Abstract
Background:
The aim of this analysis was to examine the effects of icosapent ethyl (eicosapentaenoic acid ethyl ester, IPE) on high-sensitivity C-reactive protein (hsCRP) and lipid parameters in patients with metabolic syndrome, with and without stable statin therapy.
Methods:
This post hoc exploratory analysis evaluated patients with metabolic syndrome treated with IPE 4 grams/day, IPE 2 grams/day, or placebo in phase 3, randomized, placebo-controlled studies entitled: MARINE [triglyceride (TG) levels ≥500 and ≤2000 mg/dL] and ANCHOR [TG levels ≥200 and <500 mg/dL, despite low-density lipoprotein cholesterol (LDL-C) control with stable statin therapy].
Results:
Compared with placebo in patients with metabolic syndrome in MARINE (n=204) and ANCHOR (n=645), at the approved dose of 4 grams/day, IPE significantly lowered hsCRP levels 40.0% (P=0.0007) in MARINE and 23.0% (P=0.0003) in ANCHOR. Compared with placebo in MARINE, which included patients with and without statin therapy, IPE 4 grams/day significantly reduced hsCRP levels 78.0% in statin-treated patients (P=0.0035, n=16). Compared with placebo in MARINE, IPE 4 grams/day significantly reduced TG levels (35.0%; P<0.0001), non–high-density lipoprotein cholesterol (non–HDL-C; 19.9%; P<0.0001), and apolipoprotein B levels (ApoB) (9.1%; P=0.0015) without raising LDL-C levels. Compared with placebo in ANCHOR, IPE 4 grams/day significantly reduced TG (21.7%; P<0.0001), non–HDL-C (13.5%; P<0.0001), ApoB (8.8%; P<0.0001), LDL-C (5.2%; P=0.0236), and HDL-C levels (4.0%; P=0.0053).
Conclusions:
Compared with placebo, IPE 4 grams/day significantly lowered hsCRP levels and improved lipids without raising LDL-C levels in patients with metabolic syndrome and high (≥200 and <500 mg/dL) or very high (≥500 and ≤2000 mg/dL) TG levels, with or without stable statin therapy.
Introduction
M
C-reactive protein (CRP) is a marker for inflammation. Its increase may be due to direct secretion from dysfunctional adipocytes, or, more commonly, due to an increase in hepatic secretion as the result of increased interleukin-6 (IL-6) from adiposopathic proinflammatory responses from adipose tissue. 2,10,11 An increase in CRP levels may also reflect vascular inflammation, 6 which is an integral component of the atherogenic process. 12 Thus, whether it reflects an increase in an adiposopathic proinflammatory response from an increase in body fat (which also accompanies an increase in major CVD risk factors 7 ) or due to direct responses from a proinflammatory and proatherogenic vasculature, an increase in CRP levels is a marker for increased CVD risk. 13,14
Omega-3 fatty acids are polyunsaturated fatty acids with a double bond at the third carbon from the methyl (omega) end. 15 Omega-3 fatty acids, including eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), are generally considered essential fatty acids, in that the human body has a limited ability to create an unsaturated bond at the omega-3 position. Thus, the majority of the body's EPA and DHA are derived from consumption, most commonly from marine fish. 16 Because EPA and DHA are lipophilic, these omega-3 fatty acids diffuse easily and are stored in body fat and lipid membranes. 15 Thus, consumed omega-3 fatty acids are reflected by measuring omega-3 fatty acids in plasma, cellular membranes, or adipose tissue. 17
EPA and DHA significantly reduce TG levels and may improve other CVD risk factors, 18 including markers of inflammation. When combined, EPA and DHA may reduce some inflammatory markers such as lipoprotein-associated phospholipase A2. 19 EPA combined with DHA has more inconsistent effects upon high-sensitivity CRP (hsCRP). 20 –22 Conversely, EPA alone may decrease not only lipoprotein-associated phospholipase A2 levels, but also oxidized LDL and hsCRP levels. 23 –25
Icosapent ethyl (eicosapentaenoic acid ethyl ester, IPE; Vascepa®, Amarin Pharma Inc., Bedminster, NJ) is a high-purity prescription form of EPA ethyl ester approved by the US Food and Drug Administration as an adjunct to diet to reduce TG levels in adult patients with severe (≥500 mg/dL) hypertriglyceridemia. 26 Two randomized placebo-controlled studies demonstrated the efficacy of IPE in improving lipid parameters in patients with elevated TG levels. 27,28 The pivotal MARINE study assessed the efficacy and safety of IPE in 229 patients with TG levels ≥500 mg/dL and ≤2000 mg/dL. 27 The ANCHOR study evaluated the efficacy and safety of IPE in 702 patients with TG levels ≥200 mg/dL and <500 mg/dL despite control of low-density lipoprotein cholesterol (LDL-C) levels with stable statin therapy. 28 Compared with placebo in both studies, IPE 4 grams/day significantly reduced TG levels and other lipid parameters without raising LDL-C levels.
