Abstract
Background:
To compare tumor margins and surgical outcomes between transanal minimally invasive surgery (TAMIS) and endoscopic submucosal dissection (ESD) for large or malignant rectal adenomatous polyps.
Methods:
Single institution retrospective analysis of patients who underwent TAMIS or ESD surgery.
Results:
In total, 30 consecutive patients with similar demographics who underwent either TAMIS (n = 19) or ESD (n = 11) were included. The median (interquartile range, IQR) tumor distances from the anal verge for TAMIS and ESD were 5 cm (3.5–8) and 3 cm (2–4.25) (P = 0.016). Four in TAMIS and two in ESD occupied more than half of the circumference of the bowel lumen. Five (four in situ and one stage 1) in TAMIS and two (one in situ and one stage 1) in ESD were malignant. The median specimen length, width, and height were 3.2 cm, 2.6 cm, and 1.0 cm and 3.5 cm, 2.0 cm, and 0.3 cm for TAMIS and ESD, respectively. There were no statistically significant differences in tumor circumference, malignant ratios, or specimen sizes. Resection margins were involved in two of the ESD, while none of the TAMIS were involved (P = 0.041). The median (IQR) operative time was 72 (62–89) minutes and 120 (90–180) minutes for TAMIS and ESD (P = 0.005). The median (IQR) follow-up time was 3.3 (0.3–11.7) and 0.9 (0.3–15.4) months for TAMIS and ESD. There were no morbidities, no mortalities, or local recurrences among the two groups.
Conclusions:
Both TAMIS and ESD were found to be feasible and safe in community hospital practice. Operative time was shorter, and there were no involved margins in TAMIS (versus ESD).
Introduction
The increasingly widespread application of endoscopic screening has improved the early detection rate of early (pTis and pT1) rectal cancer. 1 Early identification of a rectal precancerous tumor is usually managed by local excision to avoid morbid outcomes, such as urinary or sexual dysfunction, and the need for colostomy. However, precancerous adenomatous polyps still carry a 1–2% chance of malignant recurrence at the removal site. 2 Colonoscopic snare polypectomy is not recommended for polyps larger than 2 cm. 3 Endoscopic mucosal resection (EMR) is recommended for polyps larger than 2 cm. For those polyps larger than 4 cm or possibly malignant, a more advanced procedure is needed. Based on the technological advances in imaging systems, energy devices, and minimally invasive instruments, both transanal minimal invasive surgery (TAMIS) and endoscopic submucosal dissection (ESD) are effective alternatives for treating large or malignant rectal polyps.
TAMIS, first introduced by Atallah et al.,
4
is performed using laparoscopic instruments through a disposable single-port device inserted transanally. Currently, two ports are available: the SILS
Another surgical option is ESD. Initially applied to gastric tumors 11 and then harnessed for treating colorectal lesions,2,7,12 ESD uses an IT Knife 13 or Dual Knife 14 : adequate view of lesions hidden on the opposite side of rectal folds can be obtained with the distal attachment. 15
Both TAMIS and ESD rely on magnified vision. The only difference is that TAMIS offers two or three working channels for instruments, while only one is available for ESD.
The purpose of this study was to compare the tumor margins and surgical outcomes between TAMIS and ESD in large or malignant rectal adenomatous polyp excisions.
Materials and Methods
We reviewed the records of 30 patients who underwent TAMIS (n = 19) or ESD (n = 11) at our institution between July 2019 and February 2023 (Fig. 1). We excluded those with lesions that were not adenomatous or premalignant polyps or <2 cm in diameter.
