Abstract
The triantennary N-acetylgalactosamine (GalNAc3) cluster has demonstrated the utility of receptor-mediated uptake of ligand-conjugated antisense drugs targeting RNA expressed by hepatocytes. GalNAc3-conjugated 2′-O-methoxyethyl (2′MOE) modified antisense oligonucleotides (ASOs) have demonstrated a higher potency than the unconjugated form to support lower doses for an equivalent pharmacological effect. We utilized the Ionis integrated safety database to compare four GalNAc3-conjugated and four same-sequence unconjugated 2′MOE ASOs. This assessment evaluated data from eight randomized placebo-controlled dose-ranging phase 1 studies involving 195 healthy volunteers (79 GalNAc3 ASO, 24 placebo; 71 ASO, 21 placebo). No safety signals were identified by the incidence of abnormal threshold values in clinical laboratory tests for either ASO group. However, there was a significant increase in mean alanine transaminase levels compared with placebo in the upper dose range of the unconjugated 2′MOE ASO group. The mean percentage of subcutaneous injections leading to local cutaneous reaction was 30-fold lower in the GalNAc3-conjugated ASO group compared with the unconjugated ASO group (0.9% vs. 28.6%), with no incidence of flu-like reactions (0.0% vs. 0.7%). Three subjects (4.2%) in the unconjugated ASO group discontinued dosing. An improvement in the overall safety and tolerability profile of GalNAc3-conjugated 2′MOE ASOs is evident in this comparison of short-term clinical data in healthy volunteers.
Introduction
Targeted delivery of antisense drugs to specific tissues and cell types by conjugation of ligands that are recognized and assimilated by cell surface receptors has created the opportunity for an increase in the therapeutic index as well as a broader range of therapeutic applications. The triantennary N-acetylgalactosamine (GalNAc3) ligand is the first to demonstrate this advancement in the antisense technology by enhancing delivery to hepatocytes through the asialoglycoprotein receptor (ASGPR) [1]. ASGPR is abundantly expressed by hepatocytes and enhances uptake of GalNAc3-conjugated antisense oligonucleotides (ASOs) through the major subunit ASGR1 [2,3].
The ASO is liberated from the GalNAc3 ligand upon receptor-mediated intracellular uptake and released from the endosomal compartment for productive delivery to the target RNA [1,4]. This ligand-conjugated antisense (LICA) technology provides the opportunity to treat pathologies and conditions that stem from proteins and RNAs expressed by the liver [5–9]. GalNAc3-conjugated 2′-O-methoxyethyl (2′MOE) modified ASOs are currently in all phases of clinical development [9–13]. This integrated assessment presents a comparison of the safety and tolerability data of investigational GalNAc3-conjugated ASOs compared with the less potent unconjugated ASOs of same sequence [7,9,14,15].
Integrated assessment of clinical data across an ASO chemical class allows for identification and characterization of class effects, including clinically relevant safety signals. Our integrated safety assessments focus on markers of injury or function of organs, tissues, and cells that are considered as potential sites for antisense drug toxicity in humans. Markers are selected based on the generic disposition of ASOs (absorption, distribution, metabolism, and excretion), and prior observations from individual clinical trials and nonhuman primate studies [16–19].
Fifteen GalNAc3-conjugated 2′MOE-modified ASOs are in clinical development today. Integrated assessment of placebo-controlled phase 1 data for this chemical class demonstrated an up to 30-fold increase in potency relative to the unconjugated 2′MOE ASO, with no indication of a class effect upon analysis of standard safety laboratory tests across all doses tested [19].
The recent integrated assessment of placebo-controlled phase 2 data extended these results, finding no evidence of a class effect with up to 1 year of treatment in all monthly doses tested [20]. In this analysis, we compare the safety and tolerability profiles of a set of GalNAc3-conjugated 2′MOE ASOs and unconjugated 2′MOE ASOs at their respective dose ranges using placebo-controlled data from phase 1 dose-ranging trials in healthy volunteers.
Materials and Methods
Study design and population
The Integrated Safety Database (ISDB) consists of available clinical trial data from evaluation of Ionis investigational antisense drugs [21]. Individual study data sets were harmonized and imported into one Statistical Analysis System (SAS) data set for each laboratory test for analyses utilizing the SAS v9.4 program [21]. This ISDB analysis included data from a total of eight phase 1 dose-ranging trials in healthy volunteers.