Given that metabolic syndrome is often considered an inflammatory state that links an increase in body fat to metabolic diseases such as T2DM, 29 that an increase in inflammatory factors such as CRP may also be an important marker of CVD risk, 2,30,31 and that omega-3 fatty acids may have anti-inflammatory effects, this post hoc analysis of the MARINE and ANCHOR studies examined the effects of IPE on hsCRP and lipid parameters in patients with metabolic syndrome (a potentially proinflammatory state) to determine the effects of IPE in these patients with and without stable statin therapy.
Methods
Design and patients
This post hoc analysis evaluated patients with metabolic syndrome at baseline from the MARINE and ANCHOR studies. 27,28 Briefly, both were phase 3, placebo-controlled, randomized, double-blind, multicenter studies in eligible patients aged >18 years. Both studies included a 4- to 6-week initial stabilization period of diet and lifestyle changes and medication washout, a 2- to 3-week lipid-qualifying period, and a 12-week double-blind treatment period during which subjects received either IPE 4 grams/day, IPE 2 grams/day, or matched placebo. The MARINE study enrolled subjects with TG levels ≥500 mg/dL and ≤2000 mg/dL; patients could continue receiving a stable statin regimen with or without ezetimibe if the dose was stable for ≥4 weeks. The ANCHOR study was conducted in statin-treated subjects with high cardiovascular risk, well-controlled LDL-C (≥40 mg/dL and <115 mg/dL), and residually high TG levels (≥200 and <500 mg/dL). Patients in the ANCHOR trial were required to be on a stable statin therapy of atorvastatin, rosuvastatin, or simvastatin with or without ezetimibe for ≥4 weeks (at a stable dose such that the LDL-C qualifying criteria were met; switching between statins was prohibited). The studies were conducted in accordance with the principles of Good Clinical Practice and the Declaration of Helsinki. The protocol was approved by the appropriate institutional review boards, and patients underwent the informed consent process as documented by a written informed consent.
Patients were identified as having metabolic syndrome if they had three or more of the following at baseline: Body mass index (BMI) ≥30 kg/m2 (substituted for waist circumference); TG levels ≥150 mg/dL; high-density lipoprotein cholesterol (HDL-C) levels <40 mg/dL in men or <50 mg/dL in women; systolic blood pressure (SBP) ≥130 mmHg or diastolic blood pressure (DBP) ≥85 mmHg, or on antihypertensive medication; and fasting plasma glucose levels >100 mg/dL or on antidiabetes medication. 5,9
End points and measurements
The end points reported here include median percentage changes from baseline to week 12 in hsCRP, TG, LDL-C, non–HDL-C, apolipoprotein B (ApoB), and HDL-C levels compared with placebo. Measurements are presented herein in mg/dL, with the exception of hsCRP, which is reported in mg/L. To convert lipids from mg/dL to the International System (SI) units of millimoles/liter (mmol/L), multiply TG levels by 0.0113 and multiply cholesterol levels by 0.0259. To convert ApoB levels to the SI units of grams/L, multiply by 0.01.
