Abstract
Background:
Obesity affects ∼20% of children in the United States and reports of successful dietary treatment are lacking. This study aimed to determine the change in body weight in severely obese youth after carbohydrate-restricted dietary intervention.
Methods:
This single-center study of a carbohydrate-restricted diet (≤30 grams per day), with unlimited calories, fat, and protein for 3–4 months, examined two groups of severely obese youth of ages 5–18 years: Group A, retrospectively reviewed charts of severely obese youth referred to the Pediatric Obesity Clinic at Hoops Family Children's Hospital and the Ambulatory Division of Marshall Pediatrics, Marshall University School of Medicine, in Huntington, WV, between July 1, 2014 and June 30, 2017 (n = 130), and Group B, prospective participants, referred between July 1, 2018 and December 31, 2018, followed with laboratory studies pre- and postdietary intervention (n = 8).
Results:
In Group A, 310 participants began the diet, 130 (42%) returned after 3–4 months. Group B had 14 enrollees who began the diet, and 8 followed up at 3–4 months (57%). Girls compared with boys were more likely to complete the diet (P = 0.02). Participants <12 years age were almost twice as likely to complete the diet compared with those 12–18 years (64% vs. 36%, P < 0.01); however, the older group subjects who completed the diet had the same percentage of weight loss compared with those <12 years (6.9% vs. 6.9%). Group A had reductions in weight of 5.1 kg (P < 0.001), body mass index (BMI) 2.5 kg/m2 (P < 0.001), and percentage weight loss 6.9% (P < 0.001). Group B had reductions in weight 9.6 kg (P < 0.01), BMI 4 kg/m2 (P < 0.01), and percentage weight loss 9% (P < 0.01). In addition, participants had significant reductions of fasting serum insulin (P < 0.01), triglycerides (P < 0.01), and 20-hydroxyeicosatetraenoic acid (P < 0.01).
Conclusions:
This study demonstrated a carbohydrate-restricted diet, utilized short term, effectively reduced weight in a large percentage of severely obese youth, and can be replicated in a busy primary care office.
Introduction
The incidence of childhood obesity continues to increase in the United States. The prevalence of a body mass index (BMI; the weight in kilograms divided by the square of the height in meters) at the 95th percentile or higher among children 2 to 19 years reached 18.5% in 2015–2016. 1 A majority of children who are obese in early childhood remain obese into adolescence. 2 It has been shown that obese adolescents are likely to remain obese as adults. 3,4 Obesity is associated with comorbidities including cardiovascular and metabolic disorders in childhood, 5 and childhood obesity might lead to early death in adulthood. 6,7 It has been predicted if all obese children in the United States become obese adults, the individual average cost would be >$92,000 and the societal lifetime costs >$1.1 trillion. 8
The prevention of pediatric obesity by promoting healthful diet, activity, and environment should be a primary goal, as achieving effective long lasting results with lifestyle modification once obesity occurs is difficult. 9 In the treatment of obesity, lifestyle changes including dietary, physical activity, and behavioral modifications have been recommended with mixed success. 10 Pharmacotherapy and bariatric surgery have also been recommended in severe cases. 11,12
In these two analyses, we examined outcomes of a carbohydrate-restricted diet in a cohort of children with severe obesity. Based on previous studies from our institution and elsewhere, 13 –15 we hypothesized that implementation of this diet in children and adolescents with severe obesity would be associated with reduction in weight, BMI, and certain other adverse health markers.
Methods
This study was approved by the Marshall University Medical IRB, IRB# 1109427.
We performed a retrospective analysis of clinical data extracted from medical records from the Pediatric Obesity Clinic at Hoops Family Children's Hospital and the Ambulatory Division of the Department of Pediatrics of Marshall University Joan C Edwards School of Medicine of children referred from July 1, 2014 to June 30, 2017 (Group A). In addition, data were collected prospectively including laboratory analyses of a smaller second group of children from the obesity clinic referred from July 1, 2018 to December 31, 2018 (Group B).
