Abstract
Background:
Lymphedema is chronic limb swelling resulting from lymphatic dysfunction. It affects an estimated five million Americans. There is no cure for this disease. Assessing lymphatic growth is essential in developing novel therapeutics. Intravital microscopy (IVM) is a powerful imaging tool for investigating various biological processes in live animals. Tissue nanotransfection technology (TNT) facilitates a direct, transcutaneous nonviral vector gene delivery using a chip with nanochannel poration in a rapid (<100 ms) focused electric field. TNT was used in this study to deliver the genetic cargo in the murine tail lymphedema to assess the lymphangiogenesis. The purpose of this study is to experimentally evaluate the applicability of IVM to visualize and quantify lymphatics in the live mice model.
Methods and Results:
The murine tail model of lymphedema was utilized. TNT was applied to the murine tail (day 0) directly at the surgical site with genetic cargo loaded into the TNT reservoir: TNTpCMV6 group receives pCMV6 (expression vector backbone alone) (n = 6); TNT Prox1 group receives pCMV6-Prox1 (n = 6). Lymphatic vessels (fluorescein isothiocyanate [FITC]-dextran stained) and lymphatic branch points (indicating lymphangiogenesis) were analyzed with the confocal/multiphoton microscope. The experimental group TNT Prox1 exhibited reduced postsurgical tail lymphedema and increased lymphatic distribution compared to TNTpCMV6 group. More lymphatic branching points (>3-fold) were observed at the TNT site in TNT Prox1 group.
Conclusions:
This study demonstrates a novel, powerful imaging tool for investigating lymphatic vessels in live murine tail model of lymphedema. IVM can be utilized for functional assessment of lymphatics and visualization of lymphangiogenesis following gene-based therapy.
Introduction
Lymphedema is characterized by limb swelling due to lymphatic dysfunction. It affects 30% of patients who undergo axillary lymph node dissection in the treatment of breast cancer. 1 Lymphedema is treated with compression therapy, liposuction, and microsurgical intervention (lymphovenous bypass and vascularized lymph node transfer).2–13 These therapies show variable success rates with no cure. 14 Experimental models have attempted to address the gap in the management for lymphedema using gene-based strategies and anti-inflammatory therapies.15–19 Lymphatic visualization in experimental preclinical models is critical for the evaluation of novel lymphedema therapies. Lymphangiogenesis is prominent in many disease processes and pathological conditions. Assessment of lymphangiogenesis is essential to the discovery of therapeutic targets.
The murine tail lymphedema model is a well-established model for lymphatic research.17,20–22 This model caused sustained tail edema postsurgery resulting in histological changes consistent with lymphedema, including chronic inflammation, fibrosis, and fat deposition.21,23 We have optimized and validated gene delivery in murine tail lymphedema model using tissue nanotransfection technology (TNT).24,25 In our recent study, TNT-delivered Prox1 prophylactically prevented mice tail lymphedema and preserves lymphatic function. 26
Advanced imaging techniques have increased the understanding of vascular contributions to disease processes and effects of various vessel-targeting therapies. 27 Multiphoton laser scanning microscopy allows highly sensitive imaging of small vessels at depths up to 1.3 mm.28,29 Intravital two-photon microscopy (IVM) is a powerful imaging tool for investigating various biological processes in live animals. This technique has been used to investigate various biological tissues like kidney and brain.30,31 While the imaging depth remains a challenge, IVM enables the real-time visualization and quantification of objects and events at subcellular levels in live animals.32–34 Quantifying lymphangiogenesis for pro-lymphangiogenic therapies is necessary as well demonstrating that these lymphatic vessels are functional. IVM may provide direct visualization of functional lymphatics at multiple time points in the same live animal and help determine the functionality of newly formed lymphatics. IVM in live animal models may provide improved lymphatic visualization in real time.
In this study, we report that fluorescein isothiocyanate (FITC)-dextran-stained lymphatics in wild-type C57BL/6 background offer a valuable tool for intravital imaging of newly formed lymphatic vessels within the tail in the live animal. They can be used to observe lymphangiogenesis induced by the delivery of gene Prox1 using TNT. Time course intravital imaging in these mice experimentally evaluates the applicability of IVM to visualize and quantify lymphatics.
Methods
Animals
Mice experiments were conducted in accordance with protocols approved by the Indiana University School of Medicine Institutional Animal Care and Use Committee (IACUC #22020-Murine model of lymphedema). Adult C57BL/6 mice, 8–10 weeks from Jackson Laboratories (Jackson, Bar Harbor, ME), were used for the study. Animals were maintained in a temperature and light-controlled environment with ad libitum access to food and water.
