Abstract
Background:
Women worldwide face risks from pregnancy, HIV, and other sexually transmitted infections (STIs). To date, highly effective contraceptive methods provide no HIV/STI protection, and HIV prevention products, excluding condoms, provide no pregnancy protection. Intravaginal rings (IVRs) delivering antiretrovirals and contraceptives are a promising multipurpose prevention technology (MPT).
Methods:
Embedded within a Phase I randomized, placebo-controlled trial, we examined acceptability of continuous versus interrupted use of a 90-day MPT IVR among 47 low-risk women in Norfolk, Virginia and the Dominican Republic. A baseline survey assessed menstruation attitudes, risk perceptions and trial-related motivations. Follow-up surveys (M1/M3) examined user experiences with and preferences for IVR attributes; 18 women also participated in two in-depth interviews.
Results:
Most women rated the IVR's flexibility and smoothness (86%) and ease of insertion/removal (76%) as very acceptable. Fewer women similarly rated the IVR size (57%) and changes in color from menstruation (52%). Most participants experienced no changes or less bleeding. Those reporting more/heavier bleeding (20% M1, 15% M3) disliked the change. Overall, women preferred a 3-month (75%) to a 1-month IVR (7.5%) or a bimonthly injectable (10%). In qualitative interviews, women were willing to continuously use an IVR for 6–12 months, providing it did not “degrade” inside the body. Reasons for trial participation and prevention preferences, menstrual attitudes, and perceived IVR benefits and doubts varied by site.
Conclusions:
Findings provide strong evidence of demand for an MPT IVR that protects from pregnancy and HIV/STIs, lasts longer than 1 month, minimally disrupts menstrual bleeding, and is in women's control.
Introduction
Worldwide, sexually active women are faced with multiple health risks, including unintended pregnancy, complications from having repeated pregnancy and childbirth or at too early an age, HIV, and other sexually transmitted infections (STIs). 1 –4 Although a range of effective contraceptive methods exist, uptake and consistent and long-term use are challenged by numerous factors.
From country to country, demand for and access to various contraceptive options may vary widely and are shaped by cultural perceptions, religious norms, policy priorities, and health system structures. 5,6 Within geographic settings, women's acceptance of a specific contraceptive method is also influenced to varying degrees by the perceived fit of the method within their daily lives and sexual patterns, their own or their partners' concerns about and/or experiences with method-related side effects, the duration and perceived ease or difficulty of dosing regimens, and the ability to stop and start method use at will. 7,8
In addition, concerns about the risk of STIs may factor into women's decisions about method use. To date, highly effective contraceptive methods provide no protection from HIV and other STIs, and HIV prevention products provide inadequate (in the case of male and female condoms) or no protection (in the case of pre-exposure prophylaxis, or PrEP) from pregnancy. Clearly, a multipurpose prevention product that prevents both pregnancy and HIV or other STIs would help to address this current prevention gap. In multiple surveys in sub-Saharan Africa, women of reproductive age have clearly indicated they would prefer a multipurpose prevention technology (MPT), that is, two products in one, rather than individual products for contraception and HIV prevention. 9 –11
As a platform to deliver active compounds to the female reproductive tract, intravaginal rings (IVRs) are highly promising. Nuvaring, a monthly contraceptive ring containing a combination of etonogestrel and ethinyl estradiol is one of several IVRs currently in use. 12 Recently, the U.S. Food and Drug Administration (FDA) granted approval for a new contraceptive ring containing segesterone acetate and ethinyl estradiol that provides an entire year of protection (Annovera™). Several ring platforms have also been developed and evaluated for HIV prevention. 13 –15
The benefits to vaginal rings as a delivery system are multiple. They include being discreet, one-size-fits-most, and under the control of women themselves with the ability to use it “on-demand” for intermittent periods with little need for routine adherence behavior. 16 The ring can simply be inserted and left alone. Nevertheless, studies have also identified potential concerns about IVRs, including perceived safety of intervaginal products, personal hygiene and cleanliness, possible discomfort during sex, whether to disclose use to a partner, 17,18 and general concerns about use of hormonal methods.
In this article, we examine and compare the acceptability of and preferences for a novel MPT IVR, used either continuously or in an interrupted fashion for 90 days. Acceptability data, which focused on product attributes (e.g., size, stiffness, side effects, experience with insertion and removal) were collected from participants of a Phase 1 clinical trial, conducted in Norfolk, Virginia and Santo Domingo, Dominican Republic (DR).