Written informed consent for Group B participants was obtained before enrollment. Data from both groups were analyzed before and after the institution of a carbohydrate-restricted diet in children between 5 and 18 years of age. In both groups, children were excluded for < 95th BMI percentile for age/gender at their initial visit, for declining to participate in a carbohydrate-restricted diet, and failure to return for a 1 month follow-up visit. No weight reduction medications or psychotropic drugs were documented in any participant at initiation of the studies. Both studies were approved by the institutional review board of Marshall University before initiation or enrollment.
Subjects were referred to the Pediatric Obesity Clinic from primary care physicians throughout the tristate area of West Virginia, Ohio, and Kentucky. The clinic was staffed by a single ambulatory pediatrician. After the initial visit, participants were again seen at 1 month and at 3–4 months. The initial visit included a medical history and physical examination. If subjects were determined to have a BMI >95th percentile for their age and gender, a carbohydrate-restricted diet was discussed and a handout provided for the patient and family (Supplementary Appendix SA1).
All instructions were given to both the participants and their parent or guardian simultaneously. The diet included restriction of carbohydrates to ≤30 grams per day. No restrictions were placed on grams of fat or protein, and total calories were not discussed. The carbohydrate-restricted diet was designed to minimize intake of refined carbohydrate sources such as added sugars, high glycemic grains, and fructose.
We had four rules: (1) Limit total carbohydrates to <30 grams per day. We encouraged unlimited consumption of protein, fat, and green leafy vegetables. We limited fruit consumption to berries (strawberries, raspberries, blueberries, and blackberries). (2) Do not drink any sugar/carbohydrates. No milk, juice, and sugar-sweetened drinks (lemonade, sweet tea, and sugar-containing sports drinks). (3) Allow the children to eat when they are hungry but encourage them not to eat if they are not hungry, bored, or while watching television or playing video games. Examples of acceptable snacks include beef sticks, jerky, eggs, cheese, lunch meats, pepperoni, pork rinds, celery, nuts, broccoli, cauliflower, carrots, and peanut butter. (4) Exercise daily 30–45 min. A multivitamin was also encouraged to ensure all micronutrient requirements were met. Exercise was encouraged. Parents were instructed to pack a lunch for school.
The initial visit lasted 30 min and follow-up visits lasted 15 min. Weight, height, and blood pressure were measured at the initial visit and all subsequent visits. Weight (to nearest 0.1 kg) was measured using a digital scale and height (to nearest 0.1 cm) was determined using a wall-mounted stadiometer. Subjects and parents were instructed to keep a diary of daily food intake. BMI percentiles were calculated using Epi Info 7.2.0.1 (Centers for Disease Control and Prevention). BMIp95 was calculated by dividing the patient's BMI by the BMI at the 95th percentile for age and gender. 16 Obesity classes were defined as class 1 (100%–119% BMIp95), class 2 (120%–139%), and class 3 (≥140%). 17
In addition, subjects in Group B had fasting serum analyses of total cholesterol, triglyceride, insulin, glycated hemoglobin, blood urea nitrogen (BUN), creatinine, and 20-hydroxyeicosatetraenoic acid (20-HETE) at baseline and at 3–4 month follow-up visit. All laboratory tests were performed at a hospital-certified laboratory except the 20-HETE analysis that was performed blindly at the laboratory of one coauthor (M.L.S.).
All participating families in Group B received a $100 gift certificate to offset costs including travel associated with completing clinic and laboratory visits at baseline and follow-up (1 month and 3–4 months postinitiation of diet).
The aim of the Group A study was to retrospectively test for significant reductions in weight and BMI in obese children between initiation of a low-carbohydrate diet and at 3–4 months follow-up. The aim of the Group B study was to examine prospectively changes in weight, BMI, and multiple laboratory studies, including those associated with adverse health outcomes in obese children 3–4 months after initiation of a restricted carbohydrate diet.
Statistical methods: Column statistics [ranges, mean values, 95% confidence interval (CI)], Fishers exact test, and the Wilcoxon matched-pairs signed rank tests to determine statistical significance were performed using GraphPad Prism versions 8.0.0, and 8.3.0 for Windows, GraphPad Software, San Diego, CA. The individual scatter plot graph was created using GraphPad Prism version 8.3.0 for Windows, GraphPad Software, San Diego, CA, then overlaid into the final format using GNU Image Manipulation Program (GIMP) version 2.10.8.
