Abstract
Programmed cell death ligand-1 (PD-L1) is a type I transmembrane glycoprotein expressed on antigen-presenting cells. It is also expressed in several tumor cells such as melanoma and lung cancer cells. A strong correlation has been reported between human PD-L1 (hPD-L1) expression in tumor cells and negative prognosis in cancer patients. Here, a novel anti-hPD-L1 monoclonal antibody (mAb) L1Mab-13 (IgG1, kappa) was produced using a cell-based immunization and screening (CBIS) method. We investigated hPD-L1 expression in lung cancer using flow cytometry, Western blot, and immunohistochemical analyses. L1Mab-13 specifically reacted hPD-L1 of hPD-L1-overexpressed Chinese hamster ovary (CHO)-K1 cells and endogenous hPD-L1 of KMST-6 (human fibroblast) in flow cytometry and Western blot. Furthermore, L1Mab-13 reacted with lung cancer cell lines (EBC-1, Lu65, and Lu99) in flow cytometry and stained lung cancer tissues in a membrane-staining pattern in immunohistochemical analysis. These results indicate that a novel anti-hPD-L1 mAb, L1Mab-13, is very useful for detecting hPD-L1 of lung cancers in flow cytometry, Western blot, and immunohistochemical analyses.
Introduction
L
PD-L1 is a type I transmembrane glycoprotein expressed in several cancers including breast, gastric, ovarian, pancreatic as well as in renal cell carcinoma, melanoma, and glioblastoma.(12–18) It is also an immune regulatory molecule that limits T cell effector function.(19) Binding of PD-L1 to its receptor PD-1 inhibits activated T cell proliferation and allows cancer cells evade host immune surveillance.(20,21) Furthermore, several studies have revealed a strong correlation between PD-L1/L2 expression on tumor cells and the poor prognosis in some cancers.(22–24) To use nivolumab or pembrolizumab for lung cancer treatment, information about its effectiveness in detecting PD-L1 expression in immunohistochemistry is necessary.(25) Therefore, more sensitive and specific mAbs for detecting PD-L1 are required. In this study, we generated a novel antihuman PD-L1 (hPD-L1) antibody, L1Mab-13 (mouse IgG1, kappa).
Materials and Methods
Cell lines
LN229, CHO-K1, and P3U1 cell lines were obtained from the American Type Culture Collection (ATCC, Manassas, VA). KMST-6, EBC-1, LK-2, Lu65, and Lu99 were obtained from the Cell Resource Center for Biomedical Research Institute of Development, Aging and Cancer Tohoku University (Miyagi, Japan). PC-10 was purchased from Immuno-Biological Laboratories Co., Ltd. (Gunma, Japan). LN229/hPD-L1 was produced by transfecting pCAG/PA-hPD-L1-RAP-MAP into LN229 cells using a Gene Pulser Xcell electroporation system (Bio-Rad Laboratories, Inc., Berkeley, CA). CHO/hPD-L1 was produced by transfecting pCAG/hPD-L1-RAP-MAP into CHO-K1 cells using Lipofectamine LTX Reagent (Thermo Fisher Scientific, Inc., Waltham, MA). The stable transfectants of LN229/hPD-L1 and CHO/hPD-L1 were established by limiting dilution. PA tag system (GVAMPGAEDDVV vs. clone: NZ-1), RAP tag system (DMVNPGLEDRIE vs. clone: PMab-2), and MAP tag system (GDGMVPPGIEDK vs. clone: PMab-1) have been previously established in Tohoku University Graduate School of Medicine and described in detail.(26–28)
CHO-K1, CHO/hPD-L1, P3U1, KMST-6, EBC-1, LK-2, Lu65, Lu99, and PC-10 cell lines were cultured in RPMI 1640 medium (Nacalai Tesque, Inc., Kyoto, Japan), and LN229 and LN229/hPD-L1 were cultured in Dulbecco's modified Eagle's medium (Nacalai Tesque, Inc.) at 37°C in a humidified atmosphere containing 5% CO2 and 95% air, supplemented with 10% heat-inactivated fetal bovine serum (Thermo Fisher Scientific, Inc.). The culture medium was supplemented with penicillin (100 units/mL), streptomycin (100 μg/mL), and amphotericin B (25 μg/mL) (all from Nacalai Tesque, Inc.). Zeocin (0.5 mg/mL; InvivoGen, San Diego, CA) was added to the culture medium of LN229/hPD-L1 and CHO-K1/hPD-L1.
