Abstract
Gestational diabetes mellitus (GDM) is the most prevalent metabolic complication during pregnancy. GDM is associated with adverse perinatal, neonatal, and long-term health consequences. Studies have demonstrated that the use of continuous glucose monitoring (CGM) reduces the incidence of maternal and neonatal complications in pregnant women with type 1 diabetes. Although the use of CGM in GDM has not been well studied, a growing body of evidence is showing potential benefits in the GDM population. This article discusses the advantages and challenges of CGM and provides practical guidelines for using this technology in the GDM population.
Introduction
Gestational diabetes mellitus (GDM) is the most prevalent metabolic complication during pregnancy with reported rates from 5% to 25.5% worldwide. 1,2 Significant risk factors for GDM include a family history of type 2 diabetes (T2D), a history of gestational diabetes in a prior pregnancy, lower physical activity during at least 3 months before pregnancy, advanced maternal age, and obesity. 3 As reported by Zhou et al., the increasing prevalence of GDM appears to be proportional to the increasing rate of obesity. 4
Gestational GDM is defined as glucose intolerance that is associated with adverse perinatal outcomes. 5 GDM is generally diagnosed between the 24th and 28th week of pregnancy; however, maternal hyperglycemia can be present earlier in pregnancy. 6 GDM leads to hyperglycemia and is associated with significant maternal, perinatal, and neonatal complications 7 –19 (Table 1).
If hyperglycemia present in first trimester.
If hyperglycemia is untreated or suboptimally treated from early pregnancy.
Achieving and maintaining optimal glycemic control becomes increasingly challenging with advancing gestation as insulin resistance continues to rise from midgestation until typically 36 to 38 weeks gestation. 20 Unlike pregnant patients with preexisting diabetes, GDM is a new diagnosis for pregnant patients that may include an adjustment period and a steep learning curve of the condition, associated complications, management strategies, and goals of treatment. In addition, individuals with GDM may be unfamiliar with the role of technology in the management and care of their GDM such as fingerstick testing and/or continuous glucose monitoring (CGM).
Currently, there are three CGM systems in the United States approved for use in pregnancy,
In December 2022, a panel of expert clinicians met to discuss approaches to using CGM for patients with GDM and how best to use the data and interpret software reports. This article discusses the advantages and challenges of CGM and provides practical guidelines for using this technology in the GDM population.
Core Elements of GDM Management
Within 1 to 2 weeks of diagnosis, the goal is to provide pregnant patients with nutritional and lifestyle counseling, diabetes education, information about the risks of perinatal, neonatal complications, and the long-term health risks caused by elevated glucose levels. Initial education includes pregnancy-specific glycemic goals to optimize maternal and fetal outcomes and the importance of this glycemic control throughout pregnancy. Support includes frequent glycemic monitoring check-ins, obstetric visits, and often additional fetal monitoring. There are two classifications of GDM: A1GDM, controlled with diet and exercise; and A2GDM, treatment with antihyperglycemic medications (insulin or oral medications). Currently, insulin is the first-line medication used to treat GDM, but at times, oral medications can be used in a select population of patients who are unable to use insulin.
Glucose Monitoring
Suboptimal adherence to traditional blood glucose monitoring
Frequent glucose monitoring is essential to achieving optimal glycemic control, especially given the lower glucose targets in pregnancy. 21 –23 The American Diabetes Association (ADA) clinical guidelines recommend at least fasting and postprandial testing daily (minimum four tests per day). 24 The fasting glucose represents overnight glucose control, and the 1-h postprandial glucose is most often used to characterize the peak post meal glucose excursion. Achieving pregnancy-specific glucose metrics must occur quickly to reduce risk for complications. Chehab et al. reported an increased risk of perinatal complications among individuals with GDM with slower incremental improvements in glycemic control compared with those who more rapidly achieved glycemic goals. 25
Since its introduction more than four decades ago, self-monitoring of blood glucose (SMBG) using fingerstick blood samples, test strips, and portable glucose meters has been the standard strategy to monitor glucose levels in patients with diabetes. However, the reliability of and adherence to this method is often suboptimal. 26 There are many barriers to SMBG including pain and discomfort associated with fingersticks, inconvenience of carrying testing materials, disruption of daily activities, and social stigma. 27 –30 Fingerstick data are also limited as it only provides a static measure of glucose.
