Abstract
There has been an increased focus on the neurological sequelae of repetitive mild traumatic brain injury (TBI), particularly neurodegenerative syndromes, such as chronic traumatic encephalopathy (CTE); however, no animal model exists that captures the behavioral spectrum of this phenomenon. We sought to develop an animal model of CTE. Our novel model is a modification and fusion of two of the most popular models of TBI and allows for controlled closed-head impacts to unanesthetized mice. Two-hundred and eighty 12-week-old mice were divided into control, single mild TBI (mTBI), and repetitive mTBI groups. Repetitive mTBI mice received six concussive impacts daily for 7 days. Behavior was assessed at various time points. Neurological Severity Score (NSS) was computed and vestibulomotor function tested with the wire grip test (WGT). Cognitive function was assessed with the Morris water maze (MWM), anxiety/risk-taking behavior with the elevated plus maze, and depression-like behavior with the forced swim/tail suspension tests. Sleep electroencephalogram/electromyography studies were performed at 1 month. NSS was elevated, compared to controls, in both TBI groups and improved over time. Repetitive mTBI mice demonstrated transient vestibulomotor deficits on WGT. Repetitive mTBI mice also demonstrated deficits in MWM testing. Both mTBI groups demonstrated increased anxiety at 2 weeks, but repetitive mTBI mice developed increased risk-taking behaviors at 1 month that persist at 6 months. Repetitive mTBI mice exhibit depression-like behavior at 1 month. Both groups demonstrate sleep disturbances. We describe the neurological sequelae of repetitive mTBI in a novel mouse model, which resemble several of the neuropsychiatric behaviors observed clinically in patients sustaining repetitive mild head injury.
Introduction
T
The syndrome of CTE is now appreciated to occur in U.S. football players, boxers, wrestlers, hockey players, and even military personnel. 6 –9 The precise incidence and prevalence of CTE is unknown and difficult to glean from sporadic and selected cases. The phenomenon is caused by episodic and repetitive blunt force impacts to the head and transfer of acceleration-deceleration forces to the brain. The disease presents clinically as a composite syndrome of mood disorders, neuropsychiatric disturbance, and cognitive impairment, typically without sensorimotor impairment. It has been suggested that there are two major clinical presentations of CTE: one a behavior/mood variant and the other a cognitive variant. 10 Behavioral abnormalities, such as judgment issues, increased risk taking, and depression, are characteristic and prominent early in the disease course. 6 –9,11 Additional symptoms may include difficulty sleeping and poor concentration. Memory impairment and cognitive symptoms can occur, particularly late into the disease. 6,7 Generally, in patients with CTE, the overall clinical course is a progressively deteriorating one. 10 Definitive and confirmatory diagnoses of CTE still remain direct tissue histochemical and immunohistochemical (IHC) analyses; however, recent preliminary research shows that 2-(1-{6-[(2-[F-18]fluoroethyl)(methyl)amino]-2-naphthyl}ethylidene)malononitrile/positron emission tomography may hold promise for in vivo diagnosis. 12
Much has been garnered from previous laboratory studies regarding the consequences of repetitive head injury; however, none have fully modeled and encapsulated the syndrome of CTE. Additionally, most experimental TBI utilizes anesthesia during delivery of repetitive injury, which confounds the pathophysiology and post-traumatic outcome through both neuroprotective and -destructive pathways. 13 –19 The clinical expression of repetitive mTBI is likely completely different and deleterious, compared to single concussive injury. Additionally, an animal model of CTE is essential for learning about the pathophysiological link between repetitive head injury and the development of persistent subacute symptoms and chronic neurodegenerative disease, as well as important for discovering biomarkers of disease, improved diagnostic tools, and novel translational treatment approaches.
To address this, we sought to develop a novel model of closed-head injury that utilized some of the best facets of the most widely used animal models of TBI, 20 –22 with the purpose of creating the most detailed mechanistic and neurobehavioral model of repetitive brain injury. In addition, to make the model as clinically relevant as possible, we increased the number of hits, decreased the interinjury interval, and performed the injuries all in unanesthetized mice. We hypothesized that the combination of these variables in our novel model would result in the neurobehavioral spectrum of disease observed in patients diagnosed with CTE to include potential cognitive issues, increased risk taking, depression-like behavior, and sleep disturbances, as well as the histopathological hallmarks of CTE, including increased astrogliosis, microglial activation, and hyperphosphorylated tau protein accumulation. This article will address our behavioral results with this novel model of CTE, whereas the companion article will discuss the neuropathological aspects of the study.
Methods
Animal care and maintenance
All animals used in this study were treated in accord with the National Institutes of Health (NIH) Guidelines for the Care and Use of Laboratory Animals, and all procedures were performed under approval of the institutional animal care and use committee at the University of Rochester (Rochester, NY). Adult male C57BL/6J mice (The Jackson Laboratory, Bar Harbor, ME) were purchased and housed with 5 mice per cage under standard laboratory conditions (automatically controlled temperature, humidity, ventilation, and 12-h light/dark cycle) with unlimited access to food and water throughout the study. Mice were allowed to adapt to the vivarium for at least 1 week before experimental procedures. After injury or neurobehavioral testing, animals were returned to their home cages.
Mouse controlled, closed-head, acceleration-deceleration model of injury
A total of 280 mice were used for the study. The novel feature of this model of TBI is that the injury is inflicted without use of anesthesia. At 12 weeks of age, mice undergoing injury were placed head first into a small, clear plastic rodent restraint bag/cone (Thermo Fisher Scientific, Waltham, MA). Slits were cut at the narrow end of the cone to allow for increased head mobility and ventilation space. A twist tie was placed behind the mouse to immobilize the animal within the bag, and the ends of the twist ties were positioned to stabilize the body on the foam bed. In this manner, the animal is comfortably restrained, obviating the need for anesthesia (Fig. 1). A helmet was designed from 304 stainless steel (Millenium Machinery, Rochester, NY), measuring 3 mm in thickness and 6 mm in diameter and weighing 1.5 g (Fig. 1), which was secured to the head with an elastic band. This ultimately allows for accurate delivery of injury and diffuse spread of the force of the impacts. The undersurface of the helmet was lined with 1.0-mm double-sided gel-tape (Scotch® Restickable Tabs; Scotch Brand, St. Paul, MN). The helmet was engineered to fit the curvature of the mouse skull and cover the left hemisphere between the lambda and bregma up to, but not crossing, the mid-line. Placing the anteriormost part of the helmet 1–2 mm behind the left eye places the epicenter of the helmet over the left parietotemporal cortex. Mice were then positioned on Type E foam padding (Foam to Size, Inc., Ashland, VA) and positioned below the injury device. The mouse is placed on a foam base to allow for the acceleration-deceleration component of injury key to human concussive injury. The foam was changed regularly after each cycle of hits to ensure a similar spring constant for all impacts.
