Abstract
Objectives:
To identify predictive factors associated with limb volume reduction using different decongestive lymphatic therapy (DLT) systems in patients with lymphoedema, over a period of up to 28 days.
Methods:
A multicountry (Canada, France, Germany, the United Kingdom) prospective cohort study using (DLT): skin care, exercise, compression bandaging, and manual lymphatic drainage for up to 4 weeks. Reduction in limb volume comparing DLT with (1) standard multilayer bandaging with inelastic material, and with (2) multilayer bandaging with Coban2, together with the identification of factors associated with limb volume changes.
Results:
Out of 264 patients with upper or lower limb lymphedema, 133 used Coban2 and 131 used standard care. Following DLT, mean limb volume reduction was 941 mL using Coban2 compared with 814 mL using standard care. A difference of 127 mL was found (95% confidence interval −275 to 529 mL, p = 0.53). Of the 176 patients with leg swelling, 166 (94.3%) had a limb volume measurement after 28 days and were included in the risk factor analysis. Of these, 132 (79.5%) were female, with overall mean age of 60.1 years (standard deviation = 14.7), with secondary lymphedema in 102/163 (62.6%). Duration of lymphedema was >10 years in 75/161 (46.6%) and 99/166 (59.7%) were International Society of Lymphology late-stage II/III, indicating longstanding and/or a high frequency of patients with advanced stages of lymphedema. Ninety-one (54.8%) received Coban2 and 75 (45.2%) had standard care. Multivariable factors for a greater leg volume reduction were large initial leg volume (p < 0.001), DLT treatment duration of 4 weeks compared with 2 weeks (p = 0.01), and peripheral arterial disease (p = 0.015).
Conclusion:
Limb volume changes were found to be similar between groups. Lack of standardization of DLT makes interpretation of effectiveness problematic. There is an urgent need for randomized-controlled trials. Despite this, severe lymphedema with a large limb volume responded well to DLT in this study.
Introduction
Chronic edema is defined as edema of a body part, of more than a 3-month duration, and is manifested with pitting and/or fibrosis of the skin. 1 Chronic edema is often multifactorial and includes different pathologies, for example, primary lymphedema (“congenital”) and secondary causes that include venous insufficiency, cancer, heart failure, immobility, and obesity. It is recognized that multiple comorbidities and polypharmacy may also influence the presentation of the condition. 2 The term lymphedema, which goes under the umbrella term of chronic edema, has traditionally been used to describe edema resulting from a failure of the lymphatics, due to cancer, lymphatic malformation, filariasis, and so on. The term is often used interchangeably with chronic edema, as newer research indicates the important role of the lymphatics in all chronic edema.3,4 The increased size and restricted mobility of the affected limb influence daily life with evidence of a significant impact on health-related quality of life. 5
Traditionally, treatment of lymphedema has been organized with an acute phase, in which treatment is focused on volume reduction, stabilizing the skin condition, reducing complications such as cellulitis, and increasing mobility through decongestive lymphatic therapy (DLT).6–8 DLT includes multilayer bandaging, skin care, exercise, and manual lymphatic drainage (MLD). Following this, patients move through a transition phase into a maintenance program where they undertake self-management activities to control the swelling.
