Abstract
Bone mineral density (BMD) changes during the life span, increasing rapidly during adolescence, plateauing in the third decade of life, and subsequently entering a phase of age-related decline. In women, menopause leads to accelerated bone loss and an increase in fracture risk. Between peak bone mass attainment and menopause, BMD is generally stable and the risk of fracture is typically low. This time period is marked by life events such as pregnancy and lactation, which transiently decrease BMD, yet their long-term effects on fracture risk are less certain. BMD may also be altered by exposure to medications that affect bone metabolism (e.g., contraceptives, glucocorticoids, antidiabetic medications, antiepileptic drugs). Although oral contraceptives are often believed to be neutral with regard to bone health, depot medroxyprogesterone acetate (DMPA) and gonadotropin-releasing hormone (GnRH) agonists have been associated with decreases in BMD. Development of newer medical therapies, principally GnRH antagonists (e.g., ASP1707, elagolix, linzagolix, relugolix), for treatment of endometriosis-associated pelvic pain and heavy menstrual bleeding due to uterine fibroids has renewed interest in the short- and long-term impacts of changes in BMD experienced by premenopausal women. It is important to understand how these drugs influence BMD and put the findings into context with regard to measurement variability and naturally occurring factors that influence bone health. This review summarizes what is known about the effects on bone health pregnancy, lactation, and use of DMPA, GnRH agonists, and GnRH antagonists in premenopausal women and potential consequences later in life.
Introduction
Bone mass peaks in a woman's 20s, 1 with a subsequent plateau until the mid-40s (Fig. 1). Menopause marks a transition to accelerated bone loss; however, transient changes in bone mineral density (BMD) may occur before menopause during the “plateau” period for a variety of reasons, including biological processes (pregnancy, lactation), 2 adverse effects of certain diseases or conditions (e.g., hyperparathyroidism, hyperthyroidism, Cushing's syndrome), 3 and use of pharmacologic therapies (e.g., glucocorticoids, antidiabetic medications, antiepileptic drugs, antipsychotic agents, selective serotonin reuptake inhibitors, chronic heparin, gonadotropin-releasing hormone [GnRH] agonists). 