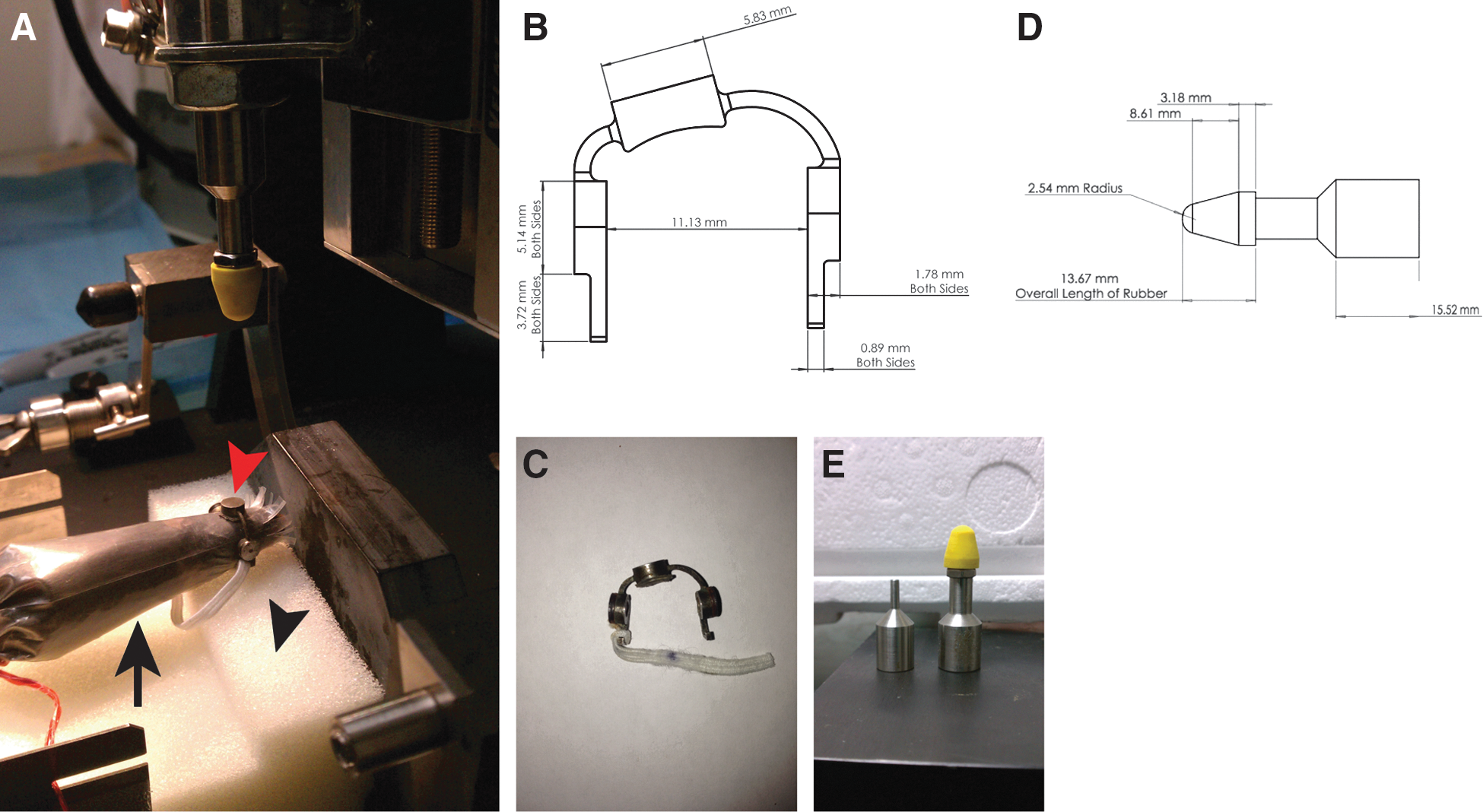
Unanesthetized, closed-head, acceleration-deceleration model of traumatic brain injury in mice. (
A controlled cortical impact (CCI) device (Pittsburgh Precision Instruments, Pittsburgh, PA) was used to deliver the head impacts with minor modifications, as noted below. The impounder was rigidly mounted at a 20-degree angle from the vertical plane. In transitioning the model to a closed head injury, the impactor tip was modified to further reduce the incidence of skull fracture. The diameter of the tip was increased to 6 mm and constructed from the vulcanized rubber of a competition lacrosse ball meeting National Operating Committee on Standards for Athletic Equipment standards (Millenium Machinery) (Fig. 1). To obtain a zero point, the impactor tip was carefully lowered until it touched the helmet surface. The impactor was angled and zeroed so that impacts occurred perpendicular to the helmet surface and thus orthogonal to the skull of the mouse, as well as to the center of gravity of the brain, thus limiting rotational acceleration. The linear acceleration forces and resultant motion of the mouse head was directly into the underlying foam. During impact, the tip was driven pneumatically to a depth of 1 cm farther than the zero point. Duration of impact was 100 ms and was delivered at a velocity of 5 m/sec. After impact, animals were then removed from the restraint bag and returned to their cage. Animals in the single mTBI group received a single head impact, whereas mice in the repetitive mTBI group received six head impacts per day (each hit separated by 2 h) for 7 days straight—for a total of 42 head impacts. The rationale to use 42 injuries is intended to resemble a clinical experience with repetitive head trauma. An interinjury interval of 2 h was chosen to maximize the reasonable number of impacts in a day and is corroborated by recent work investigating the effects of mTBI interinjury intervals. 23 There were no post-traumatic apneic episodes or seizure activity observed and no mortalities secondary to any of the impacts. Sham-injured/control animals underwent a procedure identical to trauma groups (placed into the restraint cone and had the helmet placed on); however, no injury was delivered. All control mice were age matched to account for any age-related differences in behavior or pathology. Mice were then assessed in a blinded fashion using various neurobehavioral tests, as described below. Timing for all testing was from the point of last impact (i.e., the amount of time out from the single impact for the single TBI mice and the time out from the 42nd hit for the repetitive TBI mice). In addition, all mice were used only once for an individual behavioral task at any time point.
Neurological Severity Score
To broadly characterize the effects of a single and repetitive mTBI in this model, a Neurological Severity Score (NSS) was obtained to evaluate the neurological impairment, compared to uninjured controls, as previously described.
24
The NSS is a composite clinical score consisting of 10 individual clinical parameters, including tasks on motor function, alertness, and general physiological behavior (Supplementary Table 1) (see online supplementary material at
Assessment of motor function
Vestibulomotor function was assessed by wire grip testing (WGT) 1 h after TBI and on postinjury days 1–7, as previously described, with minor modifications. 27 Briefly, mice (10 per group) were picked up by the tail and placed on a taut metal wire suspended between two upright bars 50 cm above a padded table. The time and manner in which the mouse could hold onto the wire were noted and scored on a scale of 0–5. Mice were tested three consecutive times at each time point. The score reported is the average of these individual trials. A composite group score was then calculated as the mean of these scores at each time point and then used for analysis.
Assessment of spatial learning and memory
Control, single mTBI, and repetitive mTBI groups were tested in the Morris water maze (MWM) acutely (acquisition trials on postinjury days [PIDs] 1–5 and probe trial on PID 6), subacutely (acquisition trials on PIDs 9–13 and probe trial on PID 14), and chronically at 1 (acquisition trials on PIDs 30–34 and probe trial on PID 35) and 6 months (acquisition trials on PIDs 180–184 and probe trial on PID 185) after the final head impact. Briefly, mice had to locate an invisible platform submerged 5 mm below the water level in a circular pool (dimensions, 120×30 cm; temperature, 22±1°C), based on the spatial location of five strategic visual cues fixed at distinct positions around the pool, as described previously. 28 The water was made opaque by adding nontoxic, water-soluble Tempera paint (Sargent Art, Inc., Hazleton, PA). Data were recorded with the help of video cameras (Sony Exwave HAD®; Sony Electronics Inc., Park Ridge, NJ) with a computerized video tracking system (Viewer3; BIOBSERVE GmbH, St. Augustin, Germany).
For training days (acquisition phase), all mice (14–15 per group, per time point) were given a maximum test duration of 60 sec to locate the hidden platform. The latency to reach the platform was recorded by the computerized video tracking system. Mice that failed to locate the platform within the time limit were guided to it and allowed to rest and orient themselves for 15 sec. The acquisition phase testing was conducted over 5 consecutive days, with four trials on each day, with the goal of locating the submerged hidden platform from different starting points and orientations (north, south, east, and west). On day 6 of MWM testing, all animals were tested for visual acuity and swimming speed using a visible platform paradigm. None of the animals were excluded from further testing based on the visual acuity and motor evaluation tests. On day 6, all mice also underwent a probe trial (retention phase), where the platform was removed from the pool. Mice were given 30 sec to swim, and time spent in the target quadrant (quadrant where the platform had been) versus the other quadrants was assessed as described previously. 28
Assessment of anxiety-related and risk-taking behaviors
Anxiety-related and risk-taking behavior of mice were evaluated using the elevated plus maze (EPM) test. When mice are placed in an EPM apparatus, both exploratory and fear drives are evoked, with rodents displaying a natural aversion for the open arms of the maze. 29,30 Mice (15 per group, per time point) were evaluated at 2 weeks, 1 month, and 6 months from the last head impact. The EPM consisted of two opposing open arms (35×5 cm) and two closed arms (35×5×15 cm) that extended from a central platform (5×5 cm) elevated 60 cm above the floor. A small raised lip (0.5 cm) around the edges of the open arms prevents animals from slipping off. Mice were placed individually on the central platform facing an open arm, away from the examiner, and were allowed to freely explore the maze for 5 min under even overhead fluorescent lighting, as described previously. 31 The behavior of each mouse was monitored using a video camera (Sony Exwave HAD; Sony Electronics) and the movement of the mice automatically registered and analyzed with a computerized tracking system (Viewer3; BIOBSERVE GmbH). Time spent in the open and closed arms was measured, and each mouse was only tested once in the maze. 31 The floor of the EPM was washed after each testing with 0.1% acetic acid solution to remove odors left by previous subjects. 31
Assessment of depression-like behavior
To determine the long-term effects of mTBI on depression-like behavior, mice (15 per group) were tested in the Porsolt forced swim test (FST) and the tail suspension test (TST) at 1 month postinjury, as previously described. 32 –36
Porsolt forced swim test
Briefly, mice were placed in an open glass cylinder (diameter 12 cm, height 24 cm, and water level 16 cm) containing fresh tap water (23–25°C). The duration of the entire test was 6 min, with the first 2 min used for habituation and only the last 4 min used for analysis. Two different experimenters blinded to the groups of mice evaluated the behavior of the animals manually. A mouse was judged to be immobile when it remained floating in the water, making only those movements necessary to keep its head above the water surface.
