Abstract
Acute respiratory distress syndrome (ARDS), a rapid onset inflammatory lung disease with no effective specific therapy, typically has pathogenic etiology termed pneumonia. In previous studies nuclear factor-κB (NF-κB) inhibitor α super-repressor (IκBα-SR) and extracellular superoxide dismutase 3 (SOD3) reduced pneumonia severity when prophylactically delivered by viral vector. In this study, mRNA coding for green fluorescent protein, IκBα-SR, or SOD3 was complexed with cationic lipid, passed through a vibrating mesh nebulizer, and delivered to cell culture or directly to rats undergoing Escherichia coli pneumonia. Injury level was then assessed at 48 h. In vitro, expression was observed as early as 4 h in lung epithelial cells. IκBα-SR and wild-type IκBα mRNAs attenuated inflammatory markers, while SOD3 mRNA induced protective and antioxidant effects. In rat E. coli pneumonia, IκBα-SR mRNA reduced arterial carbon dioxide (pCO2) and reduced lung wet/dry ratio. SOD3 mRNA improved static lung compliance and alveolar-arterial oxygen gradient (AaDO2) and decreased bronchoalveolar lavage (BAL) bacteria load. White cell infiltration and inflammatory cytokine concentrations in BAL and serum were reduced by both mRNA treatments compared to scrambled mRNA controls. These findings indicate nebulized mRNA therapeutics are a promising approach to ARDS therapy, with rapid expression of protein and observable amelioration of pneumonia symptoms.
Introduction
Acute respiratory distress syndrome (ARDS) is a rapid-onset, acute inflammatory lung injury with a continuing high mortality and no effective specific therapy. Current standard of care is supportive, such as fluid management and mechanical ventilation strategies [1]. It has diverse etiologies, of which pathogen-induced pneumonia is among the most common and debilitating [2,3]. The increased number of patients requiring critical care for COVID-19-mediated ARDS has placed huge emphasis on the need for improved therapies [4,5]. Lung-targeting aerosol-mediated delivery offers increased local efficacy and minimized systemic side effects [6,7]. Vibrating mesh nebulizers (VMNs) in particular provide a promising technology for targeting lung tissue in ARDS patients, offering distal penetration and no adverse effects on larger and more delicate drugs, and compatibility with all respiratory support interventions [8,9].
Recent advances in mRNA technology have paved the way for novel therapies [10,11], while the COVID-19 pandemic has accelerated the development of safe and robust mRNA vector systems [12–15]. Here, we focus on evaluating the potential for a semisynthetic mRNA technology to reduce ARDS severity.
It has previously been shown that overexpression of specific genes can reduce the severity of Escherichia coli pneumonia [16]. Nuclear factor (NF)-κB is a transcriptional regulator controlling the expression of hundreds of genes involved in inflammation and other processes [17]. The efficacy of NF-κB inhibition with inhibitor kappa B alpha (IκBα) overexpression was also reported in ventilator-induced (VILI) [18], and endotoxin-induced [19] lung injury models. In pneumonia, infiltrating neutrophils generate reactive oxygen species that can cause cellular dysfunction and cell death in adjacent tissues and organs and play a critical role ARDS pathogenesis [20].
Superoxide dismutase (SOD) oxidizes and then reduces superoxide to less reactive peroxide and thereby acts as a defense factor in the lung and other tissues. SOD overexpression decreased severity in endotoxin-induced lung injury [21] and protected the lung against emphysema in rodent models [22]. To date, viral vector delivery systems have been prevalent for preclinical overexpression studies, leading to longer therapeutic delivery to injury induction intervals. In this study, we aimed to deliver rapidly expressing IκBα super-repressor (IκBα-SR) and extracellular SOD (SOD3) mRNAs to pulmonary cells using VMN and to examine their potential to ameliorate the severity of a rodent pneumonia ARDS model. Some of the results of this study have been previously reported in abstract form [23].
Materials and Methods
Cell lines
BEAS-2B cells (ATCC®, Manassas, VA) were cultured in Dulbecco's modified Eagle's medium (DMEM)/Nutrient F12 Ham medium (Sigma-Aldrich, Dublin, Ireland), with 10% fetal bovine serum, penicillin G (100 U/mL), and streptomycin (100 μg/mL). The A549/NF-κB-luc cell line (Thermo Fisher, Waltham, MA) is the alveolar epithelial A549 incorporating a chromosomally integrated luciferase reporter of NF-κB transcription factor activity. These were passaged in RPMI-1640 medium (Sigma-Aldrich), with 10% fetal bovine serum, penicillin G (100 U/mL), and streptomycin (100 μg/mL). Human primary Small Airway Epithelial Cells (SAEC) (Lonza, Basel, Switzerland) were cultured in small airway cell basal medium with SAGM BulletKit (Lonza) during the expansion phase. All cell lines were cultured at 37°C in a humidified incubator with 5% CO2 in air.
Lipid/mRNA transfection
BEAS-2B and A549 cells were seeded at 4 × 104 cells/well and SAEC at 5 × 104 cells/well in 96-well plates to reach ∼80% confluence for transfection 48 h later. Lipofectamine 3000 (L3000; Thermo Fisher) was complexed with scrambled mRNA or mRNA encoding enhanced green fluorescent protein (GFP), IκBα wild type (WT), IκBα-SR, and SOD3 (Factor Bioscience, MA) (Supplementary Table S1) at a ratio of 3 μL L3000 per 1 μg mRNA. Complexes were prepared in OptiMEM (Thermo Fisher) as per manufacturer's instructions. Complexes incorporating GFP mRNA were passed through the Aerogen Solo VMN with Aerogen ProX controller (Aerogen Ltd., Galway, Ireland), aerosol collected by condensation and added to BEAS-2B cells. Non-nebulized GFP complexes were added directly to cells without aerosolization and all wells received 100 ng of mRNA.
