Abstract
Overexpression of human epidermal growth factor receptor 2 (HER2) in breast and gastric cancers is an important target for monoclonal antibody (mAb) therapy. All therapeutic mAbs, including anti-HER2 mAbs, exhibit adverse effects probably due to the recognition of antigens expressed in normal cells. Therefore, tumor-selective or specific mAbs can be beneficial in reducing the adverse effects. In this study, we established a novel cancer-specific anti-HER2 monoclonal antibody, named H2Mab-250/H2CasMab-2 (IgG1, kappa). H2Mab-250 reacted with HER2-positive breast cancer BT-474 and SK-BR-3 cells. Importantly, H2Mab-250 did not react with nontransformed normal epithelial cells (HaCaT and MCF 10A) and immortalized normal epithelial cells in flow cytometry. In contrast, most anti-HER2 mAbs, such as H2Mab-119 and trastuzumab reacted with both cancer and normal epithelial cells. Immunohistochemical analysis demonstrated that H2Mab-250 possesses much higher reactivity to the HER2-positive breast cancer tissues compared with H2Mab-119, and did not react with normal tissues, including heart, breast, stomach, lung, colon, kidney, and esophagus. The epitope mapping demonstrated that the Trp614 of HER2 domain IV mainly contributes to the recognition by H2Mab-250. H2Mab-250 could contribute to the development of chimeric antigen receptor-T or antibody–drug conjugates without adverse effects for breast cancer therapy.
Introduction
Human epidermal growth factor receptor 2
The major adverse effect associated with anti-HER2 therapeutic mAbs is cardiotoxicity, thereby necessitating routine cardiac monitoring in clinics. 10 Furthermore, mice lacking ErbB2 (ortholog of HER2) displayed embryonic lethality due to the dysfunctions associated with a lack of cardiac trabeculae. 11 Ventricular-restricted ErbB2-deficient mice showed the features of dilated cardiomyopathy. 12 These results indicate that HER2 is vital for normal heart development and homeostasis. Therefore, more selective anti-HER2 mAbs against tumors, which can reduce heart failures are required.
We previously established several anti-HER2 mAbs, such as H2Mab-19 (IgG2b, kappa), 13 H2Mab-41 (IgG2b, kappa), 14 H2Mab-77 (IgG1, kappa), 15 H2Mab-119 (IgG1, kappa), 16 H2Mab-139 (IgG1, kappa), 17 and H2Mab-181 (IgG1, kappa) 18 by the immunization of HER2 ectodomain (HER2ec). We further engineered the mAbs into the mouse IgG2a type (H2Mab-77-mG2a, H2Mab-119-mG2a, and H2Mab-139-mG2a, respectively), and produced the core fucose-deficient types (H2Mab-77-mG2a-f, H2Mab-119-mG2a-f, and H2Mab-139-mG2a-f, respectively) to potentiate the antibody-dependent cellular cytotoxicity (ADCC) and antitumor effect in vivo.19–21 In this study, we developed and characterized a novel HER2 mAb, named H2Mab-250/H2CasMab-2.
Materials and Methods
Cell culture
Chinese hamster ovaries, (CHO)-K1, BT-474, SK-BR-3, MDA-MB-468, MCF 10A, hTERT TIGKs, HBEC3-KT, hTERT-HME1, RPTEC/TERT1, and P3X63Ag8U.1 (P3U1), were obtained from the American Type Culture Collection (ATCC, Manassas, VA). Human keratinocyte HaCaT was purchased from Cell Lines Service GmbH (Eppelheim, Germany). The hTCEpi, hTEC/SVTERT24-B, and HCEC-1CT were purchased from EVERCYTE (Vienna, Austria).
The cDNA of HER2 (wild type; WT) and deletion mutants (dN218, dN342, and dN511) were cloned into the pCAG-nPA16 vector (FUJIFILM Wako Pure Chemical Corporation, Osaka, Japan). A HER2 point mutant (W614A) and HER2 WT were cloned into the pCAG-nPA-cRAPMAP vector (FUJIFILM Wako Pure Chemical Corporation). CHO-K1 cells were transfected with the abovementioned vectors using a Neon transfection system (Thermo Fisher Scientific, Inc., Waltham, MA). A few days after transfection, PA tag-positive cells were sorted by the cell sorter (SH800; Sony Corp., Tokyo, Japan) using NZ-1, which was originally developed as an anti-human podoplanin mAb. 22 Finally, CHO/HER2 and CHO/HER2 (dN218, dN342, dN511, and W614A) cell lines were established.
CHO-K1, CHO/HER2 (WT, deletion, and point mutants), and P3U1 were cultured in Roswell Park Memorial Institute-1640 medium (Nacalai Tesque, Inc., Kyoto, Japan), and BT-474, SK-BR-3, MDA-MB-468, HEK293T, and HaCaT were cultured in Dulbecco's modified Eagle's medium (DMEM) (Nacalai Tesque, Inc.), supplemented with 10% heat-inactivated fetal bovine serum (Thermo Fisher Scientific, Inc.), 100 U/mL of penicillin, 100 μg/mL streptomycin, and 0.25 μg/mL amphotericin B (Nacalai Tesque, Inc.). Mammary epithelial cell line, MCF 10A was cultured in Mammary Epithelial Cell Basal Medium BulletKit™ (Lonza, Basel, Switzerland) supplemented with 100 ng/mL cholera toxin (Sigma-Aldrich Corp., St. Louis, MO).
