Abstract
Background:
Our aim is to propose a framework for the development of a research case definition of lipedema, based on current available literature and those observations that can be applied to future lipedema research with the intent to standardize and strengthen the scientific evidence base.
Methods and Results:
We conducted a narrative review of the literature, and identified consensus characteristics and disputed characteristics that could be included in a research case definition of lipedema. After considering the strength of the evidence and how each characteristic might be measured in a research study, we recommended an approach for the development of a research case definition of lipedema that would be based on consideration of five agreed-upon characteristics, and five disputed, or less substantiated, characteristics as additional evidence to enhance specificity.
Conclusions:
We present a case definition framework for lipedema drawn from the scientific literature that can be applied to future studies on lipedema. Utilizing this framework should help to increase the sensitivity and specificity of case definition and provide an opportunity for meta-analysis of clinical studies and facilitate future research intercomparisons.
Background
Lipedema is a chronic disease with elements of an adipose disorder and lymphatic dysfunction with a decrease in lymph transport capacity with duration of symptoms. Lipedema affects women almost exclusively. First identified by Allen and Hines, 1 the characteristics of lipedema are poorly defined both due to the complex nature of the disorder and the lack of available preclinical investigative model systems.2–5 At the time of this writing, there is no clinically agreed-upon case definition for lipedema; concurrently, members of the research and clinical community have proposed varying approaches to ascertainment of the clinical diagnosis.6–8
Lack of consensus on a clinical definition of lipedema leads to a lack of consensus for research case definitions. This undermines the power of ongoing lipedema research.9,10 It also confounds the ability to build an evidence base in lipedema, since consistent case definitions across studies are required inclusion as case–control, cohort study, or clinical trial results in systematic reviews or meta-analyses. 11 For instance, a ketogenic diet has been hypothesized to be a potential treatment for lipedema, 9 but clinical trial results cannot be illuminative or comparable across studies without a consistent, well-characterized case definition.12,13
Our purpose is to propose a framework for the development of a research case definition of lipedema based on the current literature and authors' observations from our aggregate clinical practices and programs. The framework can be applied to future lipedema research. Because almost all lipedema cases are found in adult females,6,8,12,13 this case definition excludes children and males due to limited evidence about lipedema in these populations. Additionally, we have also excluded common comorbidities, such as simple obesity, cardiovascular diseases, diabetes, organ dysfunction, metabolic syndrome, and lymphedema, which while impacting the diagnosis and treatment of lipedema are beyond the scope of this case definition.
Methods
This framework for a research case definition of lipedema is based upon a synthesis of the characteristics of lipedema from the published body of knowledge, to build a consensus. 14 To select published articles for the synthesis, peer-reviewed publications were identified to determine the current status of lipedema research. Accordingly, a formal literature search was performed.
Search strategy and classification
The search strategy for this narrative review utilized Google Scholar (GS), as it had the greatest coverage.15,16 In a study considering 29 completed Cochrane systematic reviews, GS successfully retrieved all 738 original articles in those studies, and was recommended to be used as a comprehensive tool of identifying scientific literature on a particular topic. 16 GS search terms used to identify articles include: lipedema, lymphedema, lymphatic disorders, adipose tissue disorders, and obesity. A Zotero library was assembled, beginning in 2013, to support multiple scientific publications on lipedema. This library, which later became the property of the Lipedema Project, Inc., (LP),9,17–19 represents, in aggregate, the peer-reviewed and nonpeer-reviewed literature of adequate quality for analysis, and covers any topic relating to lipedema.
Evaluating the strength of the evidence
The following algorithm was used to evaluate the strength of the evidence. If it met all these criteria, it was classified as a “frequently agreed-upon characteristic” of lipedema. If it met only some of the criteria, it was classified as “disputed characteristic/weak evidence.”
Characteristic was reported in at least two articles authored by different author groups.
Characteristic was reported consistently over time since the publication of the original landmark articles on the topic.1,20
When the characteristic had mixed evidence, it was accepted as “frequently agreed upon” if it met either of these criteria:
(a) There was a preponderance of evidence supporting the characteristic, or
(b) There was no plausible explanation for the mixed evidence (e.g., presence of other comorbidities)
Please note that the “frequently agreed upon characteristics” may not be characteristics of all lipedema patients, but rather explain the disease entity and how it may vary in manifestation.
Results
A thorough review of the relevant literature identified eight characteristics most frequently cited, and backed by sufficient evidence, in scientific reports of lipedema. There are five characteristics that are disputed or supported only with weak evidence. These attributes are summarized in Table 1. This section enumerates the generally agreed-upon and disputed characteristics of lipedema.
Lipedema Characteristics
This is a summary of the frequently agreed-upon and disputed characteristics of lipedema arising from the peer-reviewed literature.
As shown in Table 1, frequently agreed-upon characteristics were typically female sex, disproportionate distribution of adipose tissue in limbs, onset/exacerbation associated with hormonal flux, easy bruising, heightened sensitivity/pain, limited/no response to calorie-restricted dieting or exercise regimes, unique skin and subcutaneous tissue characteristics, and ankle cuffing.7,21,22 Currently, attributes for which there is a lack of consensus, or those supported only by weak evidence, are family history and potential genetic implications, nonpitting edema coupled with negative Kaposi–Stemmer sign, onset with gynecologic or other surgery, definition of stages and evidence of the disease progression, and clinical biomarkers.23–27
Frequently agreed-upon characteristics of lipedema
Female exclusivity
Lipedema occurs almost exclusively in females.28,29 Lipedema onset strongly correlates with periods of hormone flux, for example, puberty, pregnancy, and perimenopause.30,31 To measure this characteristic, points of hormonal flux would be recorded in research studies to temporally correlate these life-instances of puberty, pregnancy, and perimenopause and the appearance and/or exacerbation of lipedema-associated symptoms or physical findings.
Distribution of adipose tissue
Many lipedema investigators have also taken note of the disproportionate distribution of adipose tissue in these patients, typically in hips, buttocks, legs, and upper arms. At diagnosis, patients consistently present with disproportionate distribution of adipose tissue with the feet characteristically spared. Schingale 32 has proposed five patterns of disproportionate adipose distribution in lipedema (Fig. 1).