Tail suspension test
Briefly, mice were suspended by the tail to a bar elevated 40 cm above the surface of a table covered with soft cloth in a soundproof room. The duration of the test was 6 min. Two different experimenters blinded to the groups of mice evaluated the behavior of the animals manually. The immobility time of the tail-suspended mice was measured and defined as the absence of limb movement.
Assessment of sleep behavior
Surgical implantation of electroencephalography and electromyography electrodes
Electroencephalography (EEG) and electromyography (EMG) data were acquired in mice at 1 month postinjury (6–9 per group) using implantable telemetry devices (Data Sciences International, ST. Paul, MN) and the Dataquest A.R.T. system (Data Sciences International). Because the F20-EET EEG telemetry device is one of the smallest commercially available devices (3.9 g), it allowed for recording of EEG and EMG in freely moving animals. Surgery was done under anesthesia (ketamine-xyalzine) 24 h before recording. The body of the transmitter was implanted intraperitoneally through a mid-line abdominal incision. EEG lead implantation was performed by insertion of leads in small burr holes overlying the cortex. EMG leads EMG electrodes were made from multi-stranded stainless steel wires and were implanted into the neck muscle. The EEG electrodes were secured with dental cement, and the EMG electrodes were secured with sutures. Mice were allowed to recover and were individually housed in a sound-attenuated and ventilated chamber on a standard light/dark cycle, with food and water available ad libitum.
Data acquisition and analysis
After a 1-day acclimation and recovery period, the telemetry EEG/EMG devices were activated and continuous recordings were obtained for a total of 24 h (6
Statistical analysis
All statistical analyses were performed using SAS statistical software (version 9.3; SAS Institute Inc., Cary, NC). All tests were two-sided and conducted at the 5% significance level, unless otherwise indicated. In particular, significance levels were adjusted to account for multiple comparisons, when deemed necessary. All data are presented as the means±standard deviation of the mean.
The NSS score is an ordinal measure taking values in {0,1,…,5}. Its variance was estimated as 0 in the control group at all time points. The assumptions required by standard analysis of variance (ANOVA) were therefore violated, and we assessed the effect of injury on these scores using a rank-based approach. We first averaged NSS scores within each mouse and assessed the statistical significance of injury on these average scores using Kruskal-Wallis' nonparametric one-way ANOVA. Next, we performed post-hoc analyses evaluating the significance of injury on NSS scores within each time point. We used Kruskal-Wallis' one-way ANOVA and adjusted p values for multiple (seven) comparisons using Bonferroni's method. Comparisons with adjusted p values that were significant at the family-wise error rate of 5% were followed by another set of post-hoc analyses comparing pairs of injury groups (e.g., control vs. repeat) at the corresponding time point using Wilcoxon's rank-sum test and applying a Bonferroni correction to each p value to adjust for the three comparisons that were made. Secondary analyses assessing whether the NSS score changed significantly over time within each injury group (single and repeat) were conducted using Friedman's nonparametric two-way ANOVA, treating each mouse as a block and time as factor. Similar analyses were conducted to evaluate the effect of injury on wire grip score.
MWM acquisition trials were performed at four time periods: acutely (PIDs 1–5); subacutely (PIDs 9–13); 1 month (PIDs 30–34); and 6 months (PIDs 180–184) postinjury. Data were analyzed using mixed-effects two-way ANOVA, including time, injury group, and an interaction between time and injury group as factors. Measurements performed in the same mouse may be dependent, and data measured in the same mouse and on the same day may also be associated. To account for such dependencies, the models included two levels of random effects: one modeling within-mouse associations and one nested within mouse describing potential intraday correlations. We assessed the effect of injury, time, and their interaction using F tests at each time period, followed by pair-wise injury group comparisons using Tukey's post-hoc analyses. Separate analyses were conducted for each time period.
MWM probe trials were also performed at four time periods: acutely (PID 6); subacutely (PID 14); 1 month (PID 35); and 6 months (PID 185). First, we compared the proportion of time spent in the four quadrants in the single and in the repeat injury group to controls using multi-variate ANOVA (MANOVA), including injury group as a factor. We first assessed the overall effect of injury irrespective of the time period by combining p values using Fisher's method. We also reported p values assessing the significance of the comparisons at each time period. To determine whether mice spent significantly more time in the target quadrant than in any of the remaining three quadrants, we use a simple adaptation of Laska and Meisner's Min test, 41 extended to allow comparison involving four groups instead of three, and performed pair-wise comparisons of the proportion of time spent in quadrants using one-sided paired t-tests. p values were Bonferroni-corrected to account for the fact that we ran one Min test for each of the four time periods. One such analysis was performed for each injury group.
For each injury group, we compared the times mice spent in the open and in the closed arms during the EPM trials using paired t-tests. To compare the time spent in the open arm between injury groups, we used a one-way ANOVA, including injury group as a factor, followed by post-hoc tests comparing pairs of groups and adjusting for multiple comparisons using Tukey's method. One such analysis was carried out for each time period. Data from the Porsolt FST and TST were analyzed using one-way ANOVA, including injury group as a factor, followed by Tukey's post-hoc analyses.
Percent wake time, percent NREM time, percent REM time, number of NREM episodes, NREM episode length, and REM EMG activity were analyzed using one-way ANOVA, including injury group as a factor, followed by post-hoc analyses based on Tukey's method for multiple comparison adjustment. Data on the prevalence of the frequency (in Hz) was analyzed using mixed two-way ANOVA, including injury group, frequency (1–15), and their interaction as factors. We first assessed the significance of each of these factors using F tests, followed by post-hoc analyses comparing groups within each frequency level, adjusting for multiple comparisons using Bonferroni's method. Finally, we compared the frequency prevalence in pairs of injury groups using one-way ANOVA, including injury group as a factor, and adjusting for multiple comparison using Tukey's method. This second set of post-hoc analyses was carried out only for frequency levels at which significant group effects were detected.
Results
Repetitive mild traumatic brain injury results in a transient Neurological Severity Score elevation and short-lived motor deficits
In NSS testing, severity of impact for both the single and repetitive TBI groups fell in the mild spectrum. We found that averaged NSS scores were statistically different between injury groups (p<0.001; Kruskal-Wallis), and that the effect of injury was significant within each time point after multiple comparisons adjustment (padj <0.05 in all cases; Kruskal-Wallis). Post-hoc analyses comparing pairs of injury groups within each time point found that all pairs of injury groups were significantly different at the 5% significance level (Kruskall-Wallis), with the exception of the single and control groups, which could not be distinguished at 48 h, 7 days, and 1 month (Fig. 2A). Single TBI mice did have an elevated NSS at 72 h, compared to control mice. Both groups of TBI mice improved over time, with the NSS score decreasing significantly over time in both the repeat and single injury groups (both groups, p<0.001; Friedman).