Human tissues
One lung AC cancer patient who underwent surgery at the Sendai Medical Center was recruited for analysis. The study was approved by the ethical committee of Sendai Medical Center.
Flow cytometry
Cells were harvested by brief exposure to 0.25% trypsin/1-mM ethylenediaminetetraacetic acid (Nacalai Tesque, Inc.). After washing with 0.1% bovine serum albumin in phosphate-buffered saline, the cells were treated either with 1 μg/mL of anti-hPD-L1 mAbs clone 29E.2A3 (BioLegend, Inc., San Diego, CA) or L1Mab-13 for 30 minutes at 4°C. The antibodies were subsequently observed using Alexa Fluor 488-conjugated antimouse IgG (1:1000; Cell Signaling Technology, Inc., Danvers, MA). Fluorescence data were acquired using EC800 Cell Analyzers (Sony Corp., Tokyo, Japan).
Western blot analysis
Cell lysates (10 μg) were boiled in sodium dodecyl sulfate (SDS) sample buffer (Nacalai Tesque, Inc.) and proteins were then electrophoresed on 5%–20% polyacrylamide gels (FUJIFILM Wako Pure Chemical Industries., Ltd., Osaka, Japan) and were transferred onto PVDF membranes (Merck KGaA, Darmstadt, Germany). After blocking with 4% skim milk (Nacalai Tesque, Inc.), membranes were incubated with 10 μg/mL of anti-hPD-L1 (clone L1Mab-13) and 1 μg/mL of anti-β-actin (clone AC-15; Sigma-Aldrich Corp., St. Louis, MO), and then with peroxidase-conjugated antimouse IgG (1:1000 diluted; Agilent Technologies, Inc., Santa Clara, CA), and were finally developed using ImmunoStar LD (FUJIFILM Wako Pure Chemical Industries Ltd.) using a Sayaca-Imager (DRC Co. Ltd., Tokyo, Japan).
Immunohistochemical analyses
Histological sections of 4 μm thickness were autoclaved in citrate buffer (pH 6.0; Nichirei Biosciences, Inc., Tokyo, Japan) for 20 minutes. The sections were then incubated with 10 μg/mL of L1Mab-13 and 2 μg/mL of 28-8 (Agilent Technologies, Inc.) for 1 hour at room temperature, and treated using an Envision+ kit (antimouse; Agilent Technologies, Inc.) and Envision+ kit (antirabbit; Agilent Technologies, Inc.) for 30 minutes, respectively. Color was developed using 3,3-diaminobenzidine tetrahydrochloride (Agilent Technologies, Inc.) for 2 minutes, and counterstaining was performed using hematoxylin (FUJIFILM Wako Pure Chemical Industries., Ltd.).
Results and Discussion
We immunized one mouse with LN229/hPD-L1 cells and performed flow cytometric screening. We previously named this cell-based strategy as the cell-based immunization and screening (CBIS) method.(29,30) Culture supernatants of 672 wells were mixed with LN229/hPD-L1 and LN229 cells, and 27 wells (27/672; 4.0%), showing a stronger reaction against LN229/hPD-L1 cells than LN229 cells, were selected. After limiting dilution, 10 clones were established, of which 7 were classified as IgG1 subclass and 3 as IgG2b subclass. Only one clone, L1Mab-13 (IgG1, kappa), reacted with lung AC tissue, which was previously diagnosed as hPD-L1-positive in an immunohistochemical analysis. In this study, we characterized L1Mab-13.
L1Mab-13 reacted with CHO/hPD-L1, but did not react with CHO-K1 (Fig. 1A). L1Mab-13 also detected endogenous hPD-L1 of human fibroblast cells (KMST-6), indicating L1Mab-13 is useful for flow cytometry. Furthermore, L1Mab-13 showed specific bands of hPD-L1 of CHO/hPD-L1 and KMST-6 in Western blot (Fig. 1B), indicating that L1Mab-13 is also useful for detecting hPD-L1 in Western blot.