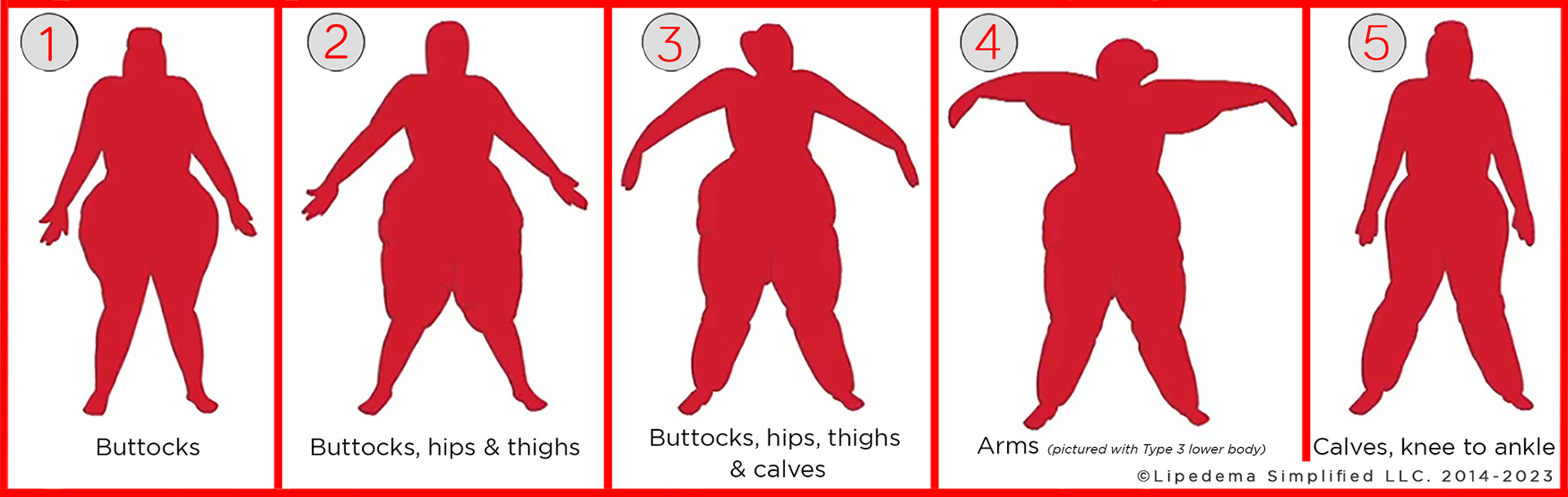
Types of lipedema (1 through 5). The types are classified according to the location of impacted adipose tissue. Type 1 shows only buttocks affected, type 2 includes buttocks as well as hips and thighs, type 3 involves all these areas plus the calves, type 4 lipedema involves the arms and is the only type that can co-occur with other types and finally, type 5 shows the calves as most severely affected. Images of courtesy Lipedema Simplified LLC. Used with permission.
As is shown in Figure 1, lipedema can be classified as Type 1, 2, 3, 4, or 5, based upon the adipose tissue distribution by body area. To classify lipedema type, the body silhouette can be visually inspected and compared with Figure 1. Type 1 involves only the buttocks, Type 2 presents in the hips and thighs as well as the buttocks, and Type 3 includes all of these plus the calves. Type 4 involves the arms extending to the wrist, with upper arms generally more severely impacted, occurring in conjunction with any of the other types (pictured with Type 3). 29 Finally, Type 5 most severely impacts the calves, although the rest of the lower body may be affected to a lesser extent.
Easy bruising
Another commonly reported attribute of lipedema is easy bruising and, specifically, frequent appearance of ecchymoses from minimal or no trauma in areas affected by lipedema.12,33 This can be assessed by a focused scrutiny of the patient's history, such as asking if they have a problem with bruising. If the answer is yes, then ask the patient to describe in more detail where in the body the bruising occurs, if there was a provoking stimulus, as well as how often this has occurred over a recent time period (e.g., past month, past 3 months). Because easy bruising is not exclusive to lipedema, it is also important to identify whether any reported easy bruising is primarily in tissue that appears to be affected by lipedema (hips, buttocks, thighs, calves, and/or arms).
Pain and/or heightened sensitivity
Additionally, the presence of moderate-to-severe pain and/or heightened sensitivity in body areas affected by lipedema, characterized as a pressure-induced and/or spontaneous burning, sharp, or stabbing ache, 34 is frequently noted in the literature,35,36 with even light pressure having been reported as a trigger for discomfort. The presence of tenderness to palpation can be confirmed upon palpation of fat in an area affected by lipedema, such as the thigh or upper arm, with verbal confirmation from the patient regarding the presence and severity of resulting sensation.
In our clinical experience, some patients with lipedema have altered recognition of pain, often believing their pain or heightened sensitivity to be a normal sensation or even that their pain is inconsequential. Additional common patient descriptors of heightened sensitivity include feeling swollen, congested, uncomfortable, and/or heavy. A demonstration of heightened sensitivity may be elicited using a provocative maneuver, such as touching, or application of a blood pressure cuff. Further questioning and the use of various self-report surveys and other diagnostic testing may be required to more accurately quantify and qualify any pain experienced.37,38
Because certain treatment interventions can influence pain patterns,39–41 the described pain may vary in intensity or frequency depending on the efficacy of the treatment modality. Additionally, patients may not realize that prior pain patterns had been linked to lipedema, especially if intervention strategies have effectively managed the pain for an extended time period. Careful questioning about a patient's pain history may be required.
Ineffective weight loss attempts
Traditional weight loss interventions may prove to be ineffective for lipedema patients, who consistently experience limited response to caloric restriction and exercise in body areas affected by lipedema.4,7 Several case reports document lipedema patients misdiagnosed with obesity; these patients, having undergone bariatric surgery experience fat mass reduction primarily in the chest and abdomen, with limited effects upon lower body fat mass.42–44 In our clinical experience, even when weight loss is achieved, the disproportion between upper and lower body does not resolve and symptoms do not improve. In cases of disproportionate reduction of the fat mass, the patient may present with a waist circumference appropriate to height, while simultaneously being classified as overweight or obese as measured by body mass index (BMI) or body composition scans. A medical history should include questions about a lipedema patient's past experiences with any dieting regimens and/or weight loss surgeries to quantify this phenomenon.