All clinical trial protocols were approved by the appropriate institutional review boards and/or independent ethics committees; and performed in accordance with the guidelines of Good Clinical Practice and Declaration of Helsinki. All subjects provided written informed consent before participation in the trial. All protocols included a placebo-controlled group. Subcutaneous injection was the route of study drug administration for each trial protocols.
Interventions and dosing protocol
The clinical trials included in this assessment evaluated four different GalNAc3-conjugated 2′MOE ASOs and four same-sequence unconjugated 2′MOE ASOs (Supplementary Table S1). Healthy volunteers received four to eight subcutaneous injections of either a GalNAc3-conjugated ASO (dose range: 10–120 mg), an unconjugated ASO (dose range: 40–450 mg) or corresponding placebo (0.9% saline) according to the dosing protocol of the specific clinical trial (Supplementary Table S2). The same dose categories were applied for this assessment of integrated clinical trial data as used in prior ISDB assessments of each chemical class [16,19].
Safety assessments
Incidence of abnormal threshold events
Incidence of abnormal threshold events were measured by sentinel laboratory tests for liver function [alanine transaminase (ALT), aspartate transaminase (AST), total bilirubin, alkaline phosphatase, and albumin], kidney function [serum creatinine, blood urea nitrogen, estimated glomerular filtration rate (eGFR), and urine protein] and hematology (platelets, hemoglobin, hematocrit, absolute lymphocyte counts, and absolute neutrophil count). eGFR was calculated using the Modification of Diet in Renal Disease equation [22]. For event criteria defined by fold change, the baseline value was used if greater than the upper limit of normal (ULN) or lesser than the lower limit of normal (LLN). If there was more than one value for a laboratory test, then the worst measurement and the corresponding ULN or LLN were used.
Abnormal laboratory values (falling outside the normal range or reaching the specified threshold) were defined by protocol stopping rules, standard reporting, or Grade 3 criteria provided by the Food and Drug Administration in Guidance to Industry for healthy adults and adolescents [23]. For all liver and kidney function and hematology tests, except for ALT and AST, a confirmed event was defined as a consecutive abnormal laboratory value on a different day. If there was no consecutive test to confirm, then the initial observation was presumed confirmed.
If there were multiple values on the same day but at a different time, the worst value was used. For ALT and AST, a confirmed event was defined as increased levels on two consecutive measurements at least 7 days apart, with all values between the initial and subsequent test also above the specified threshold. ALT, serum creatinine, and platelet levels were measured over time at each dose and included all study data up to 10 days after the last dose. Baseline was defined as the last nonmissing value before the first dose. The LLN and ULN used in the laboratory tests have been published previously [16,19].
Tolerability
Tolerability was measured according to local cutaneous reactions at injection site (LCRIS), flu-like reactions (FLRs) after injection and dose discontinuation rates. LCRIS was defined as injection site erythema, swelling, pruritus, pain, or tenderness that started the day of subcutaneous injection and persisted (start to stop) for 2 days or more. Events with onset date on the day of injection and missing resolution date were included.
FLRs were defined as either having influenza-like illness or pyrexia (including feeling hot or an increase in body temperature), plus at least two of the following: chills, myalgia, and arthralgia. Symptoms must have started on the day of the injection or the next day. The percentage of injections leading to LCRIS or FLR was also calculated as follows for each subject: (A/B)*100, where A = number of injections leading to LCRIS, and B = total number of injections.
Statistical methods
Data are presented by the incidence of events and descriptive summary statistics of laboratory test results by dose category. The data set included all healthy volunteers who received at least one dose of study drug (safety population) and had at least one post-baseline value, each data point represents the mean value from a minimum of at least six subjects and two ASOs. A meta-analysis using subject-level data was performed to compare ASO dose groups with placebo group. The endpoints evaluated were the absolute changes from baseline. The data were compared between ASO dose groups and placebo using an analysis of covariance model with the dose group and trial as the fixed factors and baseline value as a covariate.
Results
Study population
This analysis included 195 healthy volunteers; 79 subjects received GalNAc₃-conjugated 2′MOE ASOs with 24 subjects receiving placebo, and 71 subjects received the 2′MOE ASOs with 21 subjects receiving placebo. Demographics and baseline characteristics of the clinical study populations are summarized in Supplementary Table S3.