Mouse tail secondary lymphedema model
Acquired lymphedema was surgically induced in tails of C57BL/6 mice through the previously optimized and well-described lymphedema mice tail model. 24 Pain control was administered as described previously. 24 Under isoflurane-inhaled anesthesia (2%), a 3 mm full-thickness skin excision and lymphatic vessel disruption were performed 20 mm from the base of the tail in C57BL/6 mice. Underlying lymphatic vessels were visualized using isosulfan blue and disrupted under a dissecting surgical microscope. Mice tail volume was assessed by truncated cone equation.24,35 S0 indicates surgical excision of skin only. S1 indicates surgical excision of skin and lymphatic disruption.
Tissue nanotransfection technology
In vivo TNT was performed on mice tail as previously described. 24 TNT was applied to the murine tail (day 0) directly at the surgical site with genetic cargo loaded into the TNT reservoir: TNTpCMV6 (control)—TNT delivery of pCMV6 plasmid (expression vector backbone alone) (n = 6); TNT Prox1 -TNT delivery of pCMV6-Prox1 (n = 6). A gold-coated electrode (the cathode) was immersed in the plasmid solution, and a 30G needle (the anode) was inserted intradermally, juxtaposed to the TNT platform surface. To nanoporate the lymphatics, electrical stimulation (250 V, duration 10 ms, 10 pulses) was applied across the electrodes. S1+TNT Prox1 group was compared with S1+TNTpCMV6 (sham group). For IVM studies, S1+TNT Prox1 group was compared with S0+TNTpCMV6 (sham group). The S1+TNTpCMV6 group has leaky lymphatics which interfere with imaging (Supplementary Fig. S1).
Intravital reagents
Two injectable dyes were used to label the mice tail veins and lymphatics for IVM. Dextran 70 kD conjugated with Texas Red (Invitrogen, Waltham, MA) was used to label veins, and dextran 2000 kD conjugated with FITC was used to label lymphatics. Dextran 2000 kD conjugated with FITC was resuspended in sterile saline at 10 mg/mL injected in tail 90 minutes prior to imaging. Dextran 70kD conjugated with Texas Red was resuspended in sterile saline at 25 mg/mL and injected retro-orbitally (using 27-gauge needle syringe) just before imaging (Fig. 2).
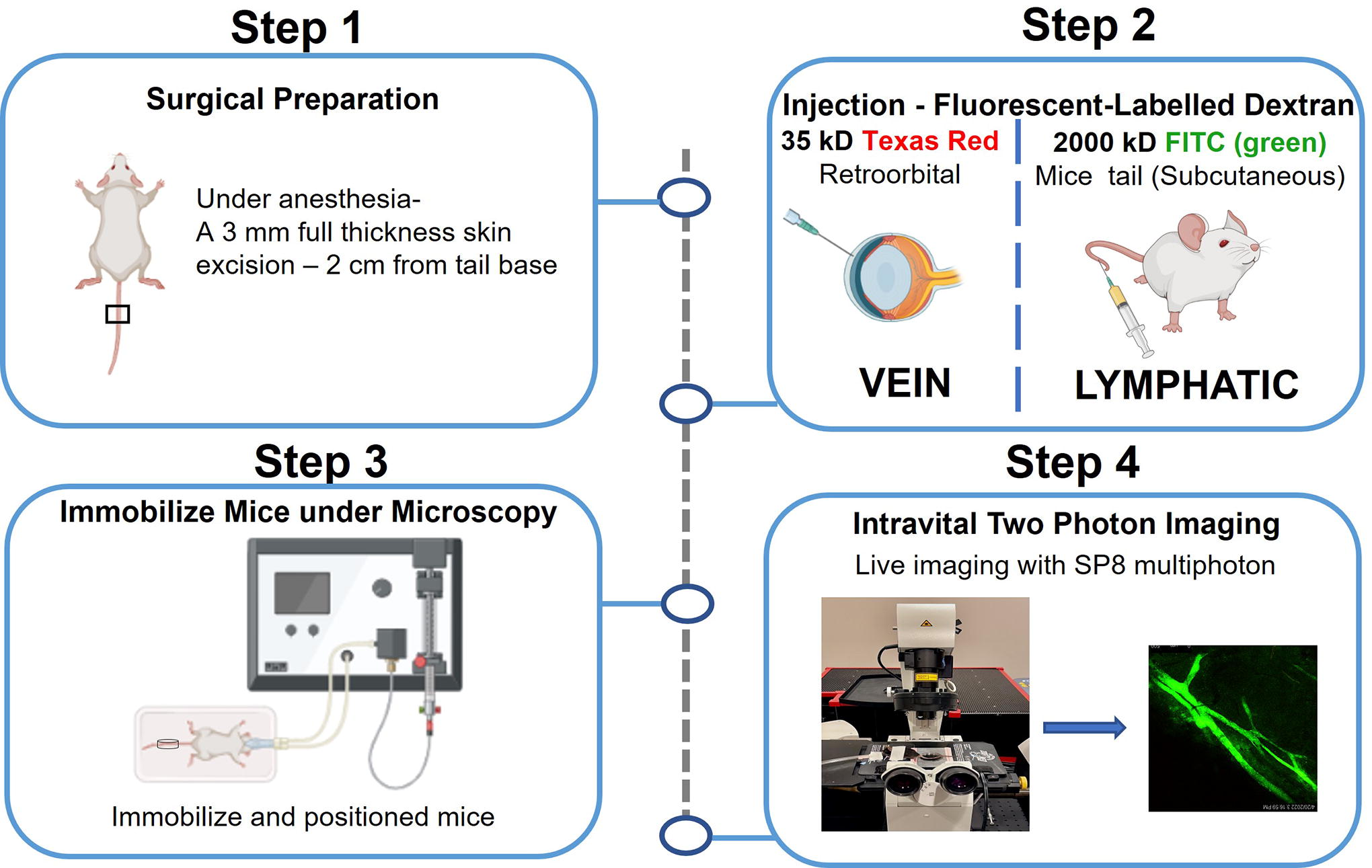
Intravital two-photon microscopy-visualization of lymphatics—a step-by-step procedure.