There is wide variation in how DLT is delivered internationally and a lack of agreement on standards or outcomes of treatment. 9 Systematic reviews have confirmed the role of DLT, however, the benefit of the different components of treatment is less well understood and significant variations in outcome occur in different populations. 6
While DLT requires the use of compression bandaging, many different materials and application methods are in use. The inability to assess the correct dosage of compression within DLT adds to the levels of uncertainty about the clinical and cost- effectiveness of the varied approaches and the contribution that each element of DLT makes to the outcome of treatment. 10
The compression bandage 3M™Coban™2 system (Coban 2 system) was initially developed for the treatment of chronic venous leg ulcers and has proven to be effective for this indication. 11 Following this, the system has been adapted for the needs of patients with different presentations of lymphedema who have more extreme limb shape and size. 12
Research has begun to unravel the correct characteristics of a compression bandage for lymphedema. Previous dogma suggested that the highest possible pressure on application was required on initial application. 7 However, research has shown that in breast cancer-related arm lymphedema, inelastic bandages with a pressure between 20 and 30 mm Hg achieved a higher degree of volume reduction than higher pressure bandages applied at a pressure between 44 and 68 mm Hg after 2 hours. 13 In patients with leg edema, compression in the range 20–40 mm Hg showed a positive correlation between exerted pressure and volume reduction. 14 However, bandages applied with an initial resting pressure of more than 60 mm Hg resulted in a less effective volume reduction. From these studies, it may be concluded that there is an upper limit, beyond which further increases of compression pressure seem counterproductive. This limit is around 30 mm Hg on the upper limb and 50–60 mm Hg on the lower extremity. 15
A study investigated the application frequency of the Coban2 system in patients with lymphedema and found a high rate of volume reduction and a good safety profile. 12 Edema reduction was still effective at 4 days after application, which may mean that bandage application can be reduced from the daily application of current practice to twice a week. Morgan et al. evaluated the experience of lymphedema patients with the Coban2 system and found that besides reducing swelling other characteristics were ease of application, lightness, neatness, flexibility, and ability to facilitate mobility. 16 The need to clarify and extend this initial work underpinned the rationale for this study.
The aim
To identify predictive factors associated with edema reduction by intensive compression bandaging within DLT in patients with limb lymphedema, and to compare the reduction of limb volume between Coban2 and standard bandage regimens.
Methods
Study design and setting
This was an international study undertaken in Canada, France, Germany, and the United Kingdom. A prospective, nonrandomized cohort design, in patients receiving DLT defined as skin care, exercise, compression bandaging, and MLD, was performed for up to 4 weeks. A maintenance period of 5 months followed, using compression hosiery within a self-management plan.
Each country had two separate centers providing DLT. Each was an expert center offering treatment for all types of lymphedema and selected in each country by the principal investigator. The first center provided care based on its standard DLT protocol (but without Coban2) and the second center offered DLT integrating the Coban2 system. An observational design was chosen for this study as it was unclear which types of patients responded best to treatment.
To avoid cross fertilization of practice, the two compression systems were used in separate sites within each country. To avoid recruitment and selection bias, all suitable patients were included in a defined period of 9 months.
Inclusion criteria
Consecutive patients from each lymphedema service in eight sites requiring DLT were screened and provided written informed consent based on the following criteria:
Aged over 18 years. Confirmed clinical diagnosis of primary and/or secondary lymphedema. Clinical judgment that DLT was required (usually International Society of Lymphology [ISL] stage II and III). Ability to complete a period of DLT. Ability to provide written informed consent.
Exclusion criteria
Previous participation in the study.
A known history of nonadherence to lymphedema treatment.
End-of-life care.
International study coordination
An international steering group, including patient representatives, supported the project. Each country had a principal investigator who ensured ethical approval was obtained. All sites used a standard protocol and operating procedures and conformed to international guidelines for research.17,18 Data were collected by trained staff using an electronic case report form. The system allowed for audit and quality control, supplemented by a 6-month monitoring of source data by an international study monitor.
Variables
Data collected included the following: demographics, body mass index (BMI kg/m2), comorbidities, duration of edema, and mobility status. Site of swelling edema was recorded using a body map and classified as primary/congenital or secondary/acquired lymphedema. Severity of edema was assessed, with the ISL-staging tool, by palpation and clinical evaluation. 19 The different elements of DLT treatment were recorded for both groups. All adverse events were recorded in both groups.
Limb volume calculation methods
Lymphedema volume was calculated for each 10cm segment of the limb using the truncated cone formula. 20 This method has demonstrated excellent inter- and intraobserver reproducibility in comparison with water displacement, which is referred to as the gold standard. 21 Absolute and percentage change was recorded on the affected and contralateral limbs at entry to the study and on a weekly basis during bandaging and at the final visit (maintenance phase). For those with bilateral disease, the limb with the largest baseline volume was selected for treatment. In the present analysis, only the patients with swelling of the leg were included.
