Abstract
Background:
This study evaluated the effect of colesevelam hydrochloride on insulin sensitivity, potential binding to glucose, and chronic effect(s) on fasting and postprandial glucose and insulin in patients with type 2 diabetes mellitus.
Methods:
Patients meeting inclusion criteria were withdrawn from all antidiabetes agents for 2 weeks and randomized to colesevelam 3.75 grams/day (n = 17) or placebo (n = 18) for 8 weeks. Hyperinsulinemic–euglycemic clamp studies were performed at baseline (week −1) and weeks 2 and 8. A meal tolerance test was conducted at weeks −1, 0, 2, and 8. The meal tolerance test and study drug were coadministered at week 0 to assess the direct effect of colesevelam on glucose absorption.
Results:
Insulin sensitivity as measured by the insulin clamp method did not change, but the Matsuda Index, a measure of whole-body insulin sensitivity calculated from postmeal tolerance test glucose and insulin levels, increased significantly within the colesevelam group from baseline to week 8 with last observation carried forward (P = 0.020). The postprandial area under the curve for glucose decreased with colesevelam versus placebo at weeks 2 and 8 with last observation carried forward (P = 0.012 and P = 0.061, respectively); the area under the curve for insulin did not decrease in concert with the decrease in area under the curve for glucose at week 2 (P = 0.585). Colesevelam had no effect on postmeal tolerance test glucose levels at week 0.
Conclusions:
These results suggest that colesevelam has no effect on peripheral insulin sensitivity or glucose absorption, but may improve glucose control by improving whole-body insulin sensitivity, although not by an acute effect on glucose absorption.
Clinical Trial Identifier:
NCT00361153
Introduction
C
T2DM is a multifactorial disease that results from a progressive insulin secretory defect on a background of insulin resistance.5 In normoglycemic individuals, exposure to acute levels of glucose results in a biphasic pattern of insulin secretion, with both an early and late phase of insulin secretion.6 In individuals progressing to T2DM, the early phase of the insulin response is significantly reduced or abolished and the late phase is reduced or delayed. Some individuals maintain normal glucose tolerance by exhibiting increased peripheral sensitivity to insulin.7 However, in those predisposed to develop T2DM, increased glucose levels are not compensated for by an increase in insulin secretion or insulin sensitivity.6
This pilot study evaluated several possible mechanisms that may underlie the improved glycemic control observed with colesevelam in patients with T2DM. Insulin sensitivity was assessed by hyperinsulinemic–euglycemic clamp studies and by the Matsuda Index calculated from fasting and postprandial glucose and insulin data. The chronic effect of colesevelam was evaluated by meal tolerance tests (MTT) administered before and after 8 weeks of treatment. Finally, a MTT administered with the first exposure to the study drug investigated the possibility of an acute or direct effect of colesevelam on the absorption of glucose or its precursors.
Materials and Methods
Study design
This 8-week, randomized, double-blind, placebo-controlled, parallel-group study (ClinicalTrials.gov identifier: NCT00361153) was conducted at a single site between May and October 2006. The study protocol was conducted in compliance with Institutional Review Board regulations, Good Clinical Practice Guidelines, and the Declaration of Helsinki. All patients provided written informed consent before participation. Patients included males and females aged 18–75 years with diagnosed T2DM (for >3 months), with an HbA1c of 7.0%–10.0% (inclusive), being treated with diet or antidiabetes agents (excluding thiazolidinediones), and with a body mass index (BMI) of 25–45 kg/m2. Patients were excluded who, at screening, had triglycerides >500 mg/dL (5.65 mmol L−1) or LDL-C <60 mg/dL (1.55 mmol L−1); T1DM; a history of diabetic ketoacidosis, allergy, or toxic reaction to colesevelam, dysphagia, swallowing disorders, intestinal motility disorders, or hyperthyroidism; lipid- or blood pressure-lowering therapy that was not stable for ≥3 months; use of any investigational drug within 30 days prior to randomization; or treatment with colesevelam, cholestyramine, or colestipol within 3 months. Exclusionary concomitant medications included all antidiabetes agents (after washout), oral corticosteroids, thyroid hormone/levothyroxine, cholestyramine, or colestipol. Patients were withdrawn from the study if their fasting plasma glucose (FPG) level was >300 mg/dL (16.65 mmol L−1) on three consecutive glucometer readings.