Surgical preparation for intravital imaging
Mice were placed under general anesthesia in an induction chamber with 3%–4% isoflurane in 100% oxygen followed by maintenance sedation at 1%–3% during the procedure. A hair removal cream (hair solution, Church and Dwight, Ewing, NJ) was placed on the mouse tail for 5 minutes. The cream was removed using gauze sponges and then rinsed with saline. Fluorescent lymphatics marker (Dextran 2000 kD, 10 mg/mL solution) was injected using 30-gauge needle syringe at the tip of the mice tail. Post-TNT (day 10), a 3 mm segment of murine tail was deskinned distal to the site of occlusion to optimize visualization. Saline was added at the skin excision site and covered with Tegaderm film (1626W) to avoid tissue dryness. A drop of ophthalmic anesthetic (0.5% proparacaine hydrochloride ophthalmic solution) was applied to the eyes prior to retro-orbital injection. A needle was inserted, bevel down, at an angle of approximately 45°, through the inferior fornix conjunctival membrane (6 o’clock position into the eye socket) to inject 100 uL of fluorescent venous marker (dextran 70 kD, 25 mg/mL solution). Vet eye ointment was applied to both eyes. The animal was transported to the multiphoton imaging room.
Intravital setup of mice
The animal was sedated with continuous flow of isoflurane (1%–3%) through nose cone and placed on a microscope stage of an inverted microscope (Fig. 3A) with the lymphatics-exposed tail positioned in a glass bottom dish and submerged in saline (Fig. 3B). The mouse was covered with a heating blanket avoiding direct contact with the heat source (37°C) and visually monitored for the respiratory rate throughout the imaging. The anesthetic was supplemented at an increased dose of -one fourth of the original dose as necessary. Mice were anesthetized with isoflurane for no longer than 30 minutes for imaging of lymphatics. Prior to imaging, mice received 200 uL of sterile saline subcutaneously to prevent dehydration.
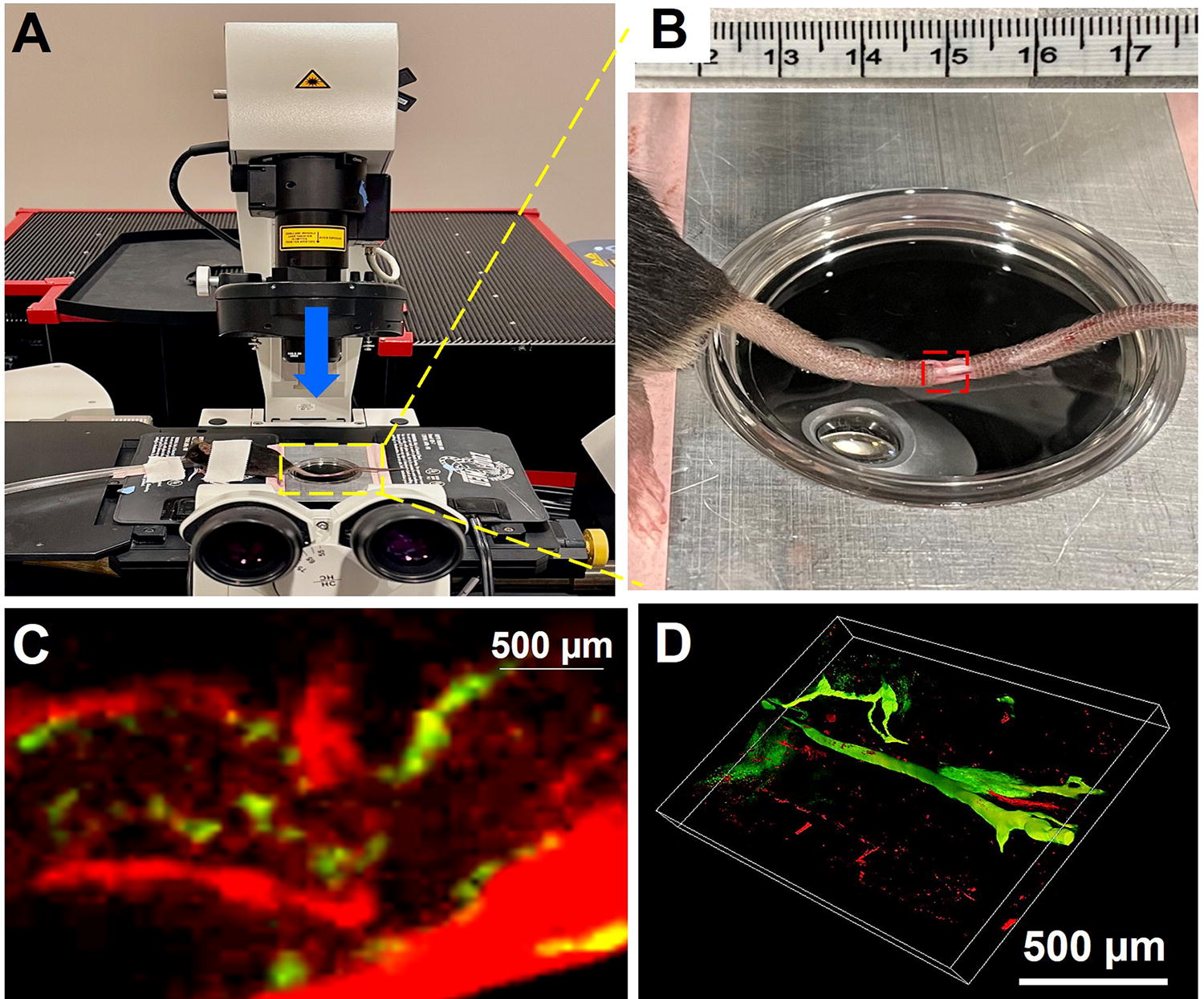
Novel intravital microscopy (IVM) of the mice tail lymphedema model.
