Abstract
Background:
Breast cancer-related lymphedema (BCRL) after primary therapy is a common condition, causing physical and psychological distress. Decongestive lymphedema therapy (DLT) using multi-layered compression bandages is an effective treatment. We conducted a randomized controlled trial evaluating the use of a specific mobilizing bandage (Mobiderm®) on lymphedema volume reduction during the intensive phase of DLT.
Methods and Results:
Fifty female BCRL patients were randomized to receive either conventional multi-layered bandages or mobilizing bandaging by using Mobiderm. Affected limb volume and excess volume were evaluated at baseline (D0) and after 15 days. The primary outcome was change in affected limb volume after adjustment for baseline. Symptom scores were evaluated by visual analogue scale (VAS); safety and tolerability were also assessed. Baseline characteristics were comparable. Affected limb volume reduction was observed in both study groups after 15 days: by 19.0% in the Mobiderm arm and 8.6% in controls (adjusted values). The between-group mean difference in adjusted volume reduction at day 15 was 256 mL (95% confidence interval [CI], 92.5 to 421.3 mL; p = 0.003) favoring Mobiderm. Reductions in excess volume of 57.3% (Mobiderm) and 25.1% (controls) were observed (adjusted values); with between-group mean difference in adjusted excess volume of 220.2 mL (95% CI, 69.3 to 371.3 mL; p = 0.006) favoring Mobiderm. Pain/heaviness VAS scores fell significantly in both groups, with mean reductions of 1.84 (Mobiderm) versus 0.83 (control; p = 0.001). Both regimens were well tolerated.
Conclusion:
The use of Mobiderm in multilayer compression bandaging shows benefit in lymphedema reduction and in alleviating functional symptoms/pain in patients with BRCL.
Introduction
With therapeutic advances and use of multimodal therapy in breast cancer (surgery, radiation therapy, and chemotherapy, targeted therapy, and hormonal therapies in various combinations), survival outcomes have greatly improved; consequently, the longer-term impact of treatment complications has assumed greater importance in a patient's well-being and quality of life (QoL). 1
Breast cancer-related lymphedema (BCRL) of the upper limb after surgery or radiation therapy to axillary lymph nodes is a common complication that can cause substantial physical and psychological distress and impaired QoL.2–4 It has been estimated that 20% of patients will develop chronic BCRL after treatment for breast cancer. 2 In India, the location of the present study, Pillai et al. reported an overall incidence of lymphedema of 41.1%, and an incidence of clinically significant lymphedema of almost 17%. 5
The incidence of BCRL and individual risk is influenced by a number of factors associated with individual patient characteristics and therapy. Treatment-associated factors include surgical procedures, radiation therapy, and adjunctive chemotherapy; in general, incidence is lower with more limited axillary nodal procedures such as sentinel node biopsy (5.6%), and it is greater in patients receiving complete axillary lymph node dissection (ALND, 19.9%), with receipt of postoperative radiation therapy or adjunctive chemotherapy further increasing incidence and risk.2,3
Preoperative high body mass index (BMI) ≥30 is a recognized risk factor, as is the presence of postoperative infection/cellulitis.3,6–9 The onset of BCRL (i.e., rate of development) is also variable; initial onset within days after the completion of primary treatment may be observed, and the majority of patients with BCRL will develop symptoms within 2 years, although more delayed onset (over many years) may also be seen.2,10,11
Complete decongestive therapy (CDT) is the foundation of BCRL management. The CDT involves a combination of manual lymphatic drainage, bandaging, skin care, and exercises to promote lymph reduction, usually delivered in two phases: an initial intensive phase to achieve optimal lymphedema volume reduction over a short period (1–4 weeks) followed by a prolonged maintenance phase. 12
As reflected in the updated best practice guidelines from the International Society of Lymphology and also the International Lymphedema Framework (ILF),12,13 the use of multicomponent compression bandaging is a key component in lymphedema treatment in both phases, and in particular during intensive phase therapy. Although different modalities exist, a combination of layers of inelastic (or “low-stretch” compressive bandages) and short-stretch elastic bandages applied to the whole limb are commonly used.