3 –5 Use of such medications is common; for example, in a longitudinal community-based study, 16% of perimenopausal and early postmenopausal women reported a history of medications associated with BMD changes. 6 In addition, a history of contraceptive use or medications to treat gynecologic conditions was high (oral contraceptives, 75%; sex steroid hormones other than birth control pills, 37%). 6 Although oral contraceptives are generally believed to be neutral with regard to bone health, 7 –9 depot medroxyprogesterone acetate (DMPA) 10 and GnRH agonists 7 are associated with decreases in BMD. The influence of these drug-related reductions in BMD on risk of fracture later in life has not been well established. 10,11
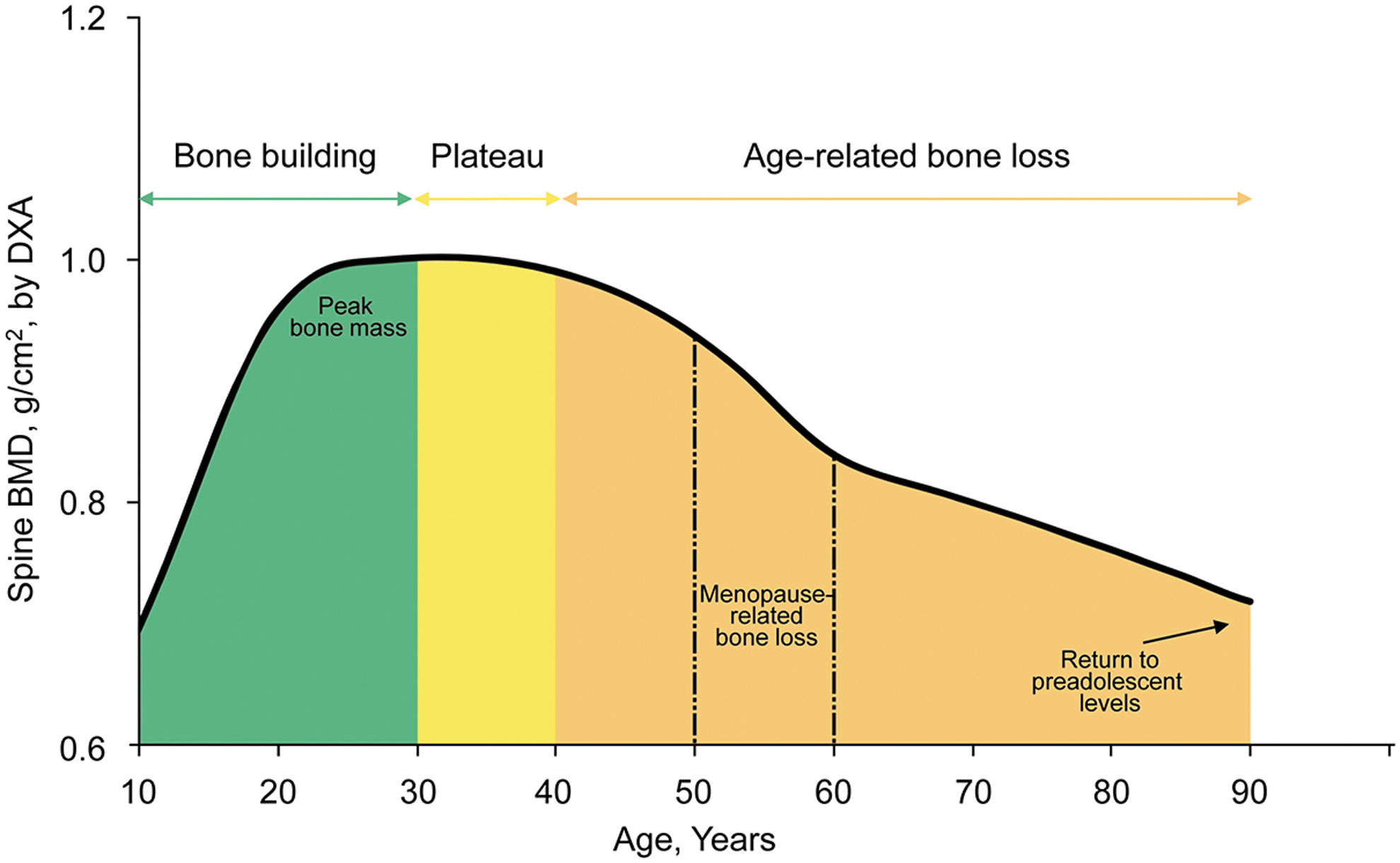
Changes in BMD in women over time. BMD, bone mineral density; DXA, dual-energy X-ray absorptiometry.
Given the increased risk with age at the population level, the bulk of evidence regarding fracture likelihood relates to older postmenopausal women. 12 However, events during the premenopausal years have the potential to influence BMD trajectory throughout the life span 13 and could affect risk of fracture later in life. Development of newer medical therapies, principally GnRH antagonists (i.e., ASP1707, elagolix, linzagolix, relugolix), for treatment of pelvic pain due to endometriosis and heavy bleeding due to uterine fibroids has renewed interest in the impact of changes in BMD experienced by premenopausal women. Salient among the questions regarding these agents are the magnitude of effect on BMD, the reversibility of changes, and if there is any impact on risk of fracture later in life. It is also important to understand these changes within the context of measurement variability, duration of use of such medical therapies, and naturally occurring factors that influence bone health. This review summarizes the effects of premenopausal life events such as pregnancy and lactation and use of DMPA, GnRH agonists, and GnRH antagonists on bone health and later life fracture risk to assist physicians and patients make informed treatment decisions.