Results
Participants
There were 449 subjects considered for participation in Group A. Twenty-one subjects were outside the age range 5–18 years at the initial visit. Thirteen were excluded for <95th BMI percentile for age/gender and 105 declined to participate in the diet. The remaining 310 children were qualified to be included. One hundred eighty children did not return for follow-up at 1 month, leaving 130 participants (42% of those commencing the diet) for inclusion.
In Group B, of the 20 potential subjects, an outcome evaluation sample of subjects who agreed to comply with the diet, have laboratory testing, and attend a follow-up visit, n = 8 (40% of potential subjects) were identified (Fig. 1). One hundred thirty-eight subjects (Group A—130, Group B—8) were followed for 3–4 months after initiating a carbohydrate-restricted diet. All subjects who agreed to participate in the diet and returned for 1 month and 3–4 months follow-up were included in these results.
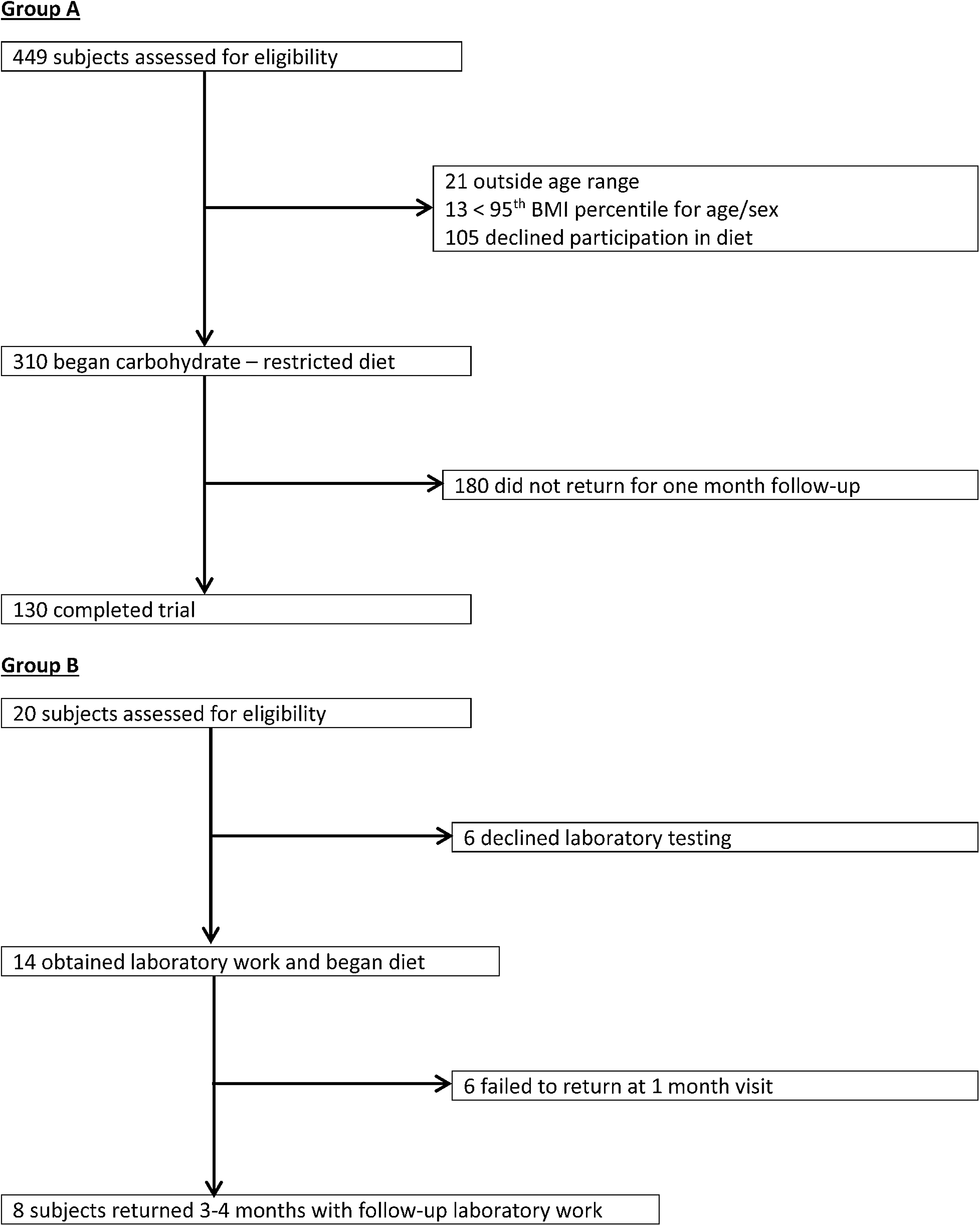
Screening and participation of Groups A and B on the carbohydrate-restricted diet.