Immortalized normal epithelial cell lines were maintained as follows; hTERT TIGKs, Dermal Cell Basal Medium and Keratinocyte Growth Kit (ATCC); HBEC3-KT, Airway Epithelial Cell Basal Medium and Bronchial Epithelial Cell Growth Kit (ATCC); hTERT-HME1, Mammary Epithelial Cell Basal Medium BulletKit without GA-1000 (Lonza); hTCEpi, KGMTM-2 BulletKit (Lonza); hTEC/SVTERT24-B, OptiPRO™ SFM, and GlutaMAX™-I (Thermo Fisher Scientific, Inc.); RPTEC/TERT1, DMEM/F-12, and hTERT Immortalized RPTEC Growth Kit with supplement A and B (ATCC); HCEC-1CT, DMEM/M199 (4:1, Thermo Fisher Scientific, Inc.), 2% Cosmic Calf Serum (Cytiva, Marlborough, MA), 20 ng/mL hEGF (Sigma-Aldrich Corp.), 10 μg/mL insulin (Sigma-Aldrich Corp.), 2 μg/mL apo-transferrin (Sigma-Aldrich Corp.), 5 nM sodium selenite (Sigma-Aldrich Corp.), 1 μg/mL hydrocortisone (Sigma-Aldrich Corp.).
All cell lines were cultured at 37°C in a humidified atmosphere with 5% CO2 and 95% air.
Development of hybridomas
The animal was housed under specific pathogen-free conditions. All animal experiments were approved by the Animal Care and Use Committee of Tohoku University (Permit number: 2022MdA-001).
Anti-HER2 mAbs, such as H2Mab-19 (IgG2b, kappa), 13 H2Mab-41 (IgG2b, kappa), 14 H2Mab-77 (IgG1, kappa), 15 H2Mab-119 (IgG1, kappa), 16 H2Mab-139 (IgG1, kappa), 17 and H2Mab-181 (IgG1, kappa), 18 were established previously. In this study, H2Mab-193 (IgG1, kappa), H2Mab-215 (IgG1, kappa), and H2Mab-250 (IgG1, kappa) were established by the same strategy. Briefly, BALB/c mice were immunized with recombinant HER2ec produced by LN229 cells together with Imject Alum (Thermo Fisher Scientific, Inc.). After several additional immunizations, spleen cells were fused with P3U1 cells. The culture supernatants of hybridomas were screened using enzyme-linked immunosorbent assay (ELISA) with recombinant HER2ec and flow cytometry with cell lines.
Production of recombinant mAb
To generate recombinant H2Mab-250 and H2Mab-119, their VH cDNAs and the CH cDNA of mouse IgG1 were cloned into the pCAG-Neo vector (FUJIFILM Wako Pure Chemical Corporation). The VL cDNAs and CL cDNA of the mouse kappa light chain were also cloned into the pCAG-Ble vector (FUJIFILM Wako Pure Chemical Corporation). The vectors were transfected into ExpiCHO-S cells using the ExpiCHO Expression System (Thermo Fisher Scientific, Inc.), and Ab-Capcher (ProteNova, Kagawa, Japan) was used to purify the recombinant H2Mab-250 and H2Mab-119.
To generate mouse IgG2a-type H2Mab-250 (H2Mab-250-mG2a), we cloned the VH cDNA of H2Mab-250 and the CH of mouse IgG2a into the pCAG-Neo vector. The mouse kappa light chain vector of H2Mab-250 is described above. To produce the defucosylated form (H2Mab-250-mG2a-f), the vectors were transfected into BINDS-09 (fucosyltransferase 8-knockout ExpiCHO-S) cells using the ExpiCHO Expression System. H2Mab-250-mG2a-f was purified using Ab-Capcher.
Flow cytometry
Cells were collected using 0.25% trypsin and 1 mM ethylenediamine tetraacetic acid (EDTA; Nacalai Tesque, Inc.). The cells (1 × 105 cells/sample) were treated with mouse anti-HER2 mAbs (10 μg/mL), trastuzumab (10 μg/mL), NZ-1 (10 μg/mL), or blocking buffer (control; 0.1% bovine serum albumin [BSA] in phosphate-buffered saline [PBS]) for 30 minutes at 4°C. Next, the cells were treated with Alexa Fluor 488-conjugated anti-mouse IgG (1:1000; Cell Signaling Technology, Danvers, MA) for mouse anti-HER2 mAbs, Alexa Fluor 488-conjugated anti-human IgG (1:1000; Sigma-Aldrich, Corp.) for trastuzumab, or Alexa Fluor 488-conjugated anti-rat IgG (1:1000; Cell Signaling Technology) for NZ-1 for 30 minutes at 4°C. The fluorescence data were collected using EC800 or SA3800 Cell Analyzer (Sony Corp), and the data were analyzed using FlowJo (BD Biosciences, Franklin Lakes, NJ).