Transfection efficiency was determined by fluorescence microscopy imaging using the CKX53 inverted microscope (Olympus United Kingdom, Southend-on-Sea, United Kingdom) at various time points followed by mean fluorescent intensity analysis using GIMP software (version 2.10.24). At 48 h following transfection, a single-cell suspension of nebulized and control GFP complex-treated cells were prepared for flow cytometry by trypsinization and resuspension in PBS. GFP fluorescence was measured on the Accuri™ C6 Plus flow cytometer (BD Biosciences, Wokingham, United Kingdom). Cytoplasmic and secreted transgene products were quantified by ELISA as per manufacturer's instructions (R&D Systems, Abingdon, United Kingdom) (n = 3–5).
Aerosol droplet size characterization
Cascade impaction was used to assess likely deposition of transfection particles in the human lung post-nebulization. The mass median aerodynamic diameter (MMAD) of the nebulized complex dispersions was measured using the Next-Generation Impactor (NGI; Copley Scientific, Colwick, United Kingdom). Liposomal GFP complexes were counted in suspensions taken from the NGI using the NanoSight NS300 (Malvern Panalytical Ltd., Malvern, United Kingdom). Complexes nebulized through the NGI were washed from the stages and quantified by quantitative polymerase chain reaction (QPCR) (n = 3). The following primers were designed using Primer blast function of NCBI Forward 5′-CAAGATCCGCCACAACATCG-3′ and Reverse 5′-CTCAGGTAGTGGTTGTCGGG-3′. The primers are specific for GFP; Accession number: U57607. Amplicon size of QPCR was 111 bp and PCR annealing temperature was set at 55°C.
In vitro assays
Pulmonary cell monolayers were injured with E. coli lipopolysaccharide (LPS, 100 ng/mL) or interleukin-1β (IL-1β) (100 ng/mL) (Immunotools Ltd., Friesoythe, Germany). After 1h of endotoxin or cytokine injury, cells were exposed to mRNA complexes or scrambled control. Supernatant was collected after 48 h for inflammatory cytokine analysis. Cell viability was assessed where appropriate. Cells were harvested and lysed for transgene expression analysis. Naive controls were not exposed to injury or transfection complexes. All in vitro experiments were carried out twice to ensure repeatability.
Cell viability
A 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay was performed to determine the cytotoxic effects of transfection and superoxide on epithelial cell viability. MTT (Sigma-Aldrich) (100 μL of 100 μg/mL in DMEM) was added to wells on a 96-well plate and the plate returned to a tissue culture incubator for 2 h. The media/MTT mixture was aspirated and formazan crystals were dissolved in 200 μL methanol and absorbance read in a VICTOR™ X plate reader (Perkin Elmer, Waltham, MA) at 595 nm wavelength. Untreated cells were regarded as 100% viable as control (n = 3).
NF-κB activation assay
After exposure to injury and treatments, A549 cells were lysed in 100 μL of Reporter Lysis Buffer (Promega Corp., Madison, WI). Lysate was mixed with 40 μL of Bright-Glo luciferase substrate (Promega) and luminescence assessed in a VICTOR X plate reader (Perkin Elmer) as an indicator of NF-κB activation (n = 3).
Superoxide assay
Cells in 96-well plates were refed before treatment with 100 μL of a 70% Hank's Buffered Solution/30% RPMI mix to limit potential antioxidant effects of complete medium. A superoxide generating hypoxanthine (X) and xanthine oxidase (XO) (1.0 mU/mL) combination was added to cells and L3000/SOD3 complexes were added after 1 h. Supernatant was collected after 48 h and SOD activity assessed using a colorimetric SOD activity assay kit (Sigma-Aldrich) (n = 3).
In vivo ARDS model
All experimental work was approved by the Animal Care Research Ethics Committee of the University of Galway and conducted under license by the Health Products Regulatory Authority (HPRA), Ireland. Adult male Sprague Dawley rats (n = 45) (Envigo, Blackthorn, United Kingdom) weighing 350–450 g were used. Animals were randomized to sham (n = 9) and E. coli pneumonia groups (n = 34). Animals in the E. coli group were anesthetized using inhalational isoflurane (Iso-Vet; Chanelle, Galway, Ireland) delivered at 4%–5%, with an oxygen flow rate of 2 L/min. Animals were orotracheally intubated under direct vision using a guidewire and a 16G catheter (BD Insyte®; BD Biosciences). An E. coli dose of 1 × 109 CFU suspended in 0.3 mL PBS was administered intratracheally. Animals remained intubated and were placed in a sealed chamber while receiving a maintenance dose of 1%–2% isoflurane for 1 h.
Nebulization
Thirty-four animals receiving E. coli were randomized and after 1 h 40 μg of mRNA encoding IκBα-SR (n = 9) or SOD3 (n = 9) or scrambled control (n = 16) mRNA (Factor Bioscience) complexed with In Vivo-jetPEI (Polyplus-transfection) at an N/P ratio of 8 and administered via the Aerogen Solo nebulizer attached to the FlexiVent small animal ventilator (Scireq, Montreal, Canada). In a second series, animals (n = 2) received 40 μg of complexed GFP mRNA but did not receive E. coli. During nebulization, animals were ventilated with 90 breaths/min, tidal volume 8 mL/kg, and positive end-expiratory pressure of 3 cm H2O. Anesthesia was maintained with 1%–2% isoflurane. Nebulization actuation (duty cycle) was set at 20%. Animals were subsequently returned to high-efficiency particulate air (HEPA)-isolated cages and underwent HPRA regulated welfare monitoring until the end of the experiment at 48 h. Sham controls (n = 9) did not undergo any procedure.
Animal harvest
Animals were anesthetized with subcutaneous injection of ketamine 75 mg/kg (Ketamidor; Chanelle Pharma, Galway, Ireland) and medetomidine 0.5 mg/kg (Domitor; Vétoquinol, Dublin, Ireland). A tracheostomy was performed and intravenous and intra-arterial access was secured. Lungs were mechanically ventilated (Harvard Apparatus, Holliston, MA) at a respiratory rate of 90 breaths/min as previously described [21]. Anesthesia was maintained with 2 mg/kg Alfaxan® (Jurox, Crawley, United Kingdom). A recruitment maneuvre consisting of positive end-expiratory pressure 15 cm H2O for 20 breaths was applied at the start of the protocol and every 7–10 min thereafter. Arterial blood pressure, temperature, and peak airway pressure were continuously measured as in previous studies [24].
