Abstract
Objective:
The main objective was to assess the correlation between antibiotic use and carbapenem-resistant Klebsiella pneumoniae (CRKP) and carbapenem-resistant Escherichia coli (CREC) induction by antibiotics.
Materials and Methods:
A retrospective cohort study was conducted from January 2017 to December 2020. This study included patients with K. pneumoniae and E. coli. Kaplan–Meier analysis and Cox proportional hazard model were used to estimate the hazard of carbapenem-resistant Enterobacterales (CRE), whereas restricted cubic spline regression was used to visualize the hazard of CRE by antibiotics at different doses.
Results:
Two thousand fifty-six K. pneumoniae patients and 3,243 E. coli patients were included. After Cox proportional hazard model analysis, carbapenems or 1st-cephalospoins or penicillin monotherapy, male and ICU admission were associated with CRKP. CREC was associated with quinolone monotherapy. Time-to-event analysis indicated that carbapenem, β-lactamase inhibitor mixtures, and quinolones were associated with higher 30-day CRKP hazards than other antibiotics (χ2 = 33.670, p < 0.001). Further restricted cubic spline regression analysis found that the hazard of CRKP induction decreased with the increased dose of β-lactamase inhibitor mixtures, but there was no significant change in the hazard ratio of CRKP induction with the increased dose of quinolones. Moreover, there was an obvious characteristic of “parabolic curve” for the hazard of CREC induction due to β-lactamase inhibitor mixtures, and the hazard value gradually increased with the dose, reached the maximum at 24 g, and finally gradually decreased from 26 g.
Conclusions:
Rational use of antibiotics should be implemented and antimicrobial stewardship policies should be adjusted according to the characteristics of each hospital.
Background
Over recent years, antimicrobial resistance (AMR) among Enterobacterales has become a serious public health problem in the world. The AMR greatly limits therapeutic options, resulting in higher morbidity, mortality and huge economic burden. 1
Carbapenem is a β-lactam antibiotic with a wide antibacterial spectrum and strong antibacterial activity, and it is often used as a last resort in the treatment of multidrug-resistant Gram-negative Enterobacterales infection.2,3 However, the incidence of carbapenem-resistant Enterobacterales (CRE) has constantly increased over past years, posing a challenge for clinical antimicrobial chemotherapy and hospital infection control.4–6 In China, Klebsiella pneumoniae and Escherichia coli were the most common Enterobacterales according to CHINET surveillance in 2018, which could cause nosocomial infections, such as pneumonia, urinary tract infection, and catheter-related bloodstream infection. 7 Previous studies had reported that CRE was associated with high mortality.8–10
Rational use of antibiotics can effectively treat bacterial infections and reduce the burden to patients. However, unreasonable use of antibiotics will increase the selective pressure of antimicrobials commonly used, which is one of the important factors leading to AMR, and some evidence showed that increased use of antibiotics could lead to the emergence of AMR.11,12 Numerous studies reported the significant relationship between antimicrobial consumption and CRE prevalence, but whether antibiotics at different doses contribute to CRE was not yet certain.
It was necessary to understand the risk factors of CRE and evaluate the correlation between antibiotic dose and CRE, to provide evidence for developing a plan to reduce selective pressure imposed by antimicrobial agents in the future. Accordingly, we conducted this study to assess the correlation between antibiotic use and isolation of carbapenem-resistant E. coli (CREC) and carbapenem-resistant K. pneumoniae (CRKP) in a large cohort.
Materials and Methods
Design and subjects
A retrospective analysis was performed in Sichuan Academy of Medical Sciences and Sichuan People's Hospital, a 3,270-bed teaching hospital in Western China for the period January 2017 to December 2020. This study population comprised all patients with K. pneumoniae or E. coli cultured from any of the clinical specimens. This study was approved by the Ethics Committee of Sichuan Academy of Medical Sciences and Sichuan People's Hospital. The Review Board exempted the requirement for informed consent because of the retrospective study and the absence of a negative impact on the patients.
Microbiology
Drug sensitivity test and result interpretation referred to the guidelines of the Clinical and Laboratory Standards Institute (CLSI) 2017. 13 Agar dilution method using Mueller Hinton agar was used for the general bacterial susceptibility test. Disk-diffusion synergy test recommended by CLSI was used to screen and detect E. coli and K. pneumoniae. The automated instrument used VITEK 2 (BioMerieux, France) compact automatic microbial identification and drug sensitivity analysis system. The disk-diffusion synergy test was the Kirby-Bauer method, and the source of the drug sensitive paper was purchased from BBL and Oxoid.