Search Methodology
We conducted a literature search using a multifile database platform (Dialog Solutions, Ann Arbor, MI), which accessed data from the Derwent Drug File, Embase, and MEDLINE. The initial broad search strategy was augmented by subsequent targeted MEDLINE searches to answer specific research questions. Articles published in English from January 1987 (when dual-energy X-ray absorptiometry [DXA] was first available) through September 2020 were reviewed for relevance; articles selected for inclusion were related to pregnancy, lactation, DMPA, GnRH agonists, or GnRH antagonists and reported DXA-measured BMD data in premenopausal women and/or bone fracture risk in premenopausal or postmenopausal women. For inclusion, studies must have specified BMD measurement or fracture location and timing relative to the exposure of interest (i.e., life event or medication use). Only fractures relevant to fragility/osteoporosis were considered (see “Evaluating fracture relevance” below). Articles describing fracture risk related to pregnancy or lactation were limited to large prospective studies published within the past 10 years. Data extracted from the identified articles included study design, patient population, relevant inclusion/exclusion criteria, percent change from baseline in BMD, odds ratios, hazard ratios, and fracture rates.
Interpreting Measures of Bone Density
BMD measurements performed using DXA (expressed in units of g/cm2) are often converted to T-scores or Z-scores for interpretation. An individual's T-score indicates how many standard deviations (SDs) their BMD measurement diverges from the mean BMD of a reference population (e.g., young Caucasian women). 14 For premenopausal women, imaging guidelines recommend use of Z-scores rather than T-scores. 3,15 The Z-score reflects the number of SDs a BMD measurement diverges from a mean BMD value derived from individuals of the same sex, age, and race. Because of differences in average BMD values among racial/ethnic groups, 14,16 the reference population used for Z-score calculation is specific to the patient's race/ethnicity. 15 A Z-score in the negative range does not signify that bone loss has occurred; a score of −2.0 or lower is interpreted as below the expected range for the patient's age. 3,15
Interpretation of BMD deviations from the norm or changes over time are subject to the inherent limitation of measurement variability. Duplicate DXA scans of the same individual may yield slightly different results due to inherent variability, generally 2%–3% for the spine and 3%–4% for the femoral neck and total hip. Variability is greater when looking at long-term changes compared with duplicate measurements made at the same time. 17 Additional variability may be introduced by use of different machines; 18 for example, GE Healthcare DXA systems are calibrated to read BMD ∼10% higher than Hologic DXA systems, but both yield similar Z-scores and T-scores. Measurement accuracy may also be compromised in those at extremes of body mass index. 3 Beyond the technical aspects of precision, there is also the need to apply the appropriate reference standards relative to the individual being assessed (i.e., use of Z-scores vs. T-scores for premenopausal women and use of a Z-score reference from a matching racial/ethnic group).
When assessing changes over time, group mean percentage change in BMD from baseline is generally reported in clinical trials. A 1% change in BMD is roughly equivalent to a 0.1-unit change in Z-score or T-score. Importantly, a group mean change from baseline in BMD of 1% may be reported in a clinical trial, but such a difference is too small to measure at the individual patient level. On an individual level, it is essential to know the DXA facility's least significant change to determine if a noted BMD “difference” is real. 15
Evaluating Fracture Relevance
When assessing the influence of BMD decreases on fracture risk, demographic factors, BMD relative to the reference standard (i.e., T-score or Z-score, as appropriate), and type of fracture inform possible associations. Although fragility fractures are often defined as any fracture resulting from low-energy trauma (e.g., a fall from standing height or lower), 19,20 the likelihood that a fracture is related to osteoporosis must take into account the individual's age and BMD, as well as fracture location. Fractures associated with osteoporosis are more likely to occur in older rather than younger individuals, are more common among those with lower versus higher BMD, and typically affect the humerus, radius, spine, pelvis, or femur. Fractures of the ankles, knees, elbows, shoulders, sternum, clavicle, or ribs have a less certain relationship to osteoporosis, 21 whereas fractures of the skull, face, fingers, toes, hands, or feet are unlikely to be related to skeletal fragility.