Analyses
Patients who met the inclusion criteria at screening were withdrawn from all antidiabetes medications until the end of the study. Those on insulin were washed out for 3 days, whereas those on oral antidiabetes agents were washed out for 2 weeks. At week 0, patients were randomly assigned 1:1 to colesevelam 3.75 grams/day or matching placebo for 8 weeks of double-blind treatment (Fig. 1). Patients took study medication as a single dose [3.75 grams (6 tablets: 625 mg/tablet)] with the evening meal, or as a divided dose [1.875 grams (3 tablets)] with the noon and evening meals; the dosing schedule was chosen by the patient and was to be maintained throughout the study.
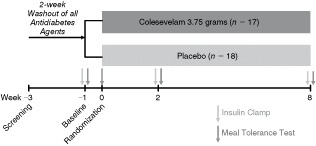
Study design.
The primary efficacy parameter was change from baseline in glucose disposal rate during the final 30 min of the insulin clamp (M-value) at week 8. Secondary efficacy parameters included: change from baseline in M-value at week 2; change from baseline in area under the curve for glucose (AUCg) and insulin (AUCi) at weeks 2 and 8; acute and chronic effects of colesevelam on postprandial glucose; change from baseline in HbA1c at weeks 0 and 8; change from baseline in FPG and fasting insulin at weeks 2, 4, 6, and 8; and change from baseline in fructosamine at weeks 4 and 8. Baseline was defined as week −1 unless otherwise specified.
Safety assessments included changes in vital signs, clinical laboratory tests, and electrocardiograms, as well as evaluation of the incidence and severity of adverse events (AEs). A complete medical and surgical history, physical examination, clinical laboratory testing, and 12-lead electrocardiograms were performed at screening. Physical examination, clinical laboratory testing, and 12-lead electrocardiograms were also performed at week 8 or upon early withdrawal. Compliance with the treatment regimen was evaluated by counting unused tablets at weeks 4 and 8, or at the early termination visit.
Hyperinsulinemic–euglycemic clamp
To evaluate insulin sensitivity, patients underwent a hyperinsulinemic–euglycemic clamp at baseline (week −1), week 2, and week 8. This method (modified from DeFronzo et al.8) allowed for the highly reproducible determination of the whole-body glucose disposal rate during a steady state of hyperinsulinemia.8 , 9 The clamp procedure was performed as follows: After priming, exogenous insulin was continuously infused at a nominal rate of 60 mU/min*m2 for 180 min (sufficient to suppress hepatic glucose production in most individuals). At the same time, exogenous glucose was infused at a rate sufficient to maintain the plasma glucose level at approximately 100 mg/dL (5.55 mmol L−1). The mean glucose infusion rate was calculated for the last 30 min of the clamp (150–180 min). Under steady-state conditions, the peripheral M-value = mean glucose infusion rate. If plasma glucose was not stable during the last 30 min, a correction was applied for appearance or disappearance of glucose. The M-value was expressed as mg glucose/min*kg body weight.
Matsuda Index
Using postprandial glucose and insulin levels derived following the MTT at week −1 and week 8, a post hoc calculation of the Matsuda Index, a measure of whole-body insulin sensitivity, was performed as follows: 10,000/√ (FPG concentration × fasting plasma insulin concentration) × (mean MTT glucose concentration × mean MTT insulin concentration).10 It should be noted that Matsuda Index values were originally calculated using glucose and insulin concentrations obtained following administration of an oral glucose tolerance test.
Meal tolerance tests
To evaluate the effects of colesevelam on postprandial glucose and insulin levels, after a 10- to 12-h fast, patients were administered a standard MTT consisting of two 8-oz cans of Ensure® Fiber (42.0 grams carbohydrates, 8.8 grams protein, and 6.1 grams fat plus 24 essential minerals and vitamins) at baseline (week −1), week 0 (after randomization), week 2, and week 8. To evaluate the potential acute binding of colesevelam to glucose, all six colesevelam or placebo tablets were coadministered with the MTT at week 0 (day 1). At weeks 2 and 8, the MTT was administered in the morning and the study drug was taken later.