Patients with GDM experience the same barriers as preexisting diabetes in pregnancy. In a study of 91 individuals with GDM, 35 (38.5%) had barriers to completing their prescribed testing regimen, which was associated with higher incidence of preeclampsia and higher HbA1c levels at delivery. 26 A diagnosis of GDM can be overwhelming for many patients during pregnancy given the need to be more precise with meal planning, exercise during pregnancy, the additional burden of frequent glucose monitoring, and taking medication if needed.
CGM overcomes the obstacles associated with SMBG
CGM is now considered the “gold standard” for glycemic management in individuals with T1D and T2D treated with intensive insulin therapy, basal insulin therapy, and all patients with a history of severe hypoglycemia including those not on insulin. 31 Unlike SMBG, CGM measures interstitial glucose levels and transmits glucose readings to the user’s smartphone app or hand-held receiver every 1 to 5 min. CGM also displays trend arrows that indicate the direction and rate of changing glucose levels and can predict glucose changes over the next 20 to 60 min, depending on the CGM system. The data are displayed in real time in numerical and graphical formats which enables the user to easily recognize glucose trajectories, detect problematic glucose patterns, and make informed therapy decisions more easily. Users have immediate feedback on both current glucose level and future potential hyperglycemia or hypoglycemia from trend arrows which allows earlier intervention and helps mitigate episodes of potential hyper or hypoglycemia. Programmable alerts can be customized based on individual needs and goals to help support glycemic control.
Numerous studies in nonpregnant diabetes populations have shown that the use of CGM lowers HbA1c, 32 –43 reduces severe hypoglycemia events, 38,39,44 increases time within target glucose range, 34 –36,42,45,46 and reduces time in hypoglycemic ranges. 35,36,47,48 A growing body of evidence now supports the efficacy and safety of CGM in GDM, including improved glycemic control, 49,50 less hypoglycemia, 51 and lower maternal weight gain. 52 Reductions in perinatal complications have also been associated with GDM use. 49 It is paramount to identify patients who would benefit from closer glucose monitoring, 53 more targeted interventions, 53 and support in addressing barriers to taking medications during pregnancy. 51 More intensive glucose monitoring than fingerstick testing provides would potentially prompt earlier intervention and less the barriers to taking medications in pregnancy. This is especially important for individuals with GDM in ethnic/racial minority populations and lower socioeconomic status, many of whom are covered by Medicaid, the public health insurance program for people with low income in the United States.
A recent report highlighted Black (32.0%) and Hispanic (30.0%) beneficiaries comprise the largest percentage of the Medicaid population, and the proportion of GDM women who require Cesarean birth is higher among Black women (45.5%) compared with White (41.3%) and Hispanic (40.6%) women. 54 Similar disparities are observed in the proportion of Black women (17.5%) whose newborns require neonatal intensive care compared with White (13.1%) and Hispanic (12.8%) women. 55 Although most state Medicaid programs do not cover CGM for individuals with GDM, 54 it is important to make patients aware of the benefits of CGM and investigate avenues to ensure technology is available to all populations in pregnancy given the more immediate potential risk mitigation for mother and infant.
All the authors with their cumulative clinical experience in diabetes and pregnancy use CGM as a tool to support glycemic management and provide patients with more information but with less distress and disruption of their daily activities. Importantly, for shared provider–patient partnership, the CGM data offer a more global assessment of patients’ 24-h glycemic status and thus more meaningful, comprehensive data that can be used to guide interventions and support patient self-efficacy and empowerment. The use of this technology can facilitate more timely adjustments of therapy (lifestyle behaviors and medication), thereby leading to optimal glucose control and ultimately help improve perinatal outcomes. 25
Practical Approaches to the Use of CGM in GDM
Patient selection
Given the demonstrated benefits of CGM in improving glycemic control 49 and promoting healthy lifestyle behaviors in GDM patients, 51 we recommend CGM to all patients who are willing and able to safely use this technology. However, when discussing CGM with patients, it is important to discuss the factors that are relevant to each patient’s clinical needs and lifestyle preferences to help determine whether CGM is right for them.