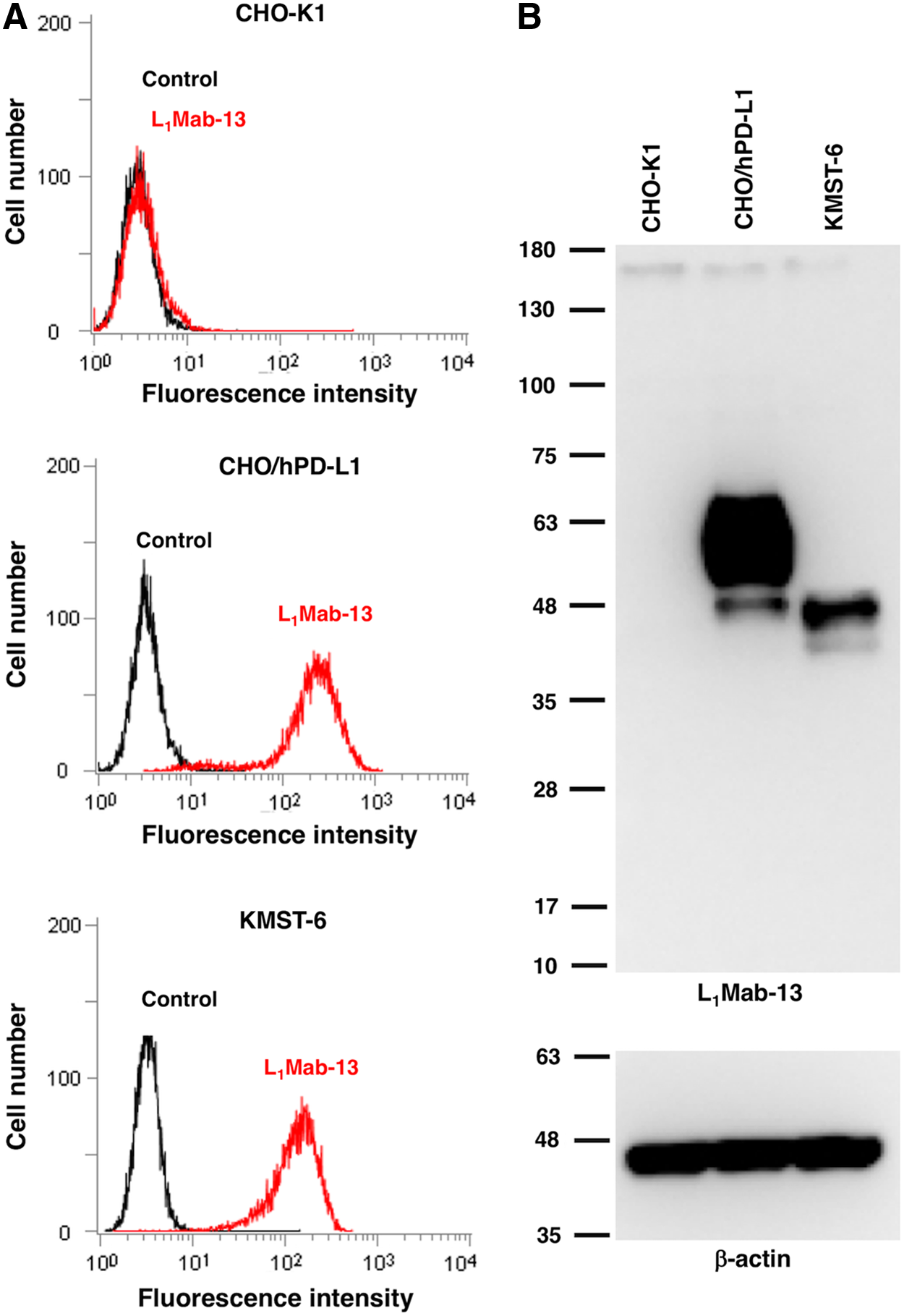
Development of anti-hPD-L1 mAbs.
L1Mab-13 recognized endogenous hPD-L1 in lung cancer cell lines, such as EBC-1, Lu65, and Lu99 in a similar pattern of clone 29E.2A3 using flow cytometry (Fig. 2). To further investigate the utility of L1Mab-13 in human lung cancers, we performed immunohistochemical analyses. Figure 3 shows that both 28-8 (positive control) and L1Mab-13 stained lung cancer cells in a membrane-staining pattern, demonstrating the utility of L1Mab-13 in immunohistochemical analysis in lung cancers.