Long-stretch elastic bandages can also be used in practice in a multilayer bandage. 14 The Mobiderm® mobilizing pad or bandage (THUASNE, Saint-Etienne, France) is composed of foam blocks lying within an adherent soft webbing, designed to apply different areas of pressure on the skin surface, which in combination with muscle movement promotes edema mobilization and evacuation. Combining Mobiderm with other multilayer bandages is associated with a shorter period of decongestive lymphedema therapy (DLT) compared with other bandages, suggesting a rapid effect in reducing edema. 14
Mobiderm technology has also been included in garments, tested in clinical studies for the maintenance phase of lymphedema treatment. 15 Data from clinical studies indicate that the use of Mobiderm garment for night-time use during the maintenance phase is well tolerated and provides clinical benefits in reducing BRCL.16–19
Although the use of quality compression bandage systems such as those using Mobiderm are widely available in many countries across Europe, North America, and Australia, their availability in other regions (including countries with a considerable burden of BCRL such as India 5 ) is more limited, and data on their use and clinical outcomes are lacking. 13 The purpose of the present study was to evaluate the effects of the use of the Mobiderm system as a component of compression bandaging in Indian patients with BCRL.
Materials and Methods
Study design and patient population
This was a single-center prospective randomized controlled trial (RCT) conducted between July 2015 and December 2017 in the department of surgical disciplines at the All India Institute of Medical Sciences (AIIMS) in New Delhi, India. Eligible patients were women aged from 18 to 60 years, with established secondary upper limb lymphedema after breast cancer surgery, considered suitable for intensive phase DLT. Exclusion criteria included primary lymphedema, co-existing cardiac or arterial disease, involvement in any other clinical study within the previous 3 months, and the inability to provide informed consent.
At enrollment, a complete medical history and examination were performed to establish baseline characteristics, including limb volume (measured via the water displacement method). The included patients were then randomly assigned to the Mobiderm or control groups in a 1:1 ratio, by using a computer-generated randomization sequence.
The study was conducted in accordance with the International Conference on Harmonization Guidelines for Good Clinical Practices, the principles of the Declaration of Helsinki, and also national ethical guidelines. The study protocol and other relevant study information was filed with the local institutional review board, and approval was granted by the ethics committee of AIIMS (IEC/NP-98/13.03.2015). Written informed consent was obtained from each patient before study enrollment.
Treatments and evaluations
Patients were assigned to receive one of two multicomponent compression bandage regimens. In the Mobiderm group, patients received a multi-layer bandage composed of: an inner layer of cotton band (Bande Coton; THUASNE); an intermediate layer (Mobiderm band; THUASNE); and an external layer of elastic short-stretch bandage (Biflexideal®; THUASNE). In the control group, the intermediate layer comprised an ortho cotton wool soft pad (as used in current treatment in India), whereas the internal and external layers were the same as used in the Mobiderm group. In both groups, the compression bandages were initially applied by Certified Lymphedema Therapist (CLT) in the AIIMS hospital team, and then reapplied three times during the first week and once during the second week by the same clinical team.
All patients were provided with clear detailed instructions on relevant exercises, skin care, and hygiene associated with intensive phase decongestive therapy. To reduce the risk of dermatolymphangioadenitis (DLA), in accordance with established protocols for the current standard of care of patients with BCRL at AIIMS, all patients received a course of Benzathine penicillin G (1.2 MU) via a deep intramuscular injection given once every 3 weeks.20,21
Patients were evaluated at baseline (V1, day 0) and after 15 days of compression bandaging (V2). Upper limb volume (in mL) was measured by using the volume displacement method, the gold standard, which involves immersing the entire upper limb in a large container, with the volume of water displaced assessed by using a large measuring cylinder. A glass chamber filled with water was used for this purpose with an outlet at the upper end to allow the water to come out once displaced by the immersed arm. This displaced water was collected in a cylinder with a marking on it for volume measurement, kept exactly below the outlet.