Arterial blood samples were drawn for analysis while animals were ventilated for 15 min at 21% O2 (room air) followed by 15 min at 100% O2. Blood gas concentrations, plasma lactate, and plasma glucose concentrations were measured using the ABL 90 Flex Plus (Radiometer, Copenhagen, Denmark) and static lung compliance was measured following administration of the paralytic agent atracurium besylate (0.5 mg/kg) while under general anesthesia (Tracrium; GlaxoSmithKline, Dublin, Ireland). Animals were euthanized by exsanguination while still under general anesthesia.
Tissue analysis
Immediately after postmortem, the heart-lung block was dissected and bronchoalveolar lavage (BAL) was performed. BAL fluid was analyzed for total white cell counts and lung bacterial colony counts and prepared for further differential staining after Cytospin concentration on glass slides. Clarified BAL samples were frozen at −80°C. Bio-Plex Pro Rat Cytokine Panel (Bio-Rad) was used to assess BAL samples for 23 different cytokines. Samples were diluted 1:4 and 50 μL of each sample loaded per well as per manufacturer's instructions. Serum and BAL concentrations of cytokine-induced neutrophil chemoattractant-1 (CINC-1), IL-6 (IL-6), and tumor necrosis factor-alpha (TNF-α) were also determined by sandwich ELISA (R&D Systems) as per manufacturer's instructions.
Lung lobes from animals administered GFP complexes were either (1) filled with and mounted in Tissue-Tek Optimal Cutting Temperature® Compound (OCT®; Sakura Finetek Europe B.V., Alphen aan den Rijn, Netherlands), snap frozen in liquid nitrogen-cooled isopentane, and stored at −80°C or (2) lobes were dissected and stored at −80°C. OCT mounted tissues were sectioned in 10 μm increments on a Leica CM1850 Cryostat (Leica Biosystem, Dublin, Ireland). Sections were imaged by fluorescence microscopy on an Olympus CKX53 inverted fluorescent microscope. A GFP ELISA (Abcam, Burlingame, CA) was performed by homogenizing 200 mg of lung tissue in Extraction Buffer and the protocol was completed as per manufacturer's instructions.
Statistical analysis
Data were analyzed using GraphPad Prism software 9.0 (GraphPad Software Ltd., San Diego, CA). Ordinary one-way analysis of variance followed by Tukey's multiple comparisons test was used to calculate the P values. Results are expressed as mean ± standard deviation. A P value of <0.05 was considered to indicate statistical significance.
Results
Semisynthetic mRNA expressed protein rapidly and efficiently in vitro and in vivo
Transgene expression was observed as early as 4 h after transfection in BEAS2B cells treated with both nebulized and non-nebulized complexes and was maintained to at least 48 h (Fig. 1A–E). While nebulization decreased fluorescence intensity at 48 h (Fig. 1E), expression was still robust and delivered dose could be increased by adjusting L3000 to a higher concentration during complex formulation. Flow cytometry measurements did not detect a significant reduction in GFP-positive cells between nebulized (54.2%) and non-nebulized (62.6%) complexes at 48 h (P > 0.05). Fluorescent images of nebulized In Vivo jetPEI/GFP complexes 48 h after administration to the rat lung showed a clear pattern of GFP fluorescence in epithelial cells (Fig. 1D) and quantitative assessment showed GFP at a concentration of 35,000 pg/g of homogenized lung tissue.
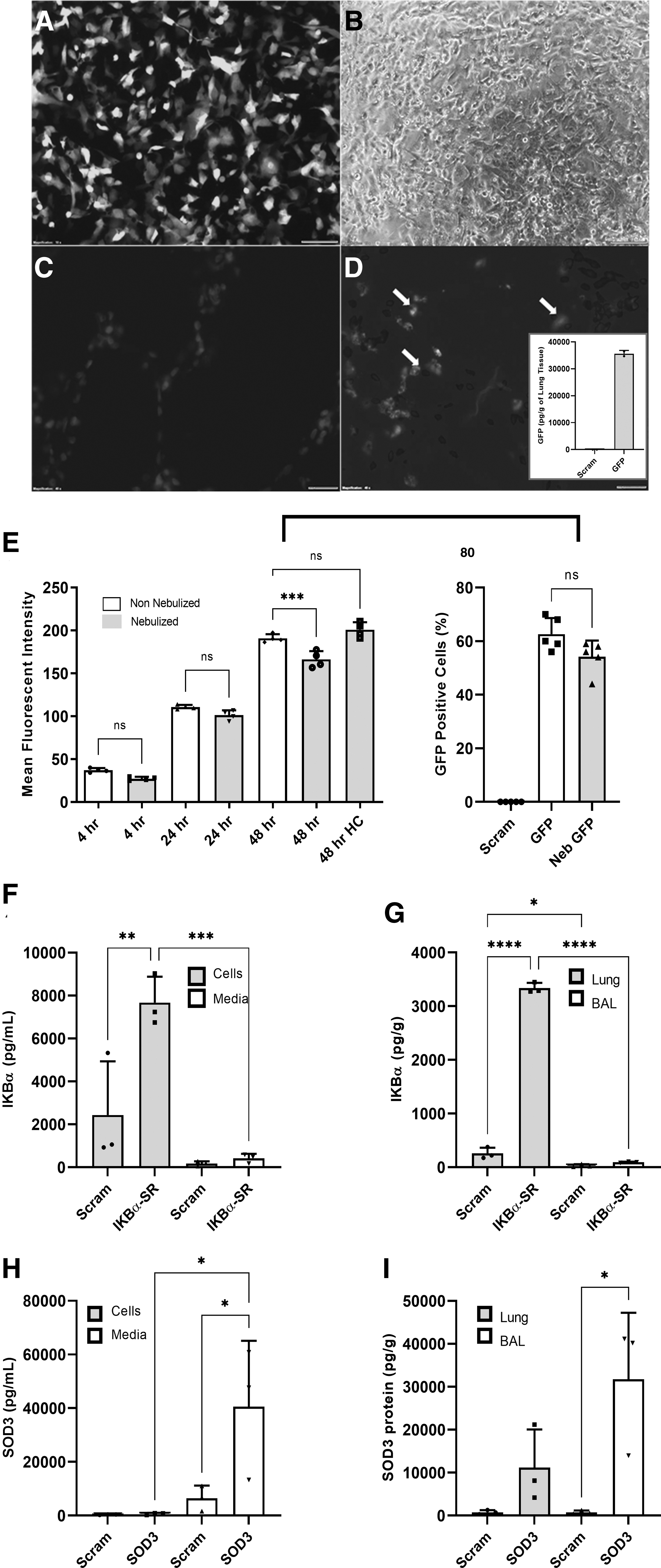
Transgene expression in vitro and in vivo.