Distinctive tissue changes
Distinctive skin and tissue changes have been documented; the resulting textures are thought to be diagnostic of lipedema.45–47 Figure 2 shows three general locations where these skin tissue changes should be evaluated.
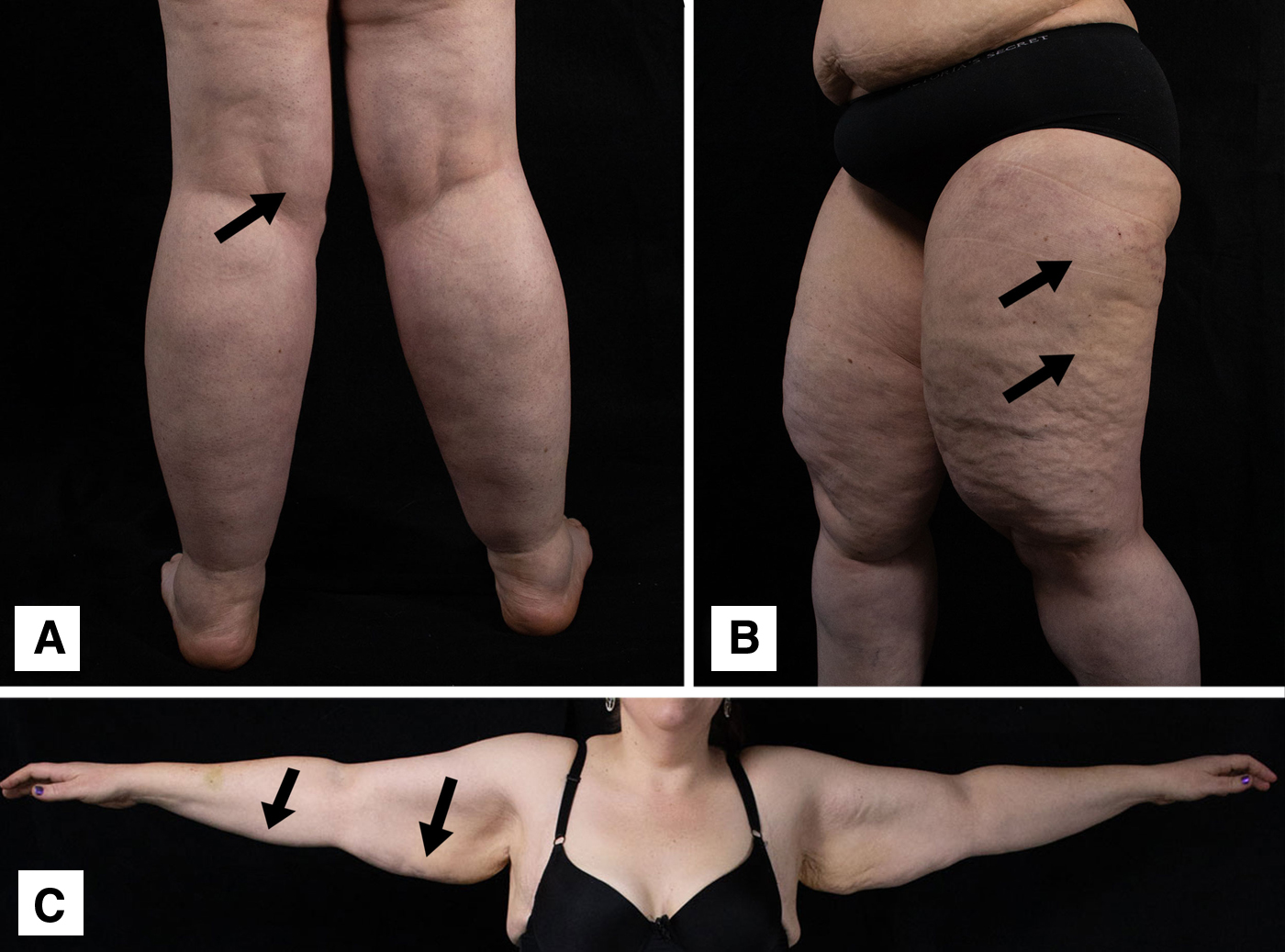
Areas of evaluation to determine location and severity of lipedema tissue changes. Arrows indicate the exact location of the examination area. Complementary sides of the body (e.g., arms, legs) should be evaluated to confirm the presence of bilateral tissue changes.
First, identified as A in Figure 2, the fat pad at the posterior medial knee should be examined by gently pressing the examiner's fingers into the tissue, and moving the length of the fingers in a circular motion. Next, skin and tissue of the thigh, especially the anterolateral aspect (labeled as B in Fig. 2), is evaluated by pressing the flat of the examiner's fingers, or deeper palpation, if necessary, by pressing with the pads of the examiner's fingers. Third, labeled C in Figure 2, the lower two inches of the subcutaneous fat of both the proximal and distal arm should be evaluated by rolling the subcutaneous adipose between the index and middle finger, and the thumb. Complementary sides of the body (e.g., arms, legs) should be evaluated to confirm the presence of bilateral tissue changes.
During examination, palpation should be performed for the purpose of classifying the subcutaneous adipose tissue in each area into one of three categories. The first category, normal fatty tissue (unaffected tissue), is characterized as smooth, firm, and resilient, without dense fibrotic texture, nodularity, or granularity. In the second category, representing mildly affected tissue, the tissue feels soft, spongy, with fluid density and/or unusually loose, and may have a granular texture, reflecting the presence of lentil- or chickpea-sized nodules within the subcutaneous adipose tissue. Finally, in the third category, representing severely affected tissue, the consistency is tough, matted, and not very pliable, with the presence of larger nodules that may be walnut-, plum-, or apple-sized. In our clinical experience, nodules palpated specifically in the thigh and knee 48 and upper arm 24 may not be as readily identified in all patients with advanced lipedema due to pathological involvement of the surrounding tissue. This may occur in later stages; thus, care should be taken to note overall tissue texture.