When comparing the 192 subjects, from Groups A and B, who failed to return at 1 month with those 138 who completed the diet, females were more likely than males to complete the diet (P = 0.02) and those <12 years of age were more likely to return compared with those ≥12 years (P = 0.004). In further comparing those who completed the diet with those who failed to return, there were no differences in starting BMI, weight, or obesity class (Table 1). However, males in Group A when compared with females lost more weight (5.9 kg vs. 5.0 kg), had a greater percentage weight loss (8% vs. 6%), and had a greater reduction in BMI (2.65 vs. 2.35), although these differences were observational only, and not statistically significant.
Comparison of Subjects Beginning Carbohydrate-Restricted Diet Groups A and B (N = 330)
BMI, body mass index; ns, not significant.
Clinical outcomes
Of the 138 total subjects, 126 (91%) lost >1.0 kg (range 1.4–20.7), 4 (3%) gained >1.0 kg (range 2.7–10.3), and 8 (6%) lost or gained <1.0 kg. All 138 participants and their families verbally confirmed compliance with the diet. In Group A, 130 subjects lost a mean of 5.1 kg (P < 0.001, 95% CI: −5.195 to −4.385) and a mean reduction in BMI of 2.5 kg/m2 (P < 0.001, 95% CI: −2.762 to −2.178).
In Group B, eight subjects lost a mean of 9.6 kg (P < 0.01) and a mean reduction of BMI of 4.0 kg/m2 (P < 0.01) (Fig. 2). In Group B, additional results of baseline and follow-up serum levels for insulin, glycated hemoglobin, total cholesterol, high-density lipoprotein (HDL) cholesterol, BUN, creatinine, and 20-HETE were obtained. Changes in HDL (P = 0.16), total cholesterol (P = 0.15), BUN (P = 0.37), glycated hemoglobin (P = 0.10), and creatinine (P = 0.24) were not significant. Changes in fasting insulin, triglyceride, and 20-HETE were significant (Table 2).
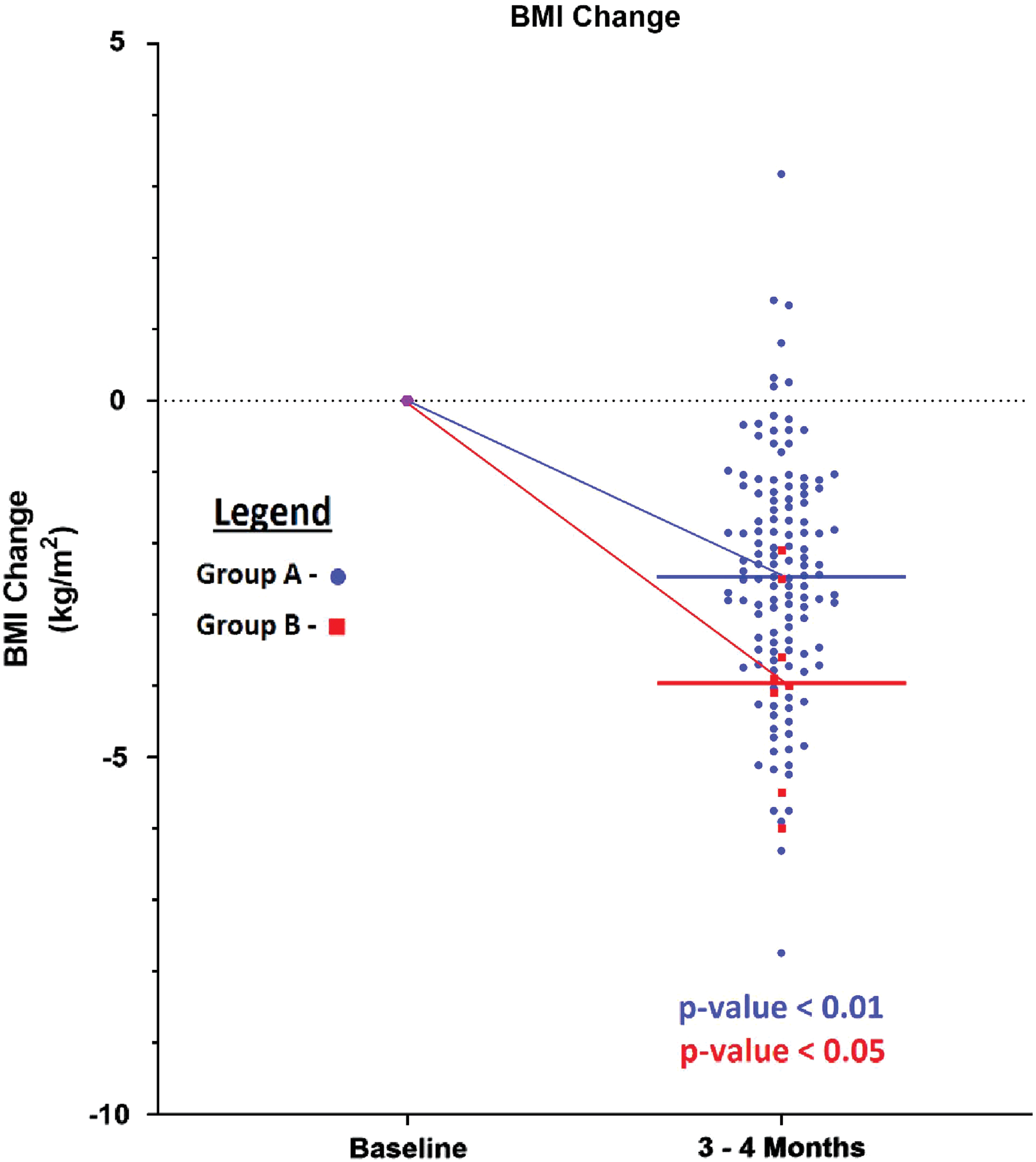
Change in BMI with carbohydrate-restricted diet for Groups A and B. Purple designates both groups at the prediet BMI change. Blue designates Group A while red designates Group B. The lines indicate the mean BMI change of each group. BMI, body mass index. Color images are available online.
Outcomes After Carbohydrate-Restricted Diet