Clinical factors
CGM is appropriate for all GDM patients treated with insulin or other antihyperglycemic medications. The following are some clinical and environmental/lifestyle characteristics of patients who would benefit from CGM use: Higher risk pregnancy due to prior history of GDM. Comorbidities such as cystic fibrosis or other medical conditions that put patients at higher risk for hypoglycemia or hyperglycemia. The use of insulin or other antihyperglycemic medication. Insufficient SMBG data or barriers/difficulty performing prescribed glucose testing. Shift work or jobs with high levels of physical activity or limit the ability to access SMBG testing supplies. Fear of hypoglycemia. Would benefit from additional support for lifestyle behavior change.
Lifestyle factors
Because most GDM patients do not require insulin or antihyperglycemic medications at diagnosis, 56 there is little or no risk of hypoglycemia. However, CGM can be beneficial for patients with barriers to performing the prescribed blood glucose monitoring plan and treatment regimens or those concerned about hypoglycemia. Some examples where CGM might be indicated to support nonmedication requiring GDM patients would be to maximize exercise and food choices through monitoring of the effects of physical activity on glucose levels and to better evaluate postprandial excursions from food selections. CGM is also useful for individuals with a history of hypoglycemia during pregnancy, particularly when taking glucose lowering medications, and those concerned about hypoglycemia as a result of activity or exercise. CGM will help support lifestyle choices for these patients and can decrease anxiety and stress associated with fingerstick testing for patients with GDM. 57,58 In addition, CGM may help identify earlier need for medication therapy and support discussions between patients and their healthcare team in which data can be reviewed together for shared decision making. CGM can also increase patient comfort with medical treatment by allowing patients to see the benefit to consistent use of a recommended medication like insulin and possibly reduce the need for escalation of therapy. For example, patients observing the impact of higher fiber foods with meals or the impact of exercise on glucose levels. The use of CGM can also benefit patients with erratic schedules (night shift, inconsistent meal timing, etc.) and reduce the burden and distress (inconvenience, social stigma, timing of glucose checks) that can be associated with SMBG.
Glucose Targets for GDM
Current ADA clinical practice guidelines recommend glucose monitoring in both GDM and preexisting diabetes. 24 Glucose targets are fasting plasma glucose <95 mg/dL and either 1-h postprandial glucose <140 mg/dL or 2-h postprandial glucose <120 mg/dL. 24 Although the ADA has not yet established glycemic time-in-range targets for the use of CGM in GDM or T2D on insulin therapy, it has adopted the clinical metrics and targets for pregnancy in T1D that were established by an international consensus panel 60 (Table 2). 31,59
International Consensus Guidelines have not defined glycemic targets for T2D and GDM.
These TBR targets may not apply to A1GDM patients without hypoglycemic symptoms because levels <63mg/dL may reflect normal glucose physiology of pregnancy patients. CGM, continuous glucose monitoring; GDM, gestational diabetes mellitus.
For patients at low risk for hypoglycemia, such as those with GDM managed with diet and T2D TIR ≥ 90% is a feasible goal. Although it is important that patients achieve optimal glycemic control as soon as possible, 25 individualizing patient’s goals based on their characteristics and initial glycemic control is also important to support the patient and not create higher levels of diabetes distress. Initial targets should be achievable with frequent intermittent goals to avoid patients from becoming discouraged. For example, an initial TIR > 80% can be set followed by progressive increase in the TIR to target levels. A stepwise approach to increasing TIR by 5% increments can be an achievable goal for most patients. Although the ADA recognizes that there are insufficient data to support the use of CGM in women with T2D and GDM, 24 in T1D pregnancies, every 5% increase in TIR is associated with improved perinatal outcomes. 60 Other metrics such as mean glucose and glycemic variability (standard deviation [SD], % coefficient of variation [%CV]) should also be considered when assessing glycemic status. Table 3 presents the established CGM metrics for general use in clinical care. 59 However, in GDM, it is common to review CGM data over shorter durations of sensor wear (e.g., 7 days), given the progressive increase in insulin resistance in the second and third trimester, requiring more rapid changes in therapies that outside of pregnancy.
International Consensus Standardized CGM Metrics for Clinical Care 59
CV, Coefficient of variation.