ADCC reporter bioassay
The ADCC reporter bioassay was performed using an ADCC Reporter Bioassay Kit (Promega Corporation, Madison, WI), according to the manufacturer's instructions. Target cells (BT-474 and HaCaT, 12,500 cells per well) were cultured in a 96-well white solid plate. H2Mab-250-mG2a-f and trastuzumab (Herceptin; Chugai Pharmaceutical Co., Ltd, Tokyo, Japan) were serially diluted and added to the target cells. Jurkat cells stably expressing the human FcγRIIIa receptor and a nuclear factor of activated T cell (NFAT) response element driving firefly luciferase, were used as effector cells. The engineered Jurkat cells (75,000 cells in 25 μL) were then added and cocultured with antibody-treated target cells at 37°C for 6 hours. Luminescence using the Bio-Glo Luciferase Assay System was measured using a GloMax luminometer (Promega Corporation).
Immunohistochemical analysis
Formalin-fixed paraffin-embedded tissue of HER2-positive breast cancer was obtained from Sendai Medical Center. 15 Informed consent for sample procurement and subsequent data analyses was obtained from the patient or the patient's guardian at Sendai Medical Center. Normal tissues were purchased from BioChain Institute, Inc. (Eureka Drive Newark, CA) or Cybrdi, Inc. (Frederick, MD). The tissue sections were autoclaved in citrate buffer (pH 6.0; Nichirei Biosciences, Inc., Tokyo, Japan) for 20 minutes. The blocking was performed using SuperBlock T20 (Thermo Fisher Scientific, Inc.). The sections were incubated with H2Mab-250 (1, 0.5, or 0.1 μg/mL) and H2Mab-119 (0.5 or 0.1 μg/mL), and then treated with the EnVision+ Kit for mouse (Agilent Technologies, Inc., Santa Clara, CA). The chromogenic reaction was performed using 3,3′-diaminobenzidine tetrahydrochloride (Agilent Technologies, Inc.). Counterstaining was performed using Hematoxylin (FUJIFILM Wako Pure Chemical Corporation) and Leica DMD108 (Leica Microsystems GmbH, Wetzlar, Germany) was used to obtain images and examine the sections.
ELISA
Synthesized peptides covering the HER2 extracellular domain IV and point mutant peptides were synthesized by Sigma-Aldrich Corp. The peptides (10 μg/mL) and HER2ec (1 μg/mL) were immobilized on Nunc Maxisorp 96-well immunoplates (Thermo Fisher Scientific, Inc.). Plate washing was performed with PBS containing 0.05% (v/v) Tween 20 (PBST; Nacalai Tesque, Inc.). After blocking with 1% (w/v) BSA in PBST, H2Mab-250 (10 μg/mL) was added to each well. Then, the wells were further incubated with peroxidase-conjugated anti-mouse immunoglobulins (1:2000 dilution; Agilent Technologies, Inc.). Enzymatic reactions were conducted using One-Step Ultra TMB. The optical density at 655 nm was measured using an iMark microplate reader.
Determination of KD through surface plasmon resonance
Measurement of KD between H2Mab-250 and the HER2 peptides was performed using surface plasmon resonance (SPR). H2Mab-250 was immobilized on the sensor chip CM5 in accordance with the manufacturer's protocol by Cytiva. Immobilization of H2Mab-250 (10 μg/mL in acetate buffer (pH 4.0); Cytiva) was carried out using an amine coupling reaction. The surface of the flow cell 2 of the sensor chip CM5 was treated with 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide and N-hydroxysuccinimide, followed by the injection of H2Mab-250. The KD between H2Mab-250 and the peptides was determined using Biacore X100 (Cytiva). The binding signals were measured using a single-cycle kinetics method. Data were analyzed by 1:1 binding kinetics using Biacore X100 evaluation software (Cytiva) to determine the association rate constant (ka) and dissociation rate constant (kd) and KD. The affinity constant (KA) at equilibrium was calculated as 1/KD.
Results
Establishment of H2Mab-250
We immunized mice with HER2ec produced by LN229 cells. The culture supernatants of hybridomas were screened using ELISA with HER2ec. We further screened the reactivity to HER2-positive breast cancers (BT-474 and SK-BR-3) and nontransformed normal epithelial cells, including HaCaT (keratinocyte) and MCF 10A (mammary gland) using flow cytometry. One of the established hybridomas, H2Mab-250, reacted with CHO/HER2, HER2-positive BT-474, and SK-BR-3 cells, but not with triple-negative MDA-MB-468 cells. H2Mab-250 did not react with HaCaT and MCF 10A cells (Fig. 1A). In contrast, H2Mab-119 showed similar reactivity to both cancer and normal epithelial cells (Fig. 1A).