Later, we discuss the concept of staging lipedema into three stages; in this staging schema, the second category of tissue would be considered stage 1 or 2 lipedema, and the third category stage 3 lipedema, respectively.4,9,17
Cuffing
Finally, “inverse shouldering,” now commonly known as “cuffing,” at the ankles was first observed by Harwood 21 as a notable, and potentially diagnostic, feature in lipedema, described by other observers in both the ankles and wrists.2,7,22 Ankle cuffing is more prominent with Types 2 and 3 while wrist cuffing is more likely with Type 4 (as shown in Fig. 1). Cuffing is discernible as the sudden transition from excessive adipose buildup to unaffected tissue (as shown in Fig. 3).
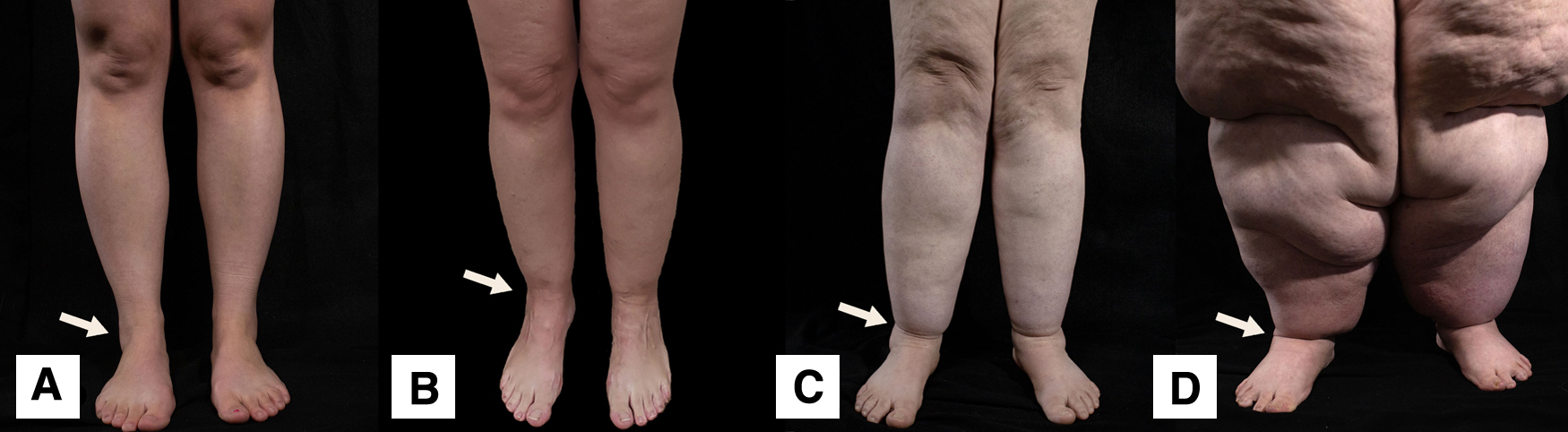
Examples of cuffing at the ankles are shown by arrows. Classifying positive or negative cuff sign should be done on the basis of the extent of inverse shouldering visible at the ankle.
As shown in Figure 3, ankles are visually inspected to classify patients as having a positive or negative ankle cuff sign. A negative cuff sign is considered present when the transition from calf to foot is smooth, with no abrupt difference in adipose deposition at the ankle. Figure 3A depicts a negative cuff sign due to lack of visible lipedema tissue in the calves; conversely 3B indicates a positive cuff sign with a mild presentation, evident by disproportionate deposition of adipose in the calves that ends abruptly at the ankle, resulting in a subtle extension of tissue at the transition point that is still smooth. Figure 3C shows a moderate presentation, where disproportionate adipose deposition in the calves is more prominent, causing a distinct ridge of adipose that extends significantly from the ankle. Finally, Figure 3D shows severe cuffing indicated by extreme disproportionate adipose deposition in the calves, resulting in a fold comprising adipose and skin at the transition point, completely obscuring the ankle.
Cuffing may not be present in all individuals with lipedema. Thus, the absence of a cuff sign would not preclude a lipedema diagnosis. For instance, ankle cuffing would not occur in those individuals with lipedema Types 1 or 2 (Fig. 1), as the affected tissue would not extend to the ankle.
Disputed characteristics of lipedema
In this section, we present characteristics of lipedema that are listed in Table 1 as disputed or having weak supporting evidence.
Family history
Family history is elicited to identify any first-, second-, or third-degree relatives who also present with features of lipedema, primarily subcutaneous fat distribution.21,49 In a study conducted by author Forner-Cordero, 85% of the patients reported a family history of lipedema, although this may have been a result of selection bias. 50 Intake interviews with study subjects should include questions about family members who may have exhibited symptoms of lipedema, descriptions of those symptoms, and timing of onset/severity. For example, a family history question might be: “Does your body look like someone in your family or a blood relative?”
Genetics
A genetic substrate for lipedema is under active investigation. 50 Although promising, no particular genetic pattern has yet been substantiated as being unequivocally linked to lipedema.51,52 To serve the research community, investigators may choose to include genetic panels in lipedema research, indicating how specific panels are chosen, the putative mechanism, and the potential relationship of gene expression profiles to the disease pathology.