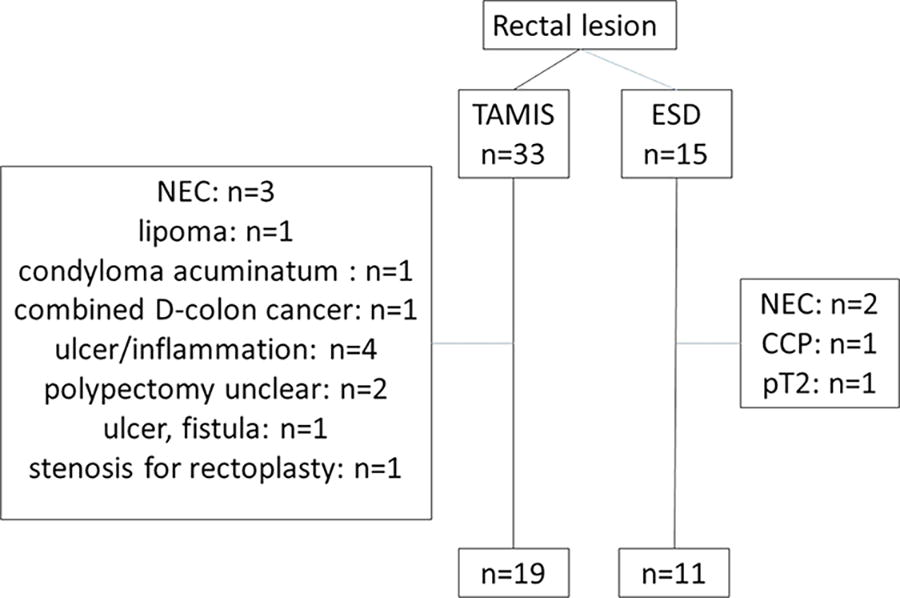
Flowchart of the patient selection. CCP, colitis cystica profunda; ESD, endoscopic submucosal dissection; NEC, neuroendocrine carcinoma; pT2, pathology T2; TAMIS, transanal minimally invasive surgery.
Demographic information including age, sex, body mass index (BMI), American Society of Anesthesiologists (ASA) class, tumor size, tumor location, surgical procedure, operative time, operative blood loss, pathological report, postoperative hospitalization, antibiotic usage, and perioperative complications were collected prospectively.
The primary end point was specimen margin status. The secondary end point was surgical complications.
The study was approved by the Department and Institutional Ethical Committees of the China Medical University, Taichung, Taiwan (Registered No: CMUH112-REC3-173).
Preparations before surgery
In addition to routine colonoscopy, magnetic resonance imaging or computed tomography were performed as necessary. The laterally spreading type polyp 16 or Japan NBI Expert Team classification17,18 class 2A or 2B lesions were included. If early malignancy was detected, re-evaluation was initiated after further discussion by the multidisciplinary team.
Depending on the clinical need, either antegrade bowel preparation 1 day before surgery or cleansing enema on the morning of the operative day was arranged.
TAMIS surgical technique
One gram of Flomoxef was administered intravenously prior to surgical incision. The patient was placed in the lithotomy position, under general anesthesia, and draped according to sterile protocol. After anal dilatation and application of a LoneStar® retractor (Cooper Surgical, US), a lubricated GelPOINT® Path (Applied Medical, Rancho Santa Margarita, CA, USA) was gently introduced into the anal canal under steady manual pressure. Once seated in position, a gauze was inserted into the upper rectum for partial barricade of the pneumorectum. Three self-retained sleeves, two 5 mm and one 12 mm cannula with access channels, were introduced into the GelSeal Cap. Next, another insufflation-stabilized bag was connected and a 12–18 mmHg pneumorectum was established. A 3D laparoscopic (Endoeye Flex 3D; Olympus, Tokyo, Japan) was used to visualize the rectal vault. A monopolar or HybridKnife® was used to mark out a 5 mm tumor margin. The waterjet system of HybridKnife® (Erbe Elektromedizin GmbH) (normal saline mixed with indigo carmine injected into the submucosa) was used to separate the surgical plane. The monopolar or VIO® (Erbe Elektromedizin GmbH) was used for dissection and coagulation. A laparoscopic grasper was used for assistance. After tumor excision and specimen removal, the wound was left open or closed with V-LOCTM or Vicryl® sutures depending on the surgical site and the discretion of the attending surgeon.
ESD surgical procedure
The patient was placed in the left decubital position and given intravenous anesthesia. The ESD procedure was carried out by a single, unassisted gastrointestinal expert using a transparent cap (D201-10704; 4 mm Olympus) fitted endoscope (PCF-H290I, Olympus, Tokyo Japan). After CO2 insufflation, a solution of Glycerol 500 cc mixed with Indigo 3 cc and Bosmin .5 cc was injected through an Injection Therapy Needle Catheter (InterjectTM, Boston Scientific, Costa Rica), to obtain adequate submucosal elevation.
