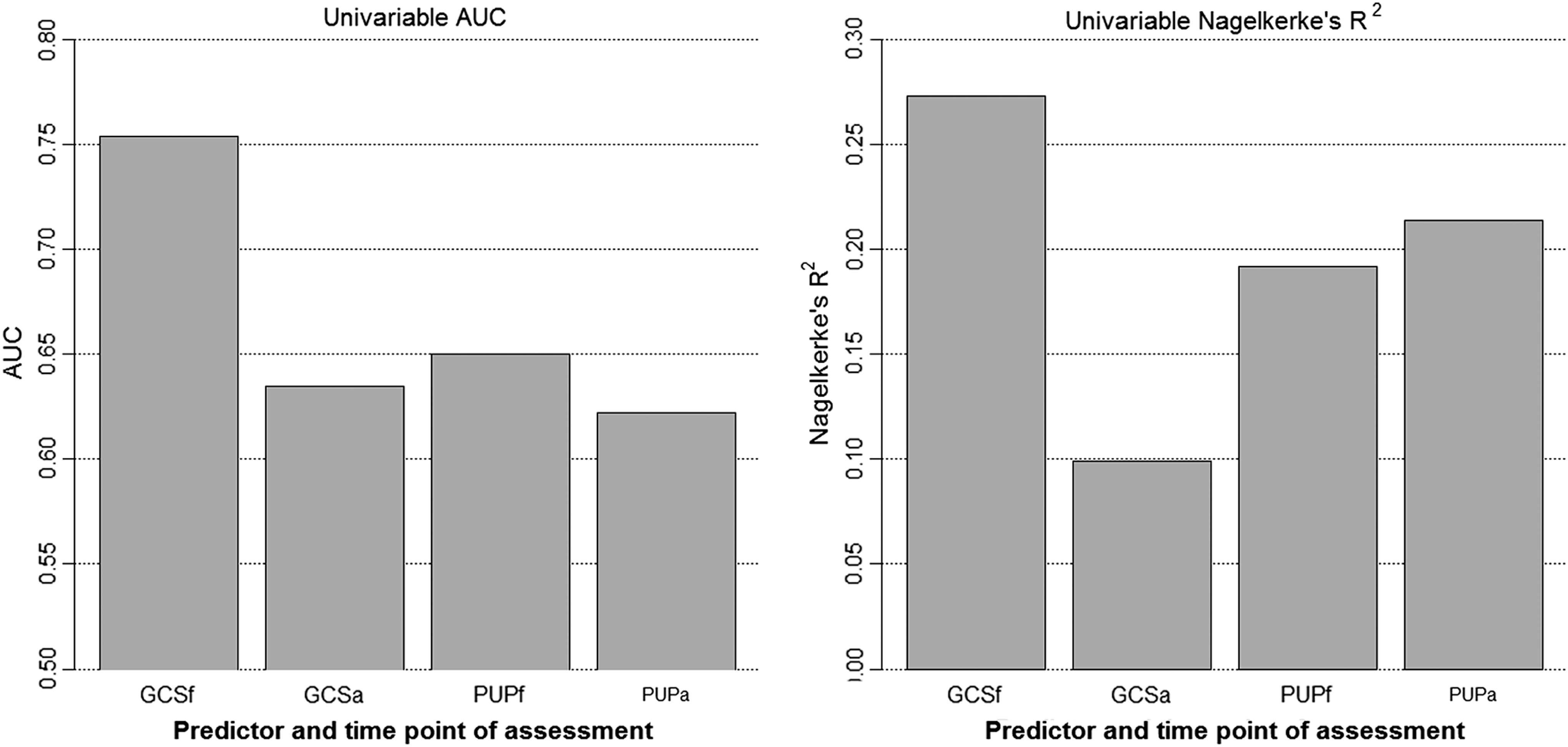
Performance of GCS and pupillary reaction assessed in field and at admission in univariate analysis. AUC, area under the receiver-operating curve; GCSf, Glasgow Coma Scale-Motor Score assessed in field; GCSa, Glasgow Coma Scale-Motor Score assessed at admission; PUPf, pupillary reactivity assessed in field; PUPa, pupillary reactivity assessed at admission.