In vivo high-resolution imaging of the mouse tail lymphatics
All IVM-based multiphoton images were captured using a Leica TCS SP8 confocal/multiphoton microscope system (Leica Microsystems Inc. Buffalo Grove, IL) available at the Indiana Center for Biological Microscopy (Indiana University, Indianapolis, IN). Images were acquired using a Leica HC PL Fluotar 5×/0.15 dry and/or HCX IR APO 25×/0.95 W objective lenses and a MaiTai DeepSee multiphoton laser (MKS Instruments, INC. Spectra Physics Lasers, Santa Clara, CA) tuned to 830 nm. Scanning was conducted in a non-discanned mode using three spectral detectors set up as follows: PMT-RLD1set to 390-440 nm to detect SHG signal of collagen, HyD-RLD2 set to 500-550 nm to detect FITC-dextran, and HyD-RLD3 set to 580-650 nm to detect Texas Red-Dextran. Images (12 bit) were acquired at 512 × 515 pixels (2.32 × 2.32 µm/466.6 × 466.6 µm), 400-600 Hz scanning speed, bidirectional X scan, and line averaging of 2. Single field images or tile acquisitions (single focal plane or Z-stacks) were acquired to obtain structural information, and time-lapse images (XY or XYZ) were captured for tracking the lymphatic fluid.
Quantitative analysis
Image quantitation and 3D reconstruction were performed using a dedicated 3D image quantitation and visualization software Imaris v. 9.7.2 (Oxford Instruments/Bitplane, Belfast, UK). 3D reconstruction was performed using a maximum intensity projection (MIP), alpha-blend, or shadow projection volume rendering algorithms. To determine the newly formed lymphatics, number of branching points/capillaries was assessed using a Filament Tracer module/ImageJ. For IVM imaging, S0 (skin excision only) animal was used to access the lymphatic flow and lymphatic branch points. The S1 mice have leaky lymphatics which interferes with the imaging.
Results
Mouse tail model of lymphedema
The murine tail model of lymphedema was utilized in this study to optimize the lymphatics visualization. Swelling of the tail as observed visually, determined through tail diameter measurement (weekly), and increase in volume are indicators of lymphedema onset (Fig. 1).