Immunonephelometry (Dade Behring BNII Nephelometer, Siemens Healthcare Diagnostics, Deerfield, IL) was used to measure hsCRP; other end points were measured using methods previously described. 27 TGs and cholesterol were measured using enzymatic colorimetric tests (Olympus AU2700 or AU5400 Analyzer, Olympus, Center Valley, PA), and non–HDL-C was calculated by subtracting HDL-C from total cholesterol.
Statistical analysis
Efficacy analyses were performed on the intent-to-treat (ITT) population, defined as all randomized patients who had a baseline efficacy measurement, received at least one dose of study drug, and had at least one post-baseline efficacy measurement.
Median differences between the IPE and placebo groups were estimated with the Hodges–Lehmann method. P values are from the Wilcoxon rank-sum test. A P value ≤0.05 was the specified alpha for significance for exploratory end points in the MARINE and ANCHOR studies and was used in this post hoc analysis; no adjustments were made for multiple comparisons. Statistical analysis was performed with SAS software version 8.2 or higher (SAS Institute, Cary, NC).
Results
Baseline characteristics
In the MARINE study, 204 of the 229 enrolled patients (89.1%) had metabolic syndrome at baseline (IPE 4 grams/day, n=65; IPE 2 grams/day, n=69; placebo, n=70). Among these patients, 153 were not receiving statin therapy and 51 were receiving stable statin therapy. In the ANCHOR study, 645 of the 702 patients (91.9%) had metabolic syndrome at baseline (IPE 4 grams/day, n=211; IPE 2 grams/day, n=219; placebo, n=215). As noted before, per protocol, all ANCHOR participants received stable statin therapy prior to study entry.
