Abstract
Abstract
Adrenomedullin (AM) and gastrin releasing peptide (GRP) are neuroendocrine peptides that have been previously implicated as regulators of angiogenesis and lymphangiogenesis. Using an immortalized human dermal microvascular lymphatic endothelial cell line stably transfected with red fluorescent protein (LEC/RFP), we demonstrate the ability of AM and GRP to augment tube formation complexity of this target cell in a dose-dependent manner. Maximum tube density was initiated at 1nM for both peptides, and as concentrations exceeded 10 nM a decrease in tube formation was noted, hence following a classic rise/fall biological response curve. In addition, we show that appropriate small molecule mimetics to neutralizing monoclonal antibodies of AM or GRP, at 1 μM concentration, can function to either inhibit (antagonist) or enhance (super agonist) peptide-induced tube formation of LEC/RFP. Our small molecule reagents by themselves have no activity, but in the presence of their respective peptides can mediate a positive or negative response, hence the super agonist designation. These compounds represent new regulatory drugs of the lymphatic system with possible patient application in the clinical management of edema and metastatic disease.
Introduction
The identification of strategic drugs that regulate the proliferative components of lymphangiogenesis has been a challenge for clinical investigators over the past several decades. Recent findings have revealed that certain neuropeptides can modulate endothelial cell growth events and may offer rational targets for drug development. Two such entities, adrenomedullin (AM) and gastrin-releasing peptide (GRP), both amidated peptides, have been found to be mitogenic for endothelial cells.21–26 The only known carboxy-terminal post-translational modification of proteins that consistently tracks with bioactivity is amidation, a process that requires a unique amino acid recognition motif in the prohormone molecule which in turn encodes for a series of consecutive enzymatic steps that ultimately leads to peptide amide formation.27–29 Figure 1 summarizes the amino acid (AA) motif encoded in the prohormones of AM or GRP that dictates the amidation process to take place via a series of enzymatic events (trypsin-like cleavage between Arg-Ser or Lys-Ser, several rounds of carboxypeptidase hydrolysis to remove the basic AA, processing of the glycine-extended intermediate compounds [-GlyTyr-Gly or -Leu-Met-Gly] by the peptidyl-alpha-amidating monooxygenase complex, and finally terminating in amidated AM or GRP) as shown.27–29 The free acid and glycine-extended intermediates of AM or GRP are several orders of magnitude less potent than the corresponding peptide amide when tested in a variety of biological systems.27–29 Drugs that target either the carboxy-terminal amide region of AM and GRP or the amide conformational recognition site on their cognate receptors will effectively block the peptide's biological activity by steric interference with ligand/receptor binding.23,30,31 AM has now been shown to be an important stimulator of lymphatic vascular development during embryogenesis and an ameliorator of lymphedema.32,33 Over two decades ago GRP was found to be a peptide product of lymphatic vessels that regulated the function of this network system in an autocrine/paracrine manner. 34 We have previously reported on the development of monoclonal antibodies that target the carboxy-terminal amide of either AM or GRP, denoted as MoAb-G6 and MoAb-2A11, respectively.30,31 MoAb-G6 did not cross react with GRP nor did MoAb-2A11 bind AM. These antibodies were shown to form immune complexes with their respective peptide immunogens and to block the biological activity of these peptides in a variety of in vitro and in vivo assay systems.30,31 As illustrated in Figure 2, we have recently utilized these neutralizing monoclonal antibodies to establish a high throughput screening strategy for identifying small molecule mimetics to these immune reagents. 35 These small molecule compounds (Fig. 3) were shown to function as augmenters or suppressors of AM or GRP bioactivity.26,35 In the following text, we will demonstrate AM and GRP as inducers of lymphangiogenesis and utilize our small molecule reagents to modulate (antagonist and super agonist) the biological activity of these peptide amides on lymphatic endothelial cells.