Treatment profiles
The mean number of doses received was 5.5 for the GalNAc3-conjugated 2′MOE ASOs and 6.5 for the unconjugated 2′MOE ASOs. The average treatment duration was similar between GalNAc3-conjugated 2′MOE ASOs and unconjugated 2′MOE ASOs (28.2 and 25.7 days, respectively). For the GalNAc3-conjugated 2′MOE ASOs, the mean exposure was approximately eightfold lower at 187 mg relative to the unconjugated 2′MOE ASOs, with a mean exposure of 1,495 mg. See Supplementary Fig. S1.
Safety laboratory tests
Liver
There were no incidences of abnormal liver function associated with measurements of ALT, AST, total bilirubin, and alkaline phosphatase (Table 1). Mean ALT levels, when analyzed by dose number, were all within normal range for both classes of ASOs (Fig. 1).
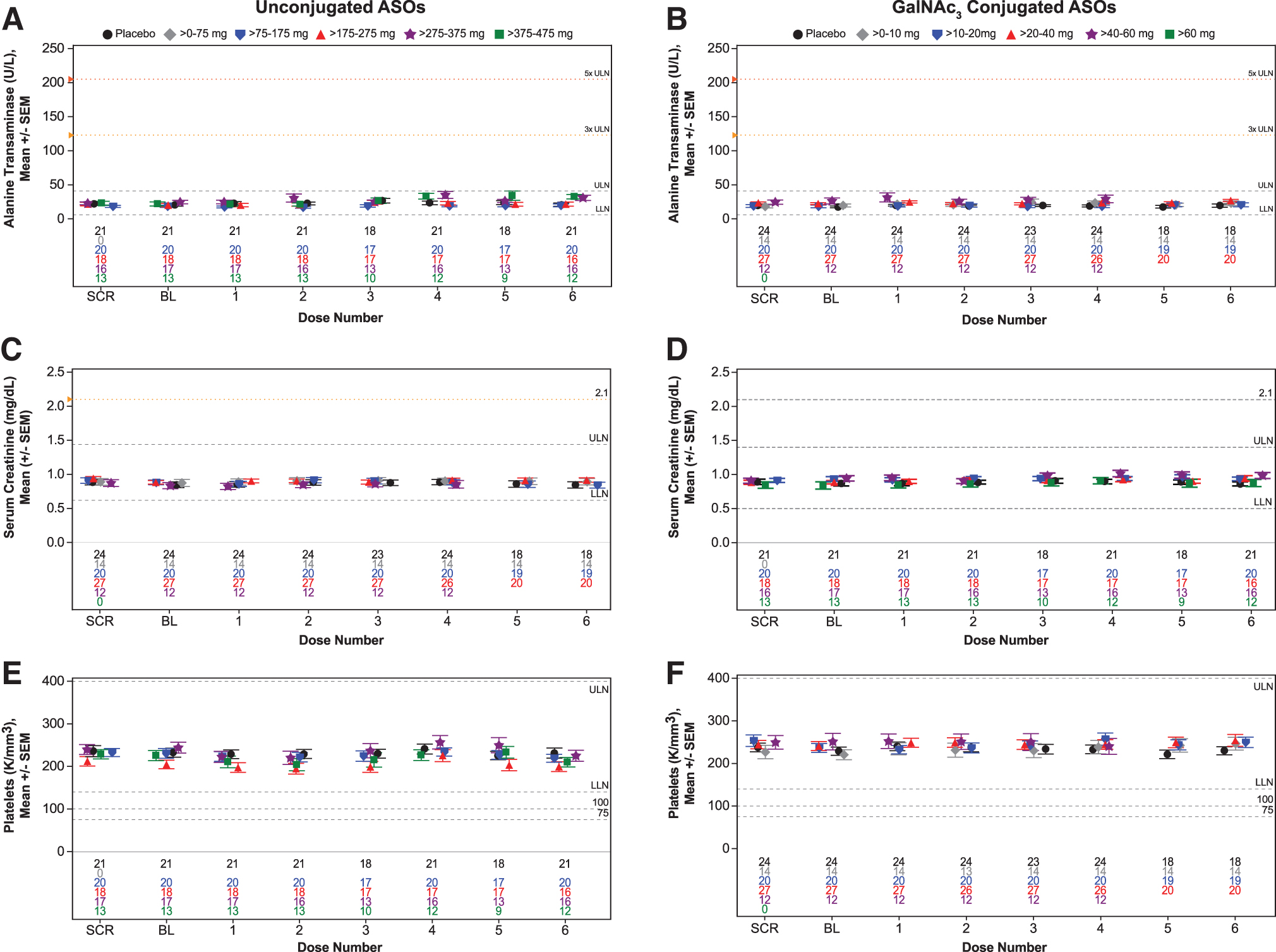
Sentinel laboratory tests results from healthy volunteers treated with multiple doses up to 6 weeks by dose category. Mean levels by dose number for unconjugated 2′MOE ASO and GalNAc3-conjugated 2′MOE ASO groups in ALT
Incidence of Confirmed Abnormal Laboratory Tests
F: female; M: male.