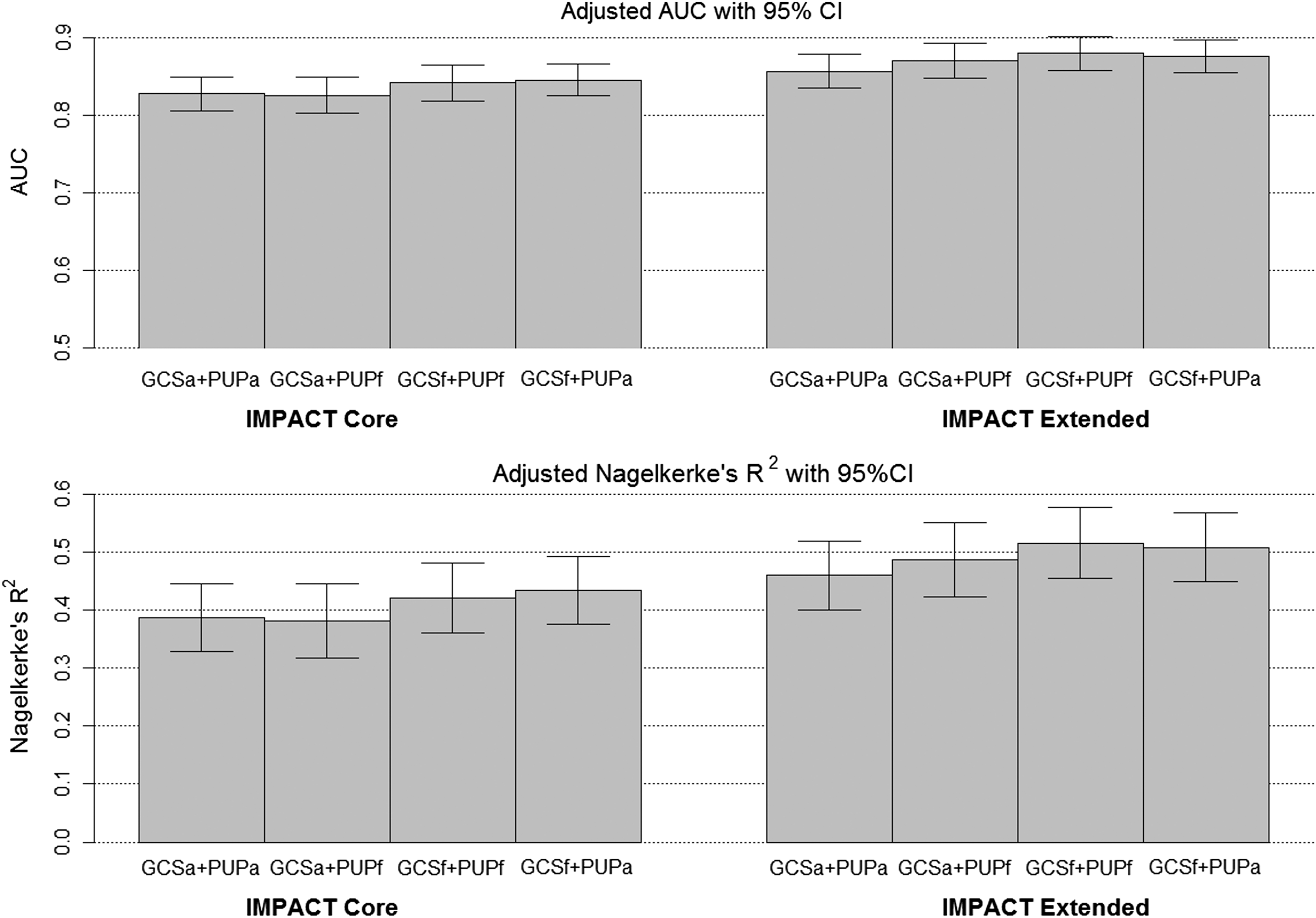
Performance of the combinations of GCS and pupillary reaction with age (IMPACT core model) and with hypoxia, hypotension, and computed tomography (CT) characteristics (IMPACT extended model). The IMPACT core models uses age, pupillary reaction and GCS motor score as additional predictors; The IMPACT extended model uses age, pupillary reaction, GCS motor score, CT classification, hypoxia, hypotension, traumatic subarachnoid hemorrhage and epidural mass as additional predictors. AUC, area under the receiver-operating curve; CI, confidence interval; GCSf, Glasgow Coma Scale-Motor Score assessed in field; GCSa, Glasgow Coma Scale-Motor Score assessed at admission; IMPACT, the International Mission for Prognosis and Analysis of Clinical Trials; PUPf, pupillary reactivity assessed in field; PUPa, pupillary reactivity assessed at admission.