Edema
Edema can be present for many reasons in both lipedema and nonlipedema patients and may be easily identified by visual inspection and palpation. If pronounced, the presence of edema may be characterized by the appearance of taut, shiny skin in the locations described in Figure 2, as well as the lower legs. Circumferential measurement of enlarged tissue may be used to identify changes in girth over a short period of time (i.e., several days). Not all edema, especially mild edema, can be objectively documented on physical examination, therefore the historical presence of edema can be supported by patient complaints of a transient sensation of tightness, fullness, and heaviness in the body areas affected by lipedema, and, more accurately, confirmed by lymphatic and tissue imaging.46,53–55
Edema is described as “pitting” if a depression is left in the tissue after pressure has been applied. Nonpitting edema is conversely defined as swollen tissue that lacks this characteristic. 56 Lipedema patients with edema have been found to display both pitting and nonpitting edema. Although nonpitting edema was one of the first characteristics ascribed to lipedema and proposed as a diagnostic characteristic, 7 the inclusion of this feature has been more recently questioned. This is because not all patients with lipedema have been observed to have nonpitting edema, and because the patients in which edema has been identified have had comorbidities that may have been responsible.13,57,58
Figure 4 demonstrates testing for nonpitting edema in the lower leg, because this is the most typical location if present. As shown in panels A through D of Figure 4, demonstration of pitting edema in the lower leg is accomplished by having the clinician apply and hold pressure to the edematous region, typically 5–10 cm above the medial malleolus, as well as on the dorsum of the foot. Pressure from the flat of the examiner's thumb should be sustained for a minimum of 10 seconds but up to 30 or more seconds may be necessary in denser tissue. As shown in panel C and D in Figure 4, pitting edema provides a persistent impression after pressure is released. Conversely, as shown in panels A and B in Figure 4, in nonpitting edema, no persistent impression remains, so differentiating pitting versus nonpitting edema can easily be established clinically. 56
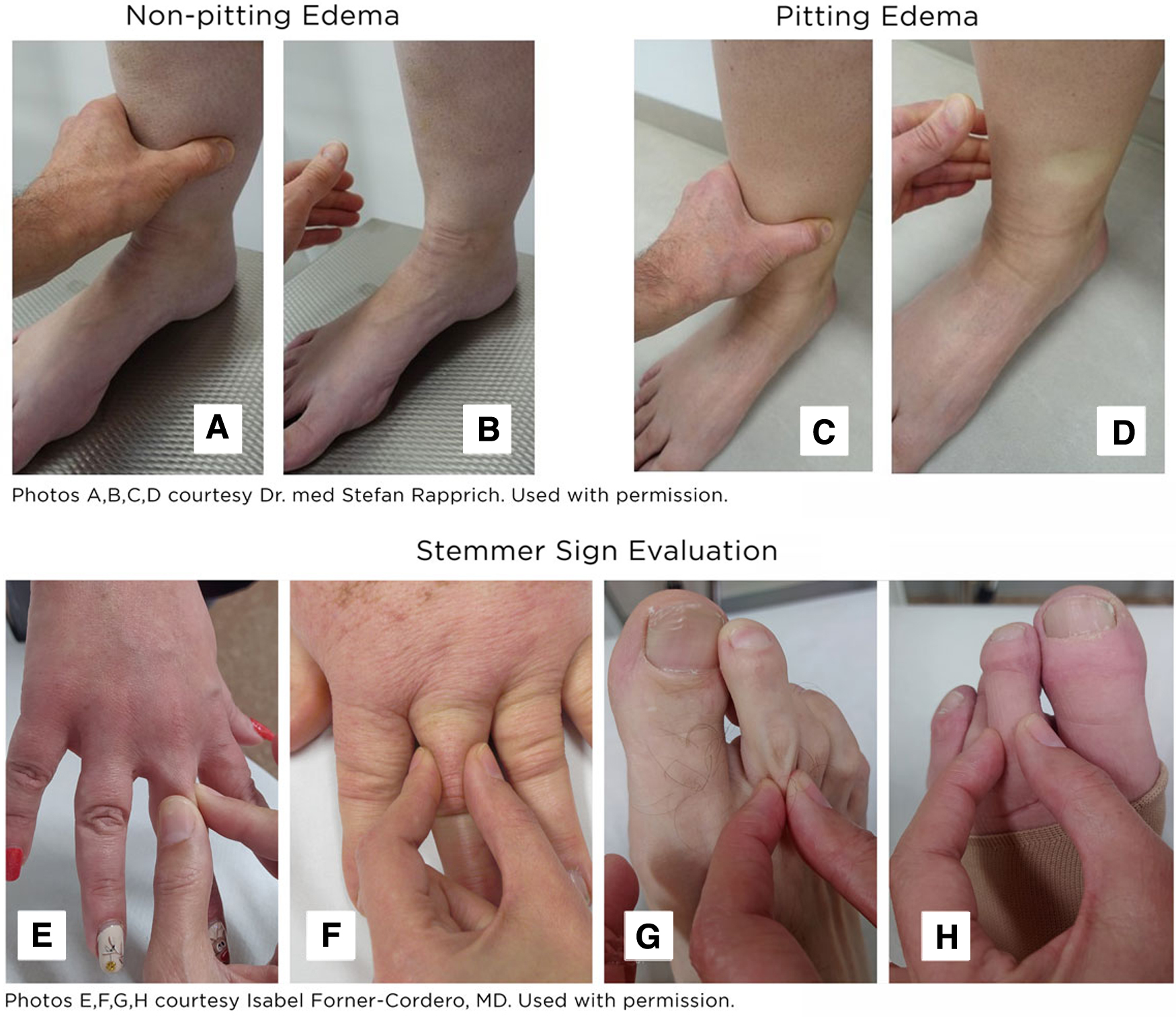
Pitting and nonpitting edema evaluation and Kaposi–Stemmer test.
Although not common, edema can also be present in the hands and feet in lipedema when accompanied with lymphedema, and this can be evaluated with a Kaposi–Stemmer sign. As shown in panels E through H of Figure 4, a Kaposi–Stemmer sign is an evaluation of how easily skin on the dorsum of the proximal end of the second digit of the hand and foot can be pinched and lifted. 56 As shown in panels E and G of Figure 4, skin that is easily lifted suggests a lack of skin or tissue changes and results in a negative Kaposi–Stemmer sign. Because the natural history that produces lower body nonpitting edema should theoretically also result in a negative Kaposi–Stemmer sign, some clinicians believe a negative Kaposi–Stemmer test coupled with nonpitting edema deserves further examination as a potential characteristic of lipedema.7,48
Whether or not edema can be seen in all lipedema patients remains controversial. However, recent studies using MR-L, near-infrared fluorescence lymphatic imaging and/or tissue biopsy have detected lymphatic anomalies as well as the presence of edema and increased fluid load in patients diagnosed with lipedema.46,53–55 This suggests that evidence of a specific type of edema in lipedema patients might be found reliably in at least a subset of patients if measured in a standardized way. Thus, this criterion might be adopted in the future if advanced technology (MR-L) was widely available to all.
Gynecological surgery
Next, a small subset of articles in lipedema point to gynecological surgery (including hysterectomy, Cesarean section), cholecystectomy, various bariatric surgeries, or other abdominal surgery as the triggering factor for the condition, but this has not been substantiated in other observations.59,60 Research should seek to clarify possible surgical induction of lipedema symptoms, utilizing a thorough review of medical and surgical history alongside questions regarding onset or exacerbation of lipedema symptoms.