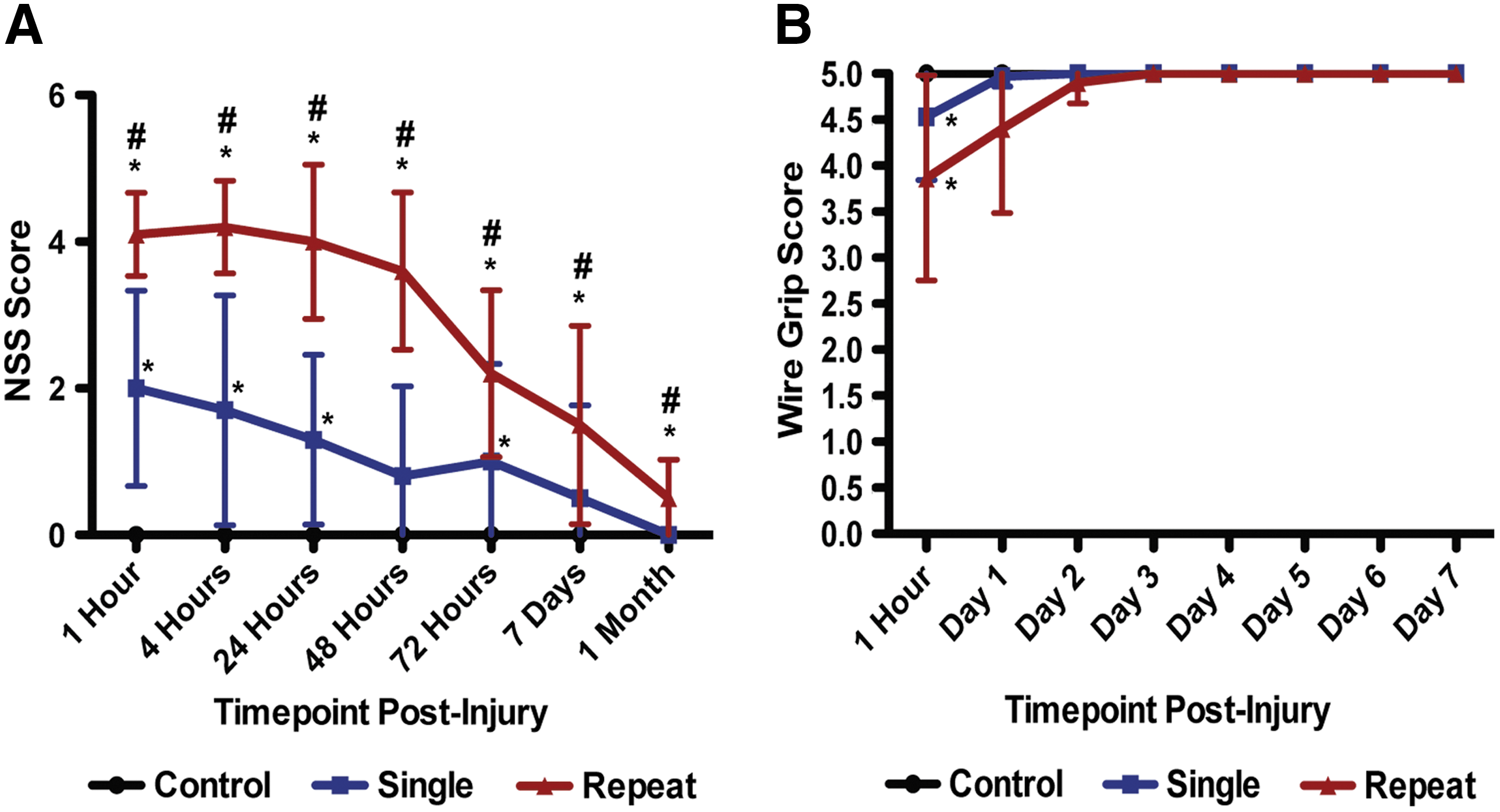
Repetitive mild traumatic brain injury (mTBI) results in elevated Neurological Severity Scores (NSS) and transient vestibulomotor deficits. (
Disruption of static and dynamic balance has also been thoroughly described in patients with concussion. 42 Vestibulomotor function was assessed by WGT, and there were significant effects of injury group on performance. We found that averaged wire grip scores were statistically different between injury groups (p<0.01; Kruskal-Wallis), and that the effect of injury was significant at 1 h postinjury, even after adjustment for multiple comparisons (padj <0.05; Kruskal-Wallis). Post-hoc analyses found significant difference at the 5% significance level, at 1 h postinjury, with both the single and repeat injury groups statistically different from the control group (Fig. 2B; Kruskall-Wallis). The single and repetitive injury groups could not be distinguished, however. We also found that the performance on the WGT improved over time, with wire grip scores increasing significantly over time in both the repeat and single injury groups (both groups, p<0.001; Friedman).
Repetitive mild traumatic brain injury causes persistent deficits with spatial learning and memory
We next assessed hippocampus-dependent spatial learning and long-term memory in the single and repetitive TBI mice using the MWM. Animals from all groups showed daily improvements in their abilities to locate the hidden platform during the acquisition phase of the MWM task; however, mTBI mice demonstrated increased latencies. At the acute and subacute time points, we found that the main effect of time, the main effect of injury group, and the interaction of time and injury group were statistically significant (acute: ptime <0.001, F(4,660) =109.9; pinjury <0.001, F(2,660) =23.3; ptime*injury <0.05, F(8,660) =2.08; subacute: ptime<0.001, F(4,788)=123.6; pinjury<0.05, F(2,788)=4.55; ptime*injury<0.05, F(8,788)=2.31; mixed two-way ANOVA; Fig. 3A,B). At 1 month postinjury, the main effect of time and the main effect of injury group were statistically significant, but their interaction did not reach significance (ptime<0.001, F(4,580)=169.1; pinjury<0.001, F(2,580)=7.69; ptime*injury>0.1, F(8,580)=0.92; mixed two-way ANOVA; Fig. 3C). At 6 months postinjury, we found that time and the interaction of time and injury group was significant, and that the main effect of injury group was trending (ptime<0.001, F(4,676)=108.5; pinjury>0.05, F(2,676)=2.71; ptime*injury<0.05, F(8,676)=2.07; mixed two-way ANOVA; Fig. 3D). Post-hoc analyses found that, at the acute time point, all pairs of injury groups (control vs. single mTBI, control vs. repetitive mTBI, and single mTBI vs. repetitive mTBI) were statistically different at the 5% significance level. At the subacute time point, the analyses found statistically significant differences between the control and the repetitive mTBI groups. At 1 month postinjury, we found statistically significant differences when comparing control to single mTBI and control to repetitive mTBI groups. At 6 months, none of the comparisons were found significant in the posthoc analyses.
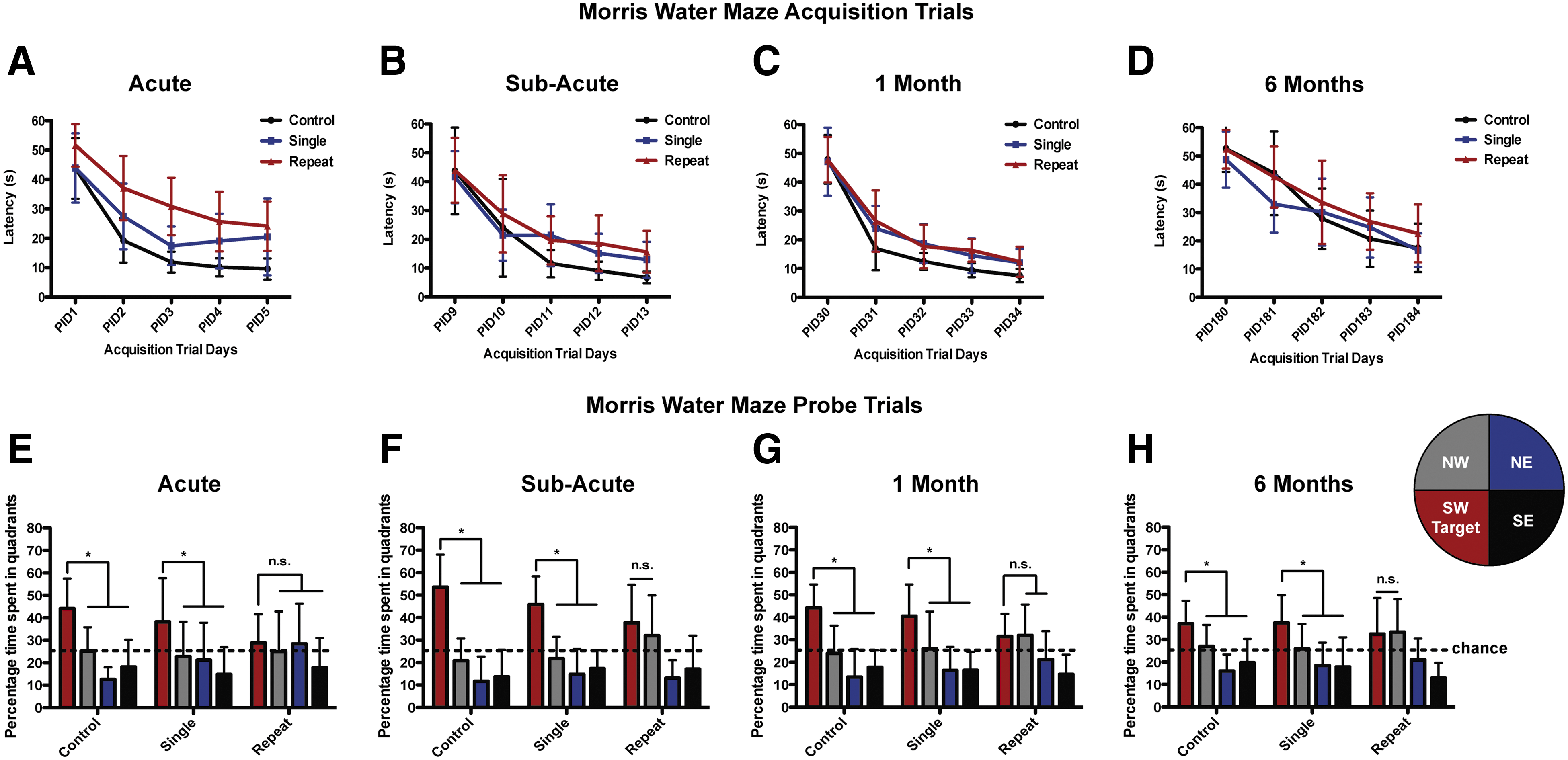
Evaluation of learning (acquisition) and spatial memory retention (probe) using the Morris water maze. (
For the probe trial testing, analyses comparing the single injury and the control groups found no significant differences in the distribution pattern of the time that mice spent in the four quadrants at any of the time points (p>0.1; MANOVA), even when combining evidence from all time points (p>0.1; MANOVA plus Fisher's combination method). When comparing the repeat and the control injury groups, we found significant differences in the distribution of the time that mice spent in the four quadrants (p<0.01; MANOVA plus Fisher's combination method). When performing such comparisons within each time point, we found significant differences between the control and the repeat groups at the acute time point (pacute <0.05; MANOVA). The difference was trending at the subacute and 1-month time points (psub-acute >0.05; p1 month >0.05; MANOVA), but it did not reach significance at 6 months (p6 months >0.1; MANOVA).