To analyze and validly compare the prognostic value of different combinations of GCS motor score and pupillary reactivity, a subset of 445 patient was used in which values of these parameters assessed both in field and at admission were available. The main reason for this was to keep the sample used for all analyses unchanged, given that differing sample composition could bias our comparisons. This subset comprised 57% of the original set of 778 patients. From the practical point of view, it is important to note that some combination of field and admission assessment of these parameters was available in a higher proportion of patients, as apparent in Figure 4. In fact, at least one value for GCS motor score and pupillary reactivity was available in almost all patients. The analysis of such a combination of values yielded an AUC of 0.872 and an R 2 of 0.506 when used in the IMPACT extended model, which suggests prognostic performance similar to other combinations. However, direct comparisons of this scenario to the scenarios primary analyzed in this article must be cautious owing to possible bias introduced by differing sample composition.
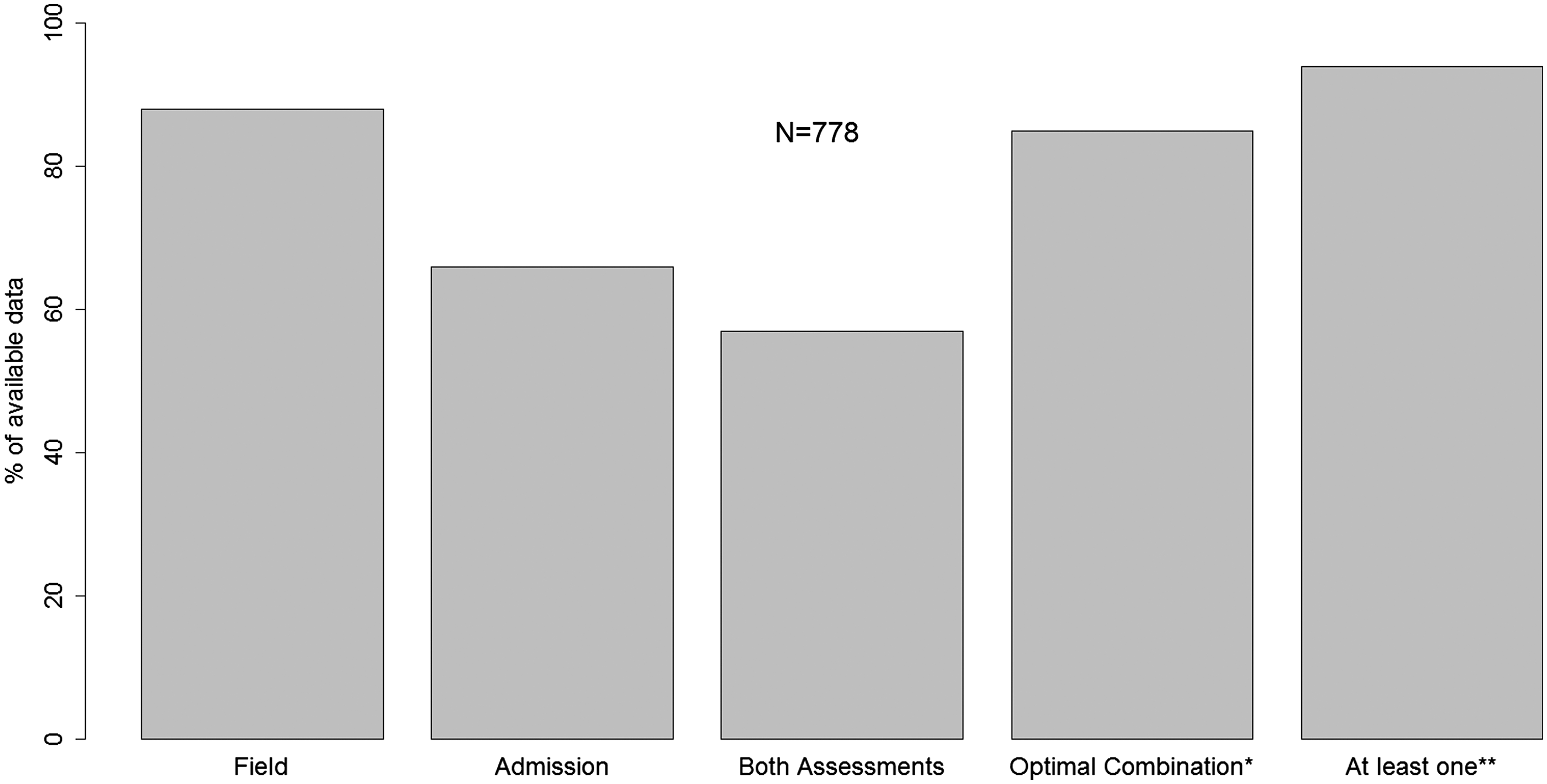
Availability of the set of GCS motor score and pupillary reaction values at different points of assessment. *Based on our analysis, this is the combination of GCS motor score assessed in field and pupillary reactivity assessed at admission, which had the best prognostic performance of all combinations analyzed. **Refers to the availability of either the set of values assessed in field or the set of values assessed at admission.