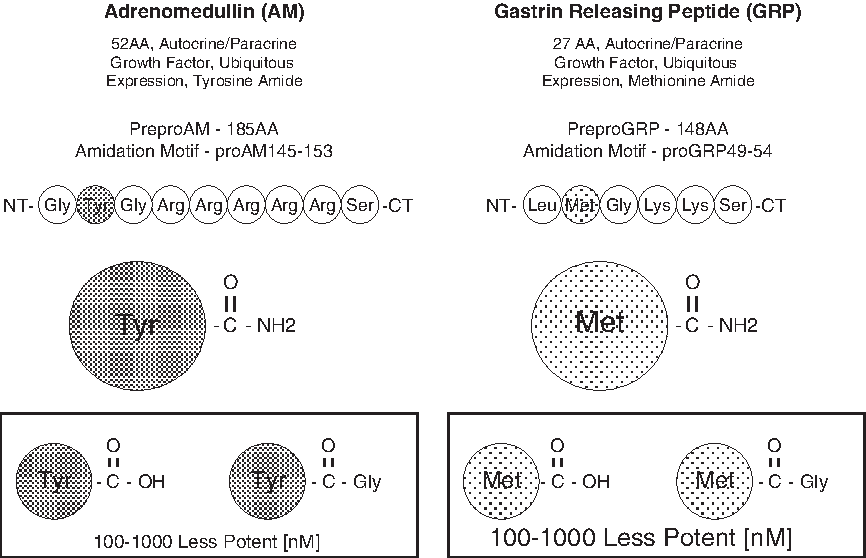
Carboxy terminal amidation of peptides: a hallmark of peptide biological activity.27–29 Two such amidated peptides, AM and GRP, are proven inducers of neovascularization and have recently been implicated in lymphangiogenesis.21–26, 32–34 A distinct amino acid (AA) motif within the prohormone signals for amidation to take place and consists of a target AA to be amidated (any AA), followed by glycine (only known amide donor AA), followed by a series of basic AAs (arginine or lysine). An initial trypsin-like cleavage event releases the amidation motif from the prohormone, followed by carboxypeptidase processing, giving rise to a glycine-extended intermediate product. This intermediate then serves as the substrate for a unique series of enzyme complexes known as peptitdyl-glycine–amidating monoxygenase that ultimately generates the amide of glycine's penultimate neighbor AA. The prohormone amidation motifs for AM (a tyrosine amide) and GRP (a methionine amide) are shown. The glycine intermediate or free-acid derivatives of both AM and GRP are 100 to 1000 time less potent on a molar basis than their respective amide counter-part depending on the biological assay used (boxed structures).
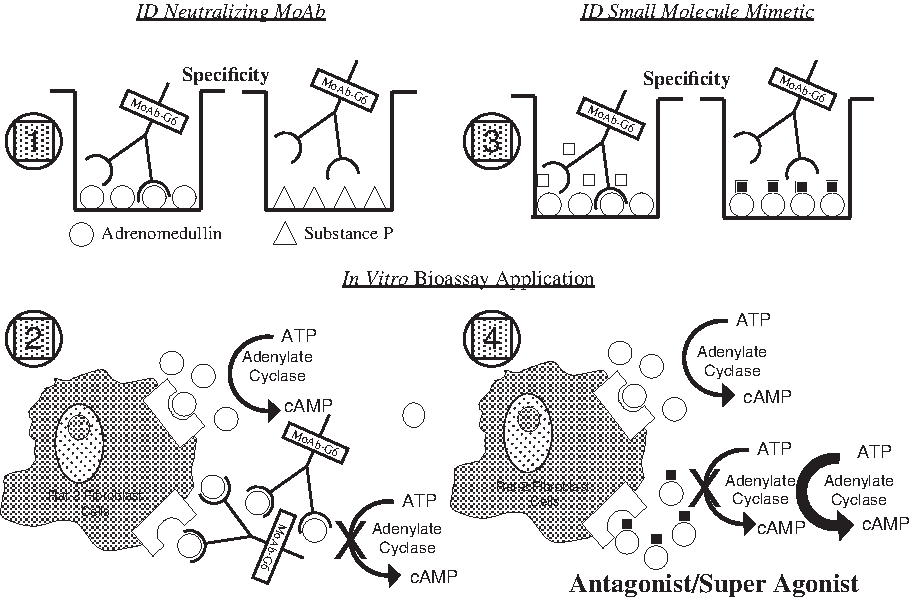