The energy device in our hospital was a DualKnifeTM (KD-655, Olympus, Tokyo Japan) or ITKnife2TM (KD-611L, Olympus, Tokyo Japan) connected to a high-frequency generator (ERBE, Elektromed-VIO300D, Tubingen, Germany). After incision and immediate submucosal access, continuous tunneling was conducted to create a flap. Submucosal dissections of the left and right margins were extended to create a pocket. Next, a marginal incision is performed on the opposite side, and dissection is continued to open the tunnel in the middle and then bilaterally. The tumor specimen is removed.
Hemostasis with the energy device in the coagulation mode or a Hemoclip (ROCC-D-26-230-C, ERBE) was employed as needed when bleeding or vascular structures are encountered. Hemoclips can also be used for wound closure.
TAMIS postoperative care
Prophylactic intravenous Flomoxef was prescribed (7 days, interquartile range [IQR], 4.5–9.5). Patients were offered liquids after recovery from anesthesia. No diet restrictions were imposed once the patients could tolerate liquids. All patients were mobilized as soon as possible. Postoperative acetaminophen (Scanol) or intramuscular meperidine was administered, as necessary. If a urinary catheter was left in place after surgery, it was usually removed on the first postoperative day.
The criteria for discharge from hospital included a tolerance of general meals 1 day after surgery, stool passage tolerance, and no signs of infection. The patient was subsequently followed up at the outpatient clinic.
ESD postoperative care
Patients were observed for 2–4 hours and discharged if no significant symptoms such as abdominal pain or abdominal fullness were seen. During this time period, they were allowed to drink fluids. There was no routine use of antibiotics. The patient was subsequently followed up at the outpatient clinic.
Results
Patients and tumor characteristics
Patient demographic data are listed in Table 1. There were no statistically significant differences in age, gender, BMI, and ASA between the TAMIS and ESD groups.
Patient Characteristics
Values are presented as the median (quartile 1 to quartile 3) unless indicated otherwise.
P values are calculated with Fisher’s test (categorical variables) or the Mann–Whitney U test (continuous variables).
ASA, American Society of Anesthesiologists; BMI, body mass index; ESD, endoscopic submucosal dissection; TAMIS, transanal minimally invasive surgery.
Tumor characteristics are listed in Table 2. The median distance between the tumor and anal verge was 5 cm (IQR, 3.5–8) and 3 cm (IQR, 2–4.25) for the TAMIS and ESD groups, respectively. This difference was statistically significant (P = 0.016). There were no statistically significant differences in specimen size. The quality of pathology completion in both group had no lesion fragment. Two patients undergoing ESD had margin involvement versus none with TAMIS (P = 0.041).
Tumor Characteristics
Values are presented as the median (quartile 1 to quartile 3) unless indicated otherwise.
P values are calculated with the Fisher’s test (categorical variables) or the Mann–Whitney U test (continuous variables).
Statistical significance (P < 0.05).
ESD, endoscopic submucosal dissection; TAMIS, transanal minimally invasive surgery.
Surgical and postoperative characteristics
TAMIS operative time was shorter than that of ESD (72 minutes [IQR, 62–89] versus 120 minutes [IQR, 90–185], respectively; P = 0.005; Table 3). Blood loss could not be evaluated in the ESD procedure. Others surgical characteristics are listed in Table 3.
Surgical Characteristics
Values are presented as the median (quartile 1 to quartile 3) unless indicated otherwise.
P values are calculated with the chi-squared test (categorical variables) or the Mann–Whitney U test (continuous variables).
Statistical significance (P < 0.05).
ESD, endoscopic submucosal dissection; TAMIS, transanal minimally invasive surgery.
Postoperative characteristics are listed in Table 4. The median hospital stay was 3 days (IQR, 3–4) for patients undergoing TAMIS, while patients undergoing ESD were discharged the same day. No complication, death, or local recurrences occurred during follow-up, in TAMIS group (3.3 months, IQR, 0.3–11.7) and in ESD group (0.9 months, IQR, 0.3–15.4).
Postoperative Characteristics
Values are presented as the median (quartile 1 to quartile 3) unless indicated otherwise.
