Abstract
Purpose:
Secondary chronic lymphedema is a complication that seriously affects the quality of life of cancers survivors which is urgent to be studied. However, the current animal models generally have some defects such as short duration that affect the research process. To acquire an animal model which is easier to accomplish as well as higher success rate is the main goal of our experiment.
Methods:
The hind limb model of rats with secondary chronic lymphedema was established by near infrared fluorescence-guided lymphatic system destruction combined with high-fat diet innovation. The model was evaluated by limb circumference, volume, near-infrared fluorescence imaging of lymphatic vessels, and a series of histopathological staining.
Results:
The model was successfully established by operation combined with a high-fat diet and kept persistent effective lasting up to 16 weeks. Compared with the normal diet group, the high-fat group received a higher success rate (77.77% vs. 55.55%), a more obvious degree of limb swelling, and a more significant level of subcutaneous inflammatory factor infiltration at the edema site. Based on it, we kept research and found out that there was no significant difference in the process of subcutaneous fibrosis between the two groups.
Conclusions:
High-fat diet may improve secondary chronic lymphedema rat model success rate as well as effectiveness by changing the level of inflammatory infiltration in the site of lymphedema.
Introduction
Lymphedema (LE) is caused by an anomaly or damage to the lymphatic system that impairs drainage, resulting in the buildup of interstitial fluid and fat, a decrease in oxygen tension, and the development of chronic inflammation and fibrosis.1,2 The most common form is secondary (acquired) lymphedema. 3 Variations in diagnostic criteria have resulted in a reported incidence range of 5%–20%.4–6 The majority of postoperative secondary lymphedema unpredictably develops within 2–10 years or much later, 7 it may cause by acute trauma or not, but there are no definitive supporting studies. 8 But what is certain is that patients with lymphedema had a worse quality of life and mental health than those without lymphedema. 9
It is worth noting that lymphedema is a class of progressive systemic diseases that cannot be simulated by a simple
For an in-depth examination of the pathophysiological mechanisms underlying secondary chronic lymphedema, it is necessary to optimize available animal models together with simple, repeatable, and sustainable. In this study, a nonradiotherapy-based model of secondary chronic lymphedema in the rat hind limb was created by surgery improved by high-fat diet. Moreover, we used this model to investigate how high-fat diet affects the development of lymphedema and its applicability for research.
Materials and Methods
Establishment of a rat model of hind limb lymphedema
In this study, thirty 6-week-old female Sprague–Dawley rats (provided by the Experimental Animal Center of Dalian Medical University) were randomly divided into two groups: the traditional diet group (TRAD) (
After receiving meticulous attention and feeding for 2 weeks, the Sprague–Dawley rats underwent the destruction of lymphatic circulation in the left limbs. The procedure was done under the close supervision of near-infrared (NIR) fluorescence imaging. The surgical protocol was as follows: Before surgery, the rats’ abdomens and lower limbs’ hair were shaved by using a razor. A 1.5-cm incision was performed in the rat’s left groin, the subcutaneous fat pads and inguinal lymph nodes were excised, and the deep inguinal lymphatic vessels were identified and ligated under the guidance of NIR fluorescence imaging. The popliteal lymph nodes were isolated and cleansed, and the lymph vessels were ligated (Fig. 1A). Without sutures, a circular incision was created around the upper middle third of the rat’s limb to counter the strong self-healing ability of rats (Fig. 1B). At 4, 8, 12, and 16 weeks postoperatively, three rats were randomly selected from each of the two groups, undergoing leg circumference measurement, limb volume measurement, and NIR fluorescence imaging, then rats were killed and skin was removed for histopathological study.