Quality control was performed according to CLSI 2017 requirements during each test and every day. 13 The quality control strains (purchased from the Clinical Laboratory Center of the Ministry of Health, PRC) included K. pneumoniae ATCC 700603 and E. coli ATCC 35218.
Outcomes and definition
CRKP isolation and CREC isolation were the primary outcome of this study. Carbapenem resistance is defined as an minimum inhibitory concentration (MIC) of ≥2 μg/mL for ertapenem or an MIC of ≥4 μg/mL for meropenem or imipenem or dolipenem, according to the guidelines of CLSI 2017. 13
Confounding factors
Potential confounders were selected based on previous literature,14–16 and they included age, gender, tumor, liver failure, kidney failure, heart failure, respiratory failure, diabetes, hypertension, chronic obstructive pulmonary disease, hemodialysis, venous catheterization, mechanical ventilation, urinary catheterization, tracheotomy, surgery, ICU admission now or in the past, and community infections.
Data collection
All the data were collected from the Data Management Platform (DMP) of the hospital, which is composed of Hospital Information System (HIS), Laboratory Information System (LIS), Nursing Information System (NIS), and Nosocomial Infection Surveillance System (NISS). Each system transmitted data in real time on the platform (Supplementary Fig. S1). To accomplish the study objectives, the data management was carried out by a full-time health statistics professional of HAI surveillance in the drug-resistant bacteria management module of NISS.
Statistical analysis
Statistical analysis of the data was performed by using STATA version 20.0 (StataCorp., College Station, TX, USA) and SPSS version 23.0 (IBM Corporation, Armonk, NY, USA). Data were summarized by using the mean and standard deviation for normally distributed variables. Categorical variables were expressed in absolute numbers and percentages. We used both univariate and multivariate methods to analyze the data. Binary outcomes were tested by using the χ 2 test, and continuous data were compared by using the T-test.
Kaplan–Meier analysis and Cox proportional hazard model were used to estimate the hazard of CRE in the time-to-event analysis. For the time-to-event analysis, the endpoints in this study were 30 days from antimicrobial therapy until CRE detected from any isolate. Restricted cubic spline regression was used to visualize the hazard of CRE by antibiotics at different doses.
To assess the hazard of CRE induction by antibiotics, the data were gradually analyzed in two parts. First, in the COX proportional hazard model, we evaluated the CRE induction effect of various commonly used antibiotics on the basis of excluding other confounding factors. Second, to analyze the dose–response relationship between the antibiotics and the CRE induction effect, we fit the curve of the specific antibiotics with the strongest CRE induction effect on the basis of excluding other confounding factors. p Values below 0.05 were considered significant.
Data availability statement
All data generated and analyzed during this study are included in this article.
Results
Patient inclusion
From January 1, 2017 to December 31, 2020, a toal of 6,339 inpatients with Enterobacterales cultured from clinical specimens, including 2,056 patients with K. pneumoniae cultured and 3,243 patients with E. coli cultured, as Figure 1 shows.