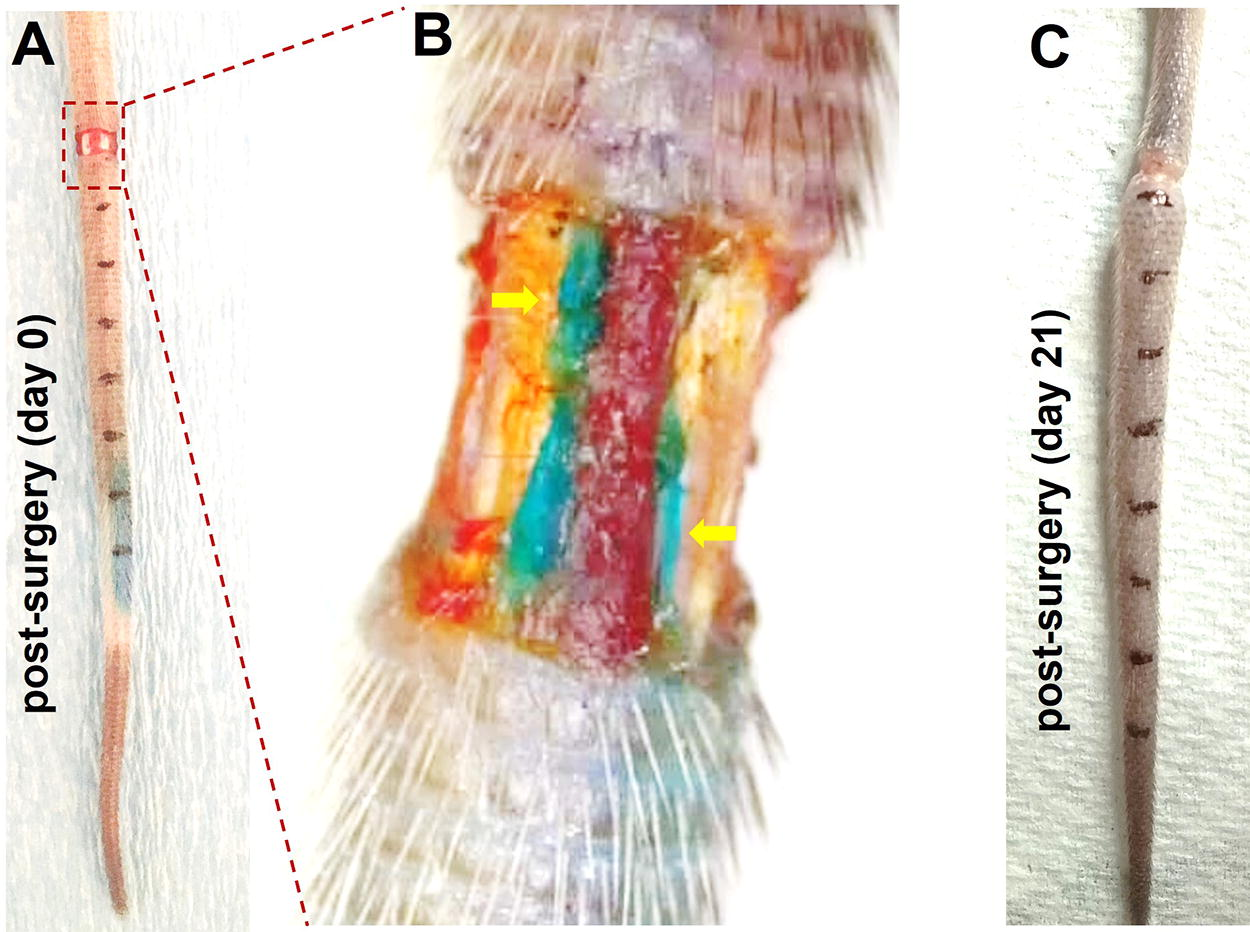
Mouse tail model of lymphedema.
Optimizing two-photon intravital microscopy in murine tail model of lymphedema
To visualize the lymphatics in live animal, we established a setup for intravital imaging using multiphoton microscope system (Fig. 2). Imaging was performed on the skin excision site of C57BL/6 mice tail by positioning on glass dish (Fig. 3B). Blood vessels were visualized by systemic administration of fluorescent tracer (dextran 70 kD) through retro-orbital injection, and lymphatic vessels were visualized by localized 2000 kD FITC-dextran injection in mice tail tip (Fig. 3C). The size differences in the linked dextrans were to prevent cross-mixing. Dextran 2000 kD would not enter the veins, whereas 70 kD dextran would not leak out of the veins. This allows for distinct visualization of lymphatics and veins. Z-stacks are collected from skin excision site and rendered as 3D MIP (Fig. 3D).