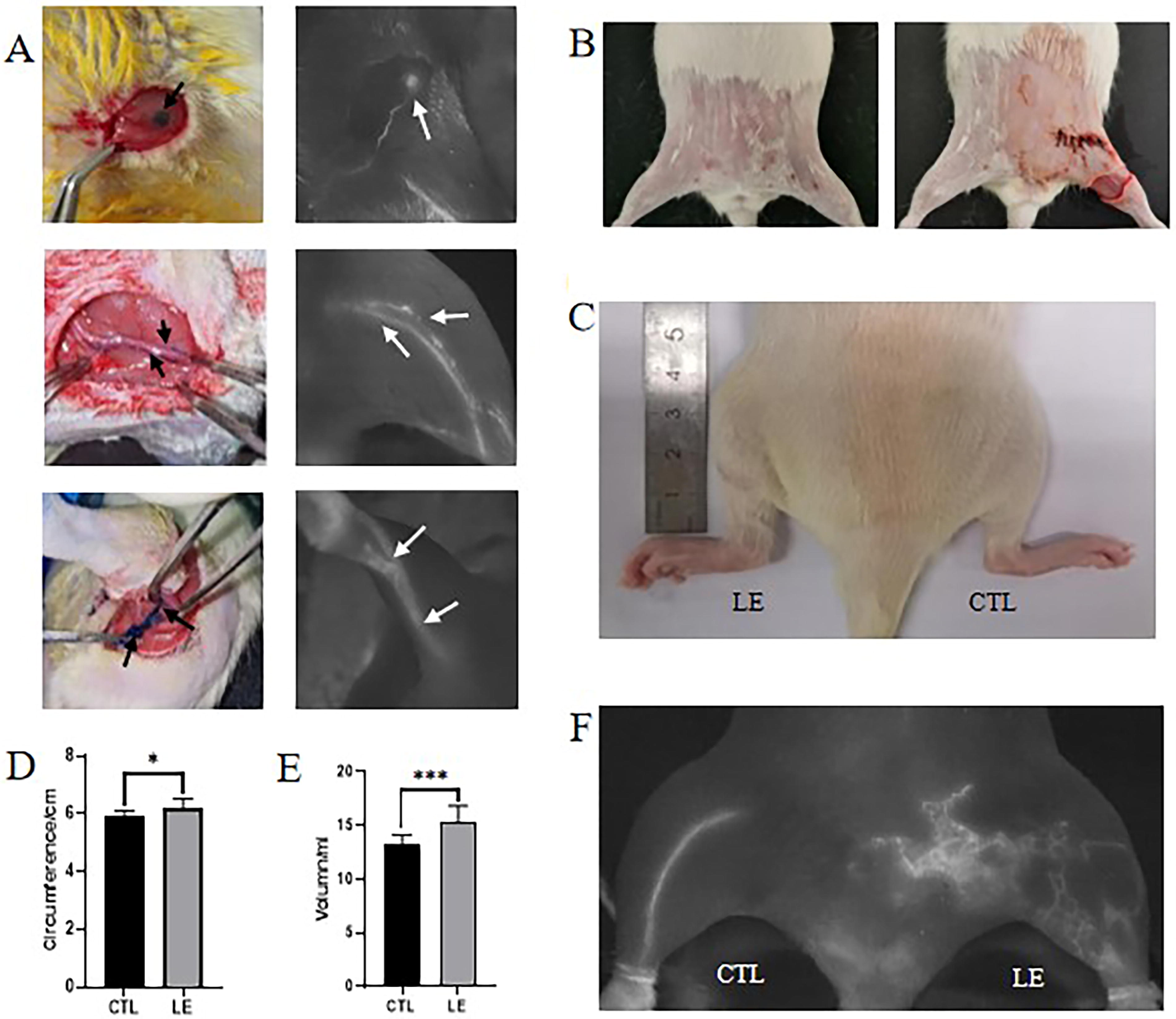
Evaluation criteria for the model success rate
The success rate of the model was determined by comparing NIR fluorescence imaging images of pre- and postoperative and lymphatic reflux in acute and chronic phase, combined with the measured values of limb volume and leg circumference in rats after operation. We regard the bilateral volume difference ≥1 mL at 12 weeks or 16 weeks after operation and the significant difference in bilateral lymphatic reflux shown by NIR fluorescence imaging as a successful model of secondary chronic lymphedema.
Quantitative evaluation of lymphedema
After 4 weeks postoperatively, we measured the upper middle third circumference and volume (water displacement method) of the hind limb of rats every 2 weeks to evaluate the progression of lymphedema. Using a soft ruler to measure the leg circumference of the middle upper third of both lower limbs. To measure the volume of the limb by the water displacement method, the rat’s limb below the surgical incision in the groin was completely submerged in water. We used the right hind limb as a reference to limit the increase in hind limb circumference and volume owing to age and individual variations. Then the difference between the experimental side and the control side was calculated.
NIR fluorescence imaging
Before imaging, the belly and lower limbs of the rats were shaved, and 200 µL of methylene blue solution (Jiangsu, China) was injected into the rat’s footpad and massaged for a few minutes to accelerate the flow of methylene blue. NIR fluorescence camera (MingDe China) were used to capture fluorescent images during operation and the 4th, 8th, 12th, and 16th weeks postoperatively.
Hematoxylin-eosin (HE) staining
Formalin-fixed, paraffin-embedded skin tissue samples were cut into 4-μm thick sections and affixed to slides (188105, CITOTEST), and the sections were dewaxed in xylene and rehydrated in gradient ethanol. Following this, HE staining (G1120, Solarbio) was conducted according to the usual protocol on the whole skin layer to detect skin tissue thickness and fat accumulation.