Subsequent analysis was performed to evaluate the preference for the target quadrant compared to the other three quadrants. Findings from the probe test indicate that mice from the single mTBI and uninjured control groups, at all time points, spent a significantly higher percentage of time in the target quadrant (the location that contained the platform during training), when compared to the other equivalent zones (Fig. 3E–H). We found that mice in the control and single injury group spent significantly more time in the target quadrant than in any of the other three quadrants at the acute, subacute, 1-month, and 6-month time points postinjury (pacute <0.001; psub-acute <0.01; p1 month <0.01; p6 months <0.05; Min test). Mice in the single injury group also spent significantly more time in the target quadrant than in any other quadrant at all time points postinjury (pacute <0.001; psub-acute <0.05; p1 month <0.05; p6 months <0.05; Min test). In contrast, repetitive mTBI mice exhibited impaired spatial memory, failing to show significant discrimination and preference for the target quadrant, compared to the other quadrants (Fig. 3E–H). We did not find that mice in the repetitive injury group spent significantly more time in the target quadrant than in any other quadrants at any of the postinjury times (p>0.1; Min test).
Mild traumatic brain injury results in subacute anxiety with repetitive head injury leading to increased risk-taking activity over time
We used the EPM to characterize the effect of single and repetitive mTBI on anxiety-related and risk-taking behaviors. One-way ANOVA revealed that the amount of time spent in the open arm differed significantly between injury groups at 14 days, 1 month, and 6 months (p14 days<0.001, F(2,32)=18.8; p1 month<0.05, F(2,38)=4.4; p6 months<0.01, F(2,42)=6.68; one-way ANOVA). At 2 weeks postinjury, mTBI mice exhibited increased anxiety-like behavior (Fig. 4A). Post-hoc analyses found significant differences at the 5% significance level at 14 days postinjury between the control and the single injury groups and between the control and the repetitive injury groups (ANOVA plus Tukey). Single and repetitive mTBI resulted in significantly reduced time spent in the open arms of the maze, consistent with increased anxiety.
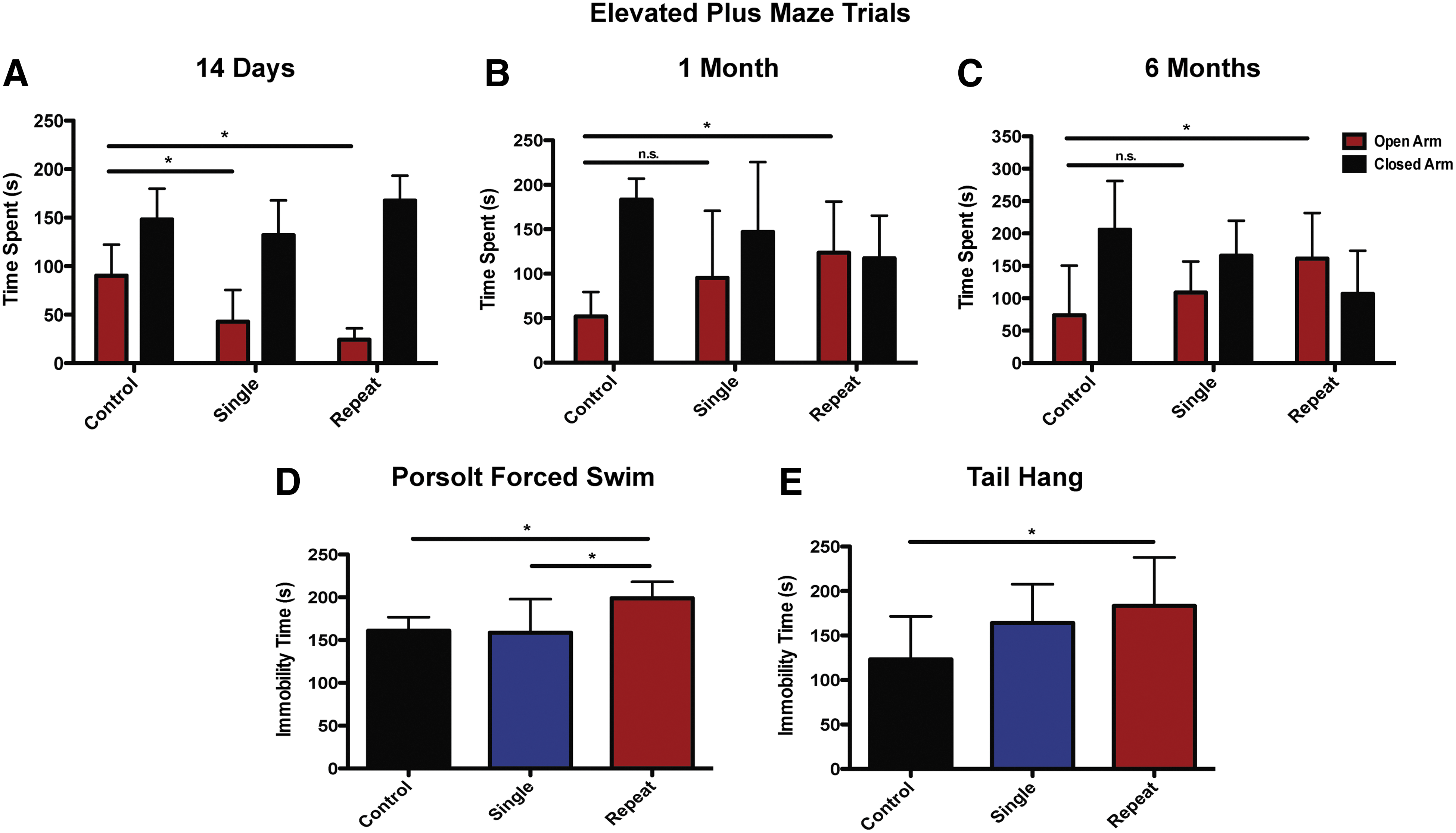
Repetitive mild traumatic brain injury (mTBI) results in increased risk-taking and depression-like behaviors. (
At the 1- and 6-month time points, significant differences were found in post-hoc analyses between controls and repetitive injury groups (ANOVA plus Tukey). Whereas there was no difference in the amount of time spent on the open arms between single mTBI and control mice at 1 and 6 months, at the 1-month time point, mice in the repetitive mTBI group spent an increased amount of time on the open arms of the EPM, compared to control mice (ANOVA plus Tukey; Fig. 4B). Such increased exploratory activity in the open arms and reduced fearfulness is consistent with increased risk taking, as noted in other studies. 30,43,44 This increased risk taking persisted and progressed in the repetitive mTBI mice out to 6 months (ANOVA plus Tukey; Fig. 4C).
Repetitive mild traumatic brain injury results in depression-like behavior at 1 month
At 1 month postinjury, in the Porsolt FST, there was a significant effect of injury severity on depression-like behavior. Data from the swim test, analyzed using one-way ANOVA, indicated significant differences between groups (p<0.01, F(2,27)=7.9; one-way ANOVA). Post-hoc analyses found significantly increased immobility time in the repetitive mTBI group, compared to the single mTBI and control groups (ANOVA plus Tukey; Fig. 4D). Single mTBI mice did not exhibit depression-like behavior. The TST revealed comparable effects of mTBI on depression-like behavior as the FST. Similar analyses carried out on data from the TST suggested that immobility times differed significantly between injury groups (p<0.05, F(2,27) =4.09; one-way ANOVA). Repetitive mTBI mice demonstrated significantly increased immobility time, compared to the control group (ANOVA plus Tukey; Fig. 4E). No other pair-wise comparisons were found significant after adjustment for multiple comparisons.