We closely examine participants' attitudes toward menstruation because contraceptive-related bleeding changes are a common side effect of levonorgestrel-only contraceptives (e.g., Jadelle, Norplant, Levonorgestrel Intrauterine system) and a common reason for discontinuation. Because Phase 1 trial participants are generally at low risk for the product indication(s) being evaluated, we also provide contextual information about participants' motivations for trial participation and their perspectives on potential acceptability within their larger communities.
Materials and Methods
Between September 2017 and December 2018, CONRAD conducted a Phase I randomized, placebo-controlled trial to evaluate the safety and tolerability of a multipurpose vaginal ring containing tenofovir and levonorgestrel that could be used continuously for 90 days (“continuous” regimen) versus cyclic use of the ring, during which the ring was removed for 3 days every 28 days during a 90-day treatment period (“interrupted” regimen). Sixty women at low risk of pregnancy and HIV were enrolled in two sites, the Eastern Virginia Medical School in Norfolk, Virginia, and Profamilia, a private nonprofit institution that provides sexual and reproductive health services and conducts contraceptive research in the DR.
Low risk was defined as women who had undergone a tubal ligation or whose partner had received a vasectomy, women who were not sexually active, or had a same-sex partner. Sexually active women were also screened at visit 1 for STIs and HIV. HIV was exclusionary. Women were randomized at a ratio of 1:1 to continuous or interrupted ring use, and 4:1 on a ring containing active versus placebo ingredient. The study was approved by the Chesapeake IRB (now Advarra; Pro00022358) at EVMS and Institutional Review Board of Profamilia (IORG0001979) and Conabios (IORG003206).
A secondary objective of the trial was to provide a more in-depth understanding of MPT IVR acceptability, including user experiences with and preferences for IVR attributes, with attention to bleeding patterns and reported adherence. All participants responded to quantitative acceptability surveys, self-administered on a computer in the clinic at 3 time points (baseline, visit 13 within first month of IVR insertion, and visit 31 after removal at 3 months) and a maximum of 10 participants in each site were invited to participate in a series of two in-depth interviews (IDI), administered in person and in Spanish in the DR and through mobile phone in English in the United States by trained qualitative interviewers.
Survey measures and analysis
The baseline acceptability assessment included: a set of 22 items assessing menstrual period beliefs and behaviors 19 ; questions related to menstrual hygiene management, pregnancy intentions, contraceptive use, and HIV/STI risk perception; as well as eight items assessing motivations for research trial participation. Altruistic motivations related to wanting to participate in the research to help researchers and/or to find new products, or because research is necessary to improve health. Other reasons included obtaining the latest information, improving one's personal health or because of free medical check-ups or money.
Two follow-up surveys included: items assessing four domains of experiences with vaginal ring use (i.e., IVR benefits, IVR doubts, IVR side effects, and removal) 20 ; questions on menstrual cycle effects; attitudes toward vaginal ring characteristics; and future use of contraceptive and/or HIV/STI prevention products.
All response options for menstrual beliefs and IVR experiences were based on a six-point scale with 1 = disagree a lot and 6 = agree a lot. Future use was measured by the statement “If this vaginal ring was shown to protect from HIV, HSV, and pregnancy, how interested would you be in using it outside of a clinical trial?” Response options were 1 = Not at all interested, 2 = Somewhat interested, and 3 = Very interested.
We disaggregated baseline data by regimen (continuous vs. interrupted), agent (active vs. placebo), and site, and follow-up data by these same categories and by visit. We examined item means and frequencies and conducted bivariate analyses (t-tests and chi-squared tests) to determine statistically significant differences by regimen, agent, site, and visit (for repeated follow-up measures).
Before finalizing descriptive tables for multi-item constructs, we used exploratory factor analysis procedures to determine the number of underlying constructs (e.g., menstrual attitudes) to verify that subsets of IVR experience items fit on their respective domain based on previous IVR experience psychometric work. We assessed individual items, dropping any items that did not load to factors or that detracted from the subscale Cronbach's alpha test of reliability, with a minimum acceptable reliability cutoff of 0.7. We then generated mean scores for each subscale by calculating the means of participants' responses to each item within a subscale, reverse-scoring item responses as necessary to ensure items loaded to subscales unidirectionally. All quantitative results were then organized into descriptive tables.