Statistical methods
Baseline variables, prerandomization assessments, and demographic characteristics were summarized for all randomized patients. The summary set included all randomized patients who received treatment, had a baseline and ≥1 scheduled postrandomization efficacy assessment, and was used to summarize all efficacy variables. The safety set included all patients who had any safety assessment on or after week 0, and was used to summarize AEs, clinical laboratory tests, electrocardiograms, vital signs, and physical examination findings.
An analysis of covariance (ANCOVA) model with treatment administered as a fixed effect and baseline M-value as a covariate was applied to test the primary efficacy parameter. The treatment difference between the colesevelam and placebo group was evaluated by least-squares means and standard errors, two-tailed 95% confidence intervals (CI), and a two-sided P value. An analysis of variance (ANOVA) model was used if the assumption of parallelism did not hold. Last observation carried forward (LOCF) analysis was used to evaluate missing week-8 efficacy values. Following the MTT, AUCg and AUCi were calculated using the trapezoid rule. Changes in these variables were evaluated by nonparametric tests, because AUC values do not follow a normal distribution. Values for AUCg and AUCi are shown as median or median change from baseline.
A total of 34 patients with T2DM were to be assigned randomly to either colesevelam or placebo in a 1:1 ratio. This sample size had 80% power at a two-sided significance level of 5% to detect a placebo-corrected mean difference of 4.5 mg/min*kg in change from baseline in M-value, assuming a standard deviation (SD) of 4.24 and a dropout rate of 10%.
Results
Patient disposition and baseline characteristics
A total of 66 patients were screened and 35 were randomized: 17 received colesevelam and 18 received placebo (Fig. 2). Approximately 94% of patients in both the colesevelam group (n = 16) and placebo group (n = 17) completed the study. One patient (6%) from each treatment group discontinued; 1 was lost to follow up (colesevelam group) and 1 met the protocol-specified discontinuation criteria of FPG >300 mg/dL on three occasions (placebo group). At baseline, there were no significant differences between the treatment groups based on age, gender, or race (Table 1). Mean height, weight, and BMI were also similar between the colesevelam and placebo groups at baseline. At randomization (week 0; day 1 predose), HbA1c was 8.2% ± 0.9 and 8.7% ± 0.9 (P = 0.097) in the colesevelam and placebo groups, respectively. Compliance with study medication was 94% in the colesevelam group and 93% in the placebo group.
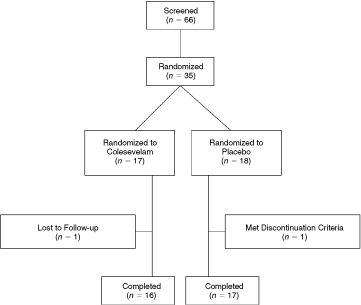
Patient disposition.
B
aWeek 0 (day 1 predose).
b n = 17.
cTo convert to mmol L−1, multiply by 0.0555.
Abbreviations: HbA1c, glycosylated hemoglobin; SD, standard deviation.
M-value (peripheral insulin sensitivity)
The mean M-value did not change significantly from baseline in the colesevelam group compared with placebo, resulting in a mean treatment difference of −0.09 ± 0.4 mg/min*kg at week 8 LOCF (P = 0.832) (Fig. 3A). There was a nonsignificant increase in the mean M-value in the colesevelam group compared with placebo at week 2 (mean treatment difference: +0.72 ± 0.5 mg/min*kg; P = 0.138).
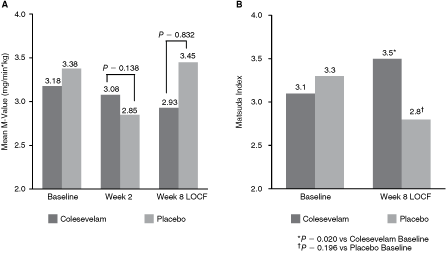
Effect of colesevelam on mean M-values (
Matsuda Index (whole-body insulin sensitivity)
Whole-body insulin sensitivity was assessed using the Matsuda Index calculated at baseline (week −1) and week 8 LOCF. There was a significant improvement from baseline of approximately 13% in the mean Matsuda Index within the colesevelam group at week 8 LOCF (P = 0.020) (Fig. 3B).