P values are calculated with the Mann–Whitney U test (continuous variables).
ESD, endoscopic submucosal dissection; TAMIS, transanal minimally invasive surgery.
Discussion
In our retrospective single-center experience, we found that both techniques were clinical feasible and safe from the surgical viewpoint. Two important differences exist between these two techniques: TAMIS was quicker and there were no involved margins.
Both of these differences may be related to exposure. Adequate traction and counter traction are critically important for exposure in order to create and carry out adequate dissection along the surgical plane. Counter traction is possible in the TAMIS procedure but not in endoscopy where there is only one working channel. Traditionally, the ESD expert used an attachment cap for elevating the flap after initial dissection, to create a traction effect and identify the surgical plane. The ESD endoscopist can also use a gravity effect, by changing patient position, to achieve counter traction (Fig. 4 in reference 19). However, in our opinion, these methods cannot achieve the same results as TAMIS. In TAMIS, additional instruments archive traction freely and dynamically in 3D: 360 degrees circumferentially and forward or backward, longitudinally along the enteric axis,20,21 as shown by Martin-Perez et al. (Fig. 8 in reference 22). Combined with a 3D flexible laparoscopic camera, blind spots are minimized. Of note, patient position does not need to be changed during the operation and traditional laparoscopic instruments, already available in any modem operating room, can be used in conjunction with TAMIS.
Newer techniques and devices have also emerged to resolve the ESD traction problem. 23 However, the clip-with-line traction 24 in colonoscopic ESD is fixed and difficult to adjust. When location of the surgical plane is uncertain in TAMIS, we can employ a counter traction force or change direction or tract forward and backward to detect the avascular plane. This improves dissection speed and facilitates identification of clear tumor margins.
In addition to traction and exposure, another difference between TAMIS and ESD may be the pneumorectum. During the TAMIS procedure, we usually insert a gauze in the upper rectum to maintain pneumorectum. This may induce a microdissection phenomenon during surgical plane dissection that improves tissue separation and exposure. This maneuver is somewhat difficult during ESD.
TAMIS also offers other technical advantages including the use of the waterjet applicator. The Waterjet Applicator (HybridKnife®) injects water under high pressure into the submucosa, separates, and elevates the resection area by the formation of a water cushion. The contrasting fluid also increases the conductivity and improves the cutting features of monopolar electrosurgical resection, especially in fat-rich tissue. 25 Baral et al. 26 stated that concurrent use of indigo carmine may highlight the submucosal vessels for better visibility and facilitates hemostasis. We found this advantage to be useful and suggest its usage especially for centers that are new to this technique.
En bloc excision and pathology quality were also improved by those advantages. Pathology quality, which includes lesion fragmentation and margin involvement, is associated with local recurrence. 27 In our study, two margins were involved in the ESD group, possibly related to tumor size. 8 Even after adjustment of confounding factors such as tumor size 7 and cohort study year influence, 27 shorter operative time with TAMIS compared with transanal endoscopic Microsurgery (TEM) is in agreement with several studies.7,27 This trend is also noted when robotic TAMIS is compared with laparoscopic TAMIS. 6 This could be due to the more stereo 3D view and more flexible and reachable angle provided by the Endowrist of the da Vinci robotic surgical system. We believe that this trend is also true for TAMIS compared with ESD.
Reviewing the literature, there are not many reports that compared these two techniques. Mao et al. compared the ESD with self-made glove-trocar. 12 Simon et al. prematurely stopped their trial because of low complication rate. 28 The TRIASSIC study is still ongoing, and the results are awaited. 29
Limitations
This is a single-institution, small sample size, retrospective cohort with a limited duration of follow-up. The anorectal function and quality of life data were not studied.
Conclusion
Both TAMIS and ESD are clinically feasible for use in community hospitals. The TAMIS procedure takes less time and procures adequate tumor resection with uninvolved margins, most likely because of adequate traction and counter traction, the pnumo-rectum effect, and 360 degrees visibility.
Footnotes
Acknowledgments
The authors thank Dr. Ming-Chih Lin for his help with the tables.
Disclosure Statement
J.-J.Y., on behalf of all authors, declares that they have no conflicts of interest or financial ties to disclose.
Funding Information
No funding was received for this article.