Intervention: treatment regimen
Decongestive lymphatic therapy
An overview of the intervention is seen in Figure 1. DLT was delivered by specialist-trained physiotherapists and nurses in all centers. Recommendations for DLT were based on published recommendations for skin care, exercise, MLD, and compression bandaging.7,8 In the standard treatment group, multilayer bandaging consisted of a tubular protective layer (cotton) and a layer of padding (foam or cotton wool), followed by layers of inelastic (short stretch) bandages applied at full stretch and adapted for limb shape and distortion.7,8 The range of inelastic bandages used in the different countries included the following: Rosidal K (Lohmann Rauscher), Comprilan (BSN), Actico (Activa Healthcare), and Fixierbinde (Rossmann). Treatment was provided daily (Monday to Friday) for a period of up to 4 weeks. Completion was either because the lymphedema was judged to be stable (based on limb volume measurements) or because the treatment episode was concluded. This occurred in some centers that had an allocated number of visits for a DLT treatment episode. Patients in the Coban2 arm had the system replaced twice a week or according to clinical need. Reasons for rebandaging were recorded at each change.
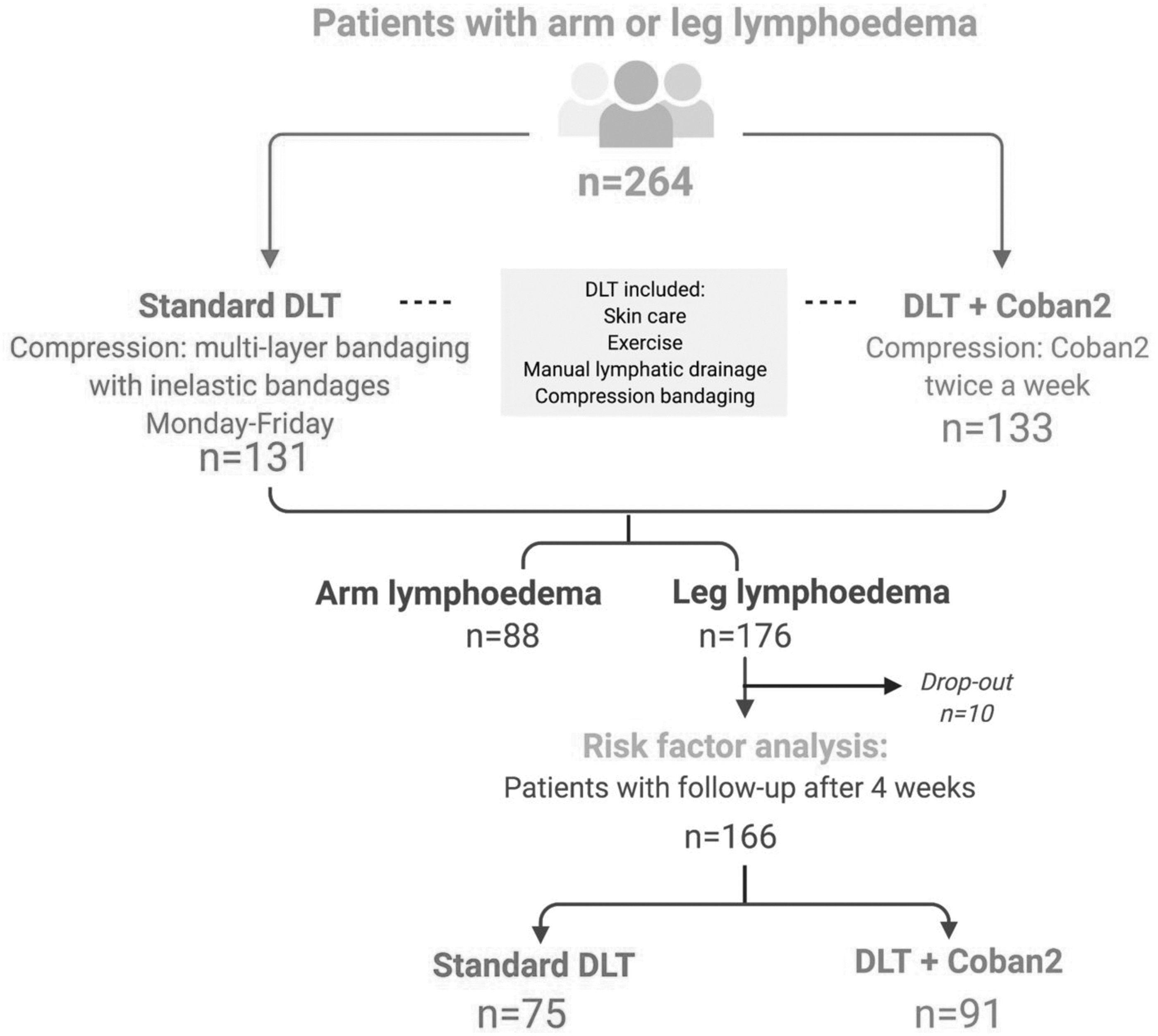
Study flow diagram. DLT, decongestive lymphatic therapy.