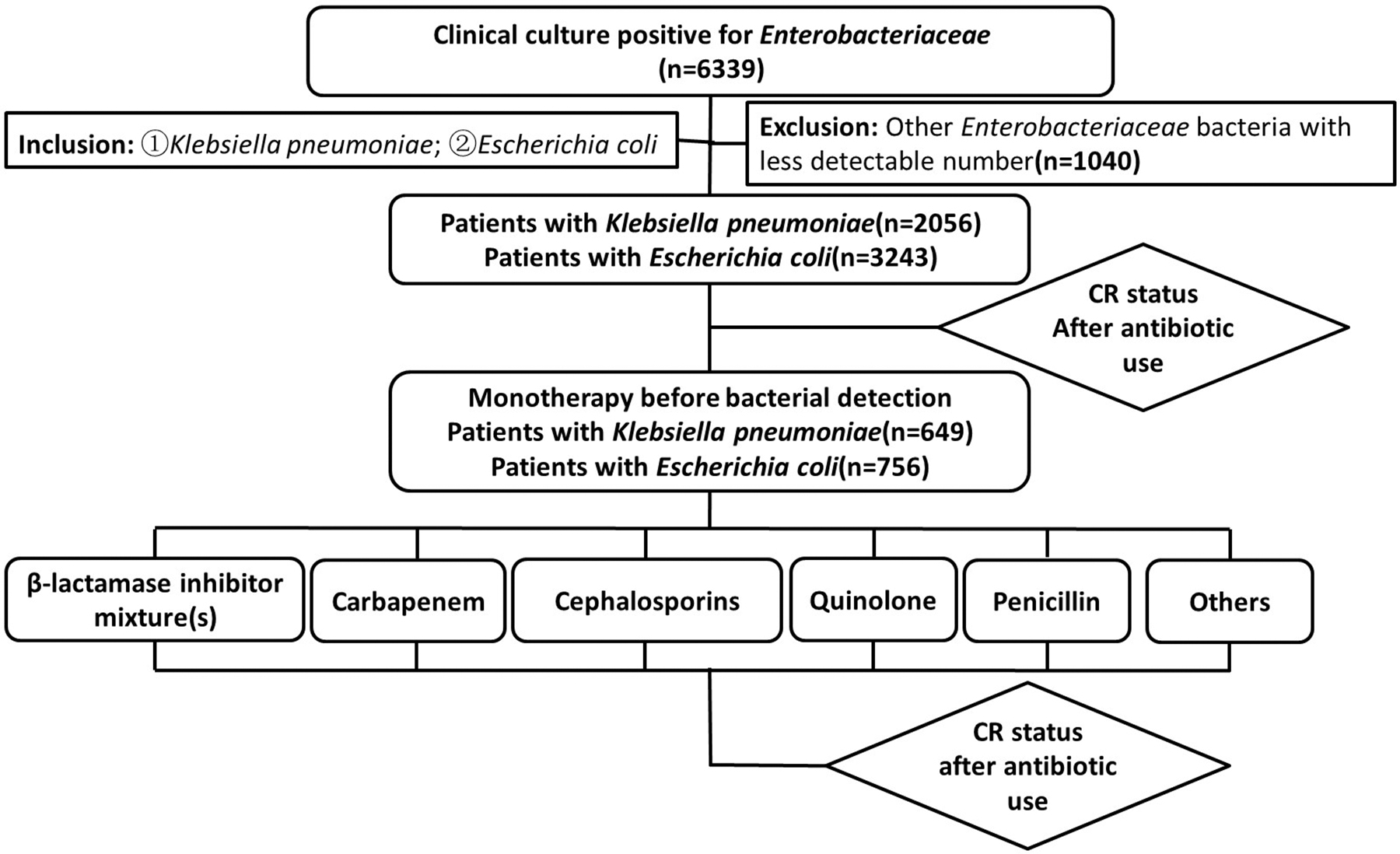
Flow chart of patients' inclusion.
Clinical characteristics
Among 2,056 patients with K. pneumoniae and 3,243 patients with E. coli, there were 308 (14.98%) CRKP and 183 (5.64%) CREC, respectively, whereas the remaining were cases with carbapenem-sensitive Enterobacterale.
Univariate analysis (Table 1) shows the following factors to be associated with a higher risk of CRKP occurrence: male, urinary cannula, vascular cannula, mechanical ventilation, tracheotomy, blood transfusion or use of blood products, respiratory failure, ICU admission now or in the past, and community infections. Moreover, the following factors are shown to be associated with a higher risk of CREC occurrence: male, urinary cannula, vascular cannula, mechanical ventilation, blood transfusion or use of blood products, respiratory failure, heart failure, ICU admission now or in the past, and community infections.
Baseline Characteristics
Data are no. (%) of objects. aStudent T-test.
Chi-square test with continuous correction.
CSKP, carbapenem-sensitive Klebsiella pneumoniae; CRKP, carbapenem-resistant K. pneumoniae; CS-E. coli, carbapenem-sensitive Escherichia coli; CS-E. coli, carbapenem-resistant E. coli; SD, standard deviation.
Antimicrobial therapy before CRE were detected
The antimicrobial therapy before the detection of bacteria in the patient's clinical specimens is shown in Table 2. There is a significant difference between the occurrence of CSPK and CRKP in antimicrobial therapy among monotherapy, combination therapy, and unused (χ 2 = 90.667, p < 0.001), and the proportion of combination antibacterial therapy in patients with CRKP is relatively high (181/308, 58.77%).