IDI topics and analysis
An unblinded study statistician purposively selected participants in each site representing the continuous and interrupted regimens and checked to ensure representation between participants on active versus placebo agents at the study midpoint. The first qualitative interview took place within the first month and explored participants' reasons for trial participation, perceived risk of HIV and pregnancy, menstrual history, prior experiences with contraceptive use, and initial experiences with the IVR, including ease or difficulty with insertion.
The second IDI took place after ring removal at 90 days, but before the final study visit. It further explored women's experiences using the IVR, including menstrual bleeding effects, impact on sex, removal circumstances, and future interest in use. (in the findings section, illustrative quotes include the participant ID with an extension of “1” or “2” to indicate when the statement was made.). IDIs in the DR were conducted in Spanish by a local researcher with expertise in qualitative data collection. IDIs with U.S. participants were conducted by phone or video by a trained qualitative researcher at FHI 360.
Audio recording was transcribed and translated and/or typed in English (in the DR) and then uploaded into NVivo 12, a qualitative software package facilitating textual data analysis. A team of 3 FHI 360 analysts followed a thematic coding process that included the following steps: (1) Reading to identify basic themes emerging in the text; (2) Coding to apply labels to text segments across all transcripts that represented the themes; (3) Memo-writing to provide a brief description of each overall theme/subtheme and examples of quotes; and (4) Data reduction by creating a matrix (in Excel) that quantitatively summarized key points across the different themes. This enabled the team to compare patterns by site and by regimen. Summary findings were shared with the DR team who provided review and feedback.
All participants signed written informed consent to participate in the clinical trial. Qualitative study participants provided a separate written informed consent that included permission for their interviews to be audiorecorded.
Results
Forty-seven participants completed baseline survey questions and 18 women participated in the qualitative substudy (11 from the DR and 7 from the U.S.), taking part in at least one IDI. A follow-up interview was conducted with 9 of the DR participants and 5 of the U.S. participants. The mean age of women in this trial was 37 years.
As expected, due to eligibility requirements, most participants perceived themselves to be at little or no risk of either pregnancy or HIV (Table 1). Altruistic motivations, including the importance of research to find effective prevention products and because it was necessary to improve health, were among the top 2 most endorsed reasons for joining this clinical trial. However, while women in the DR (60%) were also strongly motivated by the potential to learn new information about HIV, STIs, and pregnancy, almost three-quarters of women in the United States (73%) reported being motivated by the money they could get for volunteering.
Participant Characteristics at Baseline
p < 0.05.
Categories not mutually exclusive.
DR, Dominican Republic; IUD, intra-uterine device; STIs, sexually transmitted infection.
The qualitative data reveal important site-level differences in terms of perceived risk of HIV and motivations for trial participation. For example, whether they currently perceived risk to themselves, most participants from the DR strongly believed that HIV and other STIs were important threats to their community. Almost half of DR substudy participants (n = 5) knew someone personally who had been diagnosed with HIV.
In contrast, only one U.S. participant mentioned the threat of HIV within her own community. All but two U.S. participants who took part in IDIs mentioned that monetary reimbursements were a consideration for joining the trial, whereas most women from the DR were reticent to acknowledge the role that monetary incentives might have played. Many, however, were incentivized by the ability to access health care services, including receipt of pap smears, which would be too expensive outside of a trial setting. Most IDI participants (15 of 18) had participated previously in other trials.
Experiences with menstrual bleeding
The 22-item scale on menstrual period beliefs and practices factored into two subscales related to Preference for Amenorrhea (7 items, a = 0.87) and Negative Effects of Menstruation (8 items, a = 0.79). Mean scores on both subscales differed significantly by site with DR participants scoring lower on Preference for Amenorrhea and higher on Negative Effects of Menstruation than women from the U.S. (Table 2). Almost all DR participants (96%) managed their periods using menstrual pads, whereas women in the United States were likely to use a mix of hygiene products, including pads only (41%), tampons only (9%), or both pads and tampons (46%). (Data not shown).
Item and Subscale Mean Scores for Menstrual Period Beliefs and Behaviors Items, by Site, Regimen, and Agent
p < 0.05.
Items that reverse load on subscale.
Over the two follow-up acceptability surveys (V13 and V31), between 55% and 60% of participants reported at least one change to their menstrual cycles. Participants were most likely to report lighter bleeding or fewer days of bleeding, followed by heavier or more days of bleeding, or irregular bleeding (Table 3).