Mild traumatic brain injury mice exhibit sleep disturbances at 1 month
To evaluate the long-term effect of mTBI on sleep-wake behavior, we used infrared videography and electrophysiological monitoring. 40 We found that the effect of injury group on percent wake time was statistically significant (p<0.001, F(2,19) =13.34; ANOVA). Post-hoc analyses, at the 5% significance level, revealed that the percentage of wake time in the single and repeat injury groups were both significantly different from those recorded in the control group (ANOVA plus Tukey; Fig. 5A). However, no statistically significant difference was found between the single and repeat injury groups in terms of wake time (ANOVA plus Tukey). With the significant increase in wake time, we found a concomitant reduction in NREM sleep in the mTBI mice. We found that the effect of injury group on percent NREM time was statistically significant (p<0.001, F(2,19) =10.69; ANOVA). Post-hoc analyses, at the 5% significance level, revealed that the percentage of NREM time in the single and repeat injury groups were both significantly different from those recorded in the control group (ANOVA plus Tukey; Fig. 5A). However, no statistically significant difference was found between the single and repeat injury groups in terms of percentage of NREM sleep (ANOVA plus Tukey). We also found that the effect of injury group on percent REM time was not statistically significant (p>0.1, F(2,19) =1.36; ANOVA).
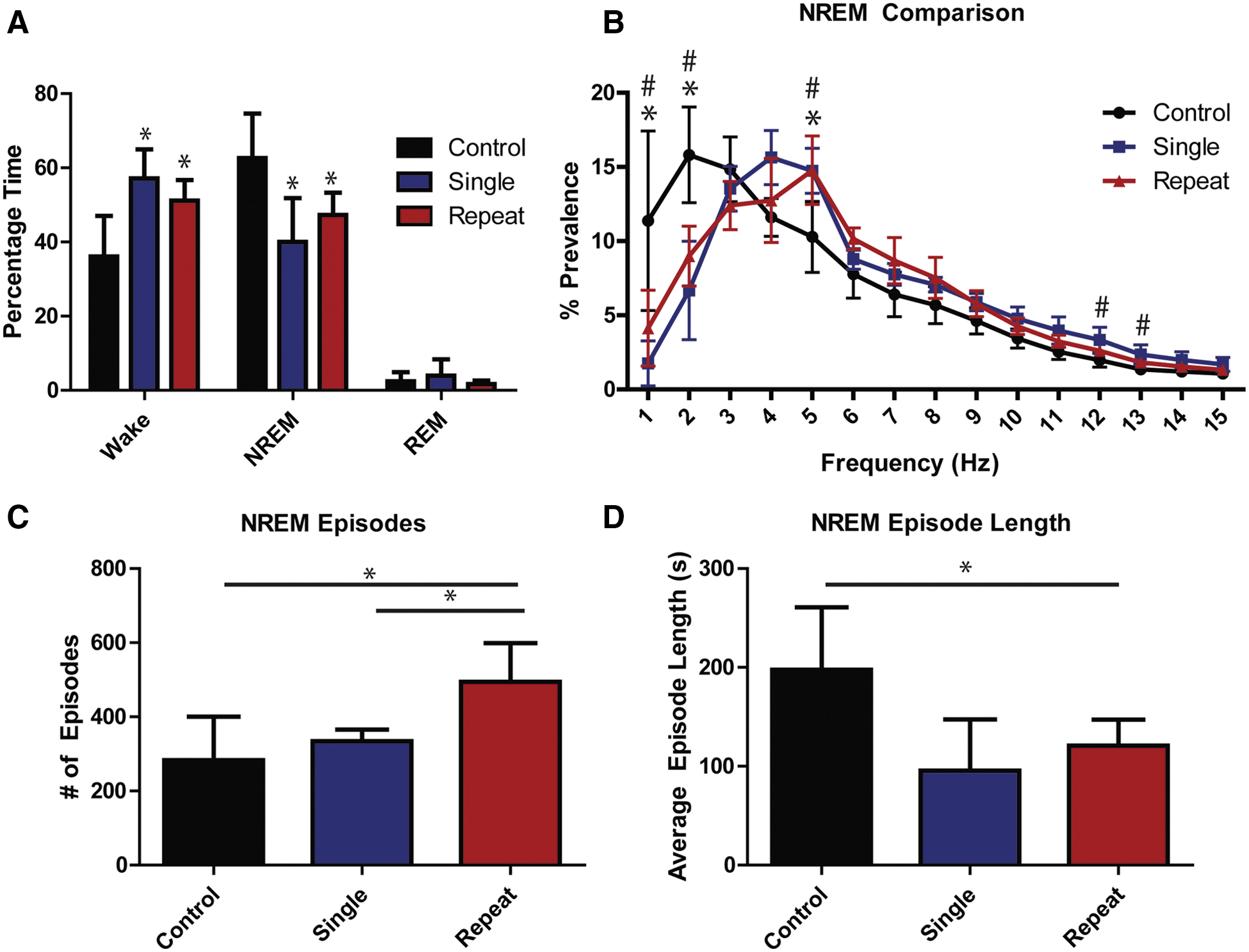
Mild traumatic brain injury (mTBI) results in sleep pattern disturbances. (
We sought to further examine the quality of NREM and REM sleep in these mice. During NREM sleep, there was an increase in cortical activity with a significant shift toward higher frequencies (Fig. 5B). Mixed two-way ANOVA suggested that prevalence was significantly different across levels of frequency (p<0.001, F(14,224) >1.000) and that the injury group had a significant effect on prevalence through its interaction with frequency (p<0.001, F(16,224) =74.6). Its main effect was not significant (p>0.1, F(1,224) =0.00). Post-hoc analyses found that the effect of injury groups on frequency prevalence was statistically significant at frequency levels 1, 2, 5, 12, and 13 Hz (ANOVA plus Bonferroni). Post-hoc analyses comparing pairs of groups at frequency levels where a significant injury group effect was found in previous analyses suggested that repetitive and single injury groups were significantly different from controls at 1, 2, and 5 Hz at the 5% significance level (ANOVA plus Tukey); no significant difference was found between the single and repetitive injury groups at these frequency levels. At 12 and 13 Hz, we found that the single injury and control groups were significantly different (ANOVA plus Tukey). No other comparisons were statistically significant.
Repetitive mTBI also caused NREM sleep fragmentation as well (Fig. 5C,D). We found that the injury group had a significant effect on both the number of episodes and average episode length (p<0.001, F(2,18) =10.53 and p<0.01, F(2,18) =8.31; ANOVA). In post-hoc analyses, the number of episodes in the repetitive mTBI mice were significantly increased from those observed in the control and single injury groups at the 5% significance level (ANOVA plus Tukey; Fig. 5C). The average episode length in the repetitive mTBI mice was also significantly reduced, compared to control mice (ANOVA plus Tukey; Fig. 5D). No significant difference could be detected between the single injury and control groups for both the number of episodes and average episode lengths. mTBI did not result in a significant difference in the quality of REM sleep, compared to control animals, with regard to power spectral analysis. Additionally, one-way ANOVA could not detect any significance difference between injury groups either in the number of episodes or in REM episode length (p>0.1, F(2,17) =2.28 and p>0.1, F(2,17) =0.14; data not shown). mTBI mice did endorse abnormal EMG activity during REM sleep, however. Effect of injury was significant on REM EMG data (p<0.01, F(2,17) =6.15; ANOVA). Tukey's post-hoc analyses revealed that the single and repetitive mTBI mice demonstrated significantly increased EMG activity, compared to controls, during REM sleep (ANOVA plus Tukey; Fig. 6).