Masson staining
After being baked for 2 hours at 58°C, the slices were dewaxed in xylene, rehydrated in gradient ethanol, and washed with PBS for two minutes. The slices were then fixed in Bouin’s solution for a further hour at 56°C. Next, they were rinsed under running water for 5–10 minutes to get rid of excess yellow color. Masson staining (CD069, ZHHC) of the whole skin layer was then performed following industry standards to identify collagen deposition in skin tissue.
Immunohistochemical (IHC) staining
The slices were quickly dewaxed in xylene, rehydrated in gradient ethanol, and then subjected to standard procedures (SP-9000, ZSGB-bio) for the detection of Cluster of Differentiation 4 (CD4), Interleukin-1 Beta (IL-1β), Interleukin-6 (IL-6), and Transforming Growth Factor-Beta 1 (TGF-β1) (Abclonal) expressions in rat hind limb skin. The skin tissue loose; therefore, prolonging the baking time can have protective effects.
Statistical analysis
The data was analyzed with SPSS 26.0 software package. Statistical data were expressed by mean ± standard deviation. Differences between groups were assessed using ANOVA and
Results
Construction of lymphedema model
Changes of bilateral limbs and NIR fluorescence imaging 12 weeks after operation
At 12 weeks following surgery, there were substantial disparities in bilateral limbs. The edema and swelling of ankle joint in LE was more obvious than that in CTL, and the skin fold was significantly larger than that in CTL (Fig. 1C). By comparing the leg circumference and volume of LE and CTL, it was observed that the LE limbs were more swollen compared with the CTL side. The limb perimeter of LE was significantly higher than that of CTL (6.209 ± 0.318 cm vs. 5.891 ± 0.212 cm), and the total volume of LE was also significantly larger than that of CTL (15.36 ± 1.47 mL vs. 13.25 ± 0.90 mL), the difference was statistically significant (Fig. 1D, E). NIR fluorescence imaging showed that there was almost no linear lymphatic vessel structure in LE limb lymphatic reflux at 12 weeks after operation, and subcutaneous lymphatic collateral reflux was significantly increased and disorderly distributed, whereas CTL limbs showed trunk lymphatic vessels regularly collecting lymph, and no obvious subcutaneous lymphatic vessel formation was found (Fig. 1F).
Success rate and changes of limb volume and leg circumference in TRAD and HFD