Maintenance treatment
On cessation of bandaging, patients were measured and fitted for an appropriate compression garment according to clinical need. Results from the maintenance arm of this study will be published separately.
Statistical analysis
Sample size calculation
To ensure 90% power, a sample size calculation for the main study of 264 (132 per group) was determined using results from a previous randomized-controlled trial in which the primary analysis was the comparison between two treatment methods (Coban2 and Comprilan). The mean reductions in the two groups were 14.56% (standard deviation [SD] = 16.72) and 9.53% (SD = 6.07), respectively, with large SDs in both groups.
Analysis plan
Statistics were prepared using Stata 12 (StataCorp, Texas). Initial univariate analysis was undertaken using standard parametric methods, either Student's t-test for comparison of two groups or one-way analysis of variance (ANOVA) for more than two groups.
The statistical method used for the analysis of risk factors was multiple regression analysis with the limb volume reduction as the dependent variable. Stepwise elimination of factors was undertaken until all variables remaining in the model achieved a probability of p < 0.05. Categorical data results were tested using the likelihood ratio test. Continuous covariates were centered around the median value as described in each of the derived models.
Independent variables considered for inclusion into the models were as follows:
Treatment group.
Demographics: age, gender.
Level of obesity (BMI).
Medical history: diabetes, renal disease, heart disease, arthritis, peripheral arterial disease (PAD), chronic venous insufficiency.
Disease factors: lymphedema classification, history of cellulitis, site of swelling, degree of fibrosis, lymphedema severity (ISL classification), presence of wound, lymphorrhea, current cellulitis.
A further similar analysis examined the effect of increased treatment duration in a subgroup of 65 patients. Missing data were not imputed and therefore remained missing.
Results
Comparison of treatment groups
Of the total 264 patients recruited into the main study, 133 were allocated to Coban2 in the four centers using this bandage, with 131 using their standard care in the other 4 centers (Fig. 1). Table 1 gives details of the presenting data in the two groups, showing a degree of difference between the groups. Osteoarthritis was more prevalent in the Coban2 group and was more likely to be treated for 4 weeks. Cellulitis was more common in this group, with an overall longer duration of lymphedema. There was a mixed pattern of severity, with more patients in stage III with Coban2, but a greater proportion with late-stage II receiving standard care. Use of multilayer bandaging was 100% in both groups, with a smaller number receiving MLD. Despite recommendations for DLT being based on international guidance, differences were also found between the elements of DLT provided in the two groups (Table 2). Higher levels of advice on exercise were delivered to the standard group compared with the Coban2 group (p < 0.001), with skin care advice recommendations higher for the standard care group (p = 0.37). Rates of MLD were similar between groups and delivered to 59.40% and 58.78%, respectively.