Expression of total IκBα and SOD3 was significantly increased in vitro and in vivo in pulmonary cells administered respective complexes compared to those receiving scrambled mRNA control (Fig. 1F–I). Increased levels of total IκBα were observed in cells (7,672 pg/mL) compared to levels detected in growth media (419 pg/mL), where a significant increase in IκBα was not detected (Fig. 1F). Similarly in rat lungs, significant IκBα overexpression was only observed in homogenized lung tissue detected at a concentration of 3,337 pg/mL compared to endogenous levels in scrambled control group of 258 pg/mL. Levels circulating in BAL were low for both controls and IκBα transfected animals at 28 and 94 pg/mL, respectively (Fig. 1G).
SOD3 detection followed an opposite trend where expression was most elevated in cell culture media in vitro, detected at a concentration of 40,592 pg/mL in SOD3-treated group compared to 6,462 pg/mL in controls. Detection of SOD3 in cells was relatively low at 387 pg/mL in controls compared to 656 pg/mL in SOD3 transfected cells (Fig. 1H). In vivo, SOD3 was found to be expressed at 31,786 pg/mL in lung BAL and detected at 11,174 pg/mL in lung tissue (Fig. 1I).
Nebulized mRNA/lipid complexes have an aerosol particle profile likely to lead to distal penetration in the human lung
mRNA encoding GFP was complexed with Lipofectamine 3000 as per manufacturer's instructions and applied to a VMN attached to a NGI. QPCR analysis of collected samples indicated that complexes would likely be deposited in bronchial and alveolar space in human subjects (Supplementary Fig. S1), with MMAD most commonly retrieved from plates corresponding to 0.3–0.6 mm thickness airways.
In vitro mRNA transfection reduced inflammation and rescued cell viability
MTT analysis of BEAS2B and SAEC cells treated with scrambled, GFP, IκBα-WT, IκBα-SR, and SOD3 mRNA compared cell viability 48 h after transfection. Most complexes reduced cell viability somewhat in relationship to naive cells while SOD3 appeared to have a protective effect in both BEAS2B (96% viability) and SAEC (94% viability) cells types, compared to GFP mRNA, which reduced viability to 77% in BEAS2Bs and 80% in SAECs, and scrambled controls, which maintained 87% viability in BEAS2Bs and 82% in SAECs (Fig. 2A, F).
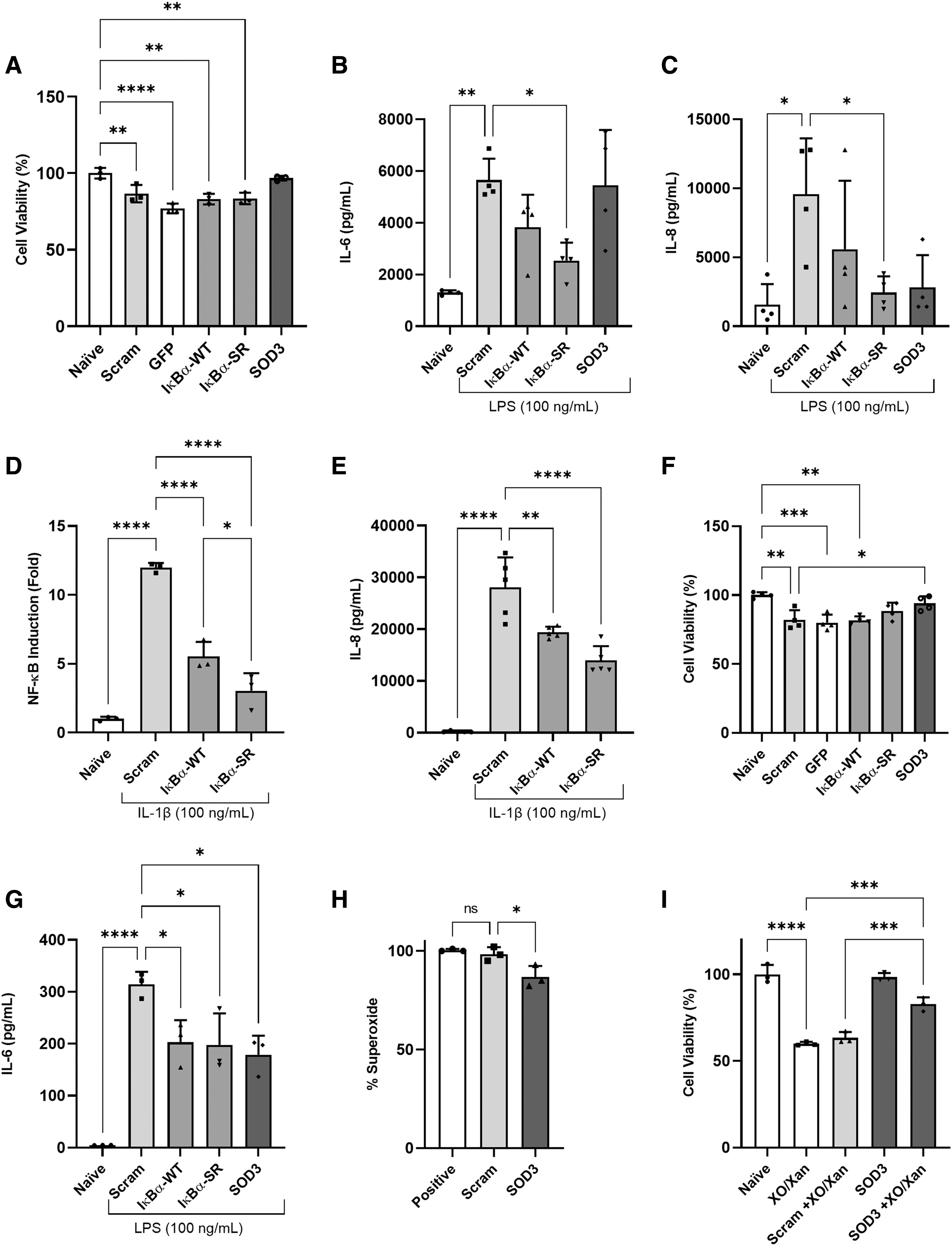
Safety and therapeutic efficacy of transgene expression in lung epithelial cell injury. Scrambled control, GFP, IκBα-WT, IκBα-SR, and SOD3 complexed with L3000 were added to BEAS2B
Importantly, there was no significant difference between scrambled and treatment mRNAs, ensuring that anti-inflammatory properties are not related to decreased cell viability in these groups. Transfected BEAS2B and SAEC treated with LPS (100 ng/mL) as an inducer of inflammatory cytokines showed effects on IL-8 and IL-6 levels measured after 48 h. Cells treated with IκBα-SR mRNA released significantly reduced levels of IL-6 (2,534 pg/mL) compared to scrambled control cells (5,651 pg/mL) (Fig. 2B) and similarly IL-8 concentration was reduced to 2,446 pg/mL from 9,564 pg/mL in cells receiving control mRNA (Fig. 2C).