2′MOE, 2′-O-methoxyethyl; ALT, alanine transaminase; ASO, antisense oligonucleotide; AST, aspartate transaminase; BL, baseline; eGFR, estimated glomerular filtration rate; GalNAc3-conjugated 2′MOE ASOs, triantennary N-acetylgalactosamine-conjugated PS 2′MOE chimeric antisense oligonucleotides; ULN, upper limit of normal.
The mean ALT change from baseline level in the unconjugated 2′MOE ASOs, however, indicated a small incremental, yet statistically significant, increase in the >275–375 mg and >375–475 mg dose groups when compared with placebo starting at Dose 4 with a maximum difference in LSM of 10.6 U/L in the highest dose group at Dose 6 (Fig. 2 and Supplementary Table S4). No significant changes in ALT levels from baseline were observed across dose groups or dose numbers for the GalNAc3-conjugated 2′MOE ASOs compared with placebo (Fig. 2 and Supplementary Table S5).
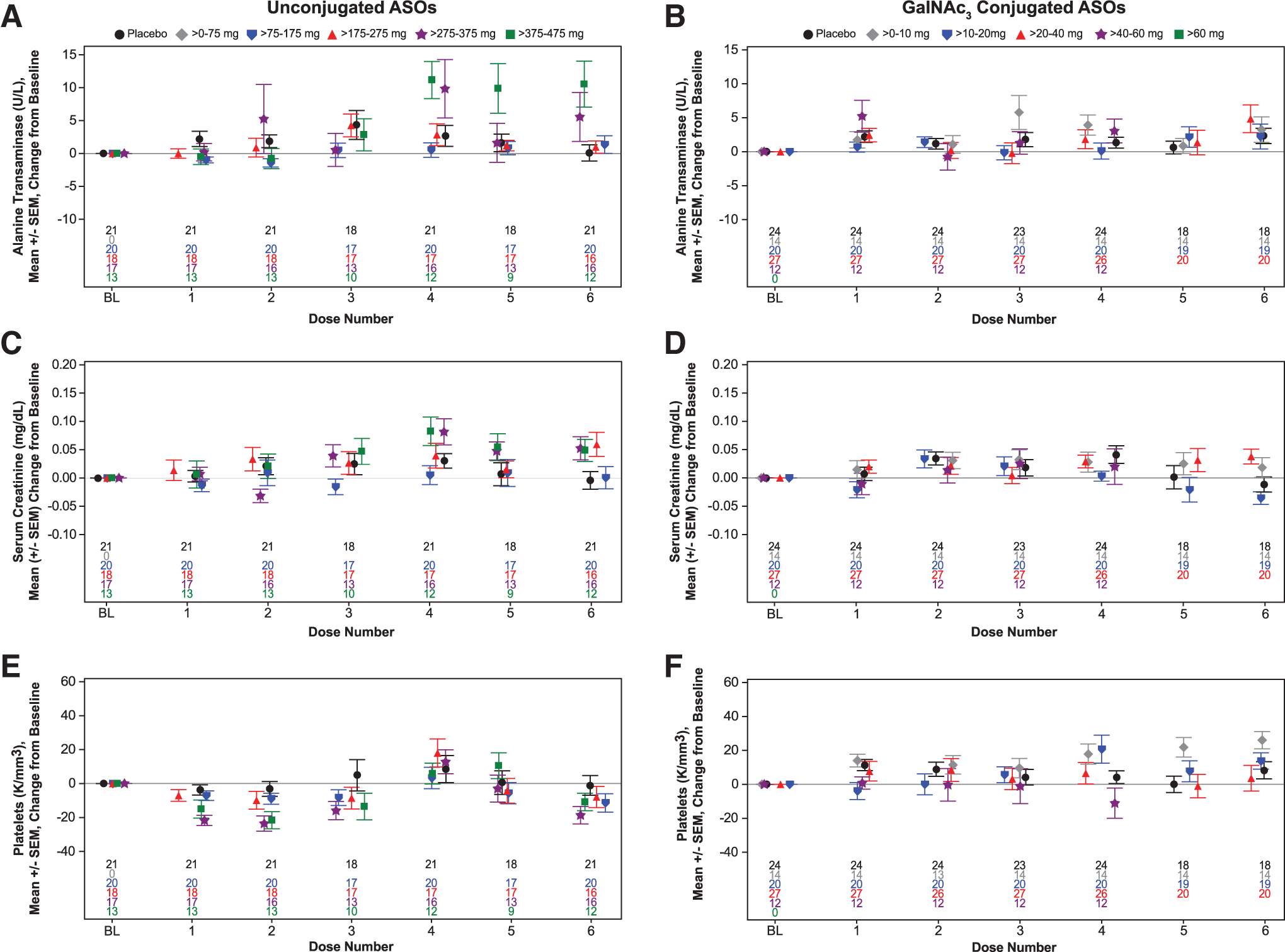
Sentinel laboratory tests results from healthy volunteers treated with multiple doses up to 6 weeks by dose category. Mean change from baseline by dose number for unconjugated 2′MOE ASO and GalNAc3-conjugated 2′MOE ASO groups in ALT
Kidney
For kidney function, there was a single incidence of an abnormal threshold event in the unconjugated 2′MOE ASO group (Table 1). An eGFR value of <60 mL/min per 1.73 m2 was observed in a female, who was <90 mL/min per 1.73 m2 at baseline with a value of 68 mL/min per 1.73 m2. Mean serum creatinine levels were consistently within the range of normal in both ASO classes by dose number (Fig. 1). Mean change from baseline in serum creatinine levels did not indicate any dose-dependent effects (Fig. 2 and Supplementary Tables S6 and S7).