TNTProx1 prevents lymphedema in murine tail model
Plasmid containing Prox1 was delivered at the surgical site of murine tail lymphedema model to induce lymphangiogenesis (Fig. 4A). In our previous report, we have established the TNT mediated gene delivery in lymphedema tail. 24 Prox1 is a master regulator for lymphangiogenesis.36–38 In our recent study, 26 TNT-delivered Prox1 prophylactically prevents postsurgical mice tail lymphedema and preserves lymphatic function. Prox1 (S1+TNT Prox1 ) delivery resulted in a statistically significant difference in relative mice tail volume compared with control animals (S1+TNT pCMV6 ) (Fig. 4B–C). In this study, we have evaluated the Prox1 mediated lymphangiogenesis through TNT. More lymphatic distribution was observed in the TNT-delivered Prox1 (S1+TNT Prox1 ) group compared to control (S1+TNT pCMV6 ) (Fig. 4D–E).
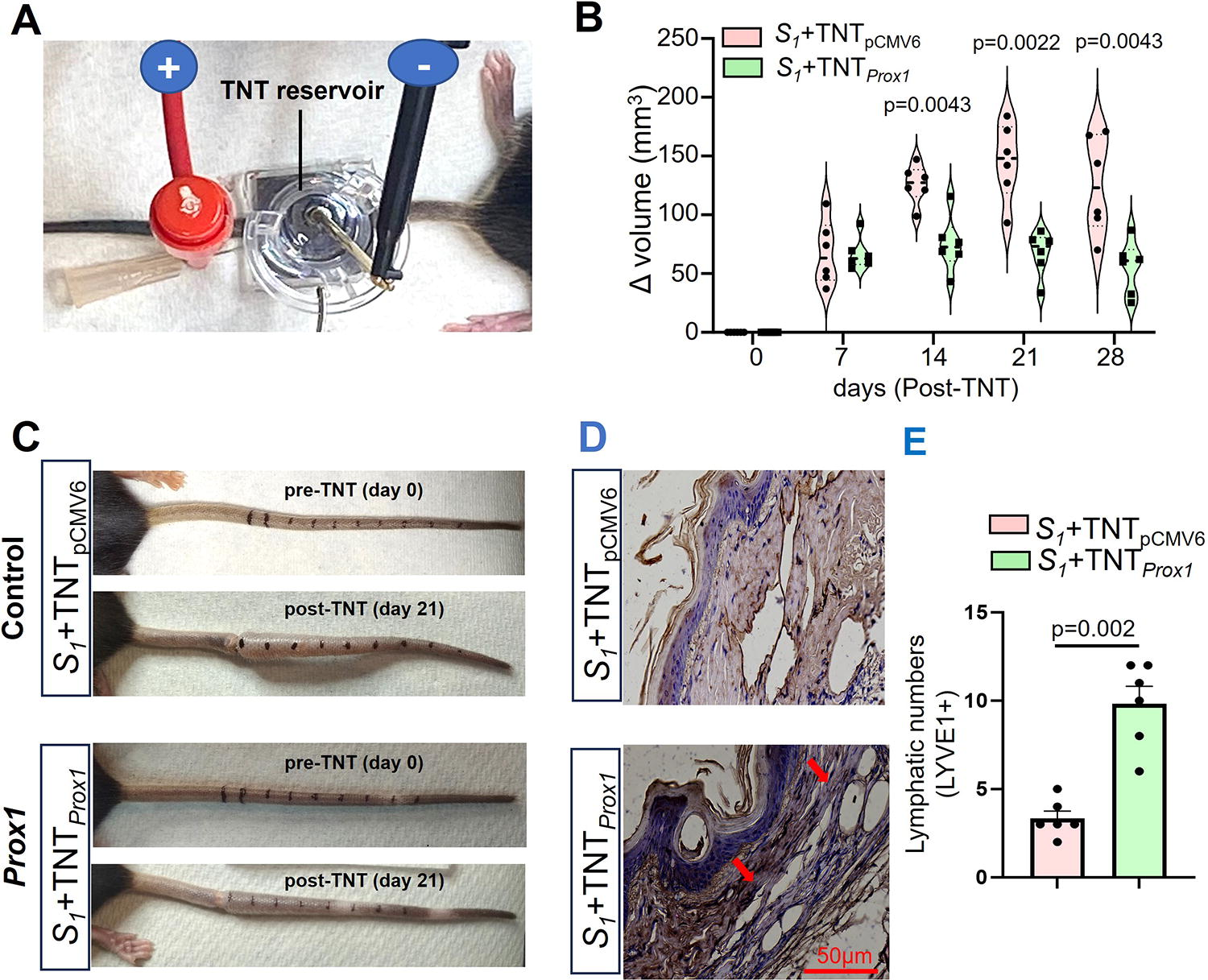
Tissue nanotransfection technology (TNT)-delivered Prox1 prevents lymphedema in murine tail lymphedema model.