Menstrual Cycle Changes Experienced at Visit 13 (1 Month) and Visit 31 (3 Months)
Response options were 1 = Disagree a lot, 2 = Disagree somewhat, 3 = Disagree a little, 4 = Agree a little, 5 = Agree somewhat, 6 = Agree a lot; lower scores indicate more disagreement with component items while higher scores indicate more agreement with component items.
Most women who reported having lighter bleeding or fewer days of bleeding after initiating IVR use liked this change. In contrast, none of the women who reported heavier bleeding or more days of bleeding liked this change (Table 4). During qualitative interviews, women's descriptions of their typical menstrual cycles varied in duration and quality. Women expressed different concerns about the potential impact of IVR use on the menstrual cycle. For example, as a woman from the DR using the continuous IVR said:
Attitudes Toward Menstruation Changes by Regimen/Agent and Site
At the beginning, I was worried like everybody. I thought, ‘’How it was going to be?’ [...] I thought to myself, ‘What could happen to me? I will have (more) bleeding or maybe my menstruation will stop.’ (36-year-old woman from DR with 3 children on continuous IVR use, #125_2)
Three women from the DR expressed hope that the IVR would reduce their menstrual bleeding while several others worried that the IVR might increase, stop, or make their periods irregular. While most U.S. IDI participants were less concerned about cycle changes, two U.S. women hoped the IVR might stop their periods altogether. As one U.S. participant explained:
I never had a cycle. […] Well, I mean, obviously it was fine on my part and they told me it was possible that, you know, they would always ask if I got my cycle at my visit and I would tell them ‘No’… So, there was nothing not normal about not getting your cycle. So, I think it could be an added advantage, in my opinion. (34-year-old woman from US on interrupted use IVR, #221_2)
Attitudes toward IVR attributes
Overall, participants found IVR attributes very acceptable (Fig. 1a, b). For example, more than 87% of women in the DR and 75% of those in the U.S. found the way the IVR was inserted and removed very acceptable. Although generally acceptable, the size and thickness of the IVR, and the change in color observed over time (especially for the continuous regimen) had more variability in responses. Initial concerns about the size and thickness of the IVR were raised during more than half of the IDIs.
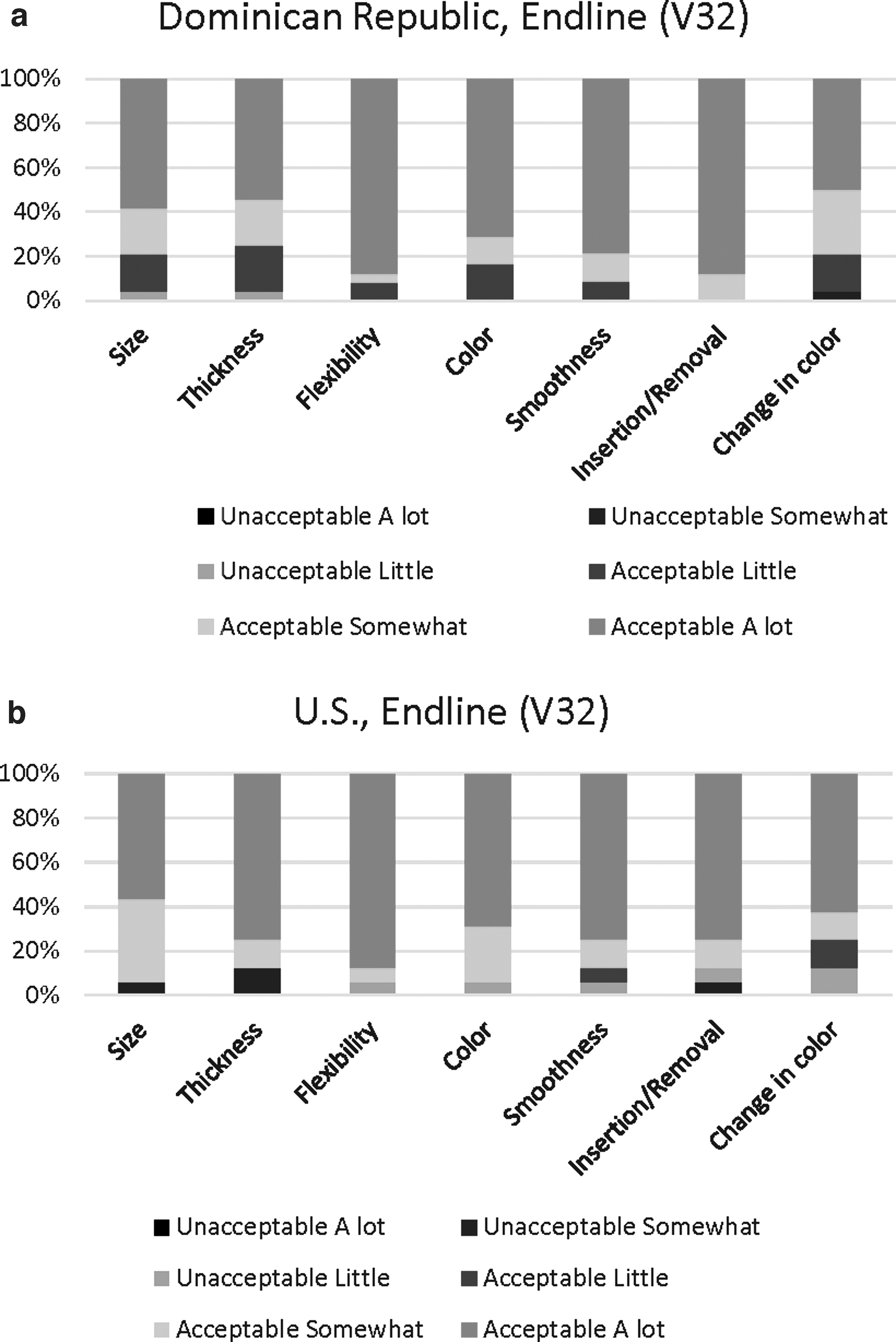
However, as suggested in the following quote, most participants concluded that such concerns were unwarranted.
Woah. That isn't a ring, it's a bracelet! [Laughter.] But afterward it was fine, once I put it in. It changed… I saw the size and I thought I wouldn't be able to insert it. But when I saw how it bent and it could be inserted, I understood. I thought it'd be smaller, a small little ring, but this was full size. I was concerned at first sight, but then it was comfortable to insert and take out. (35-year-old mother of four in the DR on continuous use, #113)