Hematology
For hematologic parameters, there were a few incidences of abnormal measurements, which differed between GalNAc3-conjugated and unconjugated 2′MOE ASOs (Table 1). In the GalNAc3-conjugated 2′MOE ASO group, one male subject experienced a hemoglobin level <10.5 g/dL; and there was one incident of a hematocrit percentage <0.85 × baseline and one incident of an absolute neutrophil count <1.0 K/mm3 in the unconjugated 2′MOE ASO group. Mean platelet levels were all within normal range by dose number, with no dose-dependent changes (Fig. 1).
Mean change from baseline in platelet counts for the unconjugated 2′MOE ASOs indicated a small incremental, yet statistically significant, decrease in the >275–375 mg and >375–475 mg dose groups when compared with placebo after the initial doses with a maximum difference in LSM of −21 K/mm3 in the highest dose group at Dose 2 and 3 (Fig. 2 and Supplementary Table S8). No dose-dependent change in platelet levels were observed in the GalNAc3-conjugated 2′MOE ASOs (Fig. 2 and Supplementary Table S9).
Tolerability
The mean percentage of injections leading to LCRIS was 0.9% in the GalNAc3-conjugated 2′MOE ASO group versus 28.6% in the unconjugated 2′MOE ASO group, an ∼30-fold reduction (Table 2). Comparison between unconjugated and GalNAc3-conjugated ASOs demonstrated a higher incidence of injection site reactions in the unconjugated ASO group than in the conjugated group owing to the lower doses administered of the GalNAc3 conjugates (Fig. 3). FLRs only occurred in the unconjugated 2′MOE ASO group, with the mean percentage of injections leading to FLR being 0.7% (Table 2).