In TRAD, the success rate of the model was 55.55% (5/9), but the effectiveness of HFD was 77.77% (7/9). According to the changes of limb volume, the limb swelling in HFD was larger than that in TRAD after 8 weeks and remained elevated after 12 weeks, while the limb swelling in TRAD decreased slightly (Fig. 2A). At 12 weeks, compared with TRAD, the volume difference between the left and right legs in HFD was larger (2.118 ± 0.998 mL vs. 1.257 ± 0.412 mL), and the difference was significant (
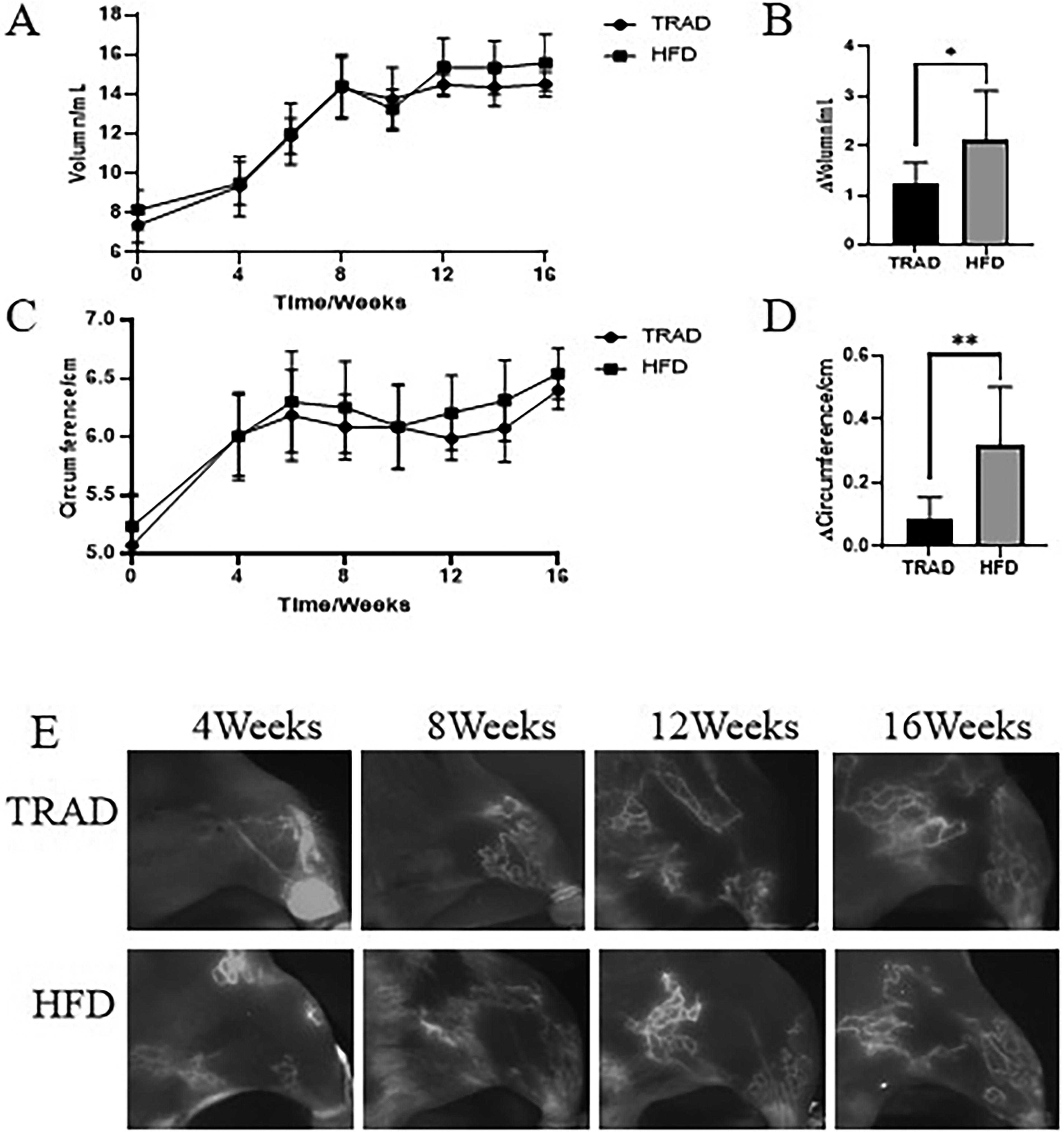
Changes of NIR fluorescence imaging with time in TRAD and HFD
In the CTL limbs, methylene blue-guided NIR fluorescence imaging showed clear lymphatic mainstem conduits, regular lymphatic fluid collection in the lower extremities, and no dermal reflux. In LE limbs, structures resembling lymphatic ducts were barely evident, along with cutaneous reflux development and lymphatic fluid collecting at certain places at 4 weeks after operation. At 8 weeks, there was almost no linear lymphatic structure, and the lymphatic reflux of lower extremities showed nebula-like reflux structure. At 12 weeks, there was basically no obvious linear lymphatic structure, and the lymph was in a state of diffuse reflux. Diffuse reflux continued to increase and the volume increased until 16 weeks after operation. The pattern of lymphatic reflux was almost consistent between the two groups (Fig. 2E).
Histological changes of lymphedema model
Changes of subcutaneous histomorphology with time in TRAD and HFD
HE staining showed that there was no significant change in the skin structure but slight disorder of the reticular layer in the two groups at 4 weeks after operation, and it suggested edematous alterations in the limbs. 8 weeks after operation, the skin thickened significantly, the area of subcutaneous fibrosis, and fat deposition increased in both groups. The subcutaneous fat lobule was obviously disordered and the interlobular septal fibrosis was changed. At 12 weeks after operation, fibrotic tissue gradually replaced adipose tissue, and the tissue between epidermis and muscle expanded obviously and showed chronic changes. Among them, the fat lobule was larger and the subcutaneous gland structure was more damaged in HFD. At 16 weeks, the degree of skin fibrosis and fat deposition in TRAD was better than that at 12 weeks. However, the degree of subcutaneous fat deposition and structural disorder were even more serious in HFD (Fig. 3).