Intravital two-photon microscopy for quantification of lymphatics
To evaluate the functionality of lymphatics generated in TNT site where Prox1 was delivered, we injected FITC-dextran (2000 kD) subcutaneously in the interstitial space at the tail tip for lymphatic uptake. In S0 group (control), we visualized lymphatic flow in the distal region (below the occlusion site) (Supplementary Movie S1). Lymphatic flow was evident in the base of the tails at the area distal to the surgical site 90 minutes after the dye injection. In lymphedema induced mice tail, a 3 mm segment of murine tail was deskinned distal to the site of occlusion to optimize lymphatic visualization. Lymphatic vessels were visualized at the second skin excision site to assess for the number of lymphatics branching points to determine the newly formed lymphatics at the S1+TNT Prox1 site (Fig. 5). The lymphangiogenesis was observed in both initial lymphatic vessels and collectors. We have observed increased lymphatic distribution in both lymphatic collectors and initial lymphatic vessels by IVM (Fig. 5A–D). TNT-delivered Prox1 mice (S1+TNT Prox1 ) tail has increased lymphatic branching points (2–3-fold) at the TNT site compared to control (S0+TNTSham) (skin incision only) (n = 5, p = 0.03) (Fig. 5).
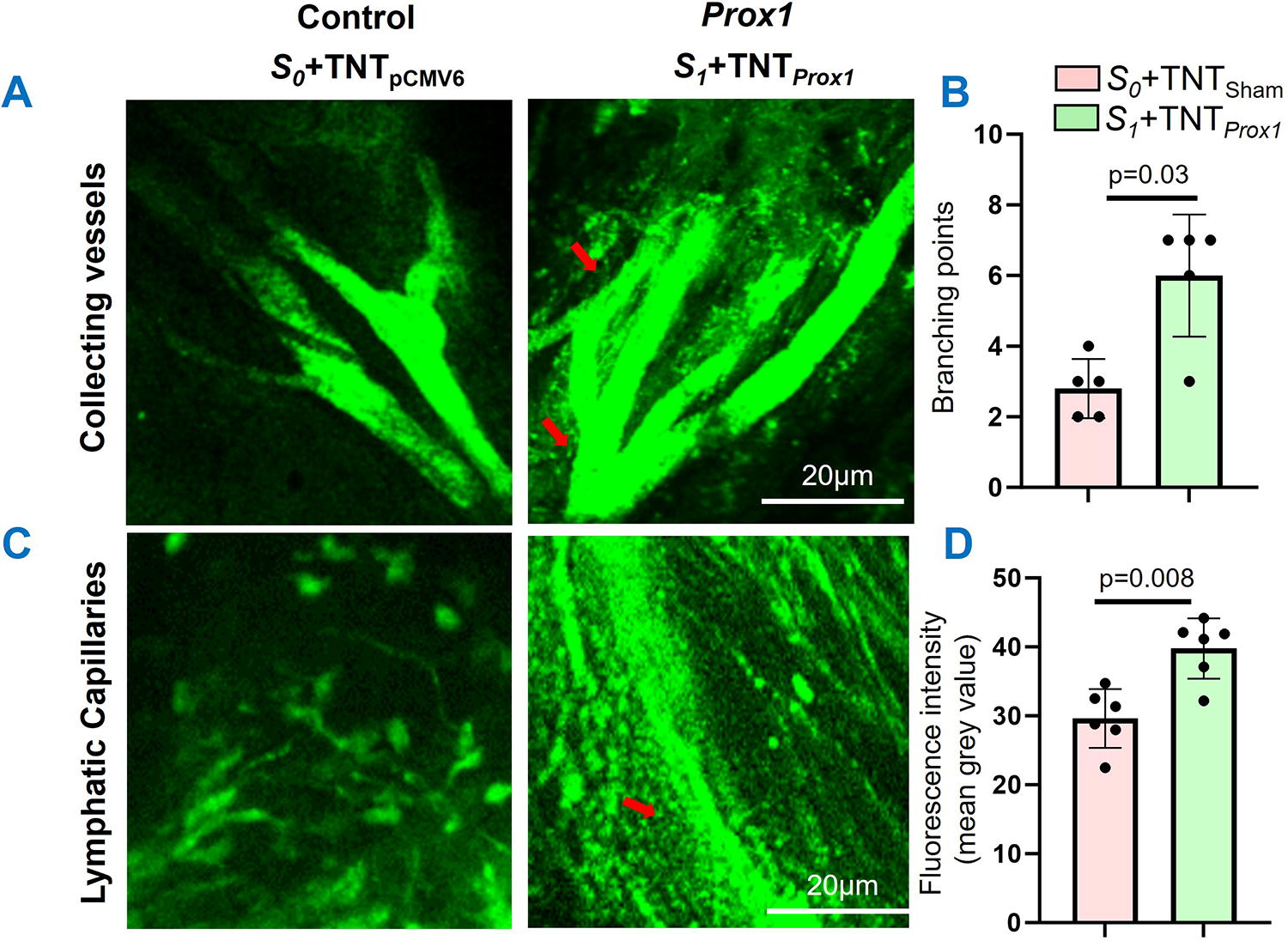
Increased lymphatic distribution at the Prox1 delivered site in the distal region of mice tail.