Discussion
Main findings
The aim of this study was to compare the GCS motor score and pupillary reactivity assessed in the field and at hospital admission and assess their prognostic value for 6-month mortality in patients with moderate or severe TBI. Our study revealed the dynamic nature of mainly GCS motor score and less of pupillary reactivity assessed at different stages after injury. As to predictive ability, our study showed that the combination of field GCS motor score and pupillary reactivity at hospital admission used in the IMPACT core and extended models allows for the best prediction of 6-month mortality. Using other combinations of GCS motor score and pupillary reactivity resulted in slightly worse prognostic performance, and therefore both the field and admission values are reasonable to use in multi-variable prediction models.
Field and admission assessments
The GCS motor score is a standardly used measure of neurological status. It is assessed at different time points after injury in patients with TBI. Significant differences between such assessments may exist. A study found that the agreement between the prehospital and emergency department (ED) GCS motor score scores was highest for mild and severe scores on the 1–6 scale, with agreement between moderate scores practically nonexistent. 12 We confirm these findings, whereas most agreements in our study were found at the extremes of the GCS motor score scale with little agreement in the range of scores 2–4.
Other studies compared the summary GCS in field and at admission. A reasonably strong correlation was typically found between values of prehospital and admission summary GCS scores (e.g., r 2=0.67). 13 Significant correlation was found also in another study, 14 and correlating field and admission summary GCS scores in our data yielded similar results: We found a correlation of 0.58 for prehospital and admission summary GCS scores and 0.51 for field and admission GCS motor scores. These findings indicate that the GCS motor score is rather dynamic and depends on the time point after injury.
One of the reasons for inconsistencies between GCS scores in field and at admission could be the problematic neurological assessment in patients arriving at the ED: intubated, paralyzed, or sedated. 8,11,15 In our data, the mean decrease of summary GCS score from field to admission was 1.7 points (p<0.001). A similar study reports a more significant decrease (5 points) 16 —however, this can be explained by inclusion of milder cases of TBI in their sample compared to our study. Despite the dynamics of the summary GCS from field to admission, the motor component of the GCS is the one that is most likely to be assessable even for patients where the full GCS score is difficult or impossible to obtain. 15 In our study, the mean decrease of GCS motor score from field to admission in intubated patients was 1 point (p<0.001) and in nonintubated patients 0.3 points (p=0.041), which suggest that field intubation (and related sedation or paralysis) could be one of the reasons for lower GCS motor scores at admission.
Inter-rater disagreement between persons performing the assessment in field and at the ED can be another cause of discrepancies and these can be a result of less experience of the assessor. 15 In our study, it has been performed by experienced emergency physicians or paramedics in the field and by an anesthesiologist at admission; therefore, we assume that all assessments are correct. This presumption is supported by similar studies, 13,17 which suggests that paramedics can accurately assess field GCS.
Given that this study was not primarily designed to compare the prognostic value of GCS motor score and pupillary reaction assessed at different time points after TBI, no specific measures were taken to tackle inter-rater disagreements. Therefore, some of the changes in time might be attributed to this factor. However, because all assessments were performed by experienced professionals, we are confident that this factor does not largely influence our results.
Contrary to the GCS, pupillary reactivity does not change significantly across assessments and thus can be considered a robust prognostic factor. 8 Additionally, pupillary reactivity may be a more objective parameter of patient status given that it is less likely to be biased by poor patient compliance. 18 Variation in pupillary reactivity at different points after injury could be a result of brain-stem flow, which could vary according to blood pressure changes if autoregulation is compromised. 8 In our study, the reactivity from field to admission remained unchanged in 84% of patients, improvement was apparent in 9%, and reactivity was worse in 7%. This supports the robustness of pupillary reactivity for prognosis.