Comparison of Treatment Groups at Entry into the Study in Patients with Arm or Leg Lymphedema (N = 264)
BMI, body mass index; CVI, chronic venous insufficiency; ISL, International Society of Lymphology; PAD, peripheral arterial disease; SD, standard deviation.
Difference in Components of Decongestive Lymphatic Therapy Delivered in Patients with Arm or Leg Lymphedema (N = 264)
MLD, manual lymphatic drainage.
In total, 126 patients in each group had completed the intensive therapy regimen using their specified bandage, Table 3. The mean reduction was 941 mL in those on Coban2 compared with 814 mL on standard care. The mean difference of 127 mL did not approach a standard level of statistical difference (p = 0.53). Three variables remained in the model following a multivariable backward elimination process, which were ISL scale, presence of osteoarthritis, and the initial limb volume. The inclusion of these three factors into the model reversed the difference between treatments giving the standard care a greater limb volume reduction of 99 mL (95% confidence interval [CI] −240 to 438 mL). Again, the result did not achieve a statistically significant result as indicated by the wide CIs (p = 0.57).
Treatment Outcome in Patients with Arm or Leg Lymphedema
CI, confidence interval.
Adverse event and loss to follow-up
In total, seven patients on Coban2 failed to complete their treatment compared with five in the standard care group. There were eight adverse events in the Coban2 group and six in standard care (Table 3). None of the adverse events was considered serious by the clinicians who managed the patients.
Factors associated with greater limb volume reduction in patients with leg lymphedema
Of the 176 (66.6%) who experienced swelling of the leg, 166 (94.3%) had a follow-up measurement after up to 4 weeks of intensive therapy and were included in this analysis.
Demographic data showed 132 (79.5%) were female with a mean age of 60.1 years (SD = 14.7) (Table 4). Mobility was poor with 88/154 (57.1%) requiring assistance. As expected, obesity and morbid obesity were common affecting 95/159 (59.89%). In total, 102/163 (62.6%) had secondary lymphedema. Swelling was of long duration with 75/161(46.6%) suffering for more than 10 years. Many had severe forms of lymphedema, with significant tissue changes with 99/166 (59.7%) classified ISL late-stage II or stage III. Complications such as a current cellulitis were present in 9/164 (5.5%), with 29/166 (17.5%) reporting a history of previous episodes of cellulitis and 15/164 (9.1%) having a concurrent wound.
Demographics and Medical History of Patients with Leg Lymphedema (n = 166)
DLT, decongestive lymphatic therapy.
Factors associated with changes in limb volume reduction: univariate analysis
Table 5 gives the univariate analysis in relation to the overall reduction in limb volume, with volume reduction given as a positive result. There was evidence of a slightly larger reduction in men compared with women although this did not achieve statistical significance (p = 0.40).
Factors Associated with Leg Volume Reduction (n = 166), Univariate Analysis in Milliliter
Factors that were associated with higher limb volume reductions included the presence of PAD (mean difference = 2530 mL, p = 0.008), osteoarthritis (mean difference = 787 mL, p = 0.028), and problems with mobility (mean difference = 889 mL, p = 0.004). There was some evidence of a greater reduction in those with heart disease, but this did not achieve statistical significance (p = 0.077).
Presence of a wound led to a greater reduction in limb volume (mean difference = 1095 mL, p = 0.032), including the presence of lymphorrhea (mean difference = 1506 mL, p = 0.0016). There was a slightly higher, although nonsignificant, reduction in volume in the Coban 2 group compared with standard care (p = 0.69).
Factors with more than two categories are presented in Table 6. There was evidence that patients with higher BMI experienced greater limb volume reduction (p = 0.031), together with those with more severe lymphedema as given by the ISL severity scale (p < 0.001). Patients with larger initial limb volume also exhibited greater limb volume reduction with a gradient from those with small limbs (<8000 mL) to those with a limb greater than 14,000 mL (p < 0.001).