Most women liked the idea of the continuous ring. Almost half of substudy participants said they would be willing to use a ring that was even longer than a 3-month duration—6 months or even a year, particularly if it were shown to be safe and effective for that long. Several women equated the concept of longer duration of use with using an implant or an intra-uterine device (IUD).
I'd leave it in there! Because it doesn't bother me. As of now, it hasn't bothered me. I haven't found anything bad about it. The time that…let's say for a year… I wouldn't…If they say it's fine… no problem. Because you don't feel anything. (32-year-old mother of two in DR, #119_1)
However, a few women worried about hygiene or whether the ring might deteriorate or become ineffective if left in the body too long. In her second interview, the U.S. participant in the above quote mused further about the ideal duration of the IVR:
I mean, I think it'll be fine because the vagina is self-cleansing. I think that it'll be okay, I just wonder what condition it will be in. I mean, I guess my only concern is if it'll break down in three months and if that is going to be an issue for my body, that the ring is deteriorating but I'm guessing it'll be able to withstand being in there for three months, but I mean, I guess we'll see. (39-year-old mother of two children from US, #218_1)
Preferences for prevention products and future use
More than 85% of participants reported being very interested in using an IVR outside of a clinical trial, if it were available, with slightly higher interest reported in the DR (92%) versus the U.S. (81%) (Table 5). Conversely, only one participant suggested during an IDI that she would consider using a ring like this one, if available, after the trial. Nevertheless, participants felt certain that other women—especially adolescents and young women would want to use it.
Pregnancy/HIV Prevention Product Preferences by Regimen and Site at V32
p < 0.05 for comparison across group (i.e., regimen, agent, or site).
Categories not mutually exclusive.
I know that this specific ring also prevents HIV. So, I think that women who are more sexually active with multiple partners, this is probably more appealing. I don't know, I don't think STDs aren't (sic) really that big of a deal if you can just get rid of it. I mean, of course I don't want an STD or anything like that but if you get chlamydia or gonorrhea, you can just get rid of it. But HIV doesn't really go away, so I think that something like this is super cool if you have a ring that prevents birth control and HIV. That's like a huge deal to me. (28-year-old mother of one in US, #217_2)
When compared with other prevention products for pregnancy and/or HIV prevention, 75% of all participants—and more than 87% of women in the DR site, preferred a continuous use of IVR. In the United States, preferences for a greater range of products were reported. Most reasons provided for preferences differed by site. In the DR, women's preferences were strongest for products that did not interrupt sex, were perceived to be under their own control and could be used discreetly, without having to inform a partner or others. In the United States, women were especially interested in products perceived as easy to use. A desire for products perceived as less likely to cause harmful side effects was identified by more than a third of participants overall (Table 5).