Prognostic value and practical implications
The GCS motor score has been confirmed as a key predictor of outcome in patients with sTBI and contains most of the predictive power of the GCS. 3,19 Previous studies report that both the ED and field GCS are strong predictors of fatal outcomes in TBI patients 20 and draw attention to the importance of the dynamics of neurological status and of assessments in multiple time points. 17 “Enrollment” GCS motor score (postresuscitation or hospital admission score based on availability) has been suggested for use in order to increase the reliability and consistency of motor score selected for prognosis. 8 Our study suggests that, when used as a single predictor, GCS motor score assessed in the field has higher predictive value in relation to 6-month mortality than at admission, which is in line with the fact that the predictive value of admission GCS has decreased since 1992 mainly as a result of aggressive prehospital treatment obscuring the assessment. 10 In the case of pupillary reactivity, our study found better performance in predicting 6-month outcome when it was assessed at admission, which also agrees with a previous finding. 8
Adding pupils and GCS motor score increases the accuracy in outcome prediction by Trauma Injury Severity Score or Revised Trauma Score, which confirms their predictive ability. 18 Indeed, both GCS motor score and pupillary reactivity have been widely used in prognostic models. 9 However, the dynamics of both parameters and the difficulties in their assessment affect predictive performance. We found that the best performance in predicting 6-month mortality (measured by AUC and Nagelkerke's R 2) was found in the models using the field GCS motor score and admission pupillary reactivity, as compared to using other combinations of these parameters.
On the other hand, the performance of the IMPACT models in predicting 6-month outcome with parameters assessed at admission was only slightly worse. Thus, the differences between the field and admission assessments did not translate into major differences in prognostic value. This suggests that a valid prognosis could be made for a patient if any of the two assessments is available. Such finding broadens the potential usability of GCS motor score and pupils as a set of prognostic variables given that predictions can already be made if only one of the observations (either from field or admission assessment) is available. In clinical practice, this will be case for most patients. In our data, at least one set of predictors was available in 94% of patients (Fig. 4). To optimize predictive performance, more predictors then only GCS and pupillary reactivity should be included. Other known strong predictors of outcome in TBI are age, CT characteristics, hypoxia, and hypotension, which are also included in the IMPACT models.
Conclusions
Motor scores after TBI are dynamic and generally deteriorated from the field to admission. Pupillary reactivity is more stable over time. Field GCS motor score and pupillary reactivity at hospital admission—compared to other combinations of these parameters—possess the best prognostic value to predict 6-month mortality in patients with moderate or severe TBI. Given that differences in prognostic performance are only small, both the field and admission values of GCS motor score and pupillary reaction may be reasonable to use in multi-variable prediction models to predict 6-month outcome.
Footnotes
Acknowledgments
The authors acknowledge the members of the Austrian Working Group on Improvement of Early TBI Care for their work in the design of the study and collection of data used for analyses in this article. Specifically, the authors thank these institutions and individuals: International Neurotrauma Research Organization (Alexandra Brazinova, MD, PhD, MPH, and Johannes Leitgeb, MD); Amstetten Hospital (Alexandra Frisch, MD, Andreas Liedler, MD, and Harald Schmied, MD); Feldkirch Hospital (Reinhard Folie, MD, Reinhard Germann, MD, PhD, and Jürgen Graber, RN, ICU); Graz University Hospital (Kristina Michaeli, MD, Rene Florian Oswald, MD, and Andreas Waltensdorfer, MD); Graz Trauma Hospital (Thomas Haidacher, MD, and Johannes Menner, MD); Horn Hospital (Anna Hüblauer, MD, and Elisabeth Nödl, MD); Innsbruck University Hospital (Christian Freund, MD, and Karl-Heinz Stadlbauer, Prof MD), Klagenfurt Hospital (Michel Oher, MD, Ernst Trampitsch, MD, and Annemarie Zechner, MD), Linz Hospital (Franz Gruber, MD, Walter Mitterndorfer, MD, and Sandra Rathmaier, MS); Linz Trauma Hospital (Norbert Bauer, MD); Salzburg Hospital (Peter Hohenauer, MD); Salzburg Trauma Hospital (Josef Lanner, MD, and Wolfgang Voelckel, Prof MD); Sankt Poelten Hospital (Claudia Mirth, MD); Schwarzach Hospital (Hubert Artmann, MD, and Bettina Cudrigh, MD); Wiener Neustadt Hospital (Daniel Csomor, MD, Günther Herzer, MD, Andreas Hofinger, MD, and Helmut Trimmel, MD); Wien Meidling Trauma Hospital (Veit Lorenz, MD, and Heinz Steltzer, Prof MD); and Wien “Lorenz Boehler” Trauma Hospital.
The project was funded jointly by the Austrian Ministry of Health (Contract: October 15, 2008) and by the Austrian Worker's Compensation Board (AUVA; FK 11/2008 and FK 11/2010). Data analysis was supported by a grant from AUVA (FK 09/13). INRO is supported by an annual grant from Mrs. Ala Auersperg-Isham and Mr. Ralph Isham and by small donations from various sources. The preparation of this article was partly done within the CENTER-TBI project that has received funding from the European Union Seventh Framework Programme (FP7/2007–2013) under grant agreement 602150.
Author Disclosure Statement
No competing financial interests exist.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