Discussion
This study demonstrates a novel, powerful imaging tool for investigating lymphatic vessels in live murine tail model of lymphedema. In this study, we present a two-photon-based intravital imaging technique and image analysis tool for the quantitative assessment of lymphatic branching in live murine tail lymphedema model. IVM was able to visualize lymphatic flow in the live animal.
In this study, Prox1 plasmid was topically delivered using TNT directly to the surgical injury site in the mouse tail. TNT facilitates direct, transcutaneous gene delivery using a silicon chip fabricated with nanochannels in a rapid, focused electric field.25,39 TNT has been used to rescue against diabetic peripheral neuropathy, repair crushed nerve, label wound-site exosomes, decrease limb ischemia, and treat ischemic stroke.25,40–43 TNT does not use a viral vector for gene delivery and avoids concerns for potential unintended global lymphangiogenesis as it is focally applied. We have recently demonstrated that TNT-delivered Prox1 prophylactically prevented mice tail lymphedema and preserves lymphatic function study. 26 In that study, immunohistochemistry indicated greater lymphatic density in the Prox1 treated mice that indicated lymphangiogenesis. RNA sequencing showed reduced inflammatory markers in those mice. Hence in this investigation, Prox1 was delivered to the mouse tail using TNT to induce lymphangiogenesis prophylactically at the time of murine lymphatic mouse tail injury.
IVM imaging can be used to quantify lymphangiogenesis regardless of the therapy or mode of delivery. Previous investigators have quantified lymphangiogenesis by branching index in the whole mount tail skin from Prox1-Green Fluorescent Protein transgenic mice. 44 Lymphatic leakiness was assessed by the injection of FITC-dextrans in Prox1-Cre-ERT2-tdTomato mouse tail. 15 In another study, lymphatic function was validated by monitoring of transport of the molecule (70 kDa-Rhodamine dextran) in mice paw by fluorescent microscopy. 45 We also found that lymphatic vessel leakiness is a limitation of IVM for assessing lymphangiogenesis for the standard murine tail model lymphedema control with skin excision and lymphatic collector clipping (Supplementary Fig. S1B). Therefore, the control used for the IVM was skin excision only leaving the lymphatic collecting ducts intact. In contrast, with the Prox1 treated mice, lymphatic leakiness was not problematic and lymphatic vessels were able to be visualized with branch points quantified. The lymphatic collecting channels that were clipped did not leak with IVM. This may be because lymphangiogenesis restored lymphatic transit through the murine tail maintaining lymphatic function.
Recent advances in high-resolution in situ imaging techniques, especially two photon Laser Scanning Microscope (2P-LSM), have been accompanied by a growing interest in the study of lymphatics in vivo. The use of 2PLSM with multiple detectors spanning the entire visible spectrum permits lymphatics imaging and quantification. This can be further improved by use of in vivo lymphatic-specific fluorescent reporter mice to monitor the lymphangiogenesis events. Intravital imaging will be useful tool for the functional assessment of lymphatics and visualization of lymphangiogenesis following gene-based therapy.
Conclusion
This study optimized the two-photon intravital imaging of lymphatics in live murine tail model. Intravital microscopy will be a useful imaging tool for assessment of lymphatic flow, functionality, and visualization of lymphangiogenesis to develop therapeutic strategies against these conditions.
Footnotes
Acknowledgments
Imaging was carried out in the Indiana Center for Biological Microscopy at Indiana University using a system purchased with funds obtained from NIH Shared Instrumentation Grant S10-OD025136. Presented at 68th Annual Meeting of Plastic Surgery Research Council, Cleveland, OH, April 15, 2023. DOI 10.1097/01.GOX.0000938196.28096.98
Author Contributions
A.H.H., M.S., and C.K.S. conceived and designed the work. G.M., I.K., S.M.D., M.M.K., C.R.N., M.D.J., M.S., and A.H.H. participated in the data acquisition and analyses. A.H.H., G.M., M.S., and C.K.S. wrote the article. A.H.H., M.S., G.M.G., and C.K.S. reviewed the article.
Author Disclosure Statement
The authors declare no competing interests.
Funding Information
This work was supported by US Department of Defense (W81XWH2110135), the National Institute of Health (R21AR082600) (K08HL167164), Indiana Clinical and Translational Sciences Institute, IU Health Values Grant, the Plastic Surgeon Foundation, and American Association of Plastic Surgeons grants and to A.H.H.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