Patient Training
Setting target ranges/alert thresholds
CGM systems offer customizable target ranges (goal glucose ranges) as well as customizable alerts that will notify the patient with an alarm and/or vibration. The target glucose range in pregnancy is 63 mg/dL to 140 mg/dL; however, CGM systems and their glucose report software default to the nonpregnant target range of is 70 mg/dL to 180 mg/dL. Target ranges may be customizable based on the CGM system or software used to view the glucose data. For example, among free platforms, LibreView can set the lower range to 63 and
Target ranges may be individualized based on each patient’s current glycemic status and treatment regimen and other factors like history of hypoglycemia, degree of diabetes-related distress, and initial glycemic control. For patients currently with glycemic control close to goal, initial targets can be more aggressive. However, for patients with more hyperglycemia or episodes of recurrent hypoglycemia with less optimal baseline glycemic control, it is advisable to start with less aggressive targets and then tighten the targets as glucose levels and patient confidence improves. Note that the target ranges and alert settings can only be set in increments of 5 mg/dL. For example, use 65 mg/dL if the desired target or alarm setting is 63 mg/dL.
For A1GDM patients, the high threshold alert can most often be set to alert for >140mg/dL to maintain the target %TIR but may need to be set at a slightly higher threshold if there is concern for alert fatigue. We recommend starting with a high threshold of 140 mg/dL while providing instruction on how to suspend or delay the alert if they experience increased diabetes distress or are in a setting where the alerts are disruptive. We advise that no low threshold alert be set in those not requiring medication management for their GDM (although some systems have a default alert of 55 mg/dL). In those only being treated with metformin, consider suspending/turning off low alerts given low risk of hypoglycemia similar to A1GDM. However, if insulin is initiated, the low threshold should be set to avoid hypoglycemia (e.g., 60–65 mg/dL), such as the example shown in Figure 1. High threshold alerts should be individualized for safety and optimization of glycemic control.
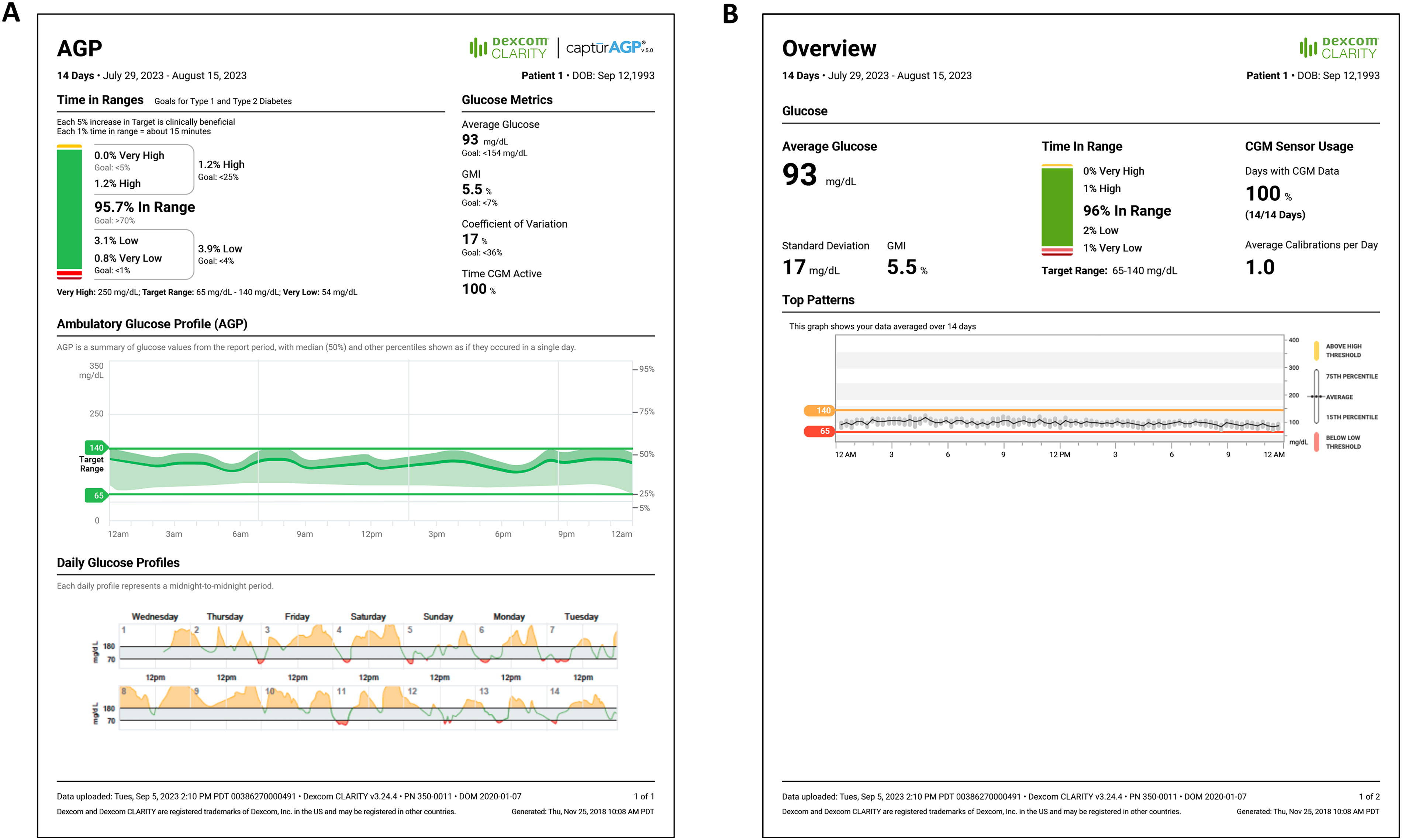
Clarity AGP and overview reports.
CGM Initiation
Upon initiation of CGM, it is important to provide patients with a basic understanding of CGM, how to adjust parameters, use the app, troubleshooting, how to maximize use, and the importance of continuous sensor wear. A checklist of fundamental principles and skills that a patient should learn when using real time CGM is shown in Table 4. Supporting patient engagement with CGM includes providing instructions and assistance with system setup (e.g., placement of sensor, use of receiver or smartphone app, and data sharing with the medical care team, and, if desired, a family or support member during pregnancy). Then, clinicians need to ensure that patients are provided information about how to interpret their CGM data and handle various issues that can arise when using the CGM system. Clinicians should strongly encourage patients to carefully review the “trouble shooting” information in their product user guide for complete information about how to solve problems if they occur. Table 5 presents major topics and issues to cover when initiating CGM.