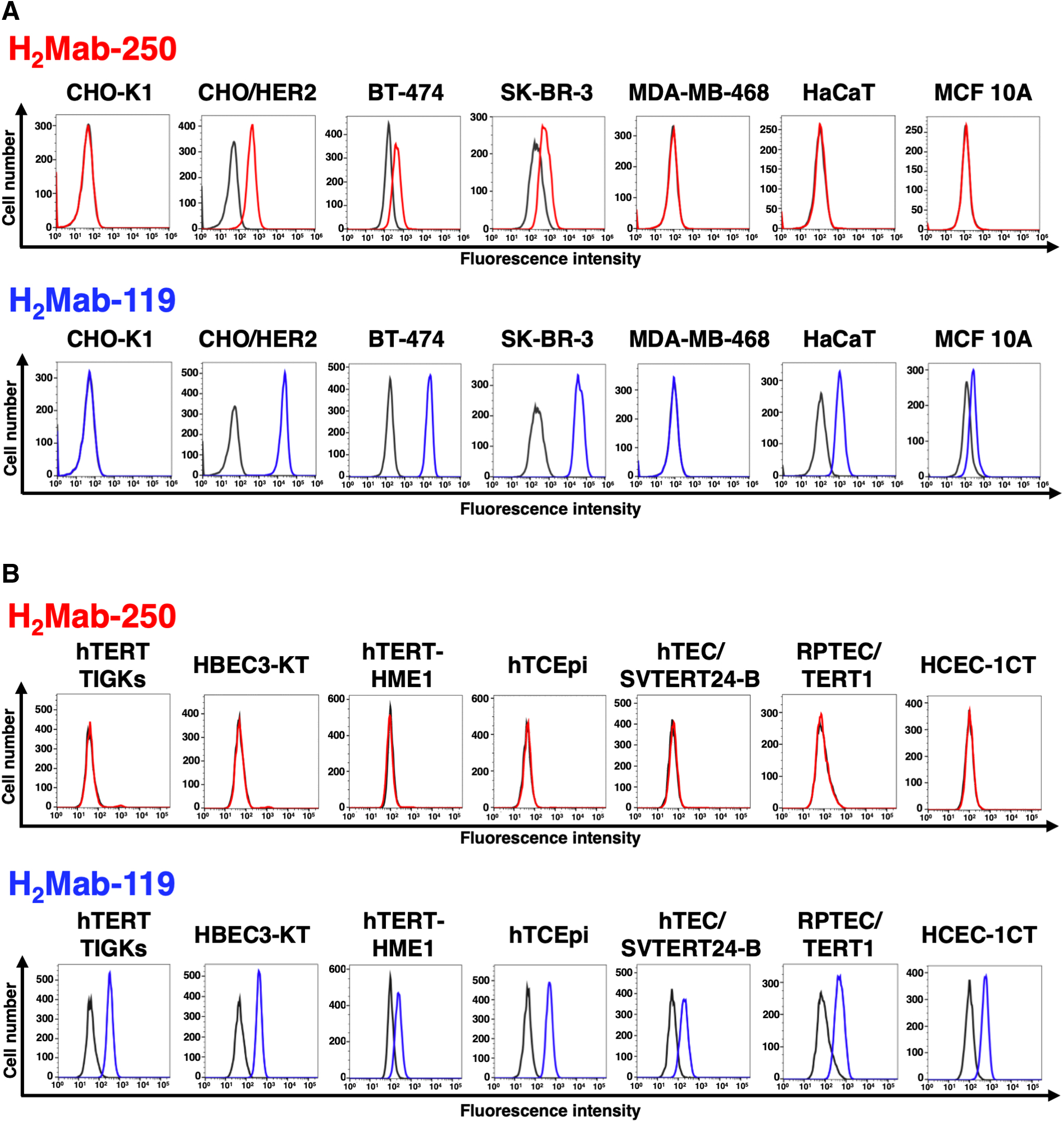
Flow cytometry using anti-HER2 mAbs.
We next investigated the difference in the reactivity to immortalized normal epithelial cells, including hTERT TIGKs (gingiva), HBEC3-KT (lung bronchus), hTERT-HME1 (mammary gland), hTCEpi (corneal), hTEC/SVTERT24-B (thymus), RPTEC/TERT1 (kidney proximal tubule), and HCEC-1CT (colon). H2Mab-250 did not react with those normal cells, while H2Mab-119 was reactive with all immortalized normal epithelial cells (Fig. 1B), indicating that H2Mab-250 possesses cancer-specific reactivity against HER2.
We further compared the reactivity of H2Mab-250 with our established anti-HER2 mAbs and trastuzumab. As shown in Supplementary Figures S1 and S2, anti-HER2 domain I mAbs (H2Mab-7715 and H2Mab-13917), anti-HER2 domain II mAbs (H2Mab-193 and H2Mab-215), anti-HER2 domain III mAbs (H2Mab-1913 and H2Mab-18118), and anti-HER2 domain IV mAbs (H2Mab-4114 and trastuzumab) reacted with HER2-positive breast cancers, nontransformed normal epithelial cells, and immortalized normal epithelial cells. 16 These results indicated that H2Mab-250 exhibits an exceptional reactivity compared with other anti-HER2 mAbs.