Categorized Continuous Variables Associated with Leg Volume Reduction (mL)
Factors associated with changes in limb volume reduction: multivariable analysis
When corrected within the multivariable analysis, only three independent factors were found to be associated with limb volume reduction: initial large limb volume (p < 0.001), treatment duration of 4 weeks (compared with 2 weeks) (p = 0.010), and the presence of PAD (p = 0.015), Table 7.
Independent Factors Associated with Change in Leg Volume (r 2 = 0.394), Multivariable Analysis
Initial limb volume fitted as a continuous variable, centered around an approximate median (10,000 mL).
A subgroup analysis
A subgroup analysis was performed in the 65 patients with leg swelling who had volume measurements at both weeks 2 and 4, Table 8. At week 2, the mean reduction was 1359 mL in the Coban group compared with 944 mL in the standard group. Between weeks 2 and 4, there was a further reduction of 145 mL in the Coban group and 221 mL in the standard group. Combining groups led to a mean reduction of 1276 mL between baseline and week 2 and 160 mL between weeks 2 and 4, with an overall reduction of 1436 mL. The additional 2 weeks of intensive treatment led to a mean decrease of 160 mL, which is a further 12.5% reduction compared with the result at week 2 (160/1276 = 12.5%). As numbers are small and CIs are wide, no inferential statistics has been performed.
Subgroup Analysis
Changes in leg volume in 65 patients with follow-up measures at weeks 2 and 4.
SD, standard deviation.
Discussion
The current study has failed to find a substantial difference between the two groups of patients being treated with standard intensive therapy compared with the Coban2 system. It has highlighted that although the mean reductions were reasonable in both groups of over 800 mL, the SDs indicate the wide spread of data around these means.
The results from the risk factor study found three independent predictive factors, associated with significant limb volume reduction after 4 weeks of treatment with DLT, being large initial limb volume, longer duration of DLT (4 weeks compared with 2), and the presence of PAD.
Levels of severity defined by the ISL staging indicated this was a complex population of long-standing duration. Despite this, those with a large increased volume at entry to the study responded well to treatment indicating that the benefit of using DLT can be maintained despite disease severity. Large initial limb volume was found to be one of the three independent factors associated with volume reduction rather than disease severity (ISL). Studies indicate an excess volume reduction after DLT ranging from 20% to 73%, however, the heterogeneity of the patient population and different methods of treatment make comparisons of studies difficult to extrapolate. 22
Satisfactory limb volume reductions could be achieved both by standard DLT with inelastic compression bandages, and DLT that included Coban2, with no statistically significant difference between the two. A direct comparison between the two groups is, however, deeply challenged, due to significant differences in the patient characteristics at baseline (such as initial limb volume) and the treatment components of the DLT, such as the component of exercise. This is likely attributed to the observational study design. Despite access to recommendations for standard DLT, this study indicates that they are being interpreted and applied differently across the world. Standardization of DLT is difficult to achieve due to the use of many different products and the inability to accurately measure the dosage of treatment.
The high proportion of females in this study (79.5%) has been reported in others using a similar methodology. 23 However, the role that gender plays in determining the outcome of DLT remains obscure. Both gender and age distribution were comparable with the POLIT study; a national study in France investigating predictive factors for lymphedema reduction. 23 A community-based service delivering DLT in the United Kingdom also reported a high percentage of females (82%), but with a significantly older age group (72.9 years). 24
Levels of obesity were high compared with other studies, but this was not an independent risk factor for treatment outcome. The role of obesity in lymphedema is complex and may be linked with many factors such as reduced mobility, inability to elevate the limb, and increased comorbidities requiring complex medication regimens that influence edema control. Obesity in a breast cancer population reported a higher risk of developing lymphedema. 25 Other studies report the negative effect of weight gain on outcomes of DLT. 26 Caution should be made of extrapolating findings from a breast cancer population to a lower limb group as the mechanisms causing edema and response to treatment may be very different. The application of effective compression is known to be more challenging to achieve in the morbidly obese due to the overall increase in limb size and shape distortion. 27
In the POLIT study, previous episodes of DLT were found to be the only independent factor associated with treatment failure (odds ratio = 2.4, p = 0.002). 23 However, the current study did not examine this factor. These results raise the question of the criteria and timing for DLT with some evidence that in those who develop rebound edema and require repeat DLT, the effects may lessen over time. This has deleterious effects for patients, families, and is a high cost to the health care systems. The reasons are unclear, but the lack of treatment adherence would be worthy of further investigations.