Small molecule drug screening approach. Screening strategy design for high throughput analysis using a 96-well microtiter plate format and based on prior NCI technology. 35 Section No. 1 indicates the initial testing of hybridoma products for ligand binding specificity. Here AM (a tyrosine amide) was used as the immunogen and the solid phased target peptide, while substance P (amethionine amide) was used as a negative control peptide. 31 Section No. 2 depicts the bioassay used to identify preselected anti-AM monoclonal antibodies (MoAb) as peptide neutralizing reagents. AM induces Rat-2 fibroblast cells to concert ATP > cAMP and MoAb-G6, identified in Section No. 1, blocks that event. Section No. 3. A secondary binding assay was established involving MoAb-G6/AM interaction and the NCI/Developmental Therapeutics Program (DTP) diversity set of 2000 small molecules family members (representing 140,000 compounds) were screened for their ability to inhibit ligand/antibody complex formation. Section No. 4. Candidate compounds making it through this initial screening were then evaluated in the Rat-2 fibroblast assay for their effects on AM-induced cAMP production. As indicated, small molecule AM antagonist and super agonist compounds were found using this collective screening strategy. 35 An identical collective screening approach was used to identify small molecule regulators of GRP bioactivity with MoAb-2A11 serving as the neutralizing anti-GRP antibody.30,35
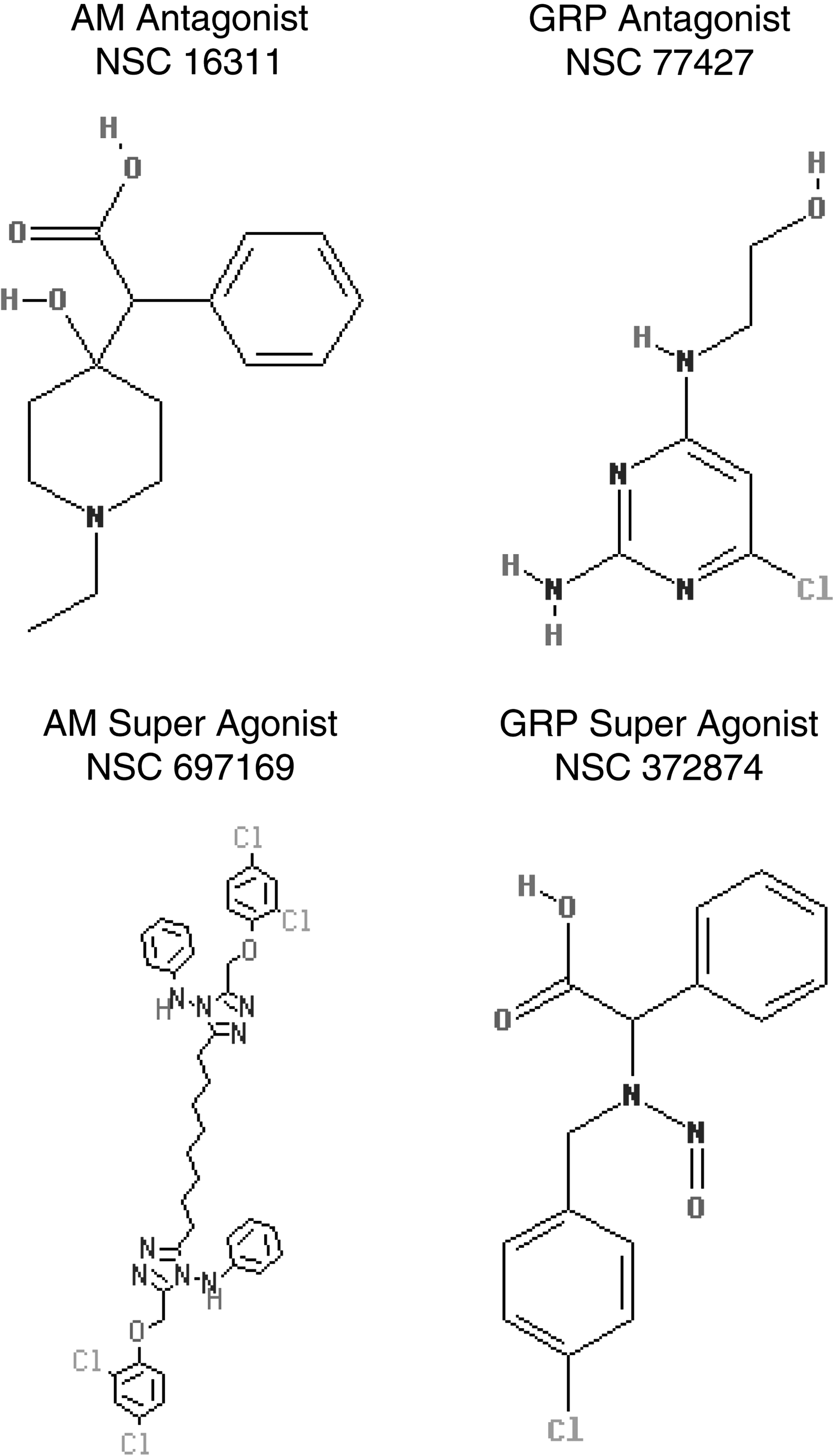
Chemical structure of small molecule AM/GRP antagonists and super agonists. Average formula weight (FW) approximately 300 daltons (NSC 697169 exception with FW 781).