IPE effects on hsCRP in patients with metabolic syndrome
Results from the MARINE study are shown in Table 1 and Fig. 1. Compared with placebo in patients with metabolic syndrome, IPE 4 grams/day significantly reduced hsCRP levels by 40.0% (P=0.0007). Compared with placebo, IPE 4 grams/day significantly reduced hsCRP levels by 27.6% (P=0.0385) in patients not receiving statin therapy and by 78.0% (P=0.0035) in patients receiving stable statin therapy. Compared with placebo, reductions in hsCRP with IPE 2 grams/day did not achieve statistical significance.
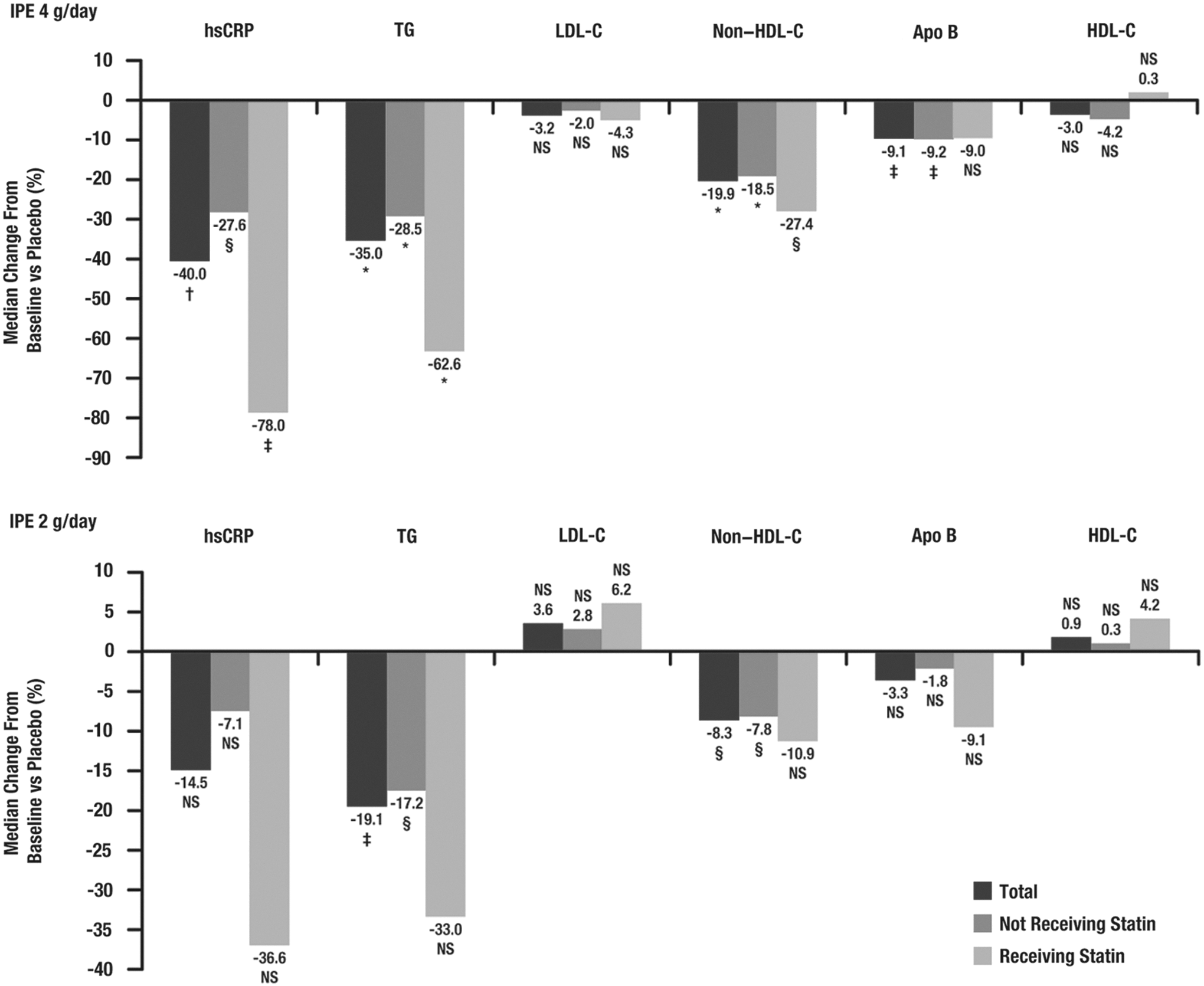
Median percentage change from baseline to week 12 versus placebo in high-sensitivity C-reactive protein (hsCRP) and lipid parameter levels in patients with metabolic syndrome from the MARINE study. (*) P ≤ 0.0001; (†) P<0.001; (‡) P<0.01; (§) P<0.05; NS, not significant (P>0.05). IPE, icosapent ethyl; TG, triglycerides; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; ApoB, apolipoprotein B.
Data are presented as median (interquartile range) for end point values. Median percent changes versus placebo are Hodges–Lehmann medians.
IPE, icosapent ethyl; hsCRP, high-sensitivity C-reactive protein; TG, triglycerides; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; ApoB, apolipoprotein B.
Results from the ANCHOR study are shown in Table 2 and Fig. 2. Compared with placebo in patients with metabolic syndrome, IPE 4 grams/day significantly reduced hsCRP levels from baseline by 23.0% (P=0.0003). Reductions in hsCRP with IPE 2 grams/day did not achieve statistical significance.