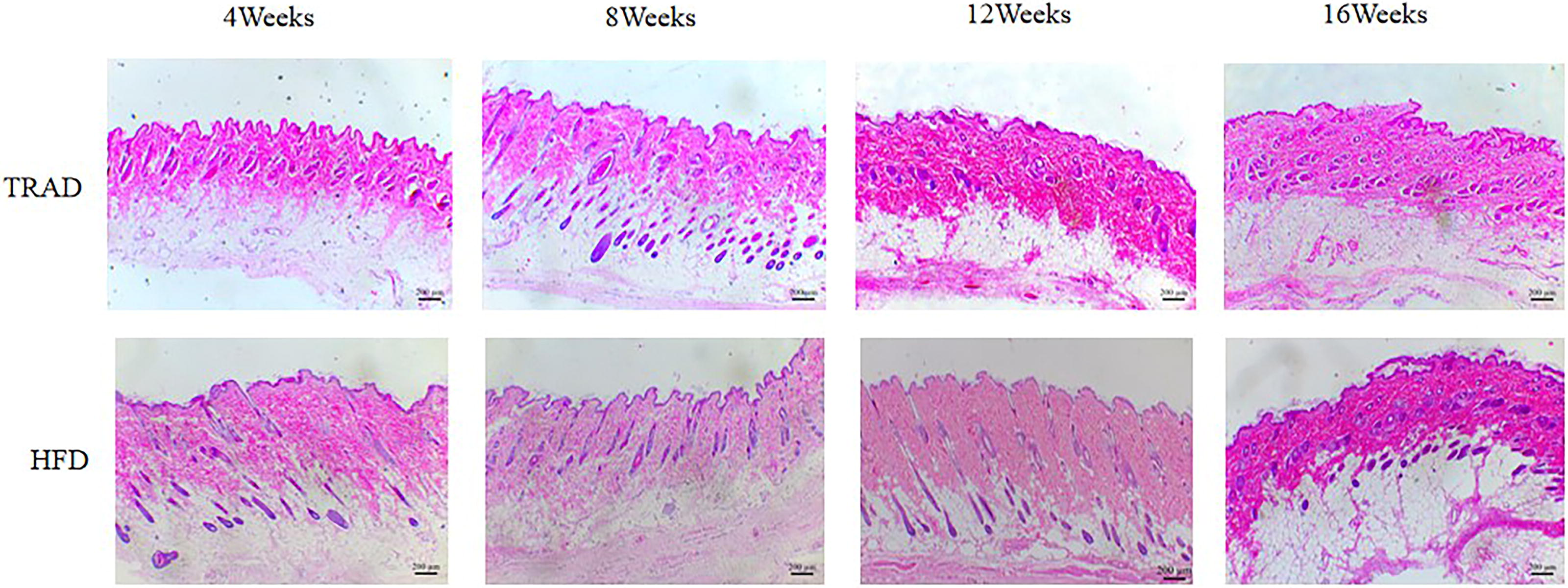
Changes of subcutaneous histomorphology with time in TRAD and HFD (40×).
The level of subcutaneous inflammation changes with time in TRAD and HFD
By measuring the expression levels of inflammatory factor expression, such as CD4 (Fig. 4A, B), IL-1β (Fig. 4C, D), and IL-6 (Fig. 4E, F), the results showed that there was a certain degree of expression of inflammatory factors in the papillary layer of dermis in both groups at the early stage of operation (4 weeks), and decreased at 8 weeks after operation. It suggested that the early inflammation caused by operation peter out within 8 weeks, and there was no significant difference between the two groups (
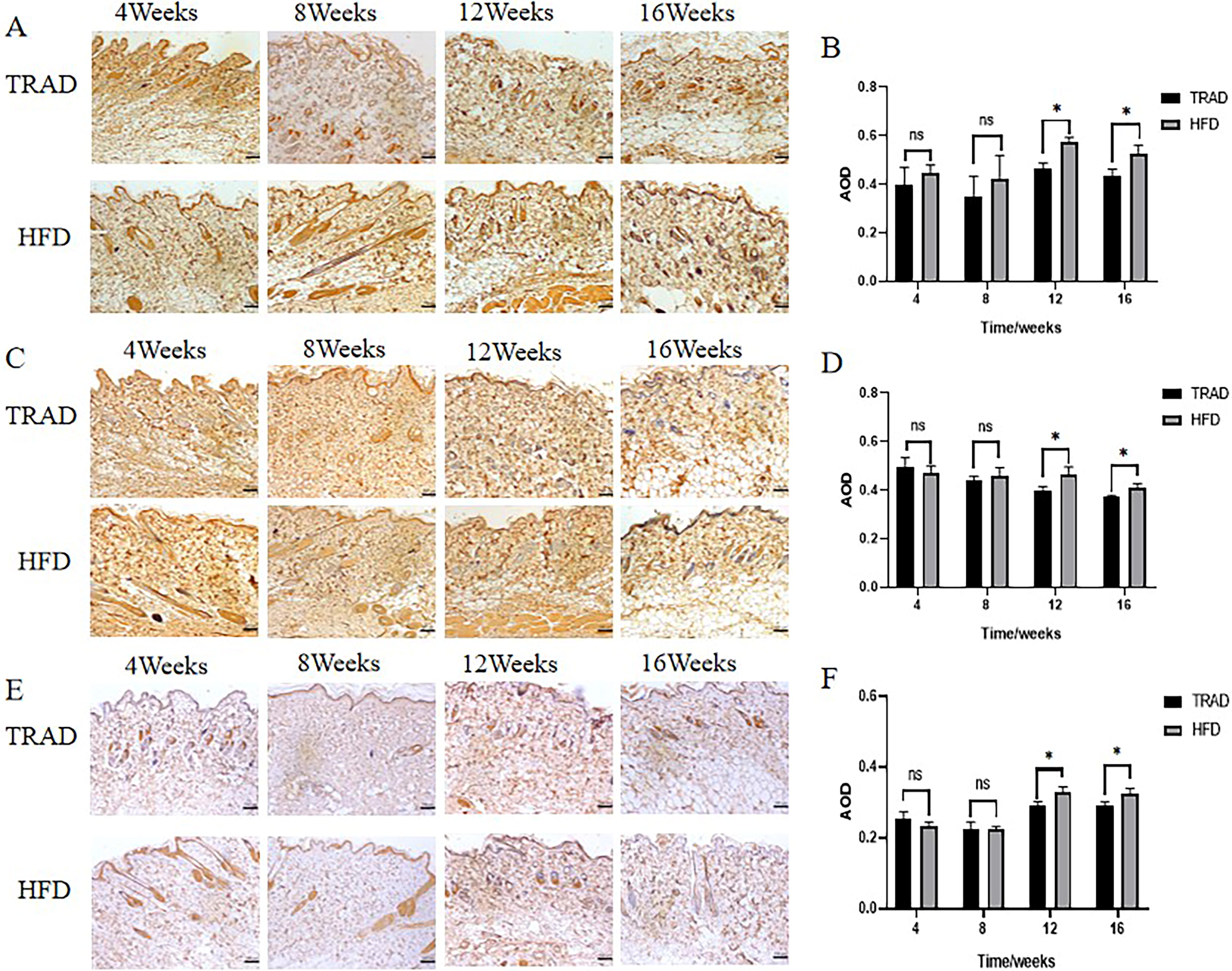
The level of subcutaneous inflammation changes with time in TRAD and HFD. Average optical density (AOD) (
Changes of subcutaneous fibrosis with time in TRAD and HFD
Masson staining showed that the subcutaneous tissue thickened significantly at 4 weeks after operation. The demarcation between the papillary layer and reticular layer of the subcutaneous dermis was still clear, and the collagen fibers were arranged neatly and in a slender shape. The collagen deposition area in HFD was significantly lower than that in TRAD (
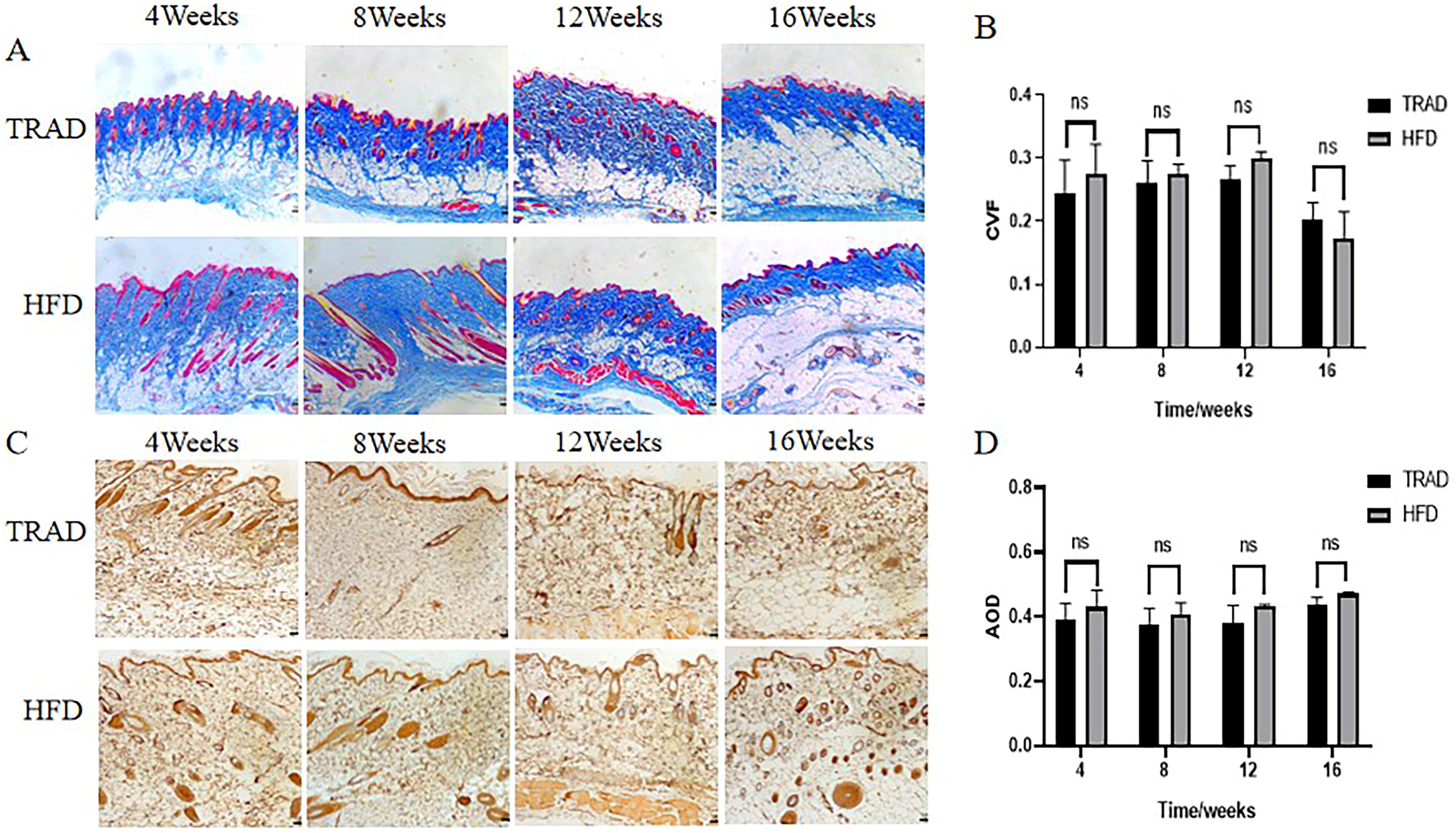
Changes of subcutaneous fibrosis with time in TRAD and HFD. Collagen volume fraction (CVF) (