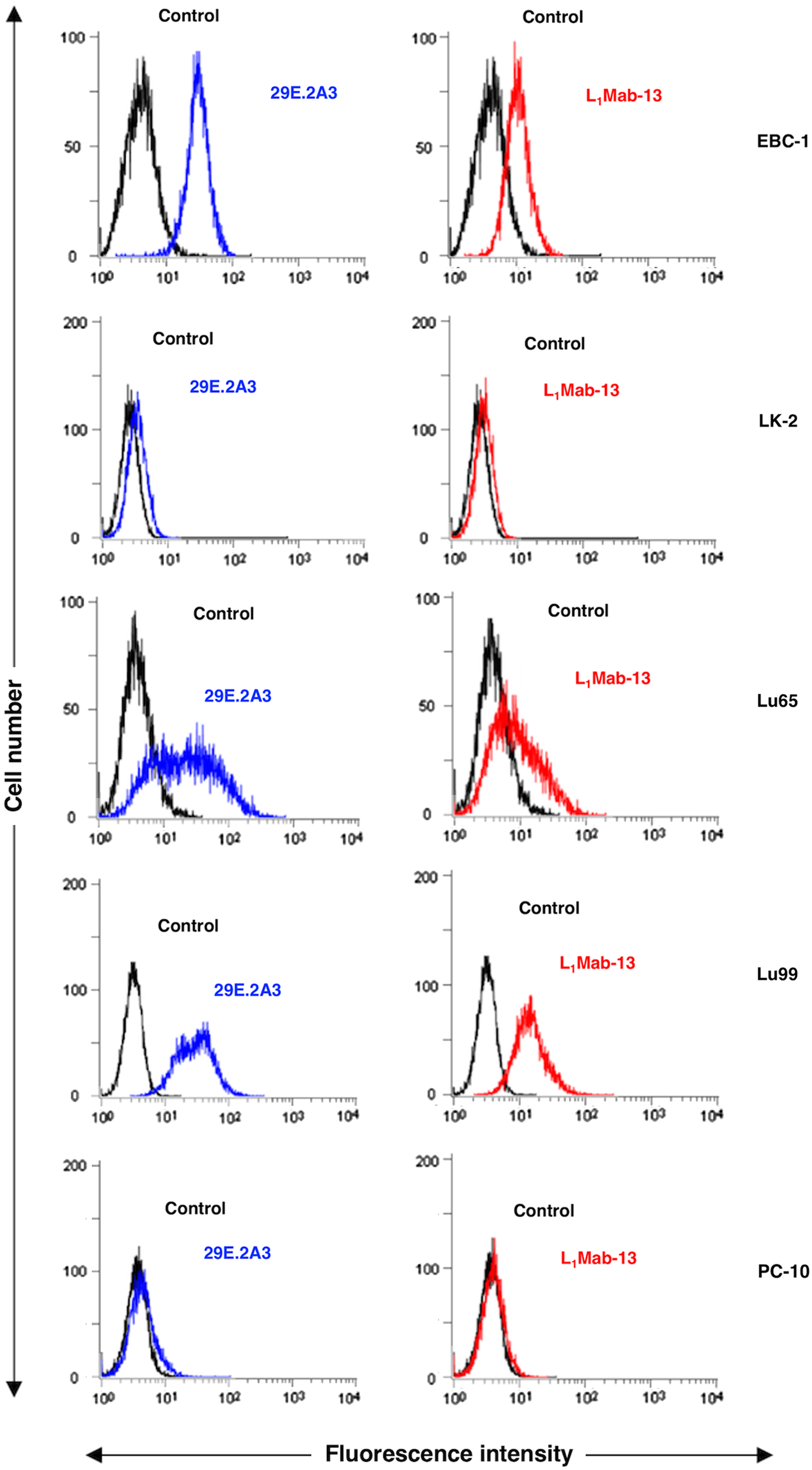
Flow cytometry. Cells were treated with 1 μg/mL 29E.2A3 (positive control; blue line) and L1Mab-13 (red line) followed by treatment with Alexa Fluor 488-conjugated antimouse IgG (negative control; black line).