Materials and Methods
Cell line
A telomerase immortalized human dermal microvascular lymphatic endothelial cell line (hTERT-HDLEC) was acquired through a NIH Material Transfer Agreement with Dr. Michael Pepper (Department of Morphology, University Medical Center, Lausanne, Switzerland). 19 hTERT-HDLEC were stably transfected with pDsRed-C1 (Clontech Laboratories, Inc., Mountain View, CA, USA) using standard AMAXA (Lonza Walkersville, Inc., Walkersville, MD, USA) protocols, G418 (Invitrogen, Life Technologies Corporation, Carlsbad, CA, USA) selected, and FACS sorted for highest percent of fluorescent cells and highest intensity of fluorescence. The resulting cell population was denoted as LEC/RFP. LEC/RFP were maintained in EMB2 complete medium (Lonza).
Small molecule drugs
Small molecules NSC 16311 (AM antagonist), NSC 697169 (AM super agonist), NSC 77427 (GRP antagonist), and NSC 372874 (GRP super agonist) have been previously described. 35
Tube formation
LEC/RFP were grown to 60% confluence in EMB2 complete medium (Lonza) and re-fed 24 h before use in the tube formation assay. 50 μl of chilled geltrex (Invitrogen) was added to each well of a pre-chilled 96-well microtiter tissue culture plate (Costar, Corning Inc., Corning, NY, USA) and transferred to a 37°C/5% CO2 incubator until solidified (approximately 45 min). Cells were trypsinized, washed three times in PBS, and resuspended in EBM2 +0.5% fetal bovine serum (FBS) at a final concentration of 380,000 cells/ml. AM and GRP were evaluated for the ability to induce LEC/RFP tube formation over a nanomolar dose range covering several orders of magnitude (0.01 nM, 0.1 nM, 1.0 nM, 10 nM, and 100 nM). Small molecule regulators of AM or GRP were used at 1 μM. 50 μl LEC/RFP cell suspensions (19,000 cells) under specified test conditions were added in quadruplicate to the microtiter plate and incubated at 37°C/5% CO2 for 3.5 h before assessing the complexity of tubes formed by fluorescence microscopy.
Results
AM- and GRP-induced dose response on tube formation
Figure 4 demonstrates AM- and GRP-mediated tube formation capabilities when tested over a dose range of 0.01 nM to 100 nM on LEC/RFP target cells. A classic rise/fall (biphasic) dose response curve was generated for both peptides (36), maximizing at 1 nM and dropping as concentrations exceed 10 nM. Although AM promotes a steeper dose response curve than GRP, the latter peptide appears to be more potent on a molar basis given the augmentation in network assembly noted at the 0.01 nM dose when compared to an equivalent AM concentration. In addition, several FBS concentrations (0, 0.5% and 5%) were evaluated to determine the ideal serum supplementation level to incorporate into the assay for maximum sensitivity of results. 0.5% FBS was chosen for all follow- up tube formation assays in these series to overcome zero serum threshhold but still offer a large dynamic range for peptide induced responses to be observed.