IHC showed that there was a certain degree of expression of TGF-β1 in subcutaneous tissues of the two groups in the acute phase (4 weeks after operation), which decreased slightly at 8 weeks after operation, whereas the expression level of TGF-β1 in chronic phase (12 weeks after operation) increased again and could be maintained until 16 weeks after operation. There was no significant difference between the two groups, which was consistent with the results of Masson staining (Fig. 5C, D).
Discussion
Although chronic lymphedema is not a life-threatening condition, persistent arm swelling is a constant reminder that cancer may recur, harming patient’s physical and mental health. 21 There are many difficulties in the construction of lymphedema animal model, which leads to the lag of technical research in this field. Olszewski et al. 11 used canines to create the first reliable model of chronic lymphedema in 1968. Later, rabbit, rodent tails and other lymphedema models successively appeared.22,23 Due to the compensatory occurrence of subcutaneous collateral lymphatic reflux, it is difficult to maintain chronic changes for a period of time. The edema duration of animal models produced by simple lymphadenectomy usually only sustained no more than 8 weeks.24,25 Therefore, to study the mechanism and treatment of lymphedema, it is urgent to establish a new compound animal model. A competent preclinical animal model of secondary chronic lymphedema should satisfy the following requirements: interruption of lymphatic reflux, clear transition from acute to chronic phase, pathological alterations similar to human lymphedema, ease of acquisition, ease of use, and the ability to maintain the state of edema for a period of time. 26
Obesity is an independent risk factor for BCRL in human, and a prospective study showed that patients with lymphedema had a higher body mass index (BMI) than those without lymphedema. 27 The increase of immune cell infiltration, the change of inflammatory stimulation level and the alterations of endothelial barrier function caused by obesity affect the ability of lymphatic transport and contraction, which leads to pathological changes such as aggravation of inflammation, increase of fibrosis, and increase of fat deposition in the process of lymphedema. 28 To improve the incidence and sustainability of secondary chronic lymphedema in animal models, we established the first animal model of secondary lymphedema with high-fat diet combined with lymph node dissection.
Under the guidance of NIR fluorescence imaging, the high-fat-fed rats destroyed the lymphatic system of their left hind limb by microsurgical technique and dissected the lymph nodes. To eliminate the possible bias caused by a high-fat diet, we chose the contralateral limb as an autologous control. The hind limb volume, leg circumference and subcutaneous lymphatic reflux pattern of rats in each period were detected, which directly showed that the lymphedema model we constructed had an obvious transition from acute phase to chronic phase.