Basic Situation of Antimicrobial Therapy
Data are no. (%) of objects.
Moreover, there is a significant difference between these two groups in monotherapy among antibiotics used (χ 2 = 38.851, p < 0.001). Among CSKP and CRKP patients, the proportion of patients using β-lactamase inhibitor mixtures is relatively high: (252/575, 43.83%) and (50/74, 67.57%), respectively.
There is a significant difference in antibacterial therapy between the CSEC group and the CREC group (χ 2 = 33.021, p < 0.001), and the proportion of combination antibacterial therapy in patients with CREC is relatively high (75/183, 40.98%). Moreover, there is also a significant difference in monotherapy between these two groups (χ 2 = 26.824, p < 0.001), Among CSEC and CREC patients, the proportion of patients using β-lactamase inhibitor mixtures is relatively high: (190/718,26.46%) and (21/38, 55.26%), respectively.
Thirty-day hazard of CRE induction by antibiotics
The results of the predicted time-to-event probability diagram indicate that the Cox proportional hazard hypothesis is valid (Supplementary Fig. S2). Omnibus Tests of model coefficients show that the Cox proportional hazards model for CRKP patients has obtained a statistically significant result (χ 2 = 40.003, p < 0.001), but the model for CREC patients has not (χ 2 = 15.555, p = 0.158).