Glycemic variables
Mean FPG significantly decreased with colesevelam at weeks 4 and 6 compared with placebo (P < 0.05) at these timepoints, whereas FPG increased significantly with placebo at week 6 compared with week 0 (P < 0.05) (Fig. 4A). Mean fasting plasma insulin levels significantly increased at week 4 from week 0 (P < 0.05), but were unchanged at week 8 LOCF within the colesevelam group (Fig. 4B). However, the change from week 0 in mean fasting plasma insulin levels was significantly different (P < 0.05) within the placebo group at week 8 LOCF (Fig. 4B). The change in mean HbA1c (%) was +0.24 (8.21 vs. 8.45; P = 0.422) with colesevelam and +0.55 (8.68 vs. 9.26; P = 0.031) with placebo, from week 0 to week 8 LOCF. Similarly, the change in mean fructosamine levels (umol/L) was +2.75 (321.28 vs. 324.03; P = 0.874) with colesevelam and +18.11 (354.37 vs. 372.48; P = 0.042) with placebo, from week 0 to week 8 LOCF.
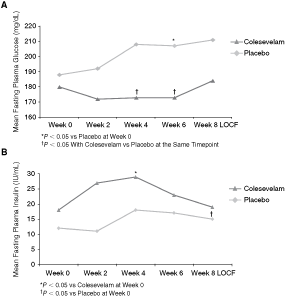
Change in mean fasting plasma glucose (FPG) (
Meal tolerance tests (acute effects)
Analysis of the potential acute binding of colesevelam to glucose upon coadministration of MTT and study drug at week 0 (day 1) revealed no significant effect of colesevelam on median AUCg values compared with baseline (week −1) or with placebo (Fig. 5A). In addition, mean plasma glucose levels were unchanged from baseline (week −1) at all timepoints before and during (30, 60, 90, 120, 240 min) the MTT in both the colesevelam and placebo groups (data not shown). In contrast, analysis of the acute effects of colesevelam on plasma insulin levels at week 0 (day 1) revealed a significant reduction from baseline (week −1) in median AUCi values (P = 0.006) within the colesevelam group (Fig. 5B). The reduction from baseline in median AUCi values with colesevelam at week 0 (day 1) was also significant when compared with placebo (treatment difference: −1702.5; P = 0.040) (Fig. 5B). The change from baseline in the incremental AUCi/AUCg ratio was significant with colesevelam compared with placebo only at week 0 (day 1) (P = 0.002; Fig. 6).
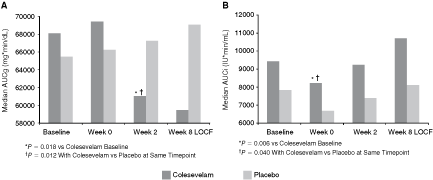
Change in median plasma glucose (AUCg) (
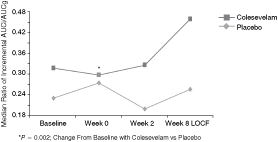
Change in incremental AUCi/AUCg ratio over time after meal replacement tests (summary set). Week 0 (day 1) = coadministration of colesevelam with meal tolerance test. Abbreviations: AUCg, area under the curve for glucose; AUCi, area under the curve for insulin; LOCF, last observation carried forward.
Meal tolerance tests (chronic effects on glucose and insulin)
Mean FPG levels immediately before the MTT were significantly reduced from baseline (week −1) at week 2 (P = 0.011) and week 8 LOCF (P = 0.04), in patients receiving colesevelam compared with placebo (data not shown). Significant reductions in postMTT mean plasma glucose levels, compared with the corresponding baseline (week −1), were seen at week 2 (30, 60, 120, 240 min; P < 0.05 for all) and week 8 LOCF (120 min; P < 0.05) within the colesevelam group (Fig. 7A). In addition, significant treatment differences with colesevelam were observed, when colesevelam was compared with placebo at each timepoint, at week 2 (60, 120, 240 min; P < 0.05 for all) and week 8 LOCF (120, 240 min; P < 0.05 for all) (Fig. 7A). Similar reductions from baseline were not seen with placebo (Fig. 7B).