By detecting the subcutaneous histological changes in different stages of the model, it was found that the pathological changes at 4 weeks after operation were similar to mild lymphedema, and tissue fluid accumulation and inflammatory factor deposition began to appear at this stage, whereas there were no obvious pathological changes. Fat deposition and fibrosis began to occur in the skin tissue at 8 weeks after operation. However, the level of subcutaneous inflammation was slightly alleviated, which was similar to that of lymphedema in II stage. At 12 weeks after operation, the level of subcutaneous inflammation was significantly higher than that at 8 weeks, and severe subcutaneous fibrosis and fat deposition occurred. The pathophysiological manifestation of this stage was very similar to that of lymphedema in III stage, and the extremely high level of inflammation made skin infection easy to occur at this site. Among them, the swelling was more obvious in HFD, which may be related to the increase of subcutaneous inflammatory structure, the impairment of lymphatic transport function, and the acceleration of fat deposition caused by long-term high-fat diet.29,30
Therefore, the animal model developed in this study can show the whole process of lymphedema from acute stage to chronic stage in terms of limb swelling, lymphatic reflux pattern, and subcutaneous pathological changes, which is consistent with the pathological changes of ISL stage. 31 The subcutaneous fat deposition, the expression of inflammatory factors, and skin fibrosis at 12–16 weeks after operation are consistent with the typical pathological changes in the late stage of lymphoedema. This implies that surgery together with high-fat diet can successfully create an animal model with persistent secondary chronic lymphedema. It is suitable for the preclinical study of the vast majority of emerging diagnosis and treatment of lymphedema.
In order to investigate the role of a high-fat diet in the development of lymphedema, the skin histology of high-fat group model was compared with that of traditional diet group model. The results showed that the expression level of inflammatory cytokines in chronic phase of HFD was higher than that of TRAD, but there was no significant difference in chronic fibrosis between the two groups. Therefore, we speculate that high-fat diet may induce the progression of late lymphedema by promoting subcutaneous inflammation to damage lymphatic transport function in rats, without significantly affecting the process of fibrosis. To some extent, this is consistent with the clinical phenomenon, which also point out the reason why high BMI index postoperation patients acquired lymphedema diagnosed by dimension difference quite earlier but still had soft skin touch for over a long period of time. 32 Recent studies have shown that chronic inflammation and lymphatic stasis are associated with abnormal lipogenesis in secondary lymphedema in animal models.33,34 Another research conducted by Nitti et al. confirmed that obesity was linearly associated with lymphatic dysfunction, perilymphatic inflammation, and iNOS expression. 35 The study of Blum et al. also showed that adipose tissue dilatation caused by chronic high-fat diet impaired the function of collecting lymphatic vessels in mice. 36 This is consistent with our results, indicating that our model is reliable as a simpler, more persistent, and more stable animal model of secondary chronic lymphedema.
However, our model still has some limitations. First of all, our study only mentioned that surgery combined with high-fat diet may increase the level of skin inflammation and accelerate the progression of lymphedema. There is no in-depth study on the molecular mechanism of high-fat diet induced inflammation and inflammation-induced lymphedema. Second, due to the difficulty and long cycle of collecting skin tissues from patients with lymphedema at each stage, our study failed to evaluate skin samples from clinical patients.
Conclusion
Although our study has limitations, high-fat combined with surgery can optimize the traditional lymphedema animal model and improve the success rate and stability of lymphedema animal model by increasing the level of subcutaneous inflammation of lymphedema. Our model is simple, durable (16 weeks), reliable, compatible with histological changes, and clinical symptoms of chronic lymphedema, and does not affect the fibrosis process of lymphedema. This modified rat model may provide a basis for the future study of the possible causes of advanced lymphedema and the possibility of developing treatment.
Footnotes
Acknowledgment
The authors thank all the authors for their efforts on this work.
Ethics Approval and Consent to Participate
Sprague–Dawley rats were purchased from the Institute of Animal Models for Human Disease Genome Engineering, Dalian Medical University. All animal procedures were approved by the Experimental Animal Research Ethics Committee of Dalian Medical University. The registration number for experimental ethics is AEE20062 (2021.02.25).
Data Availability Statement
The datasets generated during or analyzed during the current study are available from the corresponding author on reasonable request.
Authors’ Contributions
All authors: Conception and design. H.Z.: Administrative support. J.L. and H.Z.: Provision of study materials or patients. All authors: Collection and assembly of data. All authors: Data analysis and interpretation. X.C. and J.S.: Article writing. All authors: Final approval of the article.
Author Disclosure Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding Information
The article preparation supported by the National Nature Science Foundation of China [award number: 82272049].