In the COX proportional hazard model (Table 3), CRKP patients show association with carbapenem monotherapy (hazard ratio [HR], 3.252; 95% confidence interval [CI], 1.159–9.124), 1st-cephalospoins monotherapy (HR, 0.219; 95% CI, 0.085–0.564), penicillin monotherapy (HR, 0.191; 95% CI, 0.046–0.787), male (HR, 1.886; 95% CI, 1.059–3.360), and ICU admission now or in the past (HR, 1.803; 95% CI, 1.117–2.910). The CREC patients show only an association with quinolone monotherapy.
Equation Parameters of Cox Proportional Hazard Model
/There are no data in the model fitting results or the variable is not included in the model.
CI, confidence interval; HR, hazard ratio.
Time-to-event analysis (Fig. 2) indicates that the administration of carbapenem, β-lactamase inhibitor mixtures, and quinolones are associated with higher 30-day CRKP isolation hazards than other antibiotics. Log-rank test results show that the differences are statistically significant (χ 2 = 33.670, p < 0.001) (Fig. 2A). Moreover, the 30-day CREC isolation hazards are significantly higher in patients with administration of β-lactamase inhibitor mixtures and 3rd-cephalosporins than other antibiotics (χ 2 = 17.634, p = 0.003) (Fig. 2B).
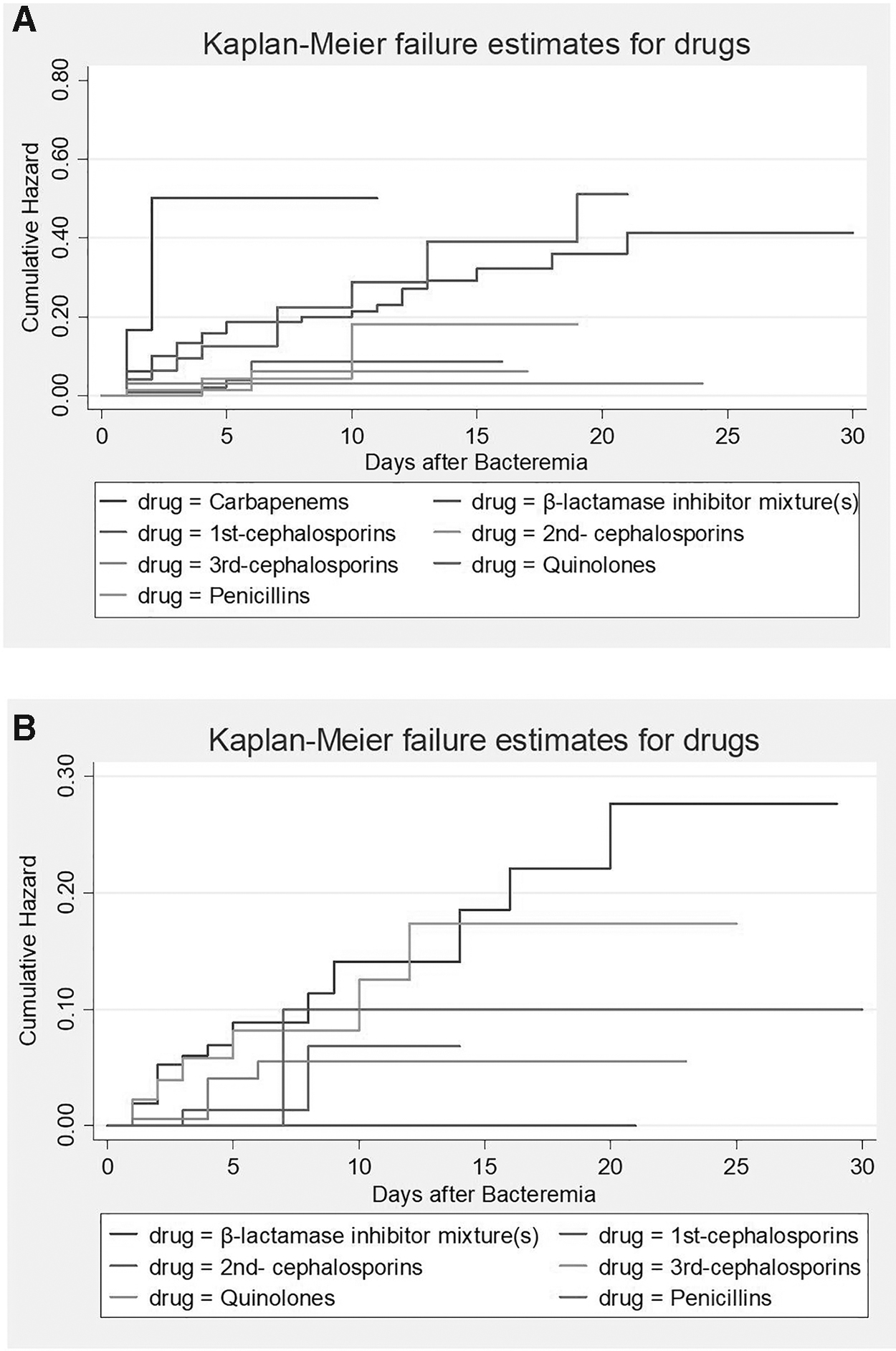
Results of Kaplan–Meier analysis:
Dose–response relationship between the hazard of CRE induction and antibiotics