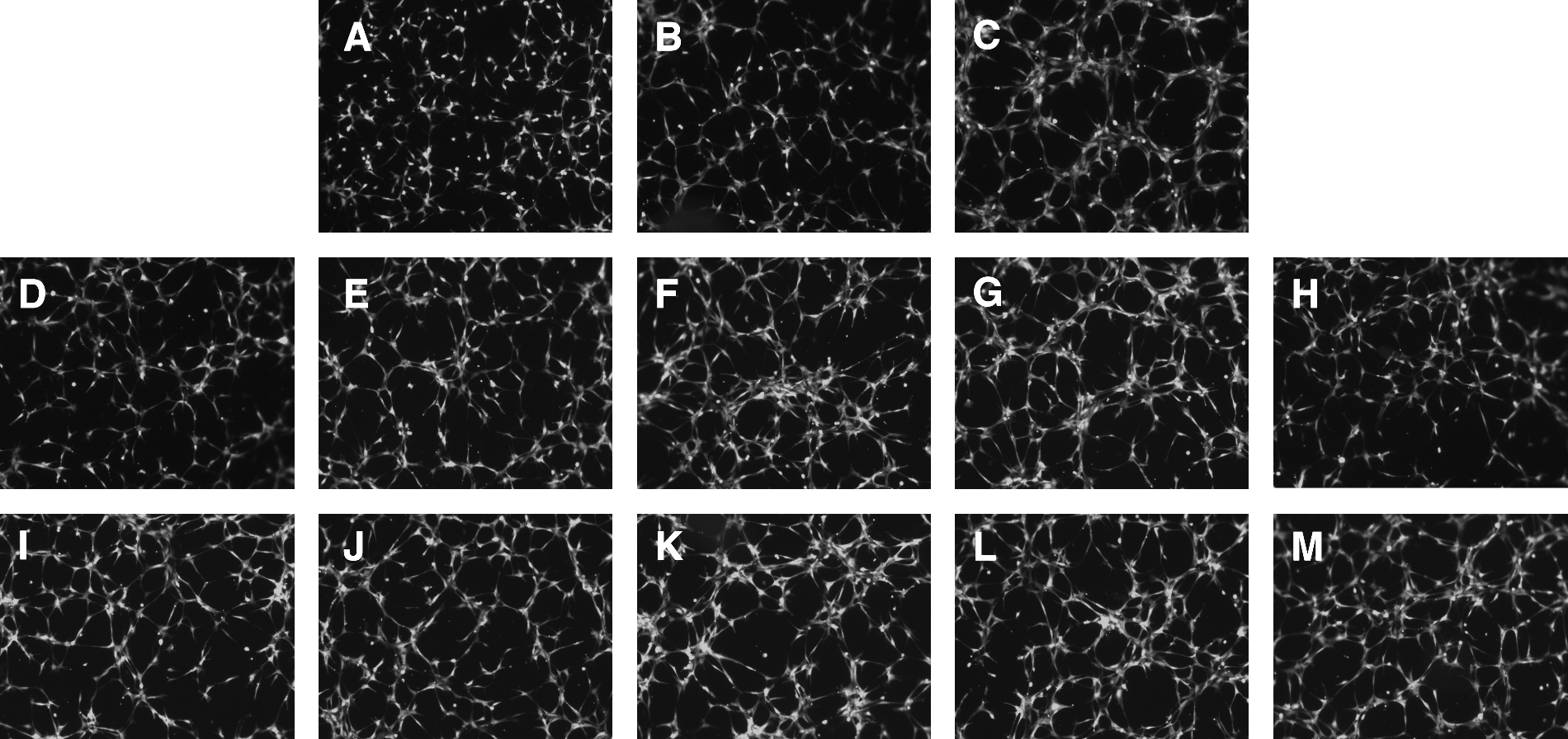
In vitro tube formation assay. Telomerase immortalized human dermal lymphatic endothelial cells stably transfected with RFP (LEC/RFP) were utilized in this 96-well analysis, seeding density of 19,000 cells/well, and evaluated after 3.5 h incubation at 37°C/5% CO2.