The whole arm was kept still inside the chamber while the patient kept holding the hand firmly at the bottom of the chamber. This prevented movement and also allowed uniformity in measurement on every occasion (Fig. 1). The volume of the unaffected contralateral upper limb was also measured at day 0, to allow an estimation of the excess limb volume due to lymphedema in the affected limb. At both visits, patients were also asked to objectively grade combined symptoms of pain and heaviness by using the visual analogue scale (VAS) in which 0 indicates “no pain or heaviness” and 10 indicates the “worst possible pain or heaviness.” 16 Information on adverse events associated with either compression bandaging regimen (in particular the presence/development of cellulitis) was also collected.

Glass chamber along with the collecting glass cylinder with marking for measurement of displaced volume.
Outcomes
The primary outcome measures were the volume reductions in the affected limb at day 15 compared with baseline (day 0); volume changes were compared within and between groups. Secondary outcomes included changes in limb “pain/heaviness” scores.
Data analysis
At the time of the study, as no comparable study using the Mobiderm band was available to quantify an appropriate sample size, no formal sample size calculation was performed. As such, a fixed number was selected for this study. A total of 50 patients were screened for eligibility and randomly assigned to the 2 study groups: Mobiderm (n = 25) or control (n = 25). One patient assigned to the control group withdrew from the study before the second study visit, and so, a total of 49 patients completed the study and were available for analysis.
Continuous variables were expressed as mean ± standard deviation (SD) or standard error; categorical variables were expressed as counts and percentage as appropriate (with percentage calculated on the basis of the per-protocol population). Changes in volume were expressed as means and associated 95% confidence intervals (CIs). Percent reductions in limb volume (LV) were calculated by using the following formula: limb volume day 0–limb volume day 15/limb volume day 0 × 100.
For categorical data, differences were evaluated by using the Fisher exact test or the Chi-square test. Continuous variables were compared by using the independent t-test (for data following normal distribution) and the Wilcoxon rank-sum test (for non-normally distributed data). Within- and between-group changes between days 0 and 15 were compared by using paired or independent t-tests or Wilcoxon rank-sum test as appropriate; repeated-measures analysis of covariance (ANCOVA) was used to adjust for differences in baseline values. For all comparisons, a p-value of <0.05 was considered statistically significant. All data analyses were performed by using STATA version 14 (STATA Corp.).
Results
Study population
Baseline characteristics of patients in the two study groups were comparable (Table 1). The majority of patients were aged >50 years and menopausal. The mean age in the Mobiderm group was 50.8 ± 10.2 years with a mean BMI of 30.0 ± 2.20 kg/m2; in the control group, the mean age was 54.9 ± 11.1 years with a mean BMI of 31.5 ± 1.5 kg/m2. More patients in the control group had co-existing diabetes (p = 0.023). From a primary treatment perspective, all patients underwent radical mastectomy and ALND; more than 60% had preoperative chemotherapy, and the great majority had postoperative radiation therapy.
Baseline Characteristics of the Study Cohort
Continuous variables compared by using the independent t-test or the Wilcoxon rank-sum test; categorical variables compared by using the Fisher exact test or Chi-square test.
ALND, axillary lymph node dissection; BMI, body mass index; SD, standard deviation.
The mean time from the last surgery to study inclusion was 12–18 months. Approximately 80% of patients in either group had evidence of partial lymphatic obstruction on lymphoscintigraphy (Table 1).
Efficacy results
At baseline, there was no significant difference in the affected limb volumes between the two groups (p = 0.677). Day 0 mean excess limb volume in the Mobiderm and control groups was 1060 mL ±619.1 and 900 mL ±582.7, respectively.