Effects of Life Events on Bone Health in Premenopausal Women
Pregnancy
Calcium metabolism and bone mineral status are markedly affected during pregnancy, as physiologic adaptations ensure that adequate mineral transfer is provided to the developing fetus and for upcoming lactation. 22 The resulting increase in bone remodeling generates a BMD decline in early pregnancy, with subsequent gains postpregnancy. An estimated BMD loss of <1%–9% occurs from before conception to immediately postpartum, the magnitude of which varies by study and skeletal location. 23 –29 In prospective longitudinal studies, the ranges of BMD decreases by skeletal location were <1%–2% for forearm, 1% for total hip, 4% for trochanter, 1%–8% for femoral neck, and 1%–9% for lumbar spine. 23 –29 Salari and Abdollahi 2 describe the confounding influences that contribute to disparate results among studies, including differences in study design, populations, densitometry, and bone sites measured.
The net influence of pregnancy-related BMD changes on long-term bone health remains to be elucidated, as the available evidence is mixed. 2 Studies evaluating the influence of pregnancy on BMD later in life generally report minimal to modest positive effects of parity history on BMD. 6,30,31 However, data are generally derived from observational cohort studies assessing historical reproductive factors such as past pregnancy, parity, and breastfeeding as contributing factors to current BMD. There is a lack of prospective, long-term studies evaluating changes in BMD in parous versus nulliparous women.
Lactation
As with pregnancy, lactation involves changes in bone metabolism to facilitate mineral transfer to the growing infant. 22 The result is a net maternal calcium loss as it is excreted in breast milk. In addition, lactation suppresses the hypothalamic-pituitary-ovarian axis, resulting in a hypoestrogenic state and consequent increased bone turnover. 32,33 In longitudinal studies, decreases in BMD during lactation, which are typically measured at ∼6 months postpartum, range from 1% to 5% for radius, 1% to 8% for lumbar spine, 3% to 6% for femoral neck, and 4% for total hip. 23 –25,32 –37 The dynamics of BMD changes during and after lactation differ by skeletal location (Fig. 2). 32 In a 2009 longitudinal study of lactating women, femoral neck and total hip BMD decreased by 4% and lumbar spine BMD decreased by 1% from 1 month (baseline) to 6 months postpartum. 32 At 18 months postpartum, femoral neck BMD was 1% below baseline, total hip BMD had returned to the baseline mean, and lumbar spine BMD had increased by 6% relative to baseline. The lumbar spine recovery data align with a previous report in which a 7% increase in BMD was observed from ∼3 days postpartum (baseline) to 18 months postpartum in women who breastfed for an average of 6 months. 35 At 3 to 4 months postpartum (during lactation), lumbar spine BMD was 3% below baseline. Holmberg-Marttila et al. 34 also noted an increase relative to baseline in lumbar spine BMD but an ongoing decrement in femoral neck BMD (−1.1%) ∼19 months postpartum in women who breastfed.