There is lack of international agreement about the optimum length of a treatment episode. Clinicians report limb volume as the main method used to establish outcome, but large variations occur in how this is applied in practice. Length of treatment episodes may be imposed due to contractual arrangements with different health care providers. Data from the subgroup analysis would suggest that although the greatest volume reduction occurs in the early stages of treatment, nevertheless, longer duration of treatment was found to be an independent factor associated with better volume reduction. However, little is known of the additional cost-effectiveness of increasing the length of time on such a labor-intensive intervention. This hypothesis requires evaluation within well-designed randomized-controlled trials, designed specifically to address these issues. As DLT is a complex intervention, research is required to determine the relative contribution each element plays in different patient populations.
Many factors may influence limb volume, including changes in weight, medication, and the diurnal influence of when the measurement is taken.2,4,6 In addition, variations may occur in measurement between clinicians even when using standardized protocols. Accurate assessment of volume in the morbidly obese with large skin folds also may contribute to inaccuracy. 28 These effects may have less influence on identifying changes in those with a large limb volume compared with those with a small degree of swelling, in whom subtle changes may be lost due to measurement error.
The presence of PAD was surprisingly found to be an independent factor associated with a large limb volume reduction in this study. The presence of PAD was defined by clinicians without the requirement for standardized methods of assessment. It is therefore possible that this provides an inaccurate profile, given the known difficulty of accurately assessing PAD in lymphedema. Two factors include tissue changes that cause difficulty in palpation of pedal pulses and inaccurate findings from the use of Doppler to record an ankle pressure due to the inability to compress arteries. Therefore, patients with large initial limb volumes (another independent risk factor found) might have been classified as having PAD, biasing our data. International guidelines recommend that compression should not be applied if PAD coexists with lymphedema, however, recent research has indicated that current recommendations are too conservative, and that compression is safe and beneficial for a much wider group of patients, providing severe limb ischemia is excluded.7,8,29
Limitations
The study has several limitations that require exploration. While use of an observational design was chosen as appropriate due to lack of clarity on factors affecting outcome, randomized-controlled trials are required to establish the efficacy of the best treatment modalities. Despite the use of inclusion criteria, important differences were present in the two groups at commencement of treatment and in the treatments used. This significantly challenges a comparison and interpretation of the treatment outcome between the two study groups. As an example, the increased frequency of acute cellulitis at baseline in the Coban2 group could lead to less intense compression bandaging due to pain, thereby with a smaller limb reduction. Separating participants according to the type of limb (upper or lower) will also inevitably lead to a reduction in power due to the smaller numbers. Despite this, independent risk factors have been identified.
Conclusions
No significant differences in terms of leg volume reduction or adverse events were found between patients receiving standard care DLT, compared with DLT with Coban2. However, due to large baseline differences between the groups, a direct comparison is greatly challenged. The study found three independent factors for a positive leg volume reduction: large initial limb volume, DLT duration of 4 weeks (compared with 2), and PAD. Our findings confirm the complexity of factors that influence outcome in patients receiving DLT. Although clinical guidelines on DLT exist, these are subject to significant variation in the way they are applied in practice. Robust prospective randomized-controlled trials are required to understand the effects of these complex interventions and to determine the relative contribution of each aspect of treatment and to define an optimum length of treatment for different patient groups. The study concludes that significant limb volume changes occur even in those with severe leg lymphedema with large increased initial limb volumes.
Footnotes
Author Disclosure Statement
The company has not been involved in the analysis of the data or writing of the article, but has been given the opportunity to review the article before submission.
Funding Information
The research reported in this article was supported by a research grant from 3M Healthcare provided to the International Lymphoedema Framework (ILF), who were responsible for the design, implementation, and evaluation.