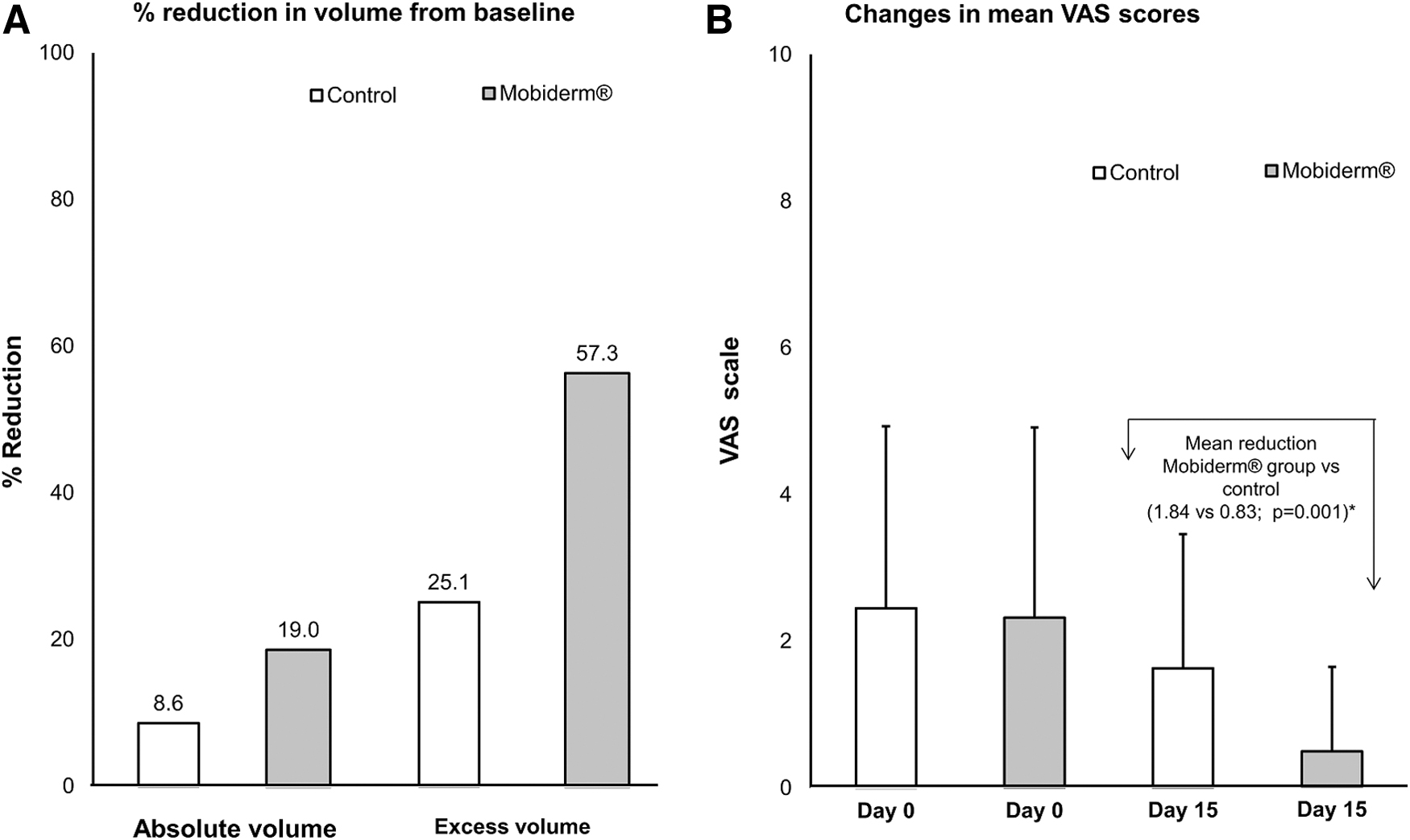
Changes in limb volume and excess volume are shown in Table 2 and Figures 2 and 3. At day 15, a reduction in affected absolute limb volume at day 15 (unadjusted values) was observed in both the Mobiderm and control groups (by 18.1% and 9.5%, respectively). When adjusting for baseline volume using ANCOVA, adjusted absolute limb volume at day 15 in the Mobiderm group was 2508 mL ±57.1 at day 15 corresponding to a 19.0% reduction in absolute limb volume from baseline (p = 0.001) (Figs. 2A and 3A). In the control group, adjusted absolute limb volume at day 15 was 2765 mL ±58.3, an 8.6% reduction (p = 0.001). The between-group mean difference in volume was 256 mL (95% CI, 92.5 to 421.3 mL) in favor of the Mobiderm group (p = 0.003).