Similarly, NF-κB (Fig. 2D) and IL-8 (Fig. 2E) induction was significantly reduced by both WT and SR transgenes in an A549/NF-κB-luc cell line. NF-Κb induction was reduced from ∼12-fold induction in control cells to a 5.5-fold induction with IκBα-WT and as low as a 3-fold induction with IκBα-SR treatment. All transgenes (IκBα-WT = 202 pg/mL, IκBα-SR = 197 pg/mL, SOD = 178 pg/mL) appeared to alleviate IL-6 inflammatory response in injured SAEC primary line compared to control mRNA (314 pg/mL) (Fig. 2G). Treatment with SOD3 mRNA also alleviated superoxide concentration from 98% to 86% (Fig. 2H) and improved cell viability in the presence of superoxide radicals from 63% to 83% (Fig. 2I).
Nebulized SOD3 and IκBα-SR complexes reduced E. coli pneumonia severity
In an animal model of E. coli pneumonia, nebulized IκBα-SR mRNA administration resulted in significantly reduced arterial carbon dioxide (pCO2) concentration from 31 mmHg in scrambled control animals to 24 mmHg in treated animals, approaching levels observed in sham (no injury) animals (22 mmHg) (Fig. 3A), as well as reduced lung tissue wet:dry ratio from 1.8 to 1.3 (Fig. 3D). SOD3 mRNA complex administration improved static lung compliance from 0.76 to 0.95 mL/mmHg (Fig. 3B), and alveolar-arterial oxygen gradient (AaDO2) from 364 to 263 mmHg (Fig. 3C). SOD3 mRNA treatment also decreased BAL bacteria load from 5,000 to 2,000 CFU/mL (Fig. 3E), and increased lung airspace percentage to 58.0% compared to scrambled mRNA controls (53.6%), the airspace of healthy animals was determined to be 61% (Fig. 3F).
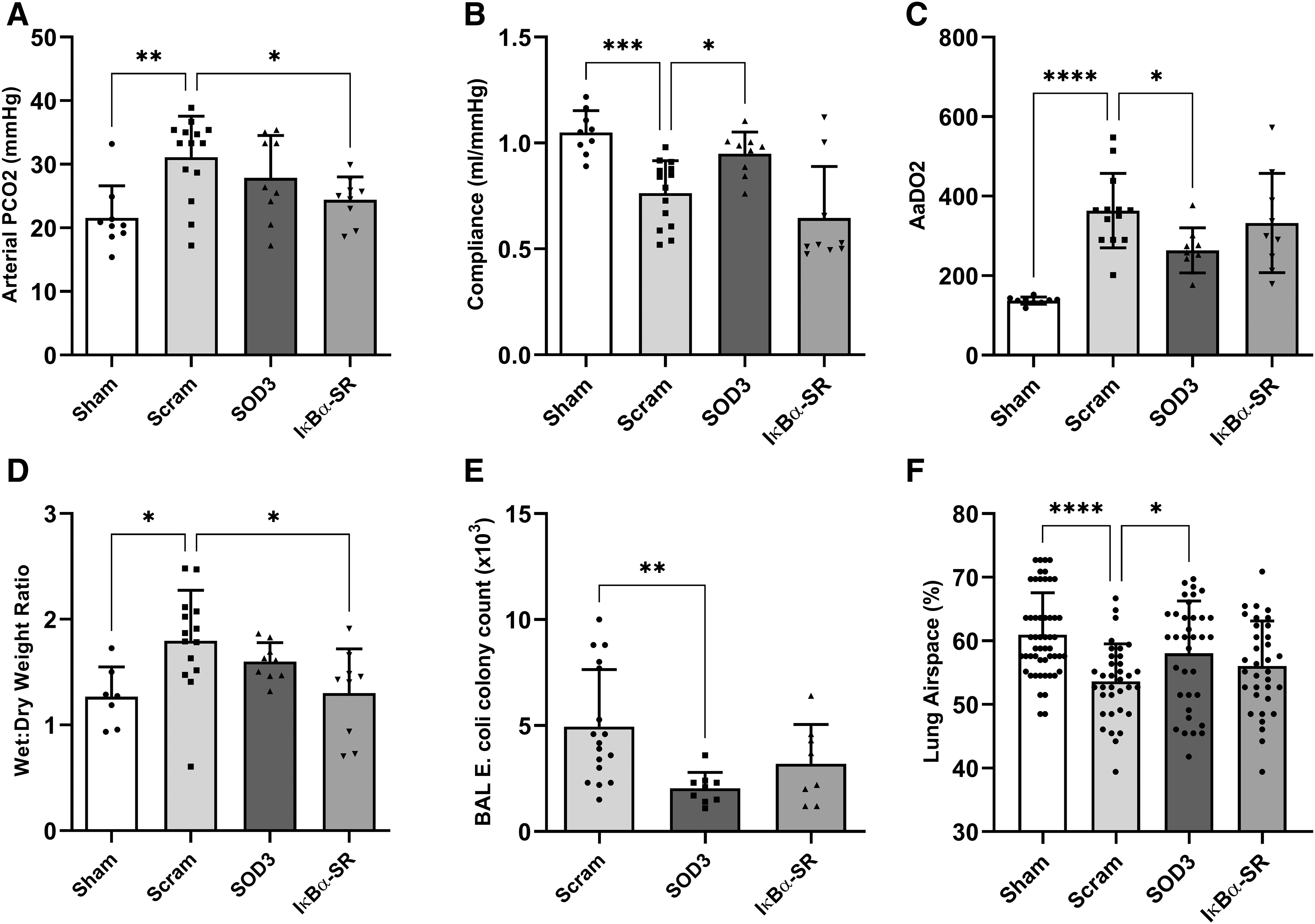
Nebulized SOD3 and IκBα-SR mRNA complexes restored physiological parameters in pneumonia. Blood gas and physiological parameters were assessed in rats 48 h after Escherichia coli pneumonia induction. Parameters measured include
Nebulized SOD3 and IκBα-SR complexes altered white cell counts
Total white cell infiltration in BAL samples was reduced from 62,433 cells/mL in scrambled control animals to 36,132 cell/mL in SOD3 and 37,333 cells/mL in IκBα-SR-treated animals (Fig. 4A). Differential staining allowed enumeration of the polymorphonuclear leukocytes (PMN) and mononuclear cells percentage in total white cell infiltrate (Fig. 4B). The neutrophil/lymphocyte ratio (NLR) was also reduced from 19.9 in control animals to 11.2 following SOD3 treatment and 13.3 after IκBα-SR mRNA treatment (Fig. 4C).