Staging
Lipedema is conventionally characterized by many clinical observers through staging on a continuum of mild to severe.4,9,17 Although the staging system presented in this study accurately describes the agreed-upon distribution of adipose tissue, whether or not the stages exist and are characterized properly is still under dispute. 61 We subscribe to a description of staging, because we observe that evidence of severity can be assessed and documented by following the patient over time. 22 Others suggest that lipedema is not a progressive disease, and therefore staging is not applicable. It should be understood, however, that staging is purely morphological and not necessarily linked to symptom severity or to the duration of the disease. Additionally, the wide variation in presentation of lipedema renders making distinctive demarcations among stages challenging.
Staging of lipedema initially relies upon a visual inspection of the body silhouette to evaluate the presence and severity of adipose disproportion, cuffing, skin dimpling or mattress-like appearance to the skin, and symmetry of the limbs. Visual inspection is followed by an examination of the tissue as described in Figure 2 and tissue is categorized according to the staging system. Edema should be evaluated, and Kaposi–Stemmer sign should be elicited, if present, as shown in Figure 4. Upon completion of visual and tissue examination, the participant should be staged according to the model shown in Figure 5.
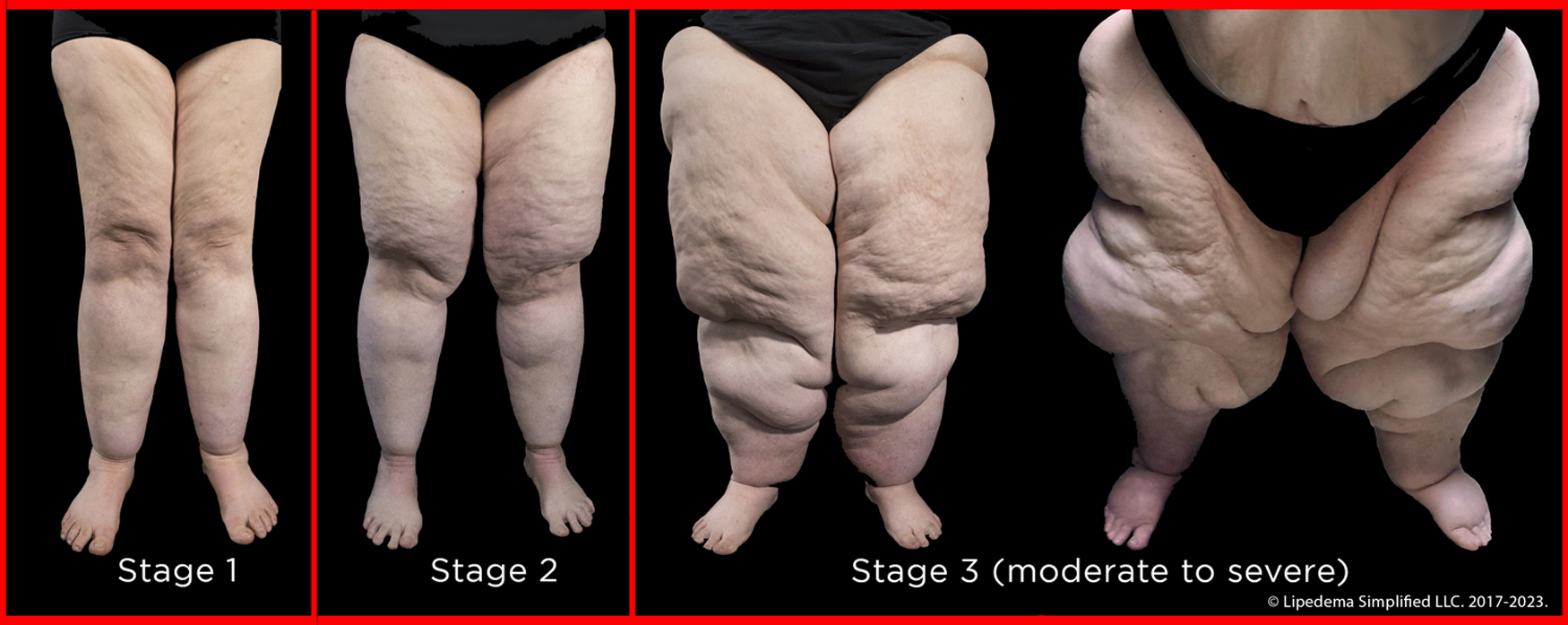
The three stages of lipedema. Although staging is disputed, those who agree observe that Stage 1 is a mild presentation, where skin changes are minimal, while Stage 2 involves more mattress-like appearance of the skin. In Stage 3, the tissue changes and level of disproportion in tissue distribution can range from moderate to severe. In the more severe presentations of Stage 3, pitting edema may be present, and the body displays severe asymmetrical disproportion, with large skin and tissue extensions resulting in protruding folds of adipose tissue. Images courtesy of Lipedema Simplified LLC. Used with permission.
As shown in Figure 5, Stage 1 shows a symmetrical distribution of the adipose tissue with smooth or mildly dimpled skin, and mild changes to the underlying tissue identified upon palpation. Feet and hands are typically unaffected, resulting in a negative Kaposi–Stemmer sign. If cuffing is present, it is typically mild. In Stage 2, the symmetrical disproportion of subcutaneous adipose tissue increases, with a dimpling appearance to the skin, and moderate changes to underlying tissue identified upon palpation.
Stage 3, as shown in Figure 5, is characterized by more pronounced changes that may present in a wide range in the degree of severity. Evident symmetrical disproportion of the adipose tissue with protrusions, mattress-like appearance to the skin, and extensions of the skin, with tissue lobules potentially overlapping the knees, can be observed. Erythema may be present in the distal calves, causing reddening or darkening of the skin. Pitting edema may be present in the dorsum of the feet, resulting in a positive Kaposi–Stemmer sign.