Small molecule regulators of AM- or GRP-induced tube formation
Figure 5 summarizes the LEC/RFP tube formation results obtained by varying the FBS concentration over a dose range of zero, 0.5% and 5%, thus serving as a comparison standard for peptide-induced network complexity with or without small molecule regulators. For all follow-up studies, a 0.5% serum supplementation was used. Peptides were evaluated at 0.1 nM, a concentration below their maximum stimulatory dose to enhance the sensitivity of the assay. Small molecules were initially tested at 1 μM concentration based on our prior working experience with these drugs for in vitro assay systems, assuring us the possibility of seeing a valid response.26,35 AM and GRP at 0.1 nM augmented the resulting network complexity of LEC/RFP over that seen for 0.5% FBS in the absence of peptide. In addition, the AM antagonist (NSC 16311) and the GRP antagonist (NSC 77427) markedly suppressed peptide induced tube formation. Conversely, the AM super agonist (NSC 697169) enhanced network complexity while the GRP super agonist (NSC 372874) appeared to do little to elevate the response over that of the peptide alone, possibly reflecting the muted biphasic response curve of this peptide.
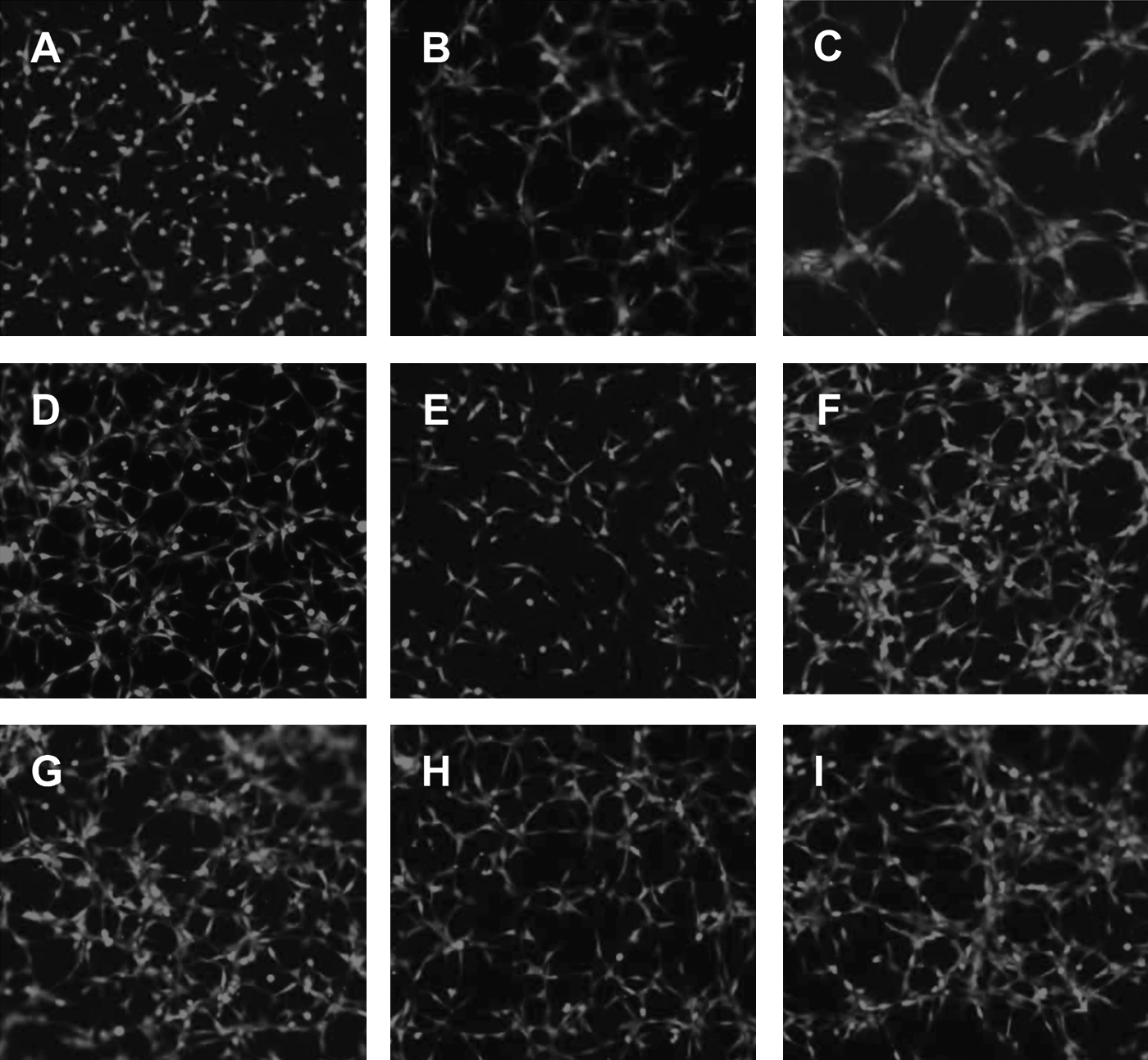
Testing small molecule regulators of AM and GRP in the tube formation assay. An identical assay as described in Figure 4 was established to evaluate the ability of small molecule antagonists and agonists to regulate peptide-induced tube formation with LEC/RFP target cells.