Checklist for rtCGM Initiation and Training
Troubleshooting Guide for Common Challenges
Anticipatory Guidance
A common concern among patients who begin using CGM is the gradual but predictable worsening of their glycemic status due to increasing insulin resistance as gestation progresses. 20 This phenomenon needs to be explained to patients to avoid anxiety and frustration. Having this discussion provides an opportunity to help patients understand that they are not failing in their treatment regimen and why insulin may be needed or increased through no fault of their own.
Download Reports
Overview of reports
The download software provides a series of reports that enable clinicians to evaluate relevant glucose patterns and trends to help make diabetes management decisions. To use the software, clinicians create patient accounts to upload, track, and review CGM data during visits. The reports enable clinicians to view an overview of glucose values, overall glucose trends, patterns, comparison charts and statistics. Patients are also able to create accounts and link them to the clinic’s account by sending an invitation or sharing a code.
Use of CGM reports
The default settings for the reports are intended for nonpregnant individuals with T1D and T2D. Therefore, the first step is to adjust the settings for pregnancy targets, whenever possible. For example, most summary reports default to 70–180 mg/dL but offer an additional summary report that offers customizable targets, allowing the provider to change to the pregnancy targets. This will allow for evaluation of the pregnancy-specific time in target glucose range (TIR). Because insulin resistance increases as pregnancy progresses, 64 it is important to review patients’ CGM data every 7 days. Given the patient recall is typically 3 to 4 days, the daily data over this time period should be reviewed with the patient and coupled to questions and discussion about activity and food choices and medication (if applicable). In addition, the Ambulatory Glucose Profile (AGP) provides a “snapshot” overview of the current glucose control. The download software features a number of different reports that facilitate both overall and more comprehensive assessment. These reports are useful in motivating patients and keeping them engaged in their therapy as well as guiding shared medical decision making.
For retrospective analysis, CGM manufacturers and independent software platforms provide reports that organize CGM data into easily understandable formats. These software programs have various benefits, but all provide the same basic information about recent glucose trends and glycemic status. There are differences between care platforms on the ability to customize to current pregnancy-specific glucose targets (63 to 140 mg/dL).