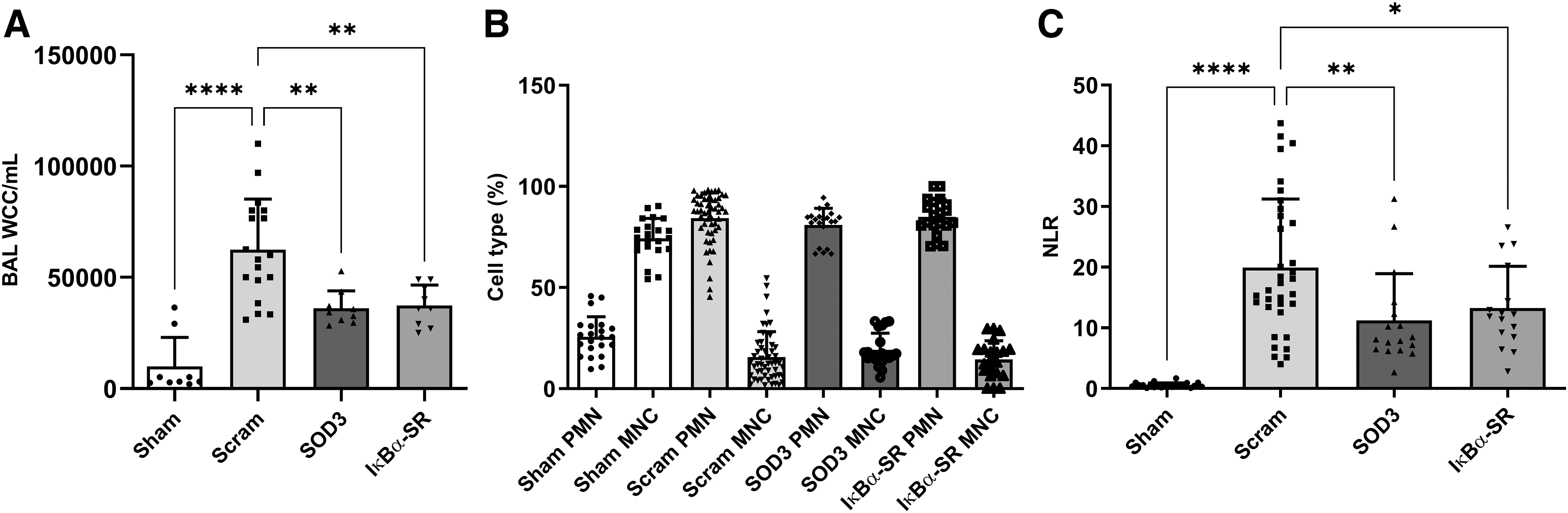
mRNA treatment effects on lung white cell infiltrates after E. coli pneumonia induction.
Nebulized SOD3 and IκBα-SR complexes reduced inflammatory cytokine concentrations
SOD3 (422 pg/mL) and IκBα-SR (453 pg/mL) mRNA complex treatment reduced inflammatory CINC-1 in BAL compared to controls (861 pg/mL) (Fig. 5A). Similarly, serum levels of CINC-1 were significantly lower in SOD3 (100 pg/mL) and IκBα-SR (117 pg/mL) treated animals compared to control group (180 pg/mL) (Fig. 5D). SOD3 and IκBα-SR both lowered circulating IL-6 levels in BAL; however, only IκBα-SR had a significant effect, reducing IL-6 concentration from 402 to 143 pg/mL, lower than that of healthy (sham) animals at 168 pg/mL (Fig. 5B). IκBα-SR similarly reduced serum levels of TNF-α from 125 to 72 pg/mL (Fig. 5F). Also, BAL IL-6 levels were found to be decreased by both SOD3 and IκBα-SR mRNA treatments in multiplex immunoassay analysis (Supplementary Fig. S2) compared with scrambled controls (P < 0.05). Interestingly, TNF-α level in BAL was not significantly affected by E. coli pneumonia induction (Fig. 5C).