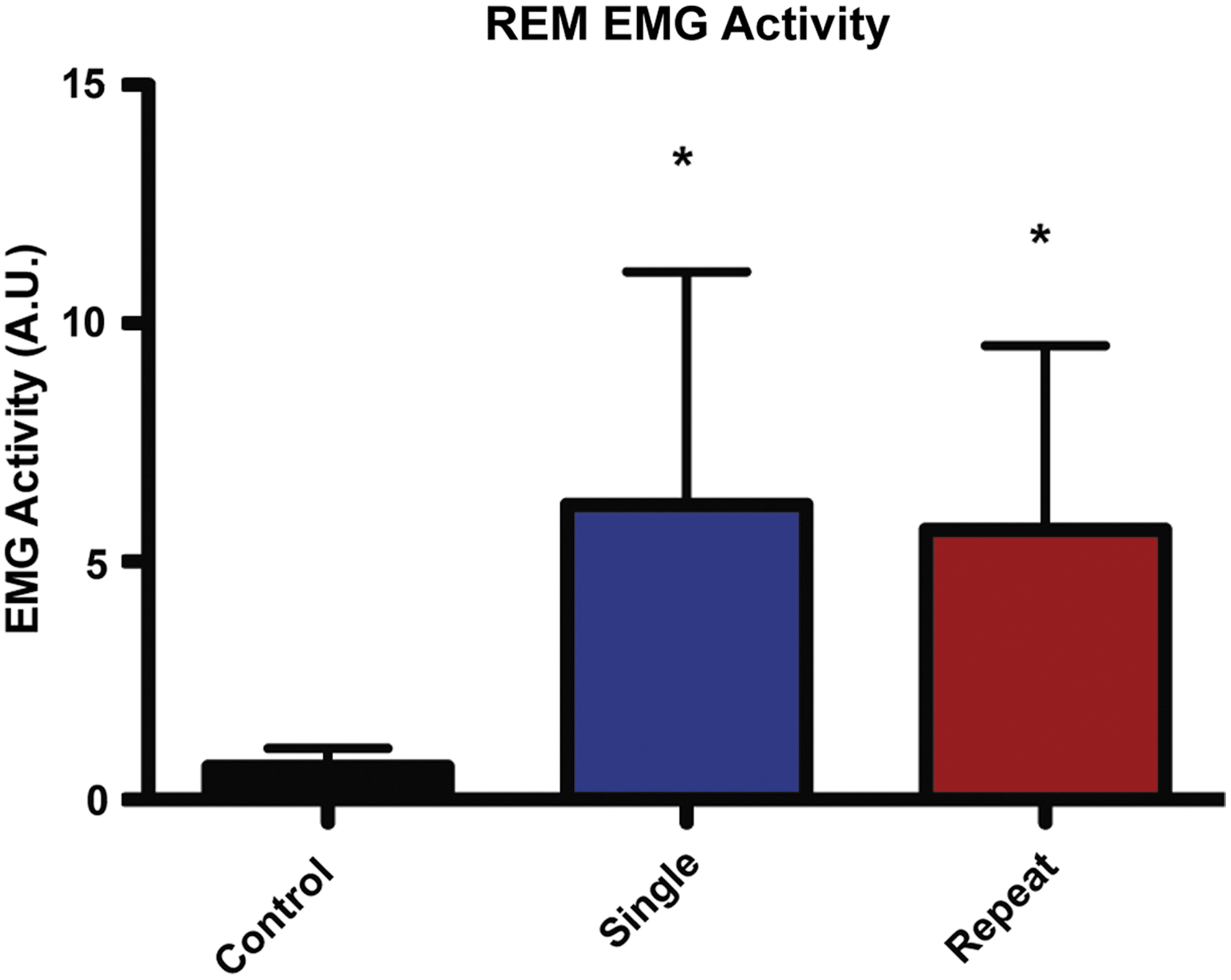
Mild traumatic brain injury (mTBI) results in abnormal REM EMG. Single and repetitive mTBI mice demonstrated significantly higher EMG activity than controls during REM sleep (Tukey). *p<0.05, compared to control mice. Values are mean±standard deviation. REM, rapid eye movement; EMG, electromyography; A.U., arbitrary units. Color image is available online at
Discussion
Novel repetitive mild traumatic brain injury model
In the last decade, there has been an increased interest in laboratory research focused on repetitive mTBI. 20,45 –57 The behavioral deficits in these studies were more pronounced and protracted in the repetitive head injury groups, although, often these deficits improve or even resolve with time. A significant challenge for investigators developing experimental models of mTBI is replicating the long-term neuropsychological and -behavioral consequences of mTBI, such as deficits in memory acquisition and retrieval, attention and executive functioning, and reduced tolerance to stress. 58 Here, we report on a novel, unanesthetized model of repetitive mTBI that is simple, practical, and has been developed with several features similar to experience in the clinical setting. Contrary to some previous studies, the changes we observed in our repetitive mTBI mice were persistent or progressed over a significant length of time.
Most experimental TBI also utilizes anesthesia during delivery of repetitive injury, which confounds the pathophysiology and post-traumatic outcome. 13 –16,19 Statler and colleagues demonstrated that isoflurane exerts neuroprotection, even when used at the lowest feasible level before TBI (i.e., when discontinued with recovery to tail pinch immediately before injury). Investigators using isoflurane must consider its beneficial effects in the design and interpretation of experimental TBI research. Experimental anesthetic exposure has been implicated in preconditioning 59 and has even been found to result in an increase in tau phosphorylation and cognitive impairment, particularly after repeated exposures. 17,18 Few studies have attempted to perform injuries in unanesthetized rodents. 60,61 The injuries in these studies were delivered to hand-held rodents and were variable with regard to severity and outcome. Our model is the first nonanesthetized animal model of TBI that consistently and efficiently reproduces the spectrum of behavioral abnormalities and neuropathological findings observed in humans after mTBI.
The adapted use of the CCI device does allow for precise control of the impact parameters, including velocity, duration, and depth of impact; however, equally as important in developing this model was maintaining many of the principles of Marmarou's weight drop model of injury. 21,22 The degree to which head motion is restricted is of chief importance in determining the relative contribution of impact and inertial components in head trauma. 62 With the head free to move into a foam base of a known spring constant, the shear forces between tissues are predominant.
Chronic behavioral sequelae after repetitive mild traumatic brain injury
Whereas most patients recover within a few days or weeks after a concussion, a number of individuals may develop long-lasting or progressively worsening symptoms, particularly if head injuries occur repetitively. 1 In some patients, repetitive mTBI may result in chronic neurodegenerative syndromes, such as CTE. 2 In the present study, we have developed and characterized a novel model of closed-head injury to investigate the neurological sequelae after repetitive mTBI. With this model, animals exposed to a single mTBI have short-term postconcussive behavioral abnormalities. By contrast, the observed cumulative effect of 42 head impacts delivered over the course of 7 days includes long-term depression-like behavior, risk-taking behavior, and persistent deficits with spatial learning and memory.
This spectrum of behavior falls in line with emerging clinical data implicating repetitive head injury in the development of subacute and chronic neurological sequelae. Previous work using survey research from the Center for Study of Retired Athletes has shown that both mild cognitive impairment (MCI) and depression are more common than expected in age-matched controls, and that there is a correlation for a higher incidence being associated with three or more concussions. 63,64 In soccer, there has been the suggestion that repetitive heading of the ball may lead to an increased risk of chronic neurological injury. 65,66 Studies in division I college football players have shown that those experiencing three or more earlier concussions had more symptoms and took longer to recover. 67 –70 Cognitive deficits and depression appear to be more common in aging former professional football players, compared to healthy controls. 71 In one recent study, these deficits were correlated with white matter abnormalities and changes in regional cerebral blood flow. 71 Another study in former professional football players found significant decreases in regional cerebral blood flow across the whole brain, especially in the prefrontal poles, temporal poles, occipital lobes, anterior/posterior cingulate gyrus, hippocampus, and cerebellum. 72 MCI screens administered to these same athletes demonstrated a significantly increased prevalence of MCI and dementia in these athletes, compared to aged-matched control patients. 72
De Beaumont and colleagues recently sought to investigate the effects of having sustained a sports concussion more than 30 years before testing on cognitive and motor functions. 73 This study demonstrated that, relative to a group of former athletes with no previous history of sports concussion, former athletes who sustained their last sports concussion more than 30 years ago exhibit cognitive and motor system alterations, show significant reductions on neuropsychological as well as electrophysiological measures of episodic memory and frontal lobe functions selected for their known sensitivity to MCI and early-onset AD, and display significant motor execution slowness. 73 The mice undergoing repetitive mTBI in this study exhibited spatial learning and memory deficits acutely, and these cognitive deficits were trending at later time points. Unlike the single mTBI and control groups, repetitive mTBI mice did not demonstrate a preference for the target quadrant, compared with other quadrants, during probe trial testing. This is consistent with the findings from clinical studies suggesting that repetitive concussive and subconcussive impacts may have long-term implications for cognitive functioning.