For clinicians who are unfamiliar with using these CGM reports, starting with the Clarity Overview report (Fig. 1A) or the LibreView (https://www.libreview.com/) AGP report 65 is helpful. These reports provide assessments of the standard CGM metrics, which include percentages of time in range (%TIR), below target range (%TBR), and above target range (%TAR) that are similar to each other and allow for quick, overall assessment of patient’s glycemic status. Both reports present the time in ranges in a color-coded graph that patients can easily understand, and both display a glucose profile, which combines all daily profiles into a 24-h average profile. In addition to assessment of glycemic variability, the AGP also presents glucose profiles for all of the days covered. The overview report provides insights regarding problematic glycemic patterns as well as motivational messages stated in simple language. For example, “[Patient Name] best glucose day was September 1, 2023.” It is important to note that the report has a default target range of 70–180 mg/L.
Review and discussion of the Clarity Overview Reports and LibreView AGP is generally all that is needed for patients with A1GDM who are mostly achieving their glycemic goals as these reports provide the most critical information, namely, %TIR and mean glucose. For patients with A2GDM, %TIR, %TBR, %TAR, mean glucose, and glycemic variability (SD, %CV) are more meaningful. For patients who are having difficulty achieving their goals, many clinicians will then view the Compare report (Fig. 2A) and/or Daily report (Fig. 2B) to facilitate informative discussions with the patient and take a “deeper dive” into the data to investigate the potential causes for the suboptimal glycemic status. Clinicians may also consider reviewing the alert settings and consider adjustments that may benefit the patient in achieving their goals.
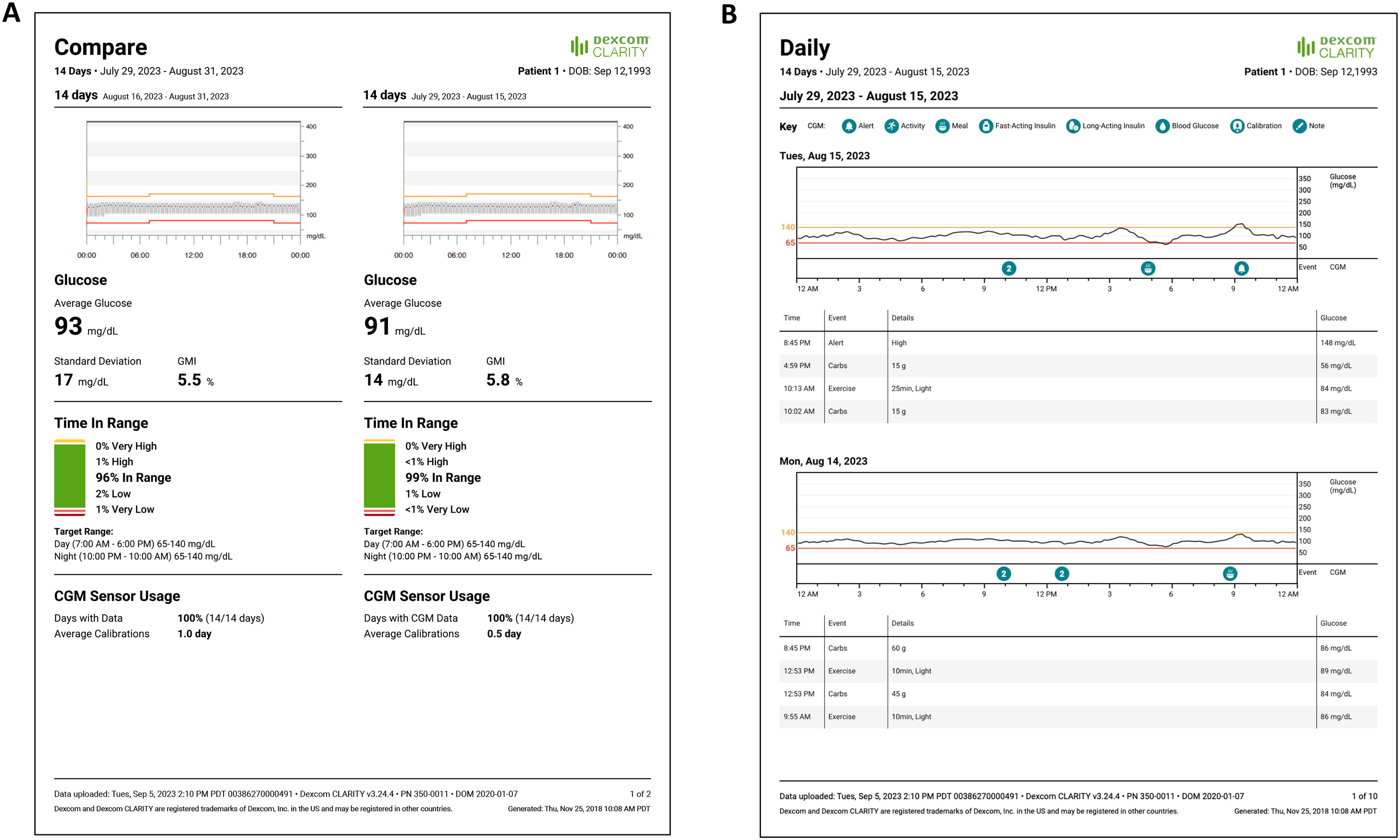
Clarity compare and daily reports.