Discussion
Here we report on the use of these small molecule compounds to enhance or suppress AM- and GRP-induced lymphatic endothelial cell tube formation. Tube formation, although not involving a proliferative component, does reflect key aspects of lymphangiogenesis, namely cell migration and organizational establishment of a network infrastructure. Figure 4 demonstrates the potential for AM and GRP to drive these aspects of lymphangiogenesis in a dose-dependent manner. Subnanomolar concentrations of either AM or GRP can mediate lymphatic endothelial cell tube formation, thus implicating a possible physiological role for these peptides in lymphatic network development in vivo. This supposition has been substantiated by published findings from other laboratories.32,33 A biphasic response curve was generated over the peptide dose range assessed. This observed rise/fall response tends to be indicative of either receptor dimerization or the presence of high/low affinity receptors.37,38 When receptor dimerization is involved, low peptide concentrations maximize receptor cross-linking, augmenting the response whereas high peptide level drive monomer formation muting the peptide induced effect. 37 Alternatively, when different receptor affinities underlie the biphasic response, low peptide concentrations preferentially triggers the high affinity receptor while high peptide levels activate the low affinity receptor. 38 Interestingly, high/low affinity receptors are known to exist for both AM and GRP.39,40
In Figure 5, when applying our small molecule regulators to AM or GRP induced lymphatic network assembly in vitro, we clearly see suppressive effects with the AM antagonist (NSC 16311) and GRP antagonist (NSC 77427) used at a 1 μM concentration.
Conversely, the AM super agonist (NSC 697169) augments tube formation while the GRP super agonist (NSC 372874) does not show an enhancement of vascular complexity over that seen by GRP alone. This NSC 372874 effect may be a reflection of the shallow dose response curve observed for GRP-mediated tube formation or possibly be indicative of suppressive effects observed at the higher peptide concentrations of the biphasic response. Evaluating GRP at a diminished concentration (0.01 nM or lower) + NSC 372874 may improve the visualization of a super agonist effect. Interestingly, small molecules we defined as super agonists by themselves have no effect; they only induce their stimulatory influence in the presence of their respective peptides. In addition, the observed super agonist's enhancement of peptide potency is not dependent on increasing ligand/receptor affinity but potently involves the masking of enzymatic degradation sites on AM/GRP thus extending the biological half-life of the peptide. 41
Recent evidence has suggested that both AM and GRP can function as cell mitogens either directly through designated receptor binding or indirectly by transactivation of an unrelated secondary receptor system.24,42 When AM binds to its respective cognate receptor AM1 (CRLR/RAMP2) on endothelial cells, a transactivation of the VEGFR2 (Flk1/KDR) takes place, triggering the VEGF-A pathway independent of the VEGF-A ligand and resulting in endothelial cell proliferation. 24 Similar transactivation effects have been identified for GRP interaction with the GRP receptor (GRPR), stimulating the epidermal growth factor receptor (EGF) independent of ligand and augmenting the growth of human squamous cell carcinomas. 42 Given that AM and GRP can influence biological events via transactivation of secondary receptor systems, our small molecule regulators may also be having both direct and indirect effects on peptide-mediated lymphangiogenesis. This becomes particularly important considering that AM and GRP are products of squamous cell carcinoma and this tumor type has recently been implicated in a VEGF–C/Flt4 autocrine loop mechanism regulating tumor growth with paracrine modulation of tumor-associate lymphangiogenesis.42–44 Hence this squamous cell carcinoma tumor model may be an ideal system to test the possible indirect influence of AM or GRP on tumor-associated lymphangiogenesis and to determine how our small molecule regulators may influence these potential interactions.