In restricted cubic spline regression analysis (Figs. 3 and 4), the fitted curve shows that the hazard of CRKP induction decreases with the increased dose of β-lactamase inhibitor mixtures, but there is no significant change in the HR of CRKP induction with the increased dose of quinolones (Fig. 3).
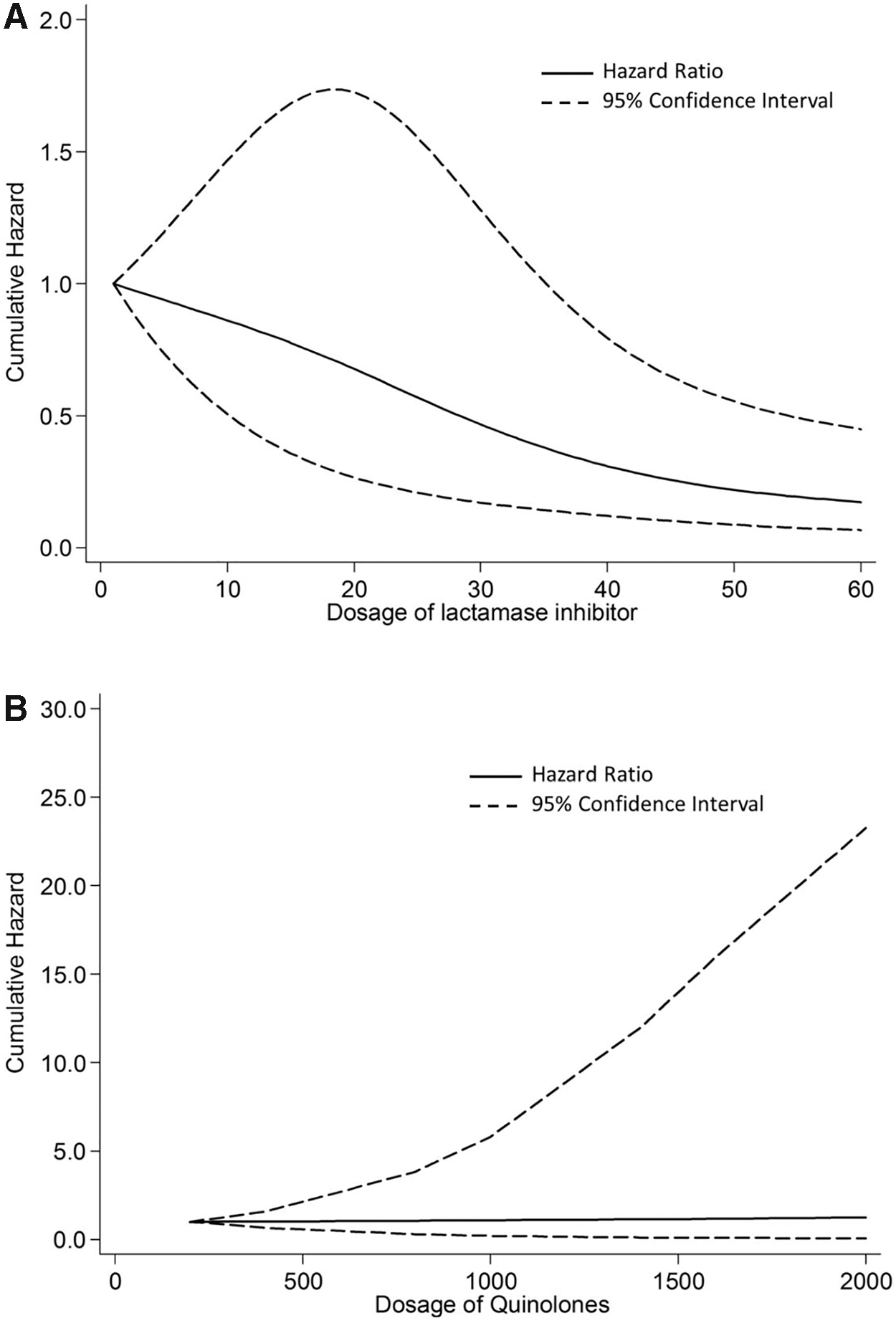
Results of restricted cubic spline regression on Klebsiella pneumoniae:
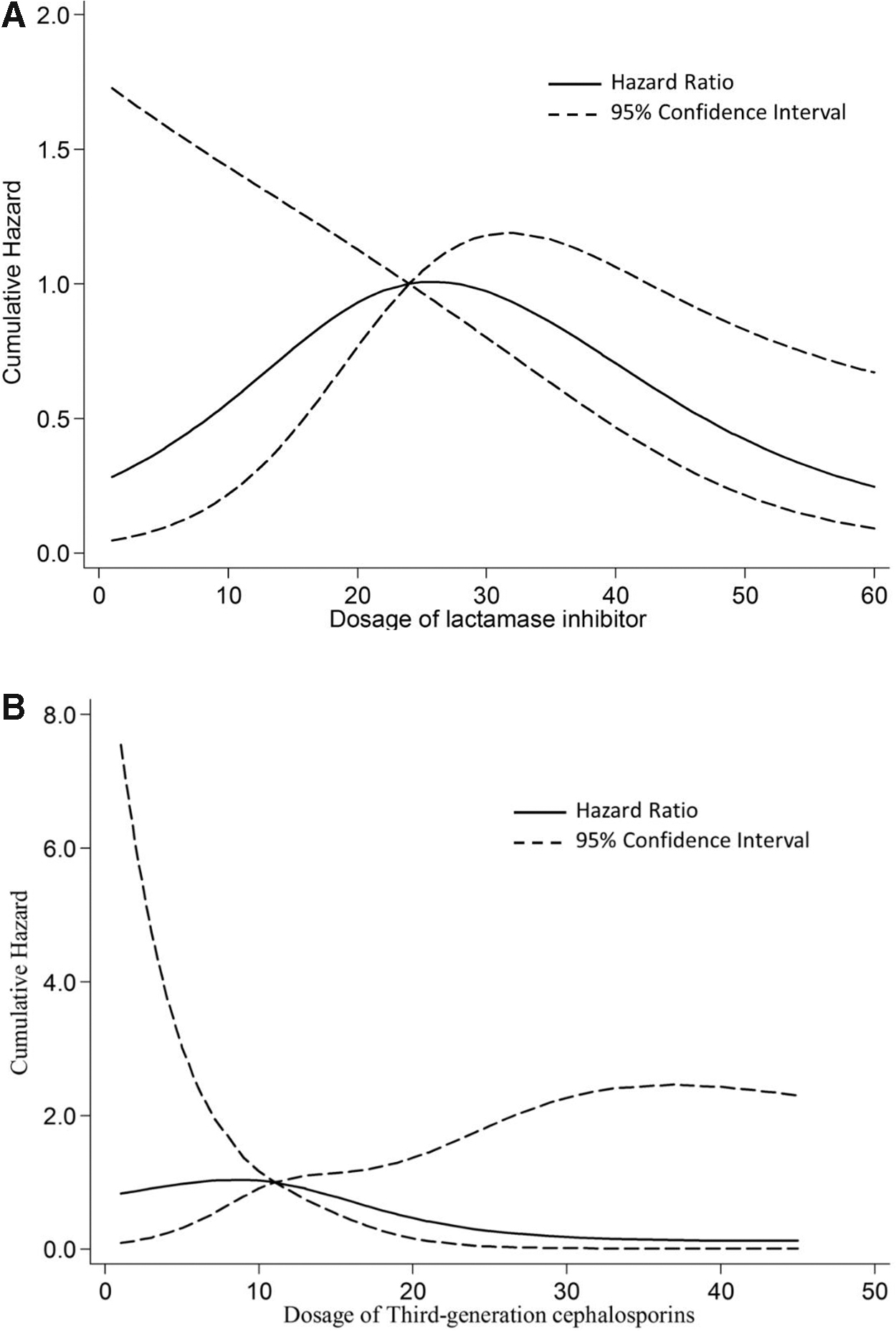
Results of restricted cubic spline regression on Escherichia coli:
Moreover, there is an obvious characteristic of “parabolic curve” for the hazard of CREC induction due to β-lactamase inhibitor mixtures, and the hazard value gradually increases with the dose, reaches the maximum at 24 g, and finally gradually decreases from 26 g. However, there is no significant change in the HR of CREC induction with the increased dose of 3rd-cephalosporins (Fig. 4).
Discussion
Carbapenems are the most effective drugs for the treatment of severe infections with gram-negative bacteria due to their broad antimicrobial spectrum and high stability for hydrolysis by most β-lactamases, including extended-spectrum β-lactamases (ESBLs) and AmpC cephalosporinases. 17 However, unreasonable use of antibiotics leads to the emergence of CRE, which is becoming more and more serious, and the treatment option is limited.7,18,19 Therefore, understanding the hazard factors for antibiotic-induced CRE is very important in the early selection of the empirical antibiotic program.
In this study, we explored the relationship between the use of antibiotics and the occurrence of CRKP and CREC, respectively. Compared with CSE, the risk factors of CRE in clinical samples were male, urinary cannula, vascular cannula, mechanical ventilation, blood transfusion or use of blood products, respiratory failure, and ICU admission.
Previous studies confirmed that antibiotic exposure was a risk factor for CRE infection.20,21 In our study, the first significant finding was that the difference in antibacterial therapy among monotherapy, combination therapy, and unused was significant between the CSKP group and the CRKP group, and the proportion of combined antibacterial therapy in the CRKP group was higher. Increased exposure to one antibiotic increased the effect of exposure to other antibiotics, at the risk of CRKP infection. 22 Therefore, combined use of antibiotics would increase the selection pressure of antibiotics, allowing carbapenem-resistant bacteria to produce various carbapenem-resistant mechanisms. 23 This result was consistent with that in the CSEC group and the CREC group. During the study, β-lactamase inhibitor mixtures were the most commonly used antibiotics.