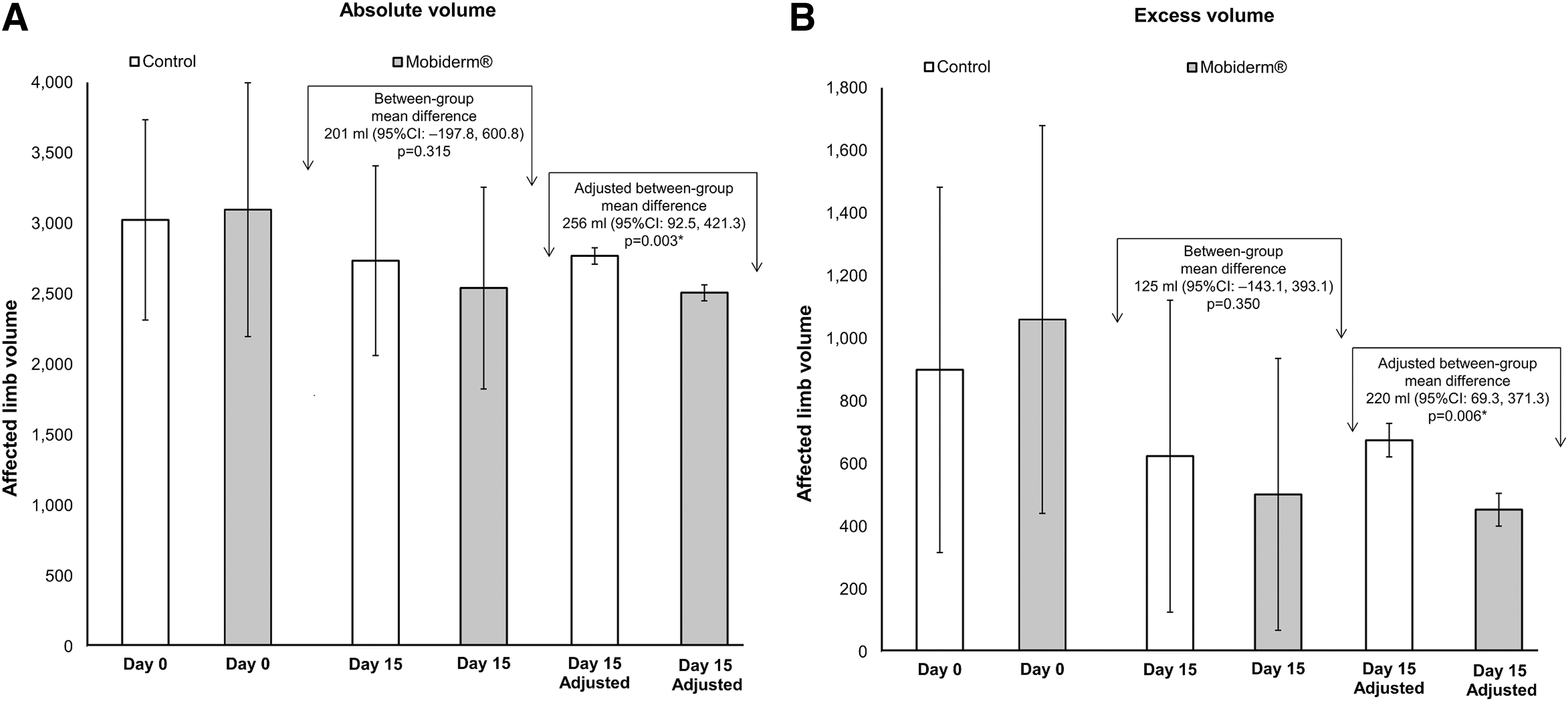
Baseline (day 0) and changes in absolute limb volumes
Change in Affected Limb Volume and Mean Affected Limb Volume Reduction Over 15 Days
Within-group p-value calculated by using paired t-test or Wilcoxon sign-rank test.