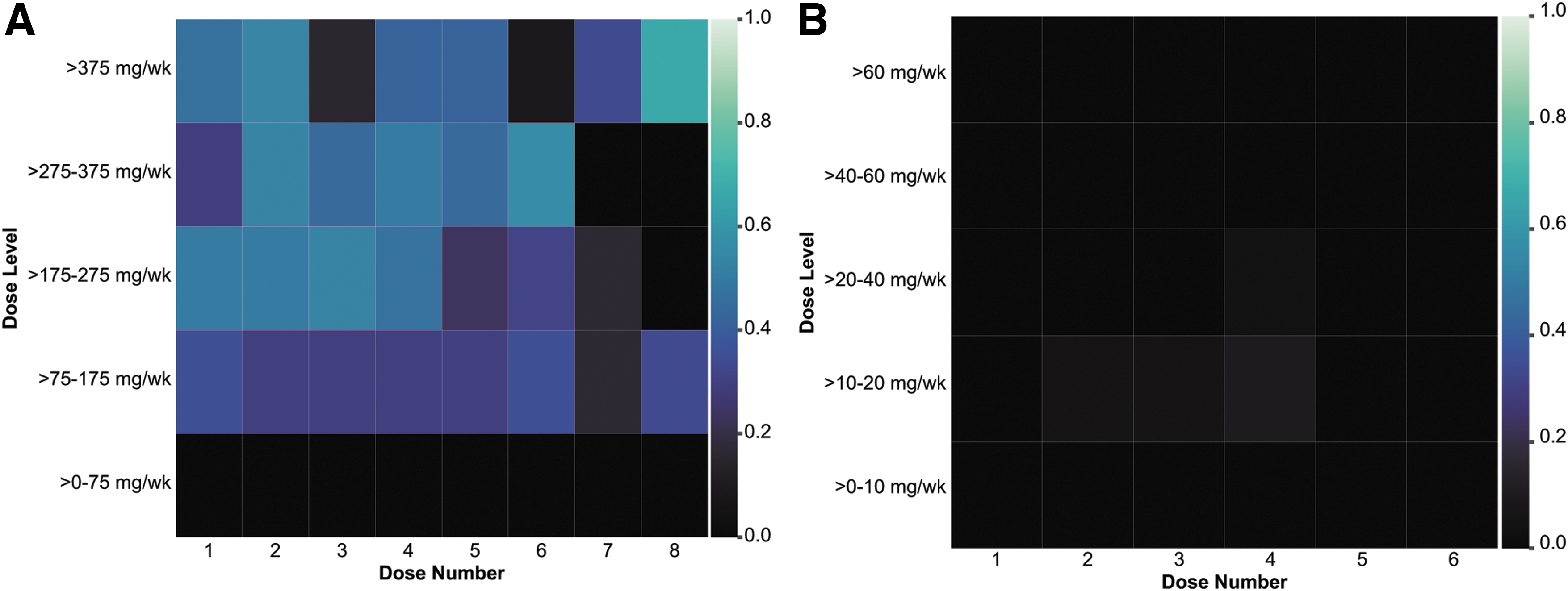
Incidence of injection site reactions by dose number and dose level in the unconjugated 2′MOE ASO group
Tolerability of GalNAc3-conjugated and Unconjugated 2′MOE Antisense Drugs
FLR, flu-like reaction; LCRIS, local cutaneous reactions at injection site.
The incidence of LCRIS was limited to three subjects (3.8%) in the GalNAc3-conjugated 2′MOE ASO group with all events reported mild in severity. The unconjugated 2′MOE ASO group exhibited an incidence of 47.9% (34 subjects) for LCRIS events of mild severity and 14.1% (10 subjects) incidence in LCRIS of moderate severity. Notably, there was no incidence of a severe LCRIS or FLR event in either ASO group. There were no dose discontinuations in the GalNAc3-conjugated 2′MOE ASO group. However, three subjects (4.2%) discontinued dosing in the unconjugated 2′MOE ASO group due to LCRIS or FLR.
Discussion
An improvement in the safety and tolerability profile of the GalNAc3-conjugated 2′MOE ASOs is evident in this head-to-head comparison with the unconjugated 2′MOE ASOs from short-term clinical studies in healthy volunteers. Although no safety signals were identified for either ASO class in this data set, as measured by the incidence of confirmed abnormal threshold values in sentinel laboratory tests for liver and kidney function, and hematology, there was a dose-dependent increase from baseline in the mean ALT level at the upper dose range of only the unconjugated 2′MOE ASOs [16].
The equivalence in pharmacological effect achieved by GalNAc3-conjugated 2′MOE ASOs at hepatocyte concentrations that are two- to threefold lower than that of the unconjugated form is likely one reason of for this differential effect over the dose ranges tested [19].
Although the fraction of dose distributed to the liver of GalNAc3-conjugated 2′MOE ASOs is about the same as the unconjugated 2′MOE ASOs, ∼80% of the total drug in the liver distributes to hepatocytes for the GalNAc3 conjugates in contrast to ∼12% with the unconjugated ASOs. This differential distribution to hepatocytes (vs. nonparenchymal cells) has been demonstrated by cellular fractionation studies in mice [24], and is also visualized by immunolocalization of unconjugated and GalNAc3-conjugated 2′MOE ASOs in monkey liver (Supplementary Fig. S2).