The second significant finding was that carbapenem, β-lactamase inhibitor mixtures, and quinolones were associated with higher 30-day CRKP isolation hazards than other antibiotics in the time-to-event analysis. Among the antibiotics, carbapenems comprised the most principal hazard factor, which was consistent with previous studies.2,24–30 The use of carbapenem may promote the production of carbapenemase, such as K. pneumoniae carbapenemase and metallo-β-lactamases, which could increase the production of CRE. 31
Other mechanisms of carbapenem resistance include outer membrane porin expression loss combined with ESBLs and AmpC enzyme, change of antimicrobial target, and high expression of efflux pump.32–34 The result suggested that the restriction of carbapenems was associated with a significant reduction in the incidence of CRKP. However, for CREC, the 30-day hazards were significantly higher in patients with β-lactamase inhibitor mixtures and 3rd-cephalosporins than other antibiotics.
Last but not least, we found that the hazard of CRKP induction decreased with the increased dose of β-lactamase inhibitor mixtures, but there was no significant change in the HR of CRKP induction with the increased dose of quinolones or carbapenems. The result indicated that the risk of CRKP induction was higher in low-dose β-lactamase inhibitor mixtures exposure. Therefore, we suggested that adequate dose of β-lactamase inhibitor mixtures should be used in the treatment of these patients. However, there was no relevant research, and further studies were necessary to explore the mechanism.
We also found that increasing the use of carbapenem or quinolones did not lead to a significant increase in the resistance of carbapenem among Enterobacterales, which was similar to a few previous studies.35–37 However, some studies showed that carbapenem or fluoroquinolone use had a positive relationship with the incidence of CRE.36,38 The result was still controversial, which might be explained by the influence of multiple interactive factors that had an impact on the induction of CRE. As a preceding result, the hazard of CRKP induction decrease might be responsible for the increasing dose of β-lactamase inhibitor mixtures.
Moreover, there was an obvious characteristic of a “parabolic curve” for the hazard of CREC induction due to β-lactamase inhibitor mixtures, and the hazard value gradually increased with the dose, reached the maximum at 24 g, and finally gradually decreased from 26 g. In other words, with the increasing use of β-lactamase inhibitor mixtures, the risk of CREC induction increased and reached the maximum when the dose of β-lactamase inhibitor mixtures reached 24 g, but it gradually decreased when the dose exceeded 26 g.
However, no existing study reported this correlation. This result may explain why the incidence of CREC was low in CRE. Although antibiotic exposure may lead to clinically significant antibiotic resistance, the effects are not consistent and vary with the organism and antibiotic resistance mechanism. Large sample studies were needed to further clarify the mechanism of the effect of β-lactamase inhibitor mixture doses on CRE.
In this study, β-lactamase inhibitor mixtures had different drug resistance, inducing effects on CRKP and CREC, which suggested that the maximum dose should not be blindly pursued in clinical medication. We should choose the appropriate treatment plan according to the specific characteristics of microbial distribution, or adjust the antimicrobial treatment strategy in real time according to the characteristics of microbial distribution, which raised higher requirements for the rational use of antibiotics.
There were several potential limitations in our study. First, this study was a retrospective design conducted in a large tertiary A-level hospital, not multicenter research. Second, due to the low prevalence of CRE and the two types of CRE that were analyzed according to the use of antibiotics, the sample size was not large enough. Third, in our study, patients with CRKP were less likely to use carbapenems before detection of CRKP.
So, we found that there was no significant change in the HR of CRKP induction with the increased dose of carbapenems. However, the effect of carbapenems on CRE was beyond all doubt. Fourth, genotypic analysis of drug resistance genes was not performed in the study, and longer observation is needed for further analysis. Therefore, multicenter studies with a large sample size are necessary in future to address these limitations to further confirm the relationship between CRE and antibiotic usage.
Conclusion
In conclusion, β-lactamase inhibitor mixtures had different drug resistance inducing effects on CRKP and CREC. The findings will be useful for identifying specific antimicrobial agents as targets for interventions. Rational use of antibiotics and infection control measures are urgently needed to reduce the selective pressure of antibiotics, delay the occurrence of CRE, and control its cross-transmission.
Footnotes
Authors' Contributions
Y.L. and J.-Y.W. designed the study. D-Q.-W. and C.W. collected the data. Y.L. and Q.X. performed the data analysis. J.C. and Y.L. wrote the article. All authors have read and critically revised the article.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Disclosure Statement
No competing financial interests exist.
Funding Information
This study was supported by the Scientific and Technology Department of Sichuan Province, the scientific and technological emergency response project (2020YFS0003).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