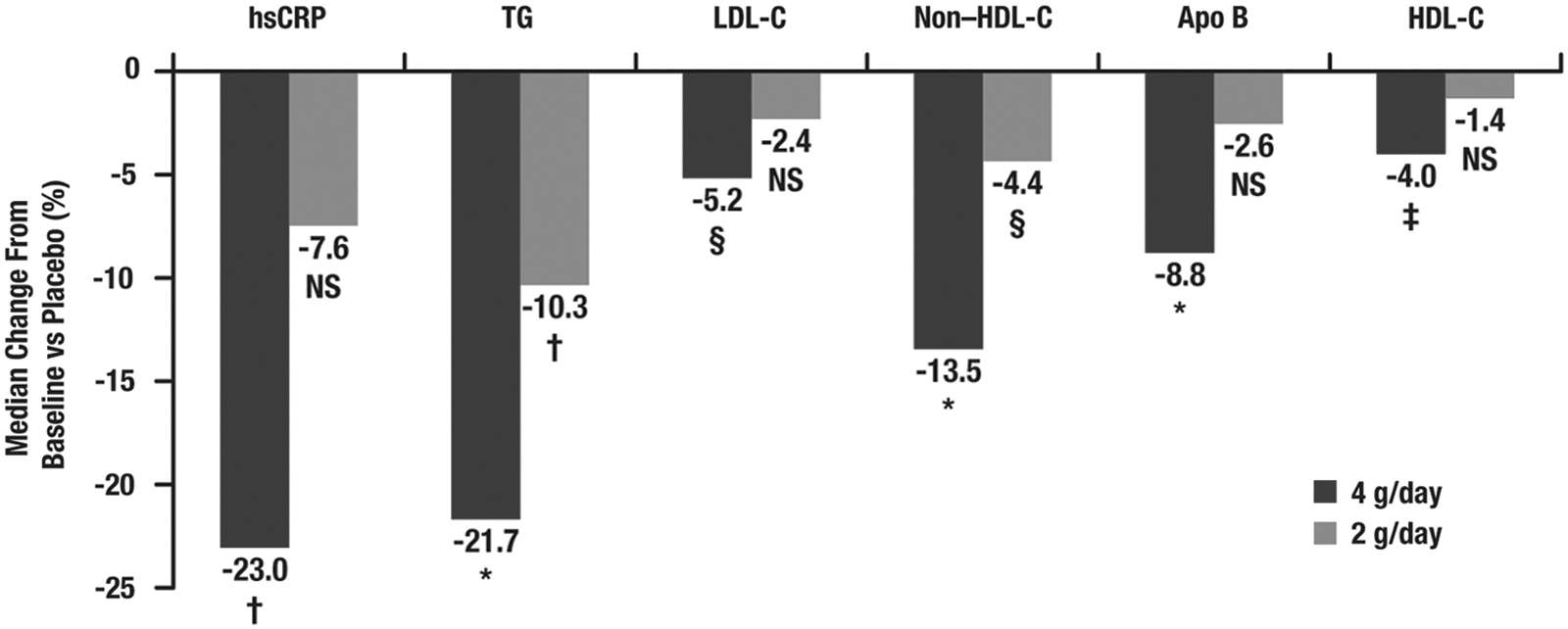
Median percentage change from baseline to week 12 versus placebo in high-sensitivity C-reactive protein (hsCRP) and lipid parameter levels in patients with metabolic syndrome from the ANCHOR study. (*) P<0.0001; (†) P<0.001; (‡) P<0.01; (§) P<0.05; NS, not significant (P>0.05). TG, triglycerides; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; ApoB, apolipoprotein B.
Data are presented as median (interquartile range) for end point values. Median percent changes versus placebo are Hodges–Lehmann medians.
IPE, icosapent ethyl; hsCRP, high-sensitivity C-reactive protein; TG, triglyceride; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; ApoB, apolipoprotein B.
IPE effects on lipid end points in patients with metabolic syndrome
Among those in the MARINE and ANCHOR studies with metabolic syndrome, IPE improved lipid levels. Compared with placebo in patients with metabolic syndrome from the MARINE study, IPE 4 grams/day significantly reduced TG (35.0%; P<0.0001), non–HDL-C (19.9%; P<0.0001), and ApoB levels (9.1%; P=0.0015), with no significant changes in LDL-C or HDL-C levels (Table 1 and Fig. 1). Compared with placebo in patients with metabolic syndrome from the ANCHOR study, IPE 4 grams/day reduced TG (21.7%; P<0.0001), LDL-C (5.2%; P=0.0236), non–HDL-C (13.5%; P<0.0001), ApoB (8.8%; P<0.0001), and HDL-C levels (4.0%; P=0.0053) (Table 2 and Fig. 2). Changes in these parameters with IPE 2 grams/day are also shown in Tables 1 and 2 and Figs. 1 and 2.