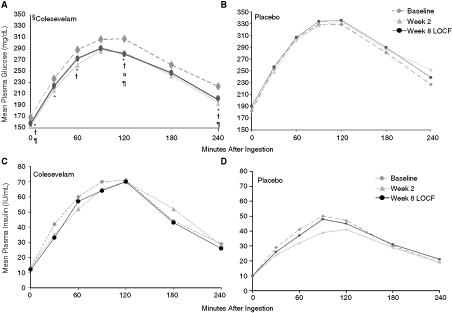
Effect of colesevelam and placebo on postprandial glucose (
After the MTT, total median AUCg levels were reduced from baseline (week −1) within the colesevelam group at both week 2 and week 8 LOCF, although the reduction was only significant at week 2 (P = 0.018) (Fig. 5A). The reduction in median AUCg at week 2 with colesevelam was significant when compared with placebo (treatment difference: −8565 mg*min/dL; P = 0.012) (Fig. 5A). At week 8, the reduction in median AUCg compared with placebo was not significant (treatment difference: −7462 mg*min/dL; P = 0.061).
Mean fasting insulin levels, obtained immediately prior to the MTT, did not change significantly from baseline (week −1) between treatment groups at week 2 or week 8 LOCF (data not shown). No significant treatment difference was observed in postMTT mean plasma insulin levels with colesevelam compared with placebo at most timepoints at week 2 or week 8 LOCF (Fig. 7C). In addition, following the MTT, median AUCi measured at week 2 and week 8 LOCF generally increased over time in both groups, with higher AUCi levels in the colesevelam group compared with placebo (Fig. 5B). The change from baseline in the incremental AUCi/AUCg ratio was not significant with colesevelam compared with either placebo or baseline (week −1) at week 2 and week 8 LOCF (Fig. 6).
Safety
Overall, colesevelam was well tolerated. A total of 14 patients (40%) reported ≥1 AE: 8 (47%) in the colesevelam group and 6 (33%) in the placebo group (Table 2). The majority of AEs reported in both treatment groups were mild or moderate in severity. One patient (6%) in each group reported a severe AE [abdominal pain (colesevelam) and diarrhea (placebo)]. There were no deaths, serious AEs, or other significant AEs. No patient discontinued treatment due to an AE.
S
Each patient is counted once.
Abbreviations: AEs, adverse events; SAEs, serious AEs.
Discussion
In this study, peripheral insulin sensitivity (M-value) did not change in either treatment group, and the change from baseline did not differ between the colesevelam and placebo groups at week 8 LOCF as measured by the insulin clamp. However, there was an effect of colesevelam on whole-body insulin sensitivity reflected by the significant increase (P = 0.02) in Matsuda Index values postMTT at week 8 within the colesevelam group compared with baseline. While there was no effect of colesevelam on peripheral insulin sensitivity, as measured by the insulin clamp, the decreased AUCg levels postMTT with colesevelam are consistent with improvement in whole-body insulin sensitivity, as indicated by the increase in the Matsuda Index at week 8.
Treatment with colesevelam resulted in a significant reduction in median AUCg levels compared with placebo at week 2 and a trend toward reduction in AUCg at week 8. Unlike AUCg levels, AUCi levels did not change significantly over time with colesevelam compared with placebo.
Coadministration of colesevelam with the MTT at week 0 (day 1), the first day of exposure to study medication, had no effect on postprandial glucose levels, indicating that colesevelam does not exert an acute binding effect(s) on glucose, its precursors, or complex carbohydrates. However, mean postprandial insulin levels decreased at some timepoints following coadministration of colesevelam with the MTT.
The effects of colesevelam on glycemic variables were similar in direction and magnitude to those observed in other studies,4 although the small sample size of this study prevented statistical significance. Overall, colesevelam was well tolerated, and the incidence of AEs was similar to placebo. Constipation and dyspepsia were the most common drug-related AEs associated with colesevelam, and there was a single report of a severe AE with colesevelam.