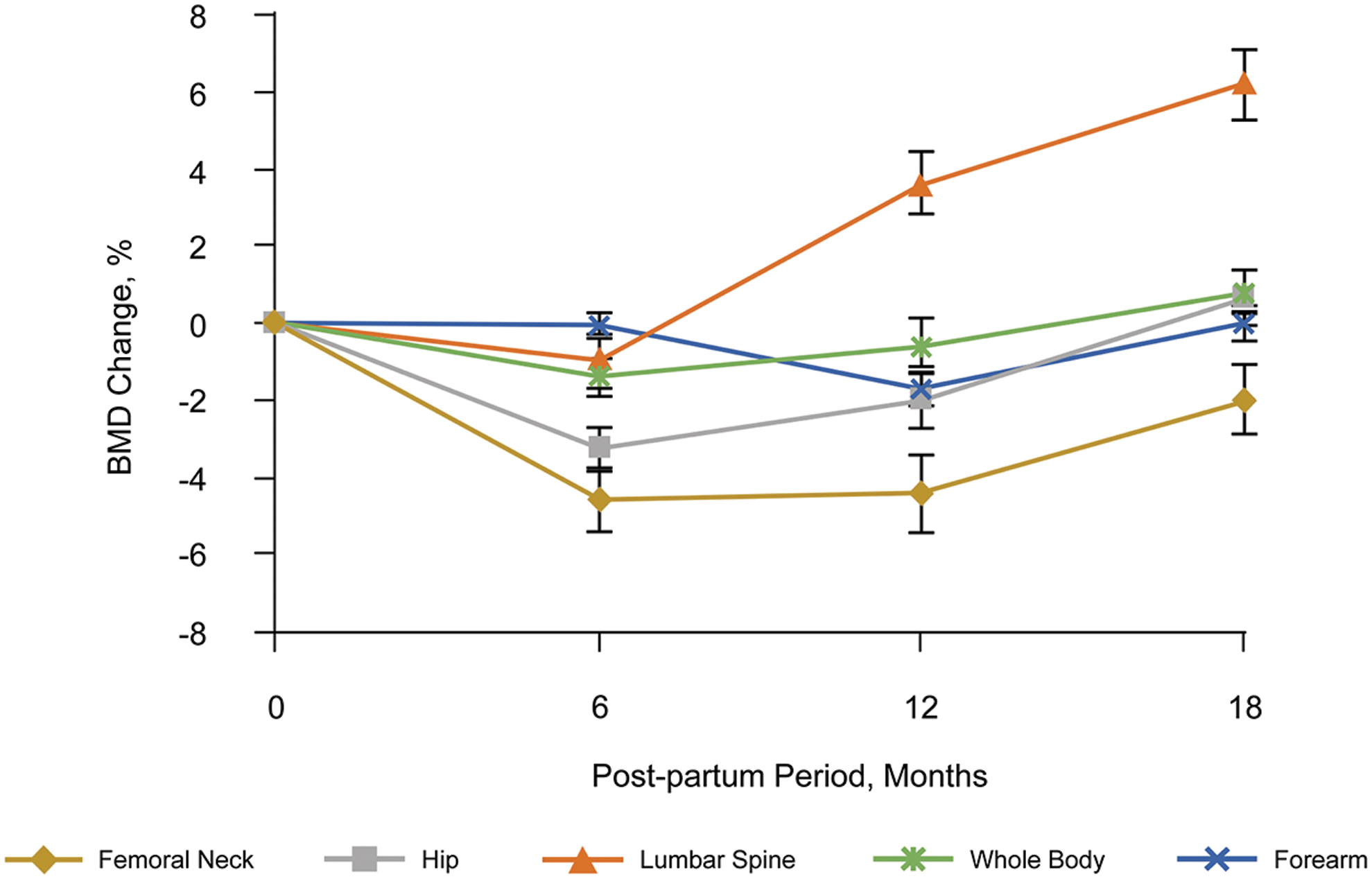
Postpartum changes in BMD in women who breastfed. Data were obtained from 36 lactating women (mean age, 23.5 years) living in a socioeconomically disadvantaged region in India who had a history of prior childbirth and were not receiving any medications known to affect bone metabolism. Femoral neck and hip, lumbar spine, whole body, and forearm measurements were available for 36, 35, 32, and 28 women, respectively. Data are mean ± standard error. Reproduced with permission from Kulkarni et al. 32
Data indicate that the recovery of lumbar spine and femoral neck BMD is slower in women with a longer breastfeeding duration. 24,25 Consistent with the direct correlation between duration of lactation and time to resumption of menses after childbirth, women with a longer postpartum duration of amenorrhea experience a lower BMD nadir and slower recovery trajectory. 24,25
Beyond the 18-month window of observation from longitudinal studies, the data for long-term effects of lactation on BMD are limited by a lack of prospective studies. Results from observational cohorts assessing the influence of breastfeeding history and duration