Discussion
This post hoc analysis from the MARINE and ANCHOR studies examined the effects of IPE on the inflammatory marker hsCRP, as well as the lipid parameters TGs, LDL-C, non–HDL-C, ApoB, and HDL-C in the subset of patients with metabolic syndrome. In patients with metabolic syndrome and high (≥200 mg/dL and <500 mg/dL) or very high (≥500 mg/dL and ≤2000 mg/dL) TG levels, IPE 4 grams/day improved lipid parameters compared with placebo and reduced hsCRP levels, a known marker for CVD risk, without increasing LDL-C levels. The results for hsCRP were similar to those observed in a previous study of markers of inflammation in all patients from the MARINE and ANCHOR studies. 23
Statin therapy has anti-inflammatory effects, as reflected by a reduction in elevated hsCRP levels. The Justification for the Use of Statins in Prevention: an Intervention Trial Evaluating Rosuvastatin (JUPITER) was a large CVD outcomes study of 17,802 “apparently healthy” subjects with LDL-C levels ≤130 mg/dL (3.4 mmol/L), but no history of CVD. 32 An entry criterion included elevated hsCRP levels ≥2.0 mg/L. Baseline median BMI was 28 kg/m2, and metabolic syndrome was present in 41% of study participants. 32 After a median follow-up of 1.9 years, rosuvastatin significantly reduced the incidence of major cardiovascular events. At 12 months, rosuvastatin reduced median hsCRP, LDL-C, and TG levels 37%, 50%, and 17%, respectively (all P<0.001). 32 Possible interpretations include a role for elevated hsCRP levels as not only a marker of vascular inflammation, but also as being directly involved in the pathogenesis of atherosclerosis and thrombosis. 2 Alternatively, the high mean BMI, high percentage of study participants with metabolic syndrome, and the elevated hsCRP levels may suggest that the disease state of adiposopathy was present at baseline. 2 Finally, a reduction in inflammatory markers (e.g., IL-6, hsCRP) with statins may, in part, be due to statin-induced reductions in adipose tissue inflammation. 33,34
Similarly, in a study of 48 dyslipidemic men with visceral obesity treated with: (1) atorvastatin alone; (2) EPA+DHA; (3) atorvastatin+EPA+DHA; or (4) placebo, atorvastatin, and atorvastatin+EPA+DHA significantly decreased hsCRP and IL-6 levels. However, EPA+DHA alone did not reduce hsCRP levels. 20 The potential for statin therapy to reduce hsCRP levels helped contribute to the study design for both MARINE and ANCHOR wherein statin-treated patients were required to be on stable statin doses prior to study entry. 27,28 Thus, the baseline hsCRP level was determined after stabilization of statin therapy in the MARINE and ANCHOR studies, which helped mitigate the possibility that the changes in hsCRP levels were statin-induced.