The ability of effector cell activation by H2Mab-250 and trastuzumab
The ADCC reporter bioassay is a bioluminescent reporter gene assay to quantify the biological activity of the antibody through FcγRIIIa-mediated pathway activation in an ADCC mechanism of action. 23 We next produced H2Mab-250-mG2a-f, the core-fucose deleted IgG2a version of H2Mab-250 using fucosyltransferase 8-deficient ExpiCHO-S (BINDS-09) cells and examined whether H2Mab-250-mG2a-f could activate ADCC program in the presence of BT-474 and HaCaT cells. To compare the ADCC pathway activation by H2Mab-250-mG2a-f and trastuzumab, we treated BT-474 and HaCaT cells with serially diluted mAbs, and then incubated with effector Jurkat cells, which express the human FcγRIIIa receptor and an NFAT response element driving firefly luciferase.
As shown in Figure 2A, H2Mab-250-mG2a-f could activate the effector (EC50: 9.9 μg/mL), but it was less effective than trastuzumab (EC50: 0.053 μg/mL). Importantly, H2Mab-250-mG2a-f did not activate the effector in the presence of HaCaT cells. In contrast, trastuzumab activated the effector with similar EC50 (0.036 μg/mL) to BT-474 cells (Fig. 2B). These results indicated that H2Mab-250-mG2a-f selectively activates the effector cells against breast cancer cells.
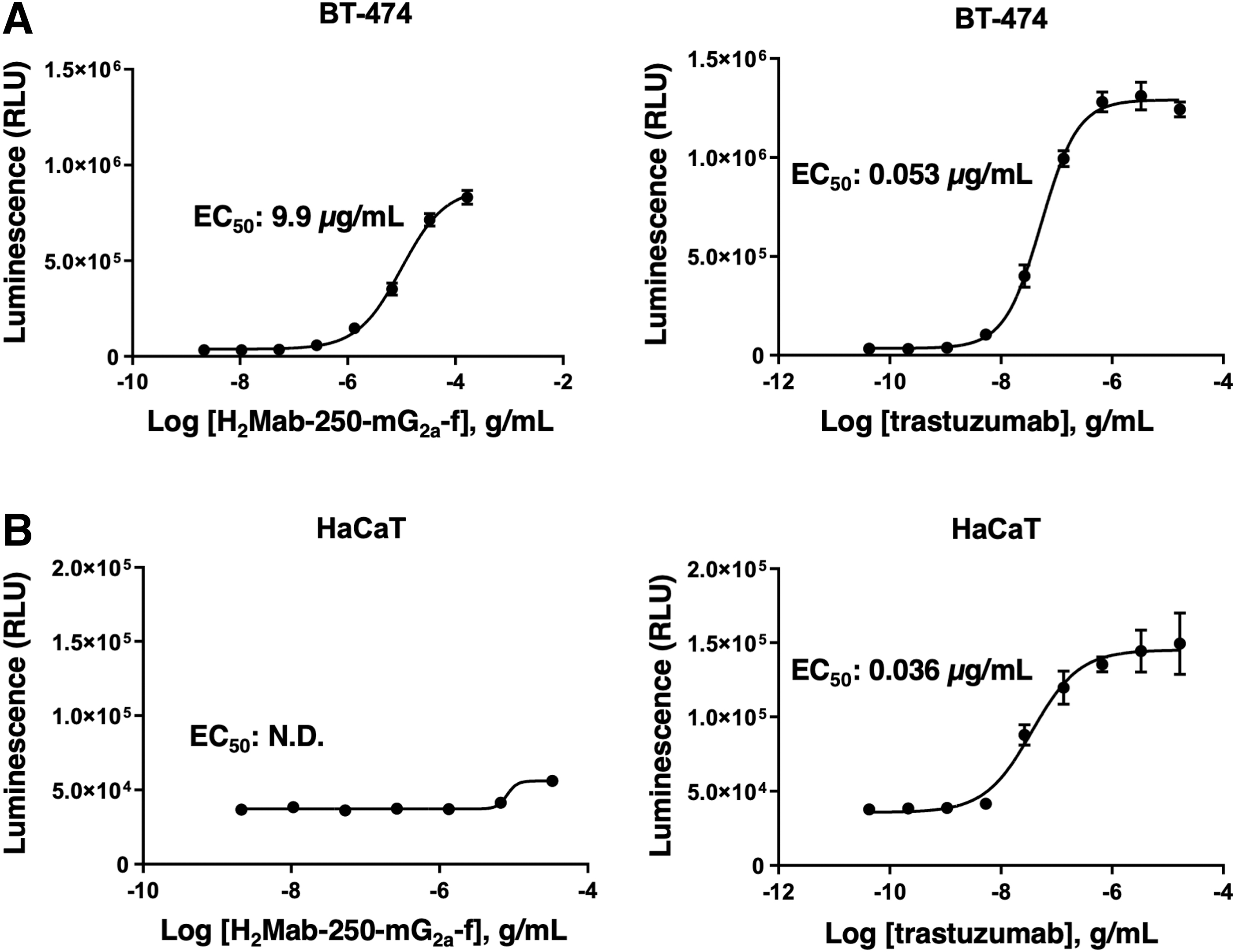
ADCC reporter assay by H2Mab-250-mG2a-f and trastuzumab in the presence of BT-474 and HaCaT cells. Target cells such as BT-474
Immunohistochemical analysis of H2Mab-250 in breast cancer and normal epithelium
Immunohistochemical analysis was performed to examine the reactivity of H2Mab-250 with normal and tumor tissue sections. In contrast to flow cytometry, H2Mab-250 exhibited more potent reactivity to the HER2-positive breast cancer section than H2Mab-119 (Fig. 3A). Since all anti-HER2 therapeutic mAbs are associated with cardiotoxicity, a major adverse effect, 10 the reactivity of H2Mab-250 to a normal heart was further investigated. Even with higher concentrations of H2Mab-250 (1 μg/mL), no reactivity with the normal heart could be detected (Fig. 3B). Finally, the reactivity of H2Mab-250 to other normal tissues was investigated. As shown in Figure 3C, no reactivity of H2Mab-250 with any normal tissues, including breast, stomach, lung, colon, kidney, and esophagus could be observed.