In patients with T2DM, intensive glycemic control and management of dyslipidemia can substantially reduce the risk of cardiovascular disease.5 Oral antidiabetes agents such as metformin and thiazolidinediones regulate plasma glucose levels by improving insulin sensitivity in patients with T2DM. However, unlike colesevelam, these drugs have only modest effects on hypercholesterolemia. Thiazolidinediones can increase LDL-C levels whereas BAS can increase triglyceride levels in patients with T2DM.11
The mechanism(s) by which BAS, including colesevelam, exert their effect on glycemic control are not well understood. Possible mechanisms for the glucose-lowering effect of colesevelam currently under investigation include effects on insulin sensitivity, insulin secretion, changes in bile acid composition,12 incretin effects,13 and effects related to intestinal/splanchnic processes.
Other potential mechanisms by which colesevelam may influence glucose levels include effects of BAS on the farnesoid X receptor (FXR/bile acid receptor), liver X receptor (LXR), and TGR5/glucagon-like peptide-1 (GLP-1) that have pivotal roles in bile acid and/or triglyceride homeostasis and are involved in the regulation of glucose metabolism.14 –16 By binding to bile acids and modifying the bile acid pool size and composition, BAS may alter expression of genes involved in glucose regulation by modulating the interaction between bile acids and FXR.14 , 17 Administration of a BAS has also been reported to result in a significant increase in the 2-h GLP-1 levels in patients with T2DM, with significant reduction in AUCg and no significant change in insulin levels, inferring enhanced insulin sensitivity and improved insulin secretion following administration of a BAS.16 A recent 16-week study with colesevelam evaluated plasma glucose, plasma insulin, total GLP-1, and total glucose-dependent insulinotropic polypeptide (GIP) levels during the fasting state and following a MTT. Compared with placebo, colesevelam significantly decreased HbA1c and FPG levels and significantly increased total fasting GLP-1, postprandial total GLP-1 AUC, and postprandial GIP AUC levels.18 Separately, exogenous GLP-1 decreased postprandial glucose by increasing the splanchnic uptake of glucose; the role of bile acid metabolism in this process is unknown.19 , 20
BAS such as colesevelam alter the bile acid pool size and composition in the gut lumen by binding bile acids, which reduces the digestion and absorption of free fatty acids (FFA) in the proximal gut and increases absorption in the distal gut, allowing for increased concentrations of FFA in the ileum to interact with the G-protein–coupled receptor 40 (GPR40).21 –24 GPR40 is expressed in β-cells of the pancreas and in the endocrine cells of the gastrointestinal tract, including those that secrete the incretin hormones GLP-1 and GIP.25 , 26 Thus, GPR40 plays an important role in the release of both GLP-1 and GIP.25
Bile acid metabolism is altered in individuals with T2DM, which may contribute to the abnormal glucose, fat, and energy metabolism characteristic of T2DM. Brufau et al.27 reported that the composition of the bile acid pool is different and more hydrophobic in patients with T2DM compared with individuals without T2DM.27 Furthermore, treatment with colesevelam resulted in a more hydrophilic bile acid pool.27 Selective modulation of bile acid pool size and composition by BAS may contribute to the improvement in both glycemic and lipid control.27 , 28 Further studies evaluating potential mechanism(s) of action are needed to fully elucidate the reduction in glycemia associated with colesevelam.
Limitations
There are several limitations to the interpretation of the data in this study. Patients with T1DM were excluded using patient history and not by an analysis of C-peptide levels. The insulin infusion rate used for the insulin clamp, 60 mU/min*m2, may not have sufficiently suppressed hepatic glucose production in all patients. The duration of the study was limited, as was the duration of washout of prior antidiabetes medications, but randomization may have neutralized the latter. Finally, the sample size was small, although there were no statistically significant differences in patient characteristics between treatment groups.
Conclusion
In this pilot study, colesevelam may have, via its dual glucose-/lipid-lowering effects and possible actions via FXR and TGR5/GLP-1 modulation, improved the overall management of T2DM by addressing two primary pathophysiological defects in T2DM—insulin resistance and defective insulin secretion. Further research is required to more fully elucidate potential mechanism(s) of action of colesevelam in T2DM.
Footnotes
Acknowledgments
Editorial assistance was provided by Luana Atherly, Ph.D., and Karen Stauffer, Ph.D. This study was sponsored by Daiichi Sankyo, Inc.
Author Disclosure Statement
Y.-L.L., S.L.A., M.R.J., S.N., and S.M. are all employed by Daiichi Sankyo, Inc. J.X. is an independent contractor for Daiichi Sankyo, Inc.