Discussion
The acceptability of a 90-day TFV/LNG IVRs was found to be high among women participating in this Phase I clinical trial. Whether continuous or interrupted regimens, women in both sites found the IVR easy to insert and wear. Women generally liked various IVR attributes, including the mode of insertion/removal, the flexibility, size, and smoothness. They appreciated the idea of having a product that provided two-in-one protection and in principle considered that a ring that could be worn for 3 months or even longer (for some, up to a year) would be beneficial.
Although about half of participants noticed some changes to their menstrual cycles from ring use, a smaller proportion of women disliked these changes. In fact, most commonly, using this ring which releases a microdose of LNG, women experienced a reduction in bleeding quantity or duration—a change that most liked. Other side effects of ring use were rarely mentioned.
While these data provide initial indications of product acceptability, our findings have some limitations. First, women in this trial were at low risk for both pregnancy and HIV infection. Many had participated in prior clinical trials and their motivations for trial participation were varied, but mostly unrelated to future access to MPT products. Consequently, their perspectives are likely to differ from women who might be eventual end users. In addition, although we were able to compare, by regimen, agent, and site, women's experiences with menstrual bleeding changes, their acceptability of product attributes and preferences for IVRs compared with other prevention products, sample sizes for most comparisons are small.
User preferences for IVRs vis-à-vis other potential MPT delivery platforms were assessed in two different crossover studies, in which women had the opportunity to use and compare four placebo vaginal products (e.g., film, tablet, ring, and gel) 21 or three potential MPT products (e.g., injectables, rings, or tablets). 22 Although the IVR was not the preferred method for most women in either study, ring acceptability and adherence improved with use over time in both studies.
Product preferences differed across country settings. In a qualitative substudy, participants emphasized the importance of having a range of prevention options to choose from because women placed different values on attributes, such as ease of use, comfort, the need for discretion, or tolerance for various perceived side effects. 18 Our study also showed differences in preferences and reaction to changes according to clinical site and country, again reinforcing the importance of developing an array of options.
As in this study, multiple studies have found that women's initial concerns about the size of the ring and use during menses or sex diminish over time. 17,18 Furthermore, there is some evidence that product-related acceptability within placebo-controlled trials may be lower than during use of a product of known efficacy outside of a trial setting. As an example, preliminary findings from an open-label extension study of a 1-month IVR for HIV prevention found high and sustained levels of acceptance for the ring and lower-than-expected HIV incidence, as compared with findings from earlier Phase III trials. 23
Conclusion
Findings from this Phase I trial of a TFV/LNG IVR suggests interest in and potential demand for a multipurpose prevention vaginal ring that provides a 3-month or longer duration of protection and can be in the control of women.
Footnotes
Disclaimer
The contents are the sole responsibility of the authors and do not necessarily reflect the views of their institutions, PEPFAR, USAID, or the United States Government.
Authors' Contributions
E.E.T. was primarily responsible for the study design, guided data analysis, and drafted the acceptable article. S.Z. conducted data management and quantitative analyses. J.T. conducted qualitative interviews for the EVMS site and participated in qualitative analysis. H.H. contributed to the study design, analysis, and interpretation of data. D.T. conducted qualitative interviews for the DR site. S.J. provided clinical coordination, whereas J.S. provided medical oversight of both sites. A.T. and V.B. were primarily responsible for all data acquisition at EVMS and DR sites, respectively. J.S., A.T., V.B., and G.F.D. contributed to study design and interpretation of data. All provided critical review of the draft article, as well as approved and agreed to be accountable for the accuracy and integrity of the final article.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This study and the clinical development of the TFV/LNG ring were supported by the United States Agency for International Development (USAID) with funds from The U.S. President's Emergency Plan for AIDS Relief (PEPFAR) under Cooperative Agreements (AID-OAA-A-10-00068, AID-OAA-A-14-00010, and AID-OAA-A-14-00011). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the article.