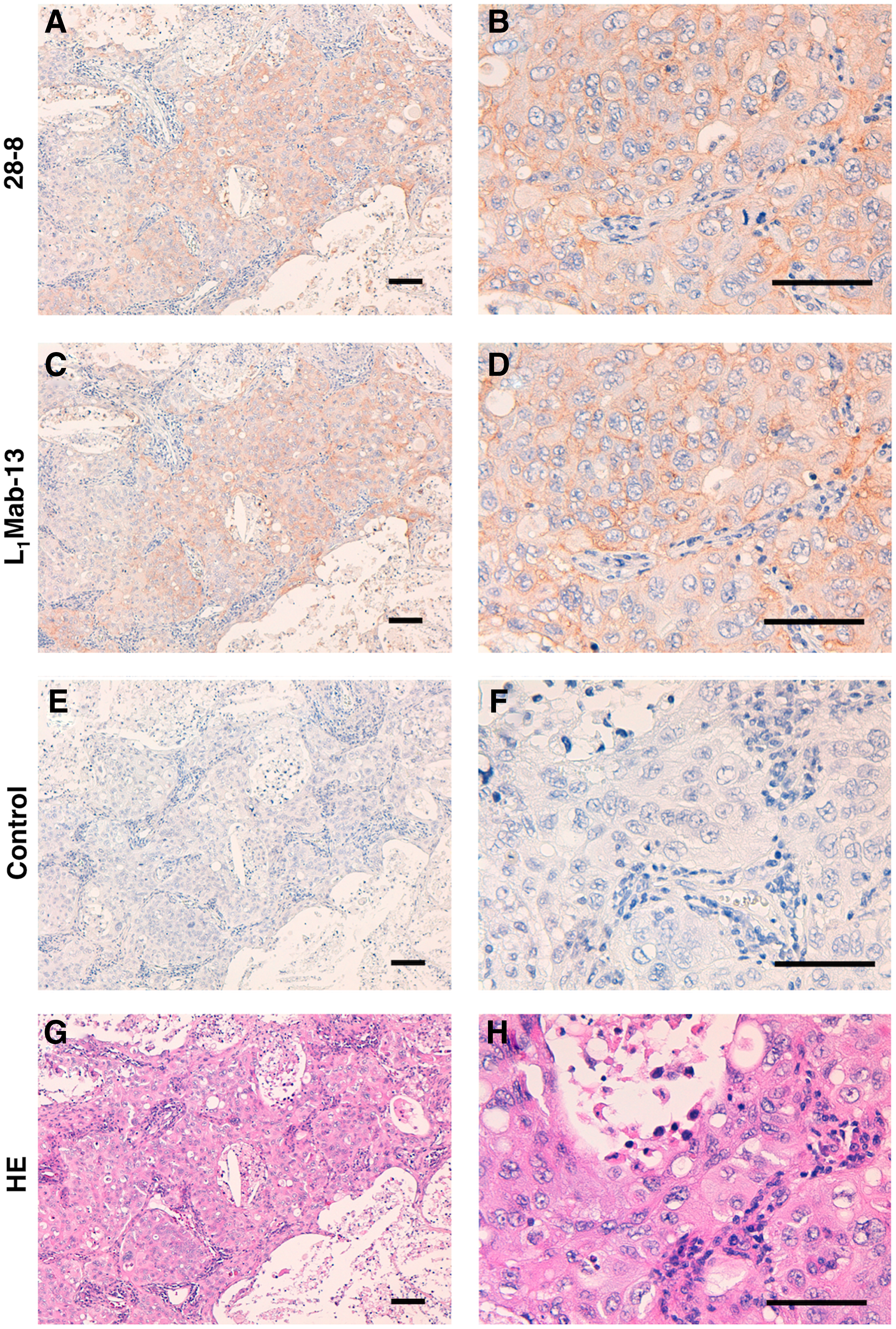
Immunohistochemical analysis by L1Mab-13 against lung AC.
In conclusion, L1Mab-13 is very useful for detecting hPD-L1 using flow cytometry, Western blot, and immunohistochemical analysis of lung cancers. Studies investigating L1Mab-13 reactivity for its applicability in other human cancers are warranted.
Footnotes
Acknowledgments
We thank Noriko Saidoh, Saori Handa, and Yoshimi Nakamura for their excellent technical assistance. This research was supported by AMED under Grant Numbers: JP17am0301010 (Y.K.), JP17am0101078 (Y.K.), and JP17ae0101028 (Y.K.).
Author Disclosure Statement
No competing financial interests exist.