The more severe presentations of Stage 3 lipedema are often characterized by asymmetrical disproportion in the lower body that is exacerbated by the presence of lymphedema. Large skin and tissue extensions can appear as protruding folds of adipose tissue or lobules, particularly over the knees and medial thighs, and hips may have a “saddlebag” appearance. 62 The affected skin has a broadly dimpled appearance and severe changes in the tissue architecture of skin and underlying tissues are present and detectable through palpation. If cuffing is present, it may be severe. Many patients in advanced Stage 3 lipedema report a severe restriction in mobility and impaired quality of life. 62
Circulating biomarkers
Finally, very little is known about how circulating biomarkers are impacted in lipedema patients, or the effect of the disease upon tissue histopathology. Historically, studies examining blood and/or tissue in lipedema patients have had small sample sizes and have suffered from a lack of comprehensive standardized reporting. Table 2 provides a greatly reduced overview of laboratory studies in lipedema.
Laboratory Studies on Lipedema
This is a summary of laboratory investigations reported in the peer-reviewed literature. Evidence is considered “weak” if study results conflict. Evidence is labeled “moderate” if we found study results pointed in a similar direction and could potentially be used for hypothesis generation.
As shown in Table 2, evidence that correlates laboratory or tissue studies to the diagnosis of lipedema is not straightforward and results may often be conflicting.23–27,40,63–74 In Table 2, the following criteria were used to evaluate the strength of evidence as weak, moderate, or strong. Evidence was considered “weak” if multiple publications were found in direct contradiction with each other (e.g., association found between elevated or decreased level of biomarker in one study but no such association found in any others). If multiple publications reported similar trends in different but small samples, the evidence was considered “moderate.” Lastly, evidence was considered “strong” if multiple publications with large sample sizes with clearly defined exclusion and inclusion criteria found similar trends. We are specifically referring to studies with epidemiologic study designs, such as cross-sectional, cohort, case–control, or clinical trial. However, no studies fit the criteria for strong evidence.
In Table 2, laboratory studies of lipedema were classified into 11 broad categories and reviewed in terms of the strength of the supporting evidence. As can be seen in the few studies that have been done, evidence is either conflicting or weakly points in the direction of the hypothesis. For this reason, clinical laboratory or tissue diagnostic techniques do not materially add to the assessment of a putative lipedema diagnosis.
In the clinical investigation of lipedema, utilization of laboratory resources can be supported with several recommendations. First, it is important to provide a description of the methodology for specimen collection and processing, the testing protocol, and how the resulting data are interpreted. Second, a clear rationale for the utilization of the laboratory approach should be provided. Third, when reporting case series of lipedema patients (where n ≤ 30), it is most helpful to report individual results in addition to population statistics to further illuminate the interpretation of the data. 75 Reporting contextual information, such as stage, BMI, weight-to-height ratio, metabolic health, inflammatory status, and current symptom severity for each individual, can enrich learning about the dataset.
Results
As a result of our review, we propose the research case definition framework for lipedema described in Table 3.
Proposed Case Definition Framework for Lipedema in Research
This table provides a framework for developing case definitions for lipedema to be utilized in research. Those with an X in the “agreed upon” column are characteristics that can be endorsed for inclusion in every research case definition of lipedema using this framework, and an X in the “less substantiated” column indicates these characteristics should be measured in all research studies using this case definition framework. X* Possessing the characteristic of “limited response to dieting” greatly increases the specificity of the classification, but if participant has no previous dieting history, it cannot be included in the case definition. X** Whereas a genetic testing panel is not yet agreed upon for lipedema, there are numerous reports of family clusters, and a genetic predisposition can be inferred.
Theoretically, the best-case definition will optimize both the sensitivity and specificity of the classification of lipedema and will need to be tailored to the research setting (e.g., in-person clinical research vs. anonymous surveys of patient groups). Abiding by this framework at the design stage when crafting a case definition for studies on lipedema will provide the foundation for clinical trials for lipedema treatments, as well as enable the scientific evidence to be aggregated in future systematic reviews and meta-analyses.
As shown in Table 3, there are 10 characteristics included in the framework. Each of the 10 characteristics is designated as “Agreed Upon” for the case definition framework, or as “Less Substantiated” and can be used for additional evidence. Five characteristics are identified as “agreed upon” and should be included in the case definition of lipedema in research studies using this framework: female sex, pain and/or heightened sensitivity with or without pressure, disproportionate distribution of adipose tissue, skin and tissue changes, and limited or no response to caloric restriction and/or exercise regimes. While the first four characteristics are relatively easy to measure for the purposes of classification and values should be available for all participants, response to dieting and exercise may not be known. Availability of this characteristic may vary, for example, by the median age of the study population. However, should this characteristic be omitted from the case definition, the specificity of the case definition will be reduced.
Three of the less substantiated characteristics are recommended for consideration of inclusion if it is felt these can improve specificity, given the particular research setting. However, all research studies using this case definition framework should seek to measure these disputed characteristics, whether they are included in the case definition or not.
The first characteristic that should be considered is cuffing, since participants who otherwise meet criteria and also have a positive cuff sign should qualify for inclusion. The rationale for not requiring a positive cuff sign in a case definition is that it would reduce sensitivity because many lipedema patients do not have cuffing. The second characteristic that can be added as additional evidence to improve specificity is nonpitting edema coupled with a negative Kaposi–Stemmer sign. Again, this should not be a required component of the case definition, as not all lipedema patients have this characteristic. Finally, the addition of the characteristic of onset or exacerbation of lipedema symptoms during period of hormonal flux could be made, as this is commonly reported in lipedema, but should not be required, as many lipedema patients do not report this characteristic.
While including these additional characteristics may introduce a lack of homogeneity in the sample, it is still possible to stratify by these characteristics in the analysis, so their inclusion can be defended and described.
While repeatedly reported in patient cohorts, we feel two other additional characteristics, easy bruising and family history of lipedema, cannot be reasonably added to the list of agreed-upon characteristics in a lipedema research case definition at this time. Due to a lack of consistent presentation and a limited number of studies that evaluate objective criteria for capillary fragility and/or family history, there is the risk of false negativity leading effectively to participant exclusion unnecessarily. To build this evidence, however, it is imperative that accurate data collection related to these characteristics be undertaken and reported.