X = no data collected for this group.
20-HETE, 20-hydroxyeicosatetraenoic acid; CI, confidence interval.
Discussion
The current United States Dietary Guidelines recommend that children of ages 1–18 years receive a diet with a content of % kcal for protein 10%–30%, carbohydrate 45%–65%, and fat 25%–35%. 18 Despite these guidelines being in place since 2006, the incidence of childhood obesity continues to rise. In this study, we present a successful diet for short-term weight loss by limiting carbohydrates without restrictions on total calories, protein, and fat.
A recent meta-analysis reported no meaningful difference in energy expenditure between low-carbohydrate and low-fat diets in adults. The studies included in that analysis, however, were short term (mostly <2 weeks), whereas the process of adapting to a low-carbohydrate high-fat diet seems to take at least 2 or 3 weeks. 19,20 Furthermore, a study in adults demonstrated meaningful weight loss with reduced calorie diets regardless of which macronutrients they emphasize. 21
RC Adkins was the first to postulate that significant fats, protein, and calories could be consumed without significant weight gain as long as insulin levels were low because of a restricted carbohydrate diet. 22 Each of our subjects in Group B had lower fasting insulin levels after 3 months of weight loss, which has been reported to be similar to a state of fasting with a high glucagon-to-insulin ratio that favors glycogenolysis, gluconeogenesis, lipolysis, and ketogenesis. 23 The proposed mechanism on the effectiveness of a ketogenic diet is maintenance of resting energy expenditure and lean muscle mass, high-fat mass loss, and increased fat oxidation rate compared with isocaloric high-carbohydrate diets. 19,24
When compared with a carbohydrate-restricted diet, reduced calorie low-fat diets have been unsuccessful in children, and we have speculated that noncompliance is a major causation factor. 25 Satiety is multifactorial. Protein is the most satiating macronutrient followed by carbohydrates and fat. 26 In this study, we allowed unlimited protein and fat and feel that the success was, in part, due to patient satiety secondary to increased protein intake.