At near equivalent number of doses and treatment duration, there is an approximate eightfold reduction in total systemic exposure with the lower doses of the GalNAc3-conjugated 2′MOE ASOs included in the current phase 1 assessment (Supplementary Table S2). Lower doses also present with lower plasma drug concentrations at peak (Cmax) and over time (area-under-curve) that are approximately two orders of magnitude lower at doses of equivalent hepatocyte target reduction for unconjugated 2′MOE ASOs, or the respective ED50 [1,19].
Effective target reduction and an acceptable safety and tolerability profile with weekly dosing over the dose ranges tested in the initial phase 1 trials, along with the long liver half-life observed in nonhuman primates [25], supported evaluation of a less frequent monthly dose schedule in the next phase 1 trials of GalNAc3-conjugated 2′MOE modified ASOs [5,6]. Phase 2 trials with up to 1 year of treatment have now provided further evidence of the improved safety profile of this chemical class with the less frequent monthly dose intervals [5,10,12,20].
Measures of tolerability also differentiated the GalNAc3-conjugated 2′MOE ASOs from the unconjugated ASOs with an ∼30-fold reduction in the mean percentage of injections leading to LCRIS, and no incidence of FLR or dose discontinuation due these events. Since the sequence and overall chemical composition of the ASOs classes are the same, the increased potency of the GalNAc3 conjugate means a lower dose for an equivalent pharmacological effect and a 10-fold reduction in injection volume at a similar concentration for the GalNAc3-conjugated versus unconjugated ASOs. The reduced amount of ASO administered by subcutaneous injection is considered the primary basis of the consequent reduction in LCRIS.
Notably, early clinical research on 2′MOE modified ASOs discovered that the degree of histological changes within skin biopsy sites correlated directly with the amount of drug present 24–48 h post-SC injection [26]. Injection site reactions have been widely reported for ASOs and are often the most common treatment-emergent adverse event [14,27–31]. As tolerability issues such as injection site reactions can lead to treatment discontinuations, a reduction in occurrence is a significant improvement for ASO therapy and especially important for diseases requiring long-term treatment.
Advances in preclinical ASO research, including assay development, have allowed for better understanding and identification of ASO structures and sequences with the potential to induce an inflammatory response, such as LCRIS and FLR [32]. Analyses of ISDB data from the unconjugated 2′MOE ASOs has found a parallel between these advancements and improvement in the tolerability profile of this class of ASOs [31,33], with the most recent analysis finding a strong correlation between FLR adverse events and the ranking of ASOs in a preclinical screening assay developed for identification of minimally inflammatory ASO sequences [33]. Accordingly, improvements in screening assays, paired with the LICA technology [34,35], could potentially lead to further improvements in the safety and tolerability profiles of ASOs.
The results presented from the current integrated data assessment are overall comparable with those reported on phase 1 studies of individual GalNAc3-conjugated 2′MOE ASOs in healthy volunteers with treatments up to 6 weeks [7–9] and in studies of the unconjugated 2′MOE ASOs in healthy volunteers with treatments up to 4 weeks [14,36].
A limitation of this analysis includes the relatively short treatment period (4–6 weeks), as it is possible that other tolerability and safety issues may present with longer ASO exposure. The number of subjects within each ASO group and dose categories was also small. The low incidence of FLRs in subjects treated with the unconjugated 2′MOE ASOs, and lack of incidence in subjects treated with the GalNAc₃ conjugates may be based in this limitation. In addition, the studies were conducted in healthy volunteers, a population that is not necessarily reflective of the intended treatment population for a given RNA target.
GalNAc₃-conjugated 2′MOE ASOs have demonstrated robust utility for targeted delivery to hepatocytes through increased potency and improved tolerability. The improved tolerability with GalNAc₃-conjugated 2′MOE ASO likely reflects the usage of lower doses with the increased ASO potency. These results suggest improvements in safety and tolerability may be observed with other LICA drugs in development for enhanced delivery to other tissue and cell compartments [34,35].
Footnotes
Author Disclosure Statement
All authors are/were employees of Ionis Pharmaceuticals at the time of this research.
Funding Information
This study was supported by Ionis Pharmaceuticals. Initial writing support was provided by Parexel International and funded by Ionis Pharmaceuticals.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