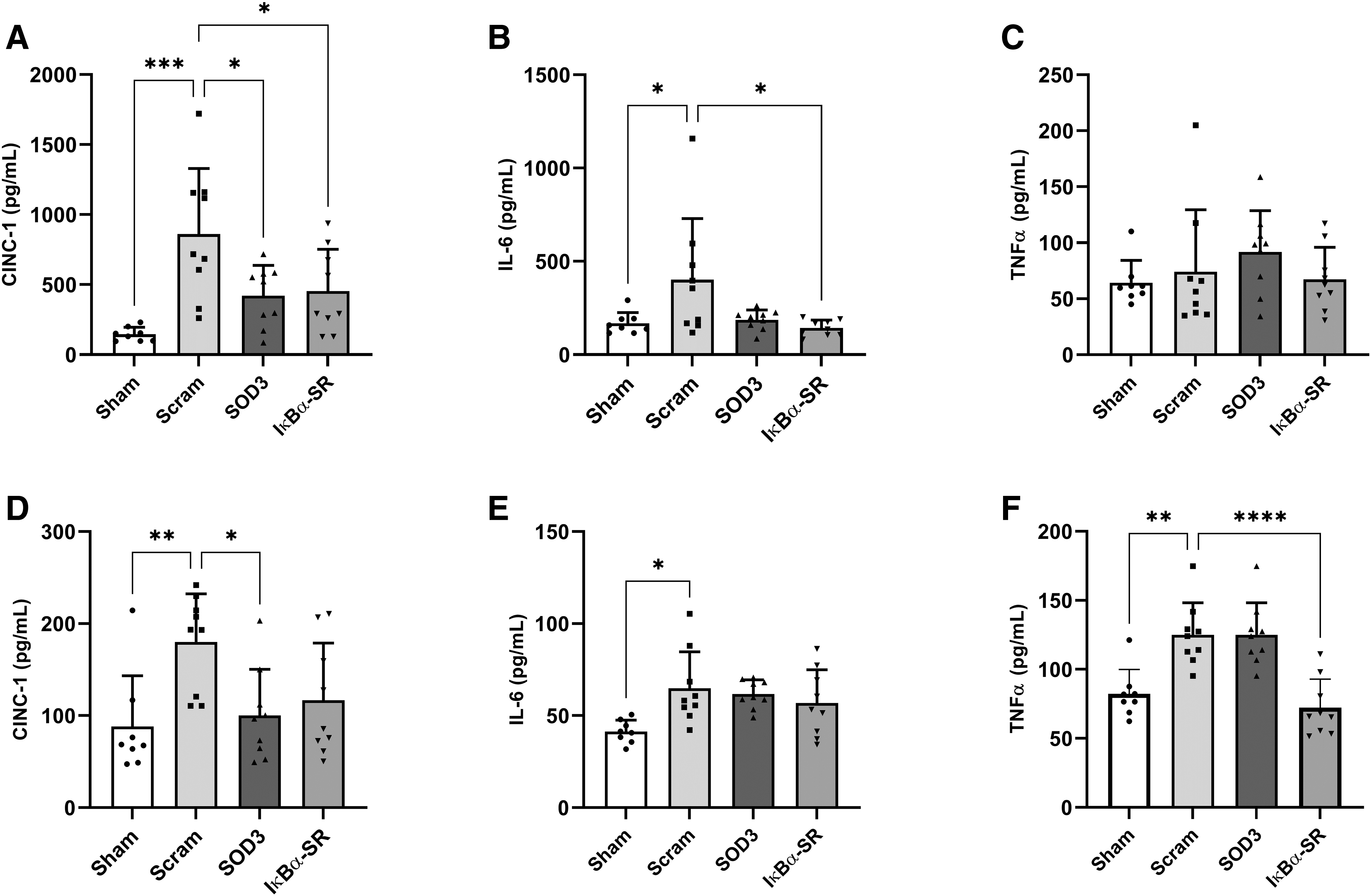
Effect of nebulized mRNA complexes on BAL and serum inflammatory cytokines. Concentrations were analyzed 48 h after mRNA complex administration to E. coli pneumonia rats. ELISA was used to determine levels of CINC-1, IL-6, and TNF-α in BAL
Discussion
ARDS has diverse etiologies, of which pathogen-induced pneumonia is among the most important and debilitating. While there are some differences between non-COVID and viral-induced ARDS, the syndrome develops in 42% of patients presenting with COVID-19 pneumonia [25] and so the requirement for effective therapies is more critical than ever. Anti-inflammatory and antioxidant therapeutic proteins have previously shown promise in preclinical ARDS models. However, recombinant proteins are limited as therapeutics, particularly due to size and stability and the requirement for post-translational modifications [26]. Gene therapy approaches have failed due to low or slow expression of transgene, so, here, we investigated whether a synthetic mRNA vector for extremely rapid production of protein, coupled with direct, large dose delivery to injured lungs by a VMN, could overcome these limitations.
VMNs have proved to be compatible with delivery of nanoparticles [27–29], biopolymers [30–32], and lipid-based carriers [33–35]. However, complexation with transfection agents is needed to substantially enhance mRNA stability and transfection efficiency [36]. Currently, there are limited reports of aerosolized mRNA to the lung airspace, but some studies have reported promising results [34,36–38]. Others have demonstrated aerosolized delivery of synthetic mRNA, which is a reliable means to transfect other tissues such as the female reproductive tract [39]. The implementation of a rapidly expressing mRNA delivery vector is likely to hold several advantages that may overcome the challenges associated with viral gene delivery, particularly delayed and low transgene expression [40]. To the best of our knowledge, this is the first instance of such technology being combined with VMN delivery to alleviate ARDS severity.
As recommended by Sedic et al. [41], we incorporated mRNA complexes loaded with scrambled mRNA as controls in this study. Complexes incorporating GFP mRNA expressed rapidly and efficiently in lung epithelial cells, where fluorescence was observed as early as 4 h post-transfection. Nebulized delivery of transfection complexes appeared to affect transfection efficiency only at 48 h postadministration, and this observation reflected what was reported by Johler et al. [37], as expression levels were similarly improved by increasing the concentration of cationic lipids complexed with nucleic acid. We report significant increases in IκBα-SR and SOD3 transgene expression in both pulmonary cell lines and tissues compared to controls, concomitant with the observation that nebulized mRNA-lipid complexes exhibited a droplet penetrations profile suitable for delivery to the alveoli.