We have described a diet wherein 138 severely obese children 5–18 years of age lost a mean of 5.5 kg over 3–4 months. Sixty-three percent of the participants (87/138) lost 5% or more of their initial body weight that in adults has been associated with health benefits including better control of existing diabetes and hypertension, reduced risk of acquiring these same comorbidities, and lower levels of risk factors for cardiometabolic disease. 25,27 –29 Fasting hyperinsulinemia and hypertriglyceridemia have been linked to metabolic syndrome in adults 29 and has been hypothesized as the earliest physiologic risk marker of cardiovascular and metabolic diseases in obese children. 30,31 All eight of our study subjects from Group B had significantly lower insulin and triglyceride levels after 3 months of weight loss.
20-HETE levels, a potent vasoactive eicosanoid and a key constituent of the microcirculation, were reduced in each subject in Group B after 3 months. Its effects on vascular function are multifaceted and include endothelial cell dysfunction and inflammation. 20-HETE and its biosynthetic enzymes have been linked to the development of hypertension, stroke, and myocardial infarction in adults. 32 To our knowledge, this is the first report to demonstrate an association between weight loss in children with reduction of 20-HETE levels.
We found that subjects <12 years age were more likely to complete the diet than those 12–18 years (P < 0.01). In elementary school-age children, parents serve to support the acquisition and maintenance of eating and exercise behaviors. 33 Although interventions with younger school-age children support strong parental involvement, the role of parents in adolescent weight control remains unclear. We, like others, speculate that our subjects <12 years age were more likely to complete the study because of greater parental involvement, whereas the majority of those in midadolescence lacked effective parental involvement and lacked concern regarding the desire to lose weight that generally increases at the time of school leaving resulting from increased autonomy, self-determination, and self-esteem. 34 –36
The strength of this study is that our approach, in contrast to many described pediatric weight loss programs, can be replicated in a busy primary care office. A multidisciplinary obesity team, including a social worker, dietician, exercise specialist, and behavioral health expert, was not utilized. Participants began the diet at their first visit, and time requirements of 30 min for the initial visit and 15 min for follow-up visits could be integrated into an ambulatory practice. Although daily exercise was encouraged, it was not mandated at the initial or follow-up visits.
Our study has limitations. Its duration was only 3–4 months. Continued weight reduction over a longer time period is uncertain. Although we could not detect any health complications, the long-term safety of a ketogenic diet in children has not been confirmed.
This is a small single-center trial. Only a limited percentage of those children who qualified for the diet actually participated. It could be inferred that those who began the diet and did not follow-up were unsuccessful in complying and did not lose weight. Although every subject in the prospective analysis lost weight; reduced BMI; and had lower insulin, triglyceride, and 20-HETE levels, our study population only included eight subjects, and the results should be considered in that context. Subject waist circumference was not measured that has been used as a proxy for visceral fat mass linked to cardiometabolic risk. 37 Finally, this is not a controlled trial and other factors in addition to the carbohydrate-restricted diet could have played a role in the weight reduction.
The American Academy of Pediatrics recommends that dietary and physical activity counseling be done for all children annually. 38 However, pediatric providers feel there are barriers interfering with obesity screening and treatment. In a survey of >200 pediatricians, 44% felt they had a lack of clinical knowledge, more than half felt there was a lack of time in their practice to manage childhood obesity, and 53% felt treatment was futile. 39
Concluding Summary
Obesity is among the greatest health challenges of the 21st century affecting ∼20% of all children and adolescents. We have presented a management strategy that effectively reduces weight in a large percentage of severely obese children and can be replicated in a busy primary practitioner's office.
Footnotes
Author Disclosure Statement
The authors declare no conflict of interest.
Funding Information
This study was partially funded through an NIH RO1 grant, # HL139793, issued to Dr. Michal Laniado Schwartzman at the New York Medical College, School of Medicine.
Supplementary Material
Supplementary Appendix SA1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