As noted before, data on the effect of EPA and/or DHA on plasma CRP or hsCRP levels are inconsistent. 20 –22,24,25,35 –46 A cross-sectional study in 124 healthy individuals suggested that plasma hsCRP concentrations had a negative correlation with plasma concentrations of total omega-3 fatty acids, EPA, and docosapentaenoic acid (DPA; C22:5). 45 In a placebo-controlled, double-blind crossover study of 14 exercise-trained men, 2.2 grams of EPA plus 2.2 grams of DHA daily for 6 weeks reduced hsCRP levels compared with placebo (P<0.05). 21 In a randomized, double-blind, placebo-controlled, three-period crossover study, compared with placebo, 8 weeks of 0.85 gram or 3.4 grams daily of an omega-3 fatty acid ethyl ester mixture (Lovaza®; each 1-gram capsule contained ∼0.465 gram EPA and 0.375 gram DHA) administered to 26 subjects with moderate hypertriglyceridemia (fasting TG levels 150–500 mg/dL) did not change hsCRP levels or levels of other inflammatory markers. 22 In a double-blind, randomized, placebo-controlled study of 34 hypertriglyceridemic men, DHA alone (3 grams/day for 90 days) produced no significant changes in hsCRP levels after 45 days compared with placebo, but reduced hsCRP levels by 15% (P<0.05) after 90 days of treatment. 46 Compared with placebo in a double-blind, placebo-controlled study of 59 patients with hypertension and T2DM, DHA or EPA alone (4 grams/day of either treatment for 6 weeks) demonstrated no change in hsCRP levels (P=0.676). 37
Regarding EPA alone, hsCRP levels were unaffected by the intervention with low/moderate-dose EPA combination treatment in a study of 51 Japanese patients with a history of atrial fibrillation who received antiarrhythmic drug treatment for 6 months followed by antiarrhythmic drug treatment plus EPA ethyl ester 1.8 grams/day for 6 months. 25 However, an earlier study of 44 Japanese patients with metabolic syndrome demonstrated that purified EPA ethyl ester (1.8 grams/day for 3 months as a supplement to dietary therapy) significantly reduced (P<0.05) CRP levels versus before treatment with EPA; statistical significance was not reached versus the control group of diet alone. 43 In a subsequent study in 92 patients with metabolic syndrome as part of a larger study examining the effects of EPA (1.8 grams/day) on arterial stiffness, CRP levels were significantly reduced from baseline levels of 1.57±0.2 μgrams/mL to 1.15±0.2 μgrams/mL after 3 months (P<0.01). 24 More recently, treatment with 1.8 grams/day EPA was found to significantly reduce hsCRP in a study of 139 Japanese chronic heart failure patients with dyslipidemia versus baseline and versus the no-EPA group (both P<0.001). 47
In this current analysis, all patients in ANCHOR were on stable statin therapy prior to study entry. However, the MARINE study included patients with and without stable statin therapy, allowing for relative comparisons of anti-inflammatory and lipid effects. Compared with placebo, among patients with metabolic syndrome in MARINE, IPE treatment produced numerically greater reductions in hsCRP levels among patients receiving stable statin therapy than in those not receiving statin therapy. However, the number of patients in the groups receiving statin therapy was small (n=16 for IPE 4 grams/day, n=17 for IPE 2 grams/day, and n=16 for placebo), and therefore those results should be interpreted with caution.
As this study evaluated a subset of patients with metabolic syndrome, it is relevant to note the potential effects of omega-3 fatty acids on the components of metabolic syndrome beyond anti-inflammatory effects. 48,49 When administered as pharmacotherapy, omega-3 fatty acids do not reduce (or increase) body weight. 50 Thus, it is unlikely that omega-3 pharmacotherapy would significantly alter WC. Although some reports suggest DBP may decrease with high consumption of C20–22 omega-3 fatty acids such as EPA, DPA, and DHA, 48,49 most controlled, randomized studies of omega-3 fatty acid pharmacotherapies have not produced significant reductions in blood pressure. Regarding glucose, in MARINE, mean changes from baseline in fasting plasma glucose were small and similar in each treatment group; in ANCHOR, there were no significant changes with IPE compared with placebo in fasting plasma glucose. 27,28