IκBα is an important protein regulating the activity of NF-κB as it interacts with REL dimers to inhibit NF-κB/REL complexes, which are involved in inflammatory responses and maintains inactive NF-κB in the cytoplasm. IκBα-SR, a mutant form of IκBα, which cannot be phosphorylated by the IκB kinase complex proteins, prevents NF-κB translocation to the nucleus and subsequent transcription of target genes [16,42]. As expected, the observed increase in expression of total IκBα in both cultured pulmonary cells and rat lung samples were found to locate to intracellular space rather than in cell media or rat BAL.
Conversely, extracellular SOD3, which catalyzes the dismutation of superoxide anions to hydrogen peroxide at the cell membrane, was predominantly detected in media from in vitro samples, and lung BAL. As stated by Hu et al. [43], SOD3 is an antioxidant immobilized in the extracellular space, which may explain the observed increased expression of SOD3 in SAEC media and BAL compared to cell homogenates.
We have previously demonstrated that novel ToRNAdo nucleic-acid delivery system (Factor Bioscience) can be used to deliver GFP mRNA to the rat lung airspace resulting in efficient and transient expression of protein in lung epithelial cells [23]. However, as the current study commenced before ToRNAdo reagent availability, commercially available In Vivo-jetPEI was implemented, which also facilitated significant levels of protein expression in transfected tissues.
Once confirmed that synthetic mRNA could be delivered to lung epithelial monolayers, expressing transgenes efficiently, and that transfection efficacy was maintained postnebulization, we set about determining the therapeutic efficacy of candidate mRNAs. Although several of the mRNAs reduced cell viability in BEAS2Bs and primary SAECs following cell transfection, the 10%–20% reduction was in line with the mild cytotoxicity effects previously reported [37]. Interestingly, the scrambled control decreased viability even in the absence of a transgene product, thus suggesting that the transfection step is itself cytotoxic rather than subsequent overexpression of the protein product.
On the contrary, transfection with SOD3 mRNA was observed to have a protective effect on cell survival, perhaps reversing an earlier loss of viability. Interestingly, SOD3 did not induce a strong anti-inflammatory effect on BEAS-2B cells stimulated with LPS, while IκBα-SR mRNA attenuated secretion of inflammatory cytokines and outperformed the WT variant of this protein providing a basis to utilize the super repressor version for subsequent in vivo studies. Nonetheless, treatment with nebulized SOD3 mRNA did reduce superoxide-free radicals and preserve cell viability in the primary cell line.
Here, we induced an E. coli pneumonia in a rodent model, characterized by impaired arterial blood oxygenation and lung compliance, and accompanied by increased white cell influx and inflammatory cytokines in BAL. Nebulized administration of SOD3 mRNA complexes to rats undergoing pneumonia resulted in improvement in lung compliance as well as an improvement in the gradient between oxygen concentration in the alveoli and arterial system (AaDO2). It was previously found that an AAV vector encoding SOD administered to the lungs via intratracheal instillation improved systemic oxygenation and lung compliance in an animal model of LPS-induced lung injury [21]. Previous investigations of SOD3 in in vivo models have established that the protein acts as an anti-inflammatory mediator [43]. Other studies have documented SOD3 gene transfer into hind limb ischemia models to significantly reduce both leukocyte migration and cytokine expression as well as conferring anti-inflammatory effects on macrophages [44].
In this study, we report that transfection with SOD3 mRNA similarly reduced white cell infiltration and CINC-1 in BAL. The study by Hasset et al. [21] also reported a reduction in IL-6 following SOD3 treatment, however, we only found significant reduction of this cytokine in multiplex data. While the antibacterial effect of SOD3 complex delivery in the in vivo model were not entirely expected, a 2009 in vitro study did document that a peptide corresponding to the C-terminal region of extracellular SOD confers antimicrobial activity against E. coli [45]. Similarly, in a mouse model of E. coli-induced pneumonia, it was reported that SOD3 knockout impaired phagocytosis of bacteria by macrophages [46]. These findings are congruent with the reduction in BAL bacterial load following SOD3 mRNA treatment, which in turn, may be instrumental in the observed improvement in lung histology observed with this treatment.
Inflammation is a critical contributory factor to COVID-19 pathophysiology, where there is an excessive and uncontrolled release of proinflammatory cytokines, including IL-6 and IL-8, in the lung [47]. Both IL-6 [47,48] and IL-8 levels [49] are significantly higher in patients with COVID-19 who subsequently die compared to patients with better outcomes. As discussed by Cron et al., [50] blocking the cytokine storm by implementation of anti-inflammation therapy is critical for reducing COVID-19 pneumonia mortality. Adenoviral-mediated overexpression of the SR form of Iκβα administered to the lungs via aerosolization decreased the inflammatory mechanism of action, whereby NF-κB modulates a variety of proinflammatory cytokines in an animal model of LPS-induced acute lung injury [19].
Here, we observed broadly similar findings, with the Iκβα-SR variant mRNA complexes recapitulating the earlier viral vector-based findings, with improved lung function as represented by a decrease in CO2 levels and lower indices of inflammatory injury in the lung tissue, such as reduced wet weight of lung tissue, white cell infiltration and inflammatory cytokine concentration in BAL and serum. NLR was implemented here as marker of physiological stress and has been proposed as a critical predictor for assessment of disease severity in patients with COVID-19 and mortality in pneumonia patients [51]. The improvements we observed in several indices of lung injury may be reflective of the reduction in NLR across both treatments.
Conclusion
Semisynthetic mRNA analogs can be delivered by VMN to the septic rat lung producing proteins rapidly, efficiently, and with therapeutic effect. This provides a platform for anti-inflammatory therapy using candidate targets combined with easy pulmonary administration and also potential for investigation of other mRNA molecules to treat a variety of lung diseases. Beyond the implications that these data hold for ARDS therapeutics, they also provide a basis for future investigation in the developing area of mRNA vaccine technology combined with an aerosolized delivery system.
Footnotes
Author Disclosure Statement
Christopher B. Rohde and Matt Angel are employees of Factor Bioscience Ltd., Ronan MacLoughlin is an employee of Aerogen Ltd. No other author has any competing financial interest.
Funding Information
D.O.T. and J.G.L. are supported by Science Foundation Ireland (SFI) and the European Regional Development Fund (13/RC/2073). D.O.T. is supported by the Health Research Board Ireland (ILP-POR-2017-024). This project is partly funded by Factor Bioscience Ltd.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