Adjusted for baseline values by using ANCOVA.
Between-group p-values calculated by using independent t-test or Wilcoxon sign-rank test.
ANCOVA, analysis of covariance; CI, confidence interval; SE, standard error.
Changes in excess volume (i.e., lymphedema) were also observed (Table 2 and Figs. 2B and 3A). Between days 0 and 15, the unadjusted excess volume reduced significantly in the Mobiderm group control (−560 mL) and also in the control group (−275 mL), both p < 0.001. Unadjusted excess volume reductions from baseline in the Mobiderm and control groups were 52.8% and 30.6%, respectively. After adjustment for baseline excess volume, adjusted excess limb volume at day 15 in the Mobiderm group was 453.4 mL ±52.2; a 57.3% reduction in excess limb volume from baseline (p = 0.001). In the control group, adjusted excess limb volume at day 15 was 673.6 mL ±53.4; a 25.1% reduction (p = 0.001). The between-group mean difference in volume was 220.2 mL (95% CI, 69.3 to 421.3 mL) in favor of the Mobiderm group (p = 0.006).
Mean VAS scores for pain/heaviness were comparable in both groups at day 0, with mean ± SD scores of 2.32 ± 2.59 (Mobiderm) and 2.45 ± 2.48 (control). After 15 days of DLT, VAS scores were significantly lower in both groups: 0.48 ± 1.15 (Mobiderm; p = 0.001) and 1.62 ± 1.83 (control; p = 0.004), with significantly greater mean reductions observed in the Mobiderm group compared with the control group (1.84 vs. 0.83; p = 0.001) (Fig. 3B).
Safety and tolerability
Cellulitis was the most frequently observed event during the pre-bandaging period that was observed in 11 patients (45.8%) of the control group and 15 (60%) of those receiving Mobiderm. There was no new episode of cellulitis during the study treatment period, and no patient discontinued treatment due to cellulitis. Both compression regimens were well tolerated.
Discussion
The role of compression therapy in the treatment of lymphedema has been extensively discussed.13,22,23 The CDT performed under the supervision of a trained lymphedema nurse or physiotherapist is currently the gold standard of care for the management of upper lymphedema, including BCRL.12,13 In the intensive phase, CDT treatment goals are to stop lymphedema progression, reduce lymphedema volume, and train patients for their subsequent life-long home self-care and educate them in risk reduction considerations.
Studies evaluating the benefit of CDT on reducing lymphedema in BRCL typically evaluate volume reductions from two perspectives: a reduction in affected limb absolute volumes or a reduction in excess volume (a proxy measurement for the lymphedema volume). 24 In the present RCT, for our primary outcome we evaluated changes in absolute limb volumes after 15 days of intensive phase CDT; and after adjustment for baseline differences, we found a mean reduction of 19.0% in absolute volume in subjects treated with the Mobiderm compression stocking and of 8.6% in those receiving a standard domestic multi-layered compression bandaging system.