IPE did not increase HDL-C levels. Thus, the only diagnostic component of the metabolic syndrome that substantially changed with IPE was a reduction in TG levels. Other lipid effects often clinically relevant to patients with metabolic syndrome include a reduction in non–HDL-C levels. According to recommendations from some national lipid organizations, non–HDL-C should be considered the primary lipid treatment target, especially among patients with elevated TG levels. 51,52 In both MARINE and ANCHOR, IPE significantly reduced non–HDL-C levels. 27,28
Limitations of this report include that it was a post hoc analysis, it used BMI rather than WC as a criterion for metabolic syndrome, and elevated TG levels were a requirement for entry into the clinical trials evaluated. Thus, the metabolic syndrome component of elevated TG levels may be of higher prevalence compared with metabolic syndrome in a general population. It is also unclear whether therapies that lower hsCRP reduce cardiovascular events. Finally, neither the MARINE nor ANCHOR study assessed cardiovascular outcomes. However, the ongoing Reduction of Cardiovascular Events with EPA–Intervention Trial (REDUCE-IT; NCT01492361) is evaluating the effects of IPE 4 grams/day in combination with statin therapy on preventing the occurrence of a first major cardiovascular event in high-risk patients with mixed dyslipidemia. 53
Conclusions
Compared with placebo, IPE 4 grams/day improved lipid parameters and reduced hsCRP levels without raising LDL-C levels in statin-treated patients with metabolic syndrome and high (≥200 mg/dL and <500 mg/dL) or very high (≥500 mg/dL and ≤2000 mg/dL) TG levels. Taken together, the data suggest that IPE may offer beneficial effects in patients with metabolic syndrome, including those on statin therapy. The hypothesis as to whether these potentially beneficial effects will translate to cardiovascular benefit is being tested in the ongoing cardiovascular outcomes REDUCE-IT study.
Footnotes
Acknowledgments
Medical writing assistance was provided by Elizabeth Daro-Kaftan, PhD, of Peloton Advantage, Parsippany, NJ, and funded by Amarin Pharma Inc., Bedminster, NJ.
Author Disclosure Statements
H.E.B.'s research site has received research grants from Alere, Amarin Pharma Inc., Amgen, Ardea, AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Catabasis, Cymabay, Eisai, Elcelyx, Eli Lilly, Esperion, Forest, Gilead, Given, GlaxoSmithKline, Hanmi, Hisun, High Point Pharmaceuticals LLC, Hoffmann LaRoche, Home Access, Janssen, Merck, Metabolex, Nektar, Novartis, Novo Nordisk, Omthera, Orexigen, Pfizer Inc, Pronova, Regeneron, Sanofi, Takeda, TIMI, Transtech Pharma, Trygg, VIVUS, and WPU Pharmaceuticals. H.E.B. has served as a consultant and/or speaker to Amarin Pharma Inc., Amgen, AstraZeneca, Bristol-Meyers Squibb, Catabasis, Daiichi Sankyo, Eisai, Isis, Merck, Novartis, Omthera, VIVUS, and WPU Pharmaceuticals. C.M.B. has received research/grant support from Abbott Diagnostic, Amarin Pharma Inc., Amgen, Eli Lilly, Esperion, Genentech, GlaxoSmithKline, Merck, Novartis, Pfizer Inc, Regeneron, Roche, Roche Diagnostic, and Sanofi-Synthelabo; is a consultant for Abbott Diagnostics, Aegerion, Amarin Pharma Inc., Amgen, Arena, Cerenis, Esperion, Genentech, Genzyme, Kowa, Merck, Novartis, Pfizer Inc, Resverlogix, Roche, and Sanofi-Synthelabo; and has received honoraria from Abbott, Amarin Pharma Inc., AstraZeneca, Bristol-Myers Squibb, Cerenis, Esperion, Genentech, GlaxoSmithKline, Kowa, Merck, Novartis, Omthera, Regeneron, Resverlogix, Roche, and Sanofi-Synthelabo. W.G.S., R.T.D., S.P., and R.A.J. are employees and stock shareholders of Amarin Pharma Inc. S.P. is also affiliated with Chilton Medical Center. R.A.B. and P.N.S. are former employees of Amarin Pharma Inc.
Author contributions: H.E.B., C.M.B., R.A.B., and P.N.S. designed the study. All authors analyzed and interpreted the data and made meaningful contributions to the final manuscript. H.E.B. is guarantor for the article.