Critically important in moving the lymphatic field forward is the development of new assay systems representing in vitro or in vivo approaches to lymphangiogenesis. Several new discoveries have exemplified these types of investigative advancements incorporating three-dimensional designs of lymphatic endothelial cells in gel matrices to mimic physiological conditions or identifying anatomical sites in host animals readily assessable for monitoring inducible lymphangiogenesis.16,17,45 Such test systems offer investigators the opportunity to identify novel drugs that modulate lymphatic network complexity or uncover naturally occurring mechanisms regulating lymphangiogenesis as was recently seen with the discovery of a soluble alternatively splice variant of VEGFR-2 (sVegfr-2) that inhibits lymphatic hyperplasia but not angiogenesis.46,47 It should be noted that our high throughput screening strategy for identifying small molecule mimetics to neutralizing monoclonal antibodies can also be applied to ligand/receptor interactions, and the VEGF-C/sVegfr-2 model would be an interesting system to examine for antagonist/agonist small molecule regulators.
In conclusion, we have demonstrated the ability of AM and GRP to mediate LEC/RFP tube formation in a dose-dependent manner. Subnanomolar concentrations of both peptides clearly augment lymphatic vessel density over that observed in the absence of peptide, hence implicating a possible physiological role for these peptides in lymphangiogenesis in vivo. This hypothesis has in fact been validated by published studies from other laboratories.32–34 We have also shown that small molecule mimetics to neutralizing monoclonal antibodies for AM/GRP can regulate peptide-induced LEC/RFP tube formation. The AM antagonist (NSC 16311) and GRP antagonist (NSC 77427), at a 1 μM dose, suppressed peptide mediated enhancement of LEC/RFP vessel density. It would be interesting to evaluate these reagents for their anti-metastatic effects on murine models of breast or lung cancer, given the known lymphatic dissemination events underlying the spread of these tumors to distal anatomical sites and the evolutionary conservation of AM/GRP among mammalian species. In addition, our small molecule super agonist for AM (NSC 697169) was shown to enhance peptide-induced LEC/RFP tube formation while a parallel reagent for GRP (NSC 372874) was not effective in stimulating network density. This negative result may be caused by a variety of influencing factors previously discussed in this section, and the drug should not be discarded without further testing, especially since it was proven to be an enhancer of GRP activity in other biological systems. Given the established mouse model used to validate AM's influence on adult lymphangiogenesis and lymphedema,32,33 it would be interesting to see if our small molecule regulators of AM bioactivity can augment or suppress AM-mediated lymphatic events in this in vivo system. Small molecule compounds represent an ideal drug format because of their extremely diffusible size (usually <400 daltons), resistance to proteolytic degradation in the circulation, ease of generating structural modifications via combinatorial chemistry, and low GMP cost for certified FDA therapeutic production compared to neutralizing monoclonal antibodies. To summarize, we have identified several small molecule compounds that effectively modulated the stimulatory effects of AM/GRP on lymphangiogenesis, thus having potential clinical use in combating lymphatic disorders.
Footnotes
Acknowledgments
This research was supported by the intramural research program of the NCI, NIH, DHSS under Project No. BC010964-01 CCRO. NCI small molecule intellectual property is covered under U.S. patent application 10/57 1,012. The content of this publication does not necessarily reflect the views or policies of the DHSS nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. Government.
Disclosure Statement
The authors of this article, Changge Fang, Marta Aparicio Miguel, Ingalill Avis, Alfredo Martinez, Enrique Zudaire, and Frank Cuttitta, have no conflicts of interest or financial ties to disclose.