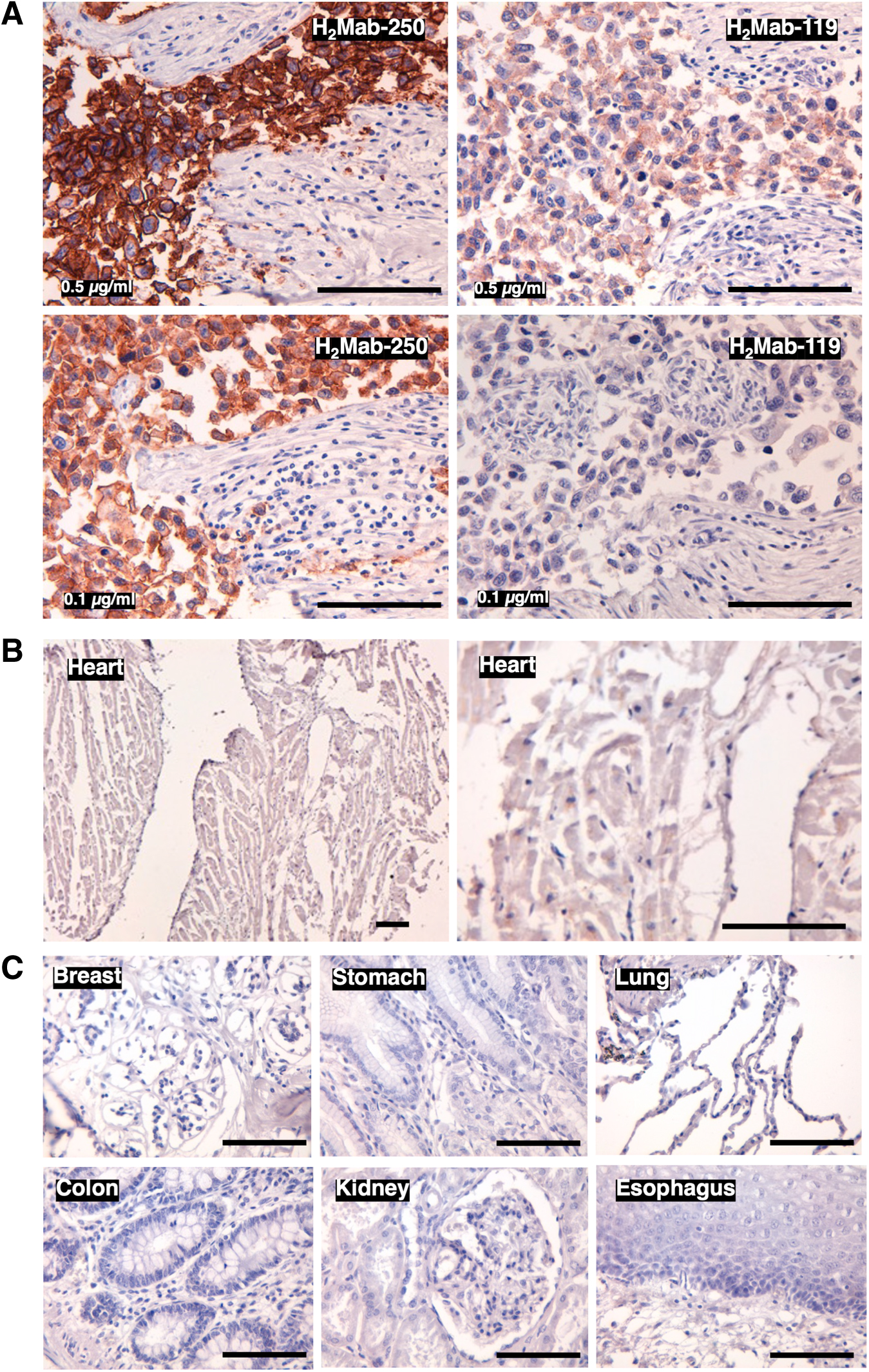
Immunohistochemical analysis of H2Mab-250 in breast cancer and normal epithelium.