In addition to cognitive impairment, patients sustaining mTBI can endorse emotional symptoms, particularly when head impacts occur repetitively. 1 Behavioral disinhibition has been reported as a consequence of mTBI in humans. Patients diagnosed with CTE have been noted to exhibit increased risk taking and poor judgment. 7,74 Depression is also common in postconcussive patients and is a prominent feature in many of those patients diagnosed with CTE. 1,6,8,9,75,76 A recent study evaluated a population of retired professional football players in order to study the relationship between white matter integrity and the manifestation of depressive symptoms. 77 The researchers utilized diffusion tensor imaging to assess white matter integrity and correlated that measure with overall depression scores as well as each of the three subfactors derived from the Beck Depression Inventory II. They found a significant association between white matter integrity and the presence, as well as severity, of depressive symptoms in retired athletes with a history of concussive or subconcussive impacts. 77
The mice sustaining repetitive mTBI in this model demonstrated depressive-like behavior, as manifested in the FST and TST at 1 month from the last injury. This behavior was not observed in control mice or mice receiving a single mTBI, underscoring the potential deleterious, cumulative effect of repetitive mild head trauma. Both the single and repetitive mTBI mice showed signs of increased anxiety on the EPM 2 weeks after their last head trauma. Whereas the behavior of single mTBI mice on the EPM normalized just 2 weeks later (at the 1-month time point), the repetitive mTBI mice demonstrated significant behavioral disinhibition and decreased fear avoidance, spending significantly more time on the open arms of the maze. At the 6-month time point, mice that had sustained repetitive head impacts continued to exhibit increased risk-taking behavior and more time on the open arms, compared to the closed arms, of the maze.
Sleep disturbances can often occur acutely after a concussion and are a source of significant morbidity in patients with protracted postconcussive symptoms, particularly when head impacts occur repetitively. 1,75,76 It has been well documented that many of the patients that have been diagnosed with CTE may have difficulty falling asleep or staying asleep or may suffer from insomnia. 6,8,78 Recent studies have suggested that TBI may be associated with virtually all common sleep-related symptoms and disorders, including insomnia, daytime somnolence, parasomnias, and sleep disruptions, such as excessive stage 1 sleep, excessive awakenings, excessive stage shifts, and reduced delta sleep. 79 –83 mTBI may even be more strongly correlated with sleep disturbances than sTBI. 84 Kaufman and colleagues characterized the long-term effects of mTBI on sleep in adolescents. 79 Nineteen adolescents without any discernible clinical sequelae, who had suffered mTBI 3 years before, were investigated in the sleep laboratory by whole-night polysomnography (PSG) recordings. In comparison to controls, mTBI was associated with lower sleep efficiency, more wake time, and with more sleep awakenings lasting greater than 3 min. In another study, parasomnias were a presenting complaint in approximately 25% of patients, with an increase in EMG tone and/or frank REM behavior disorder diagnosed in 13% of those who had PSG. 80
Until recently, there was a paucity of data on how cumulative mTBIs affect sleep. Bryan and colleagues demonstrated that multiple head injuries were associated with an increased risk for, and severity of, sleep disturbance among active male military personnel, even when controlling for depression, post-traumatic stress disorder, and concussion symptom severity. 85 Patients were categorized into three groups based on TBI history: no TBIs; single TBI; and multiple TBIs. The study found that 47.6% of patients with multiple TBIs exceeded the threshold for clinical insomnia, compared to 22.4% of those with a single TBI and only 5.6% of those with no history of TBI. Sleep disturbances might be most vulnerable to disruption after an initial TBI, and disruption of sleep maintenance might intensify after additional TBI, although further studies are needed to better understand how repetitive head injury might be related to different types of sleep disturbances. Whether impaired sleep is the result of, or the cause of, other TBI-related symptoms is yet to be determined as well.
Mild TBI mice in this study spent more time awake and less time in NREM sleep over the course of 24 h. Consistent with the overall reduction in sleep amounts, mTBI caused more NREM sleep fragmentation. Repetitive mTBI resulted in an increased number of short NREM segments. Taken along with the spectral analysis during NREM sleep, whereas control animals spent a significant portion of NREM time in deeper (stage 3/4) sleep, mTBI mice spent more time in lighter (stage 1/2) stages of NREM sleep, failing to spend a lot of time in deeper, quality NREM sleep. We also found that mTBI mice had significantly increased EMG tone during REM sleep, compared to control mice. This REM phenotype is similar to the types of mild parasomnias reported in patients with chronic sleep disturbances after mTBI. 79,80,82,83
Analyses of data from early nonhuman primate studies of concussion led investigators at the time to believe that approximately half of the potential for concussion during impact to the unprotected movable head was related to head rotation, with the remaining brain injury potential of the blow being related to the contact phenomena of the impact. 86 –89 Although the biodynamics in these primate models were likely more similar to human concussion, such studies were limited to small group size. With regard to modern-day athletes and the biophysics of mTBI, data garnered through helmet accelerometer studies at the youth, high school, and college level have shown that athletes may experience a wide range of head impacts, as well as types of forces, and this research has also suggested that it may be more likely be that the cumulative number of head impacts best correlates with the potential for concussion occurrence or chronic effects. 2
Although there was no overt rotational motion of the mouse head or body observed during or after impact, there are likely small components of rotational acceleration that occur in the sagittal and coronal planes. The almost lissencephalic rodent brain can tolerate much greater acceleration and deceleration forces than nonhuman primates and humans. 90 Additionally, their almost linear neuroaxis may impede rotational shearing after head injury and render them less vulnerable to these traumatic forces. 90 –92 Thus, unlike human concussive injury, rotational forces may contribute less in this model of injury; however, these forces are likely present. Future studies should be performed with the use of accelerometers to more clearly characterize and systematically analyze the injury biomechanics and real-time measurement of forces, velocities, and accelerations of head impacts in this new model.
The single mTBI in this model can be classified as a concussion, given the few postconcussive behavioral abnormalities observed on the NSS and sleep studies. Future studies should aim to vary the injury severity, specifically lowering the severity further such that we can investigate the long-term effects of repetitive subconcussion. In addition, more research will be needed to investigate the effect of different interinjury intervals within this model. We chose six hits per day in an attempt to simulate the more-frequent head contact observed in contact sports. It may or may not be that less-frequent mTBI would result in the same outcomes. Varying the overall number of head impacts will need to be studied in a dose-dependent fashion as well.
Although the repetitive TBI mice in our study did show improvement in their NSS score over 1 month, other behavioral assessments revealed persistent behavioral deficits. Though we did not compare the repetitive TBI mice between time points, group performance on the EPM demonstrated increasing time spent on the open arms of the maze (increasing risk-taking behavior) over the time points in the study. Progressively worsening encephalopathy and neurological deterioration is a characteristic feature in CTE. In this regard, one limitation is the distinctive difference between the results observed in this study and the human disease phenotype. Whereas there were persistent deficits at chronic time points in the study, the statistical significance of the MWM probe test performance was lost, indicating possible improvement over time in this test. We did not observe a progressive behavioral deterioration across all behavioral tests. Possible explanations for not observing a worsening encephalopathy may include issues with the biomechanical aspects of the model itself, the number of hits, initial age of trauma, the interinjury interval chosen, species differences, the robust ability of mice to recover from experimental injury in general, or even the length of follow-up. Future studies should continue to investigate longer lengths of follow-up, and additional behavioral studies are needed to validate the initial findings we report. Not all cases of CTE are alike and there are several varying histomorphological, as well as clinical, phenotypes for this spectrum of disease. 7,10,74
Most of our knowledge regarding CTE has come from postmortem analyses and retrospective, demographic data. The risk factors implicated in the development of CTE and its variability in presentation after repetitive mTBI, as well as the underlying pathophysiological mechanisms, have yet to be clearly elucidated and are difficult to discern from autopsy studies. It is difficult for any animal model to perfectly resemble human disease; however, clinically relevant models are necessary for developing an understanding of the behavioral sequelae of concussion and repetitive mTBI, as well as how they relate to the numerous complex histopathological cascades. The spectrum of behavior observed in these mice, with this model of injury, recapitulates some of the neurobehavioral signs and symptoms noted in patients diagnosed with CTE. Repetitive mTBI in this model also resulted in some of the neuropathological features observed in CTE, and these results are described in our companion article. The majority of individuals who suffer from persistent postconcussive symptoms ultimately do not develop CTE. This novel model could allow for not only a controlled, mechanistic analysis of CTE in the future, but also the neurobehavioral sequelae of repetitive mTBI in general because of its ability to capture the spectrum of human disease, as well as not require the use of anesthesia. Continued investigations with this model may contribute to our understanding of factors that distinguish those patients with persistent post-traumatic behavioral symptoms from those that develop long-term neurodegenerative syndromes, such as CTE.
Footnotes
Acknowledgments
The authors are thankful to Ken Kellerson of Millenium Machinery for his assistance in developing the helmet and modified rubber impactor. This work was supported, in part, by the NIH (grant nos. NIH-R25-NS-065748 [to A.L.P.] and NIH-R01-NS-067435 [to J.H.H.]), as well as a University of Rochester Institutional grant (to J.H.H.). Additionally, the project described in this publication was supported by the University of Rochester (CTSA award no. UL1 TR000042) from the National Center for Advancing Translational Sciences of the NIH (to A.L.P.). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Author Disclosure Statement
No competing financial interests exist.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