Finally, several disputed or less substantiated characteristics, including genetics, exacerbation of symptoms associated with gynecologic surgery, staging/evidence of progression, and laboratory biomarkers continue to be of research interest, but the current evidence is not strong enough to include them in a research case definition of lipedema. Ancillary data collection of these variables will help to build the necessary evidence base for future revisions of the framework, leading to potential future inclusion in the research case definition.
Discussion
After undertaking a thorough review of the published lipedema literature, it would appear that the evidence base for lipedema diagnosis and therapeutics is limited. Many of the reviewed publications were either case studies or small case series. Additionally, there was a general lack of detail regarding the inclusion and exclusion criteria such that bias could not be assessed. Assessment of bias became particularly important in the context of the several publications that represented convenience sampling from a single clinic. Only those publications that were deemed appropriate were included in this analysis. This approach might be interpreted as “purposive sampling” with only one criterion. 14
Our analysis has resulted in a literature-supported, flexible framework for developing a research case definition of lipedema in adult women that can be applied to current and future research and can help guide the strengthening of evidence behind lipedema research case definitions in general. This proposal considers not only the level of consensus on characteristics observed and reported in the literature, but also recommends practical methods of measurement for each, to facilitate accurate classification and reporting. Adoption of this framework entails a heightened level of standardization with respect to the data collected in lipedema research studies. In addition to standardizing variables and measurements, those applying our case definition in research should also seek to better characterize the patients' natural history of lipedema symptoms, comorbidities, and symptomatology. Better understanding the temporality of these presentations should foster a web of causation with respect to natural history.
We describe in Table 4 a practical application of this research case definition framework in order to facilitate the acceptance of participants into a lipedema research study.
Application of Proposed Case Definition Framework for Participant Selection Criteria
We suggest that a patient can enter as study subject if one of the following three pathways are present: (1) all five Agreed-Upon Characteristics are present, (2) four of the five Agreed-Upon Characteristics (two of which must be “Female” and “Pain and/or heightened sensitivity - current or historical”) are present and at least two characteristics from the Additional Evidence column are present, or (3) three of the five Agreed-Upon Characteristics are present (two of which must be “Female” and “Pain and/or heightened sensitivity - current or historical”) along with four characteristics in the Additional Evidence column.
We suggest three possible pathways for inclusion in a lipedema research study to increase specificity while still maintaining sensitivity. Inclusion is obligatory if all five agreed upon characteristics are present, including female sex, pain/heightened sensitivity (current or historical), disproportionate deposition of adipose tissue, skin/tissue changes, and nonresponsiveness to diet.
When all agreed upon characteristics are not present, two alternatives are suggested for study inclusion. All alternative options mandate that the study participant be biologically female and have pain and/or heightened sensitivity (current or historical), for homogeneity. One alternative pathway for inclusion requires only four out of the five agreed-upon characteristics be present, along with at least two of the five additional evidence characteristics (wrist/ankle cuffing, nonpitting edema with negative Kaposi–Stemmer sign, onset/exacerbation with hormonal flux, family history, and easy bruising). This option will capture lipedema patients who may no longer exhibit disproportionate adipose tissue deposition due to a comorbidity of obesity, yet present clinically with those characteristics that most contribute to increased specificity. The final option for study inclusion requires a combination of at least three agreed-upon characteristics along with four of the five characteristics in the Additional Evidence column. This option may be best in capturing those early-stage patients who do not exhibit skin and tissue changes and who have not established a dieting history.
Using this research case definition framework provides new opportunities in framing lipedema research. Subpopulations can be targeted based on type and/or stage, and inclusion and exclusion criteria can be accordingly established. This strategy could be used to standardize lipedema screening of young women to facilitate lipedema diagnosis at earlier time points in its natural history. Earlier diagnosis of patients would facilitate natural history investigations and might help to characterize the incidence and timing of relevant comorbidities (such as comorbid osteoarthritis). This ability would contribute to improving the strength of evidence behind the proposed staging system, and to resolving the mechanistic complexity that characterizes many of the reported comorbidities associated with lipedema diagnosis.
Limitations
One limitation of our framework is that it does not permit inclusion of all the characteristics that might be important and should be the subject of future research. Onset or exacerbation of symptoms coinciding with times of heightened physical or psychological stress has been noted frequently in the literature but evidence for a causal link between stress and onset or exacerbation of lipedema symptoms is limited. 12 This is an important area for future research to help clinicians more accurately identify potential triggers of lipedema onset or exacerbation. Continued investigation of the genetic substrate of lipedema will continue to be important but it is not feasible to focus upon individual causal genes. As many research groups propose, there are a variety of biological processes involved in lipedema.3,9,73,76
Because our process involved evaluation of existing research, the strength of the framework is limited by the strength of the available evidence. For instance, because many publications regarding lipedema suffer from limitations, including small sample sizes, unclear inclusion and exclusion criteria, lack of an epidemiological study design, and missing information on natural progression, future research may result in a need to reevaluate our proposed framework.
Furthermore, the lack of a pathognomonic test makes it impossible to validate this framework. Additionally, the framework is limited to the adult female population due to a lack of research on younger populations and males. If research on these populations becomes available, then additional frameworks would be necessary. Moreover, not all publications included a discussion of every possible characteristic of lipedema; therefore, our framework may suffer from information bias. Lastly, although efforts were made to include differing perspectives on lipedema, the possibility of researcher bias always exists. In an effort to combat this, further discussion and critique of our framework is encouraged.
In conclusion, we present this framework for a research case definition of lipedema based on our literature review and observations. We provide practical guidance on how to apply it to develop research case definitions for future lipedema studies. Our hope is that our framework provides a structure around which to build future scientific evidence in lipedema to strengthen the entire body of scientific work related to this complex disease.
Footnotes
Authors' Contributions
L.K., C.S., M.M.W., S.H., and M.C.: Drafted original article, contributed to the design of the article, compiled research for article, developed figures and tables, and approved the final version.
G.F., I.F.-C., S.M., S.F., and S.G.R.: Provided scientific review and advice, contributed to figures and tables, guided scientific decisions about presentation of information, edited and revised the article, recommended sources, and approved the final version.
Ethics Statement
As this article represents a review of published literature, this article is exempt from ethical review. All photographs in this article are used with permission.
Author Disclosure Statement
The authors have no conflicts of interest with respect to this article.
Funding Information
No funding was received for this article.