Epitope identification for H2Mab-250
To determine the epitope for H2Mab-250, we examined the reactivity to CHO/HER2 (WT) and the N-terminal HER2 deletion mutant (dN218, dN342, and dN511)-expressed CHO-K1 cells (Fig. 4A, left). H2Mab-250 reacted with dN218, dN342, dN511, and HER2 (WT). In contrast, H2Mab-119 reacted with only WT, but not with dN218, dN342, and dN511. Since HER2 (WT) and the deletion mutants possess PA16 tag at the N-terminus, all expressions on the cell surface could be confirmed by anti-PA16 tag mAb, NZ-1 (Fig. 4A, right). These results suggest that H2Mab-250 and H2Mab-119 recognize the domain IV and domain I, respectively.
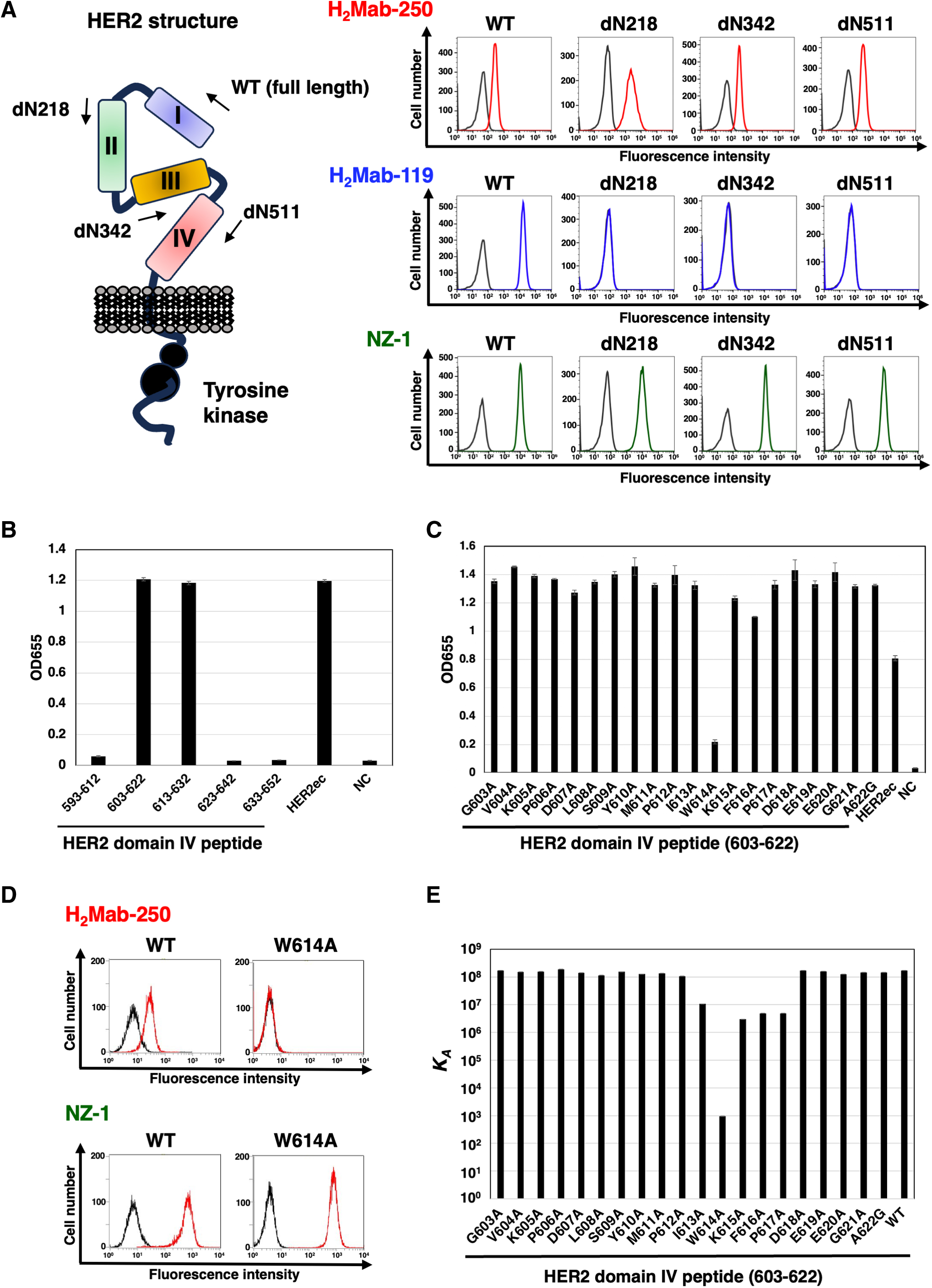
Epitope identification for H2Mab-250.