These results are comparable with those reported by others. For example, Sezgin Ozcan et al. reported an 8.9% reduction in absolute upper limb volume in patients with BRCL after 3 weeks of intensive phase CDT (comprising manual lymphatic drainage, exercise, and compression bandages) 25 ; however, Zasadzka et al. reported an absolute volume reduction of 13.9% after 3 weeks of intensive phase CDT using compression bandages in patients with lower limb lymphedema. 26
We also evaluated changes in excess volume reduction to examine effects on lymphedema per se, where we found substantial reductions with both compression regimens after 15 days bandaging, although again significantly greater in patients using Mobiderm bandage (57.3%) than control (25.1%). Again, these reductions are comparable to or even greater than those found in previous studies reporting the impact of compression bandaging on excess volume in intensive phase CDT. In their recent study, Sezgin Ozcan et al. reported a 38.1% reduction in excess volume due to BRCL. 25
However, a 33.0% reduction in excess volume in BRCL was reported in the observational POLIT study, 14 and a 37.9% mean reduction after the intensive phase was reported by Vignes et al. in a large cohort of 537 breast cancer patients.27,28 Other studies have also reported higher excess volume reductions, although these often involve additional elements of CDT or are conducted over longer treatment periods.29,30
In addition to volume reduction, we also assessed the impact of compression bandages on symptoms of pain/heaviness, and significant improvement in VAS scores after 15 days with both bandaging regimens. There was a greater reduction in VAS score in subjects treated with the Mobiderm bandage. Similar improvements in upper limb symptoms have also been reported by others after compression bandaging for BRCL in intensive phase CDT.14,17,25
Our results suggest that compression bandaging is a practical and effective approach to CDT for patients with BRCL. Although we found substantial reductions in volume and pain VAS scores using conventional compression bandages, significantly greater reductions were observed with the regimen incorporating the Mobiderm bandage as the intermediate “padding” in a multi-layered compression bandage. When used as a second layer overlying a cotton bandage, covered with a short-stretch bandage, this combination would seem an effective practical approach to achieve compression and lymphatic decongestion.
This is particularly relevant for CDT in countries such as India, where access to the broader range of quality compression products and bandages may be more limited than in other regions such as North America and Europe. The use of a simpler system such as Mobiderm offers a pragmatic approach to realize improved lymphedema than conventional compression bandages. This bandage is reusable and washable, thus offering important utility in developing countries.
Our study has several limitations. Our study cohort was relatively small, and the treatment period was relatively brief, without longer term follow-up. Consequently, we cannot comment on whether the benefits obtained with either bandaging regimen were maintained. It is recognized that volume reductions gained during intensive phase CDT may diminish despite subsequent maintenance phase treatment,14,28 and a longer study with more extensive follow-up and/or a formal maintenance phase study would be helpful to inform us of any sustained benefits of Mobiderm.
In addition, our study size and design did not allow us to evaluate any predictive factors that may have influenced our results. Obese patients are believed to be at a higher risk for lymphedema because they have baseline impaired lymphatic function,22,31 and some data indicate that higher BMI is associated with greater volume reductions in intensive phase CDT. 27 In the present study, the mean BMI was between 30.0 and 31.5 kg/m2, indicating a high level of obesity in the study population (although comparable in both groups) that may have influenced the magnitude of the volume reductions we observed.
However, our study also has some strengths: chiefly its randomized design, with comparable patient demographics in both study arms, where we evaluated clinically meaningful outcomes (volume reduction, excess volume reduction, and functional impairment). In addition, from a narrower local perspective, although previous studies have evaluated the benefit of intensive phase CDT in Indian patients with BRCL,32–34 these involved an extensive multicomponent program, performed at home, and chiefly reported on QoL outcomes. To date, none have specifically evaluated the impact of different compression bandage regimens as reported in the present study.
Conclusion
In this RCT, we found that the use of Mobiderm as a mobilizing compression bandage in intensive phase CDT resulted in significantly greater volume reductions and improvements in pain/heaviness VAS scores than a conventional compression bandage regimen in patients with BRCL.
Footnotes
Acknowledgments
The authors would like to thank all physicians who participated in the study. Medical editorial support in article development was provided by NewMed Publishing services.
Authors' Contributions
All authors were involved with the study design and data interpretation, and with the development, writing, and review of the article.
Author Disclosure Statement
P.S. and S.V. are employees of THUASNE. All other authors declare that no competing financial interests exist.
Funding Information
This study and the costs associated with the development of this article were supported by THUASNE.