The Compare report allows clinicians and their patients to assess the impact of therapy from one week to the next. This report is useful both in determining the need for adjusting therapy and in motivating patients to either continue or change their behaviors. The Daily report provides more detailed information about specified days, correlating events (e.g., alarms, meals, exercise) with glucose levels throughout each day. This information can be used to identify trends or behaviors that can be targeted in the coming week. This is why it is important to emphasize that patients enter these events in the app.
The LibreView weekly summary is used to review overall trends and highs and lows, while the Daily Patterns and Glucose Pattern Insights are helpful to identify further areas of intervention for patients. Daily data targets can be adjusted from the default range (70–180 mg/dL) to 65–140 mg/dL to more clearly assess glucose values in pregnancy-specific ranges.
The immediate feedback that CGM provides patients enables them to identify and modify specific behaviors that are adversely impacting their glycemic control and make changes on their own to modify these behaviors. When reviewing data with patients, clinicians can further reinforce the benefits of these changes and provide advice for overcoming any obstacles patients may be having with their therapy.
For patients treated with insulin, retrospective analysis of patient data enables clinicians to make more informed decisions about therapy adjustments relative to dosages, timing of insulin administration, and the type of insulin that is most appropriate for each patient. A recent article by Aleppo et al. provides an in-depth discussion on how patients can use their trend arrows to avoid glycemic events and use insulin safely and effectively. 66
Summary
Frequent SMBG has been the traditional method used for glucose measurement in GDM patients, but adherence to prescribed testing regimens is often poor, 26 due the discomfort, inconvenience, disruption of daily activities, potential for social embarrassment, and difficulty interpreting the data. 27 –30 SMBG also fails to identify many problematic glycemic patterns, particularly at night when hyperglycemia often occurs.
The use of CGM in conjunction with the CGM reports addresses these issues, thereby lessening the burden of managing GDM. Moreover, the immediate feedback CGM provides to patients encourages immediate remedial actions to address acute glycemic events and/or avoid future events; learning to manage glucose is an ongoing process. Retrospective review using the CGM reports simplifies data interpretation and creates opportunities to teach and motivate patients while providing critical information that guide therapy.
Given the significant and increasing prevalence of GDM, clinicians should use all available tools for to help their patients reduce the risk of severe adverse maternal and fetal outcomes that can result from suboptimal glycemic control. CGM offers a patient-centered behavior feedback approach to managing diabetes.
Although the use of CGM in the GDM population has not been well studied, the risk of poor maternal and fetal outcomes due to hyperglycemia is universal in all pregnancies complicated by diabetes. Additional research is needed to identify normative values in pregnant individuals who do not have GDM and to more fully elucidate the benefits of CGM in managing GDM.
Footnotes
Acknowledgments
The authors thank Christopher G. Parkin for editorial assistance.
Authors’ Contributions
All authors contributed equally to content development and article writing/revisions. All authors approved the submission of the article.
Author Disclosure Statement
K.C. receives research support provided to her institution from
Funding Information
Dexcom, Inc., provided funding for editorial assistance in developing this article.