For further assessment of the H2Mab-250 epitope, ELISA was performed using synthetic peptides that cover the HER2 domain IV. As shown in Figure 4B, H2Mab-250 reacted with HER2 domain IV peptide, amino acids 603–622 and 613–632, but not with 593–612, 623–642, and 633–652, indicating that H2Mab-250 recognizes the 613–622 of HER2 domain IV. We further used alanine-substituted peptides of the 603–622 in HER2 domain IV. A potent reduction of the reactivity was observed in the W614A peptide (Fig. 4C). We confirmed that the reactivity of H2Mab-250 completely disappeared in CHO/HER2 W614A cells in flow cytometry (Fig. 4D).
The KD of H2Mab-250 with the alanine-substituted peptides of HER2 domain IV (603–622) was measured using Biacore X100 (Table 1). The affinity constant (KA) at equilibrium was calculated as 1/KD (Fig. 4E). Compared with the KA of the 603–622 (WT) peptide, decreased KA values were observed from the 613–617 region, suggesting that the 613–617 region is involved in the binding to H2Mab-250. A remarkable reduction was measured in the W614A peptide, indicating that Trp614 is mainly involved in the recognition by H2Mab-250.
Identification of H2Mab-250 Epitope Using Point Mutants by Biacore
WT, wild type.
Discussion
In this study, we developed a cancer-specific mAb targeting HER2. H2Mab-250 can recognize breast cancer cells, but not normal cells in flow cytometry (Fig. 1) and immunohistochemistry (Fig. 3). H2Mab-250-mG2a-f could activate ADCC against breast cancer cells, but not against normal epithelial cells (Fig. 2). We also identified the H2Mab-250 epitope sequence (613-IWKFP-617) by SPR analysis (Fig. 4). The 613-IWKFP-617 sequence is partially included with the wider binding epitope of trastuzumab (residues 579–625). 24 Furthermore, no reaction was observed between H2Mab-250 and CHO/HER2 W614A in flow cytometry (Fig. 4), indicating that Trp614 plays a central role in recognition by H2Mab-250. Although H2Mab-250 possesses a high affinity to epitope-containing peptide (603–622) in SPR analysis, the recognition in flow cytometry using cell lines was lower compared with H2Mab-119 (Fig. 1). In contrast, H2Mab-250 exhibited a higher reactivity than H2Mab-119 in the immunohistochemical analysis using breast cancer tissues (Fig. 3).
This discrepancy might be induced by the possibility that the epitope sequence is partially exposed in cancer cells, but not in normal cells in clinical cancer tissues. The mechanism of recognition by H2Mab-250 should be further investigated in future studies.
For the clinical treatment of metastatic breast cancer, trastuzumab is administered in patients with HER2-overexpressing tumors, which are defined by strong and complete IHC membranous staining of more than 10% of cells (IHC 3+) and/or in situ hybridization (ISH) amplified. Furthermore, trastuzumab-based antibody–drug conjugates (ADCs), such as trastuzumab–deruxtecan (T-DXd) have been evaluated in various clinical trials. Based on the studies, T-DXd has been approved in not only HER2-positive breast cancers,25,26 but also HER2-mutant lung cancer 27 and HER2-low (IHC 1+ or IHC 2+/ISH-nonamplified) advanced breast cancer. 28 A significant number of patients can benefit from T-DXd therapy since approximately half of all breast cancers are classifiable as HER2-low. 29 Meanwhile, cardiotoxicity is the most significant toxicity associated with T-DXd. 30 Further studies are essential to evaluate in vivo toxicities of H2Mab-250.
H2Mab-250-mG2a-f could trigger the ADCC activity to BT-474 selectively (Fig. 2). Although the effect of H2Mab-250-mG2a-f is lower compared with trastuzumab, we should consider that effector Jurkat cells express human FcγRIIIa receptor. In contrast, H2Mab-250 exhibited a superior reactivity to HER2-positive breast cancer tissue sections in immunohistochemistry (Fig. 3). Chimeric antigen receptor (CAR)-T cell therapy against HER2 has been evaluated in clinical studies. 29 It would be worthwhile to investigate the cancer specificity of H2Mab-250 scFv and the efficacy of CAR-T against HER2-positive tumors in future studies. In the future, H2Mab-250 could contribute to the development of CAR-T or ADCs without adverse effects for breast cancer therapy.
Footnotes
Authors' Contributions
M.K.K. and H.S. performed the experiments. M.K.K. and Y.K. designed the experiments. H.S. and M.K.K. analyzed the data. H.S. and Y.K. wrote the article. All authors have read and agreed to the published version of the article.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This research was supported in part by the Japan Agency for Medical Research and Development (AMED) under grant nos.: JP23ama121008 (to Y.K.), JP23am0401013 (to Y.K.), 23bm1123027h0001 (to Y.K.), and JP23ck0106730 (to Y.K.), and by the Japan Society for the Promotion of Science (JSPS) Grants-in-Aid for Scientific Research (KAKENHI) grant nos. 22K06995 (to H.S.), 21K07168 (to M.K.K.), and 22K07224 (to Y.K.).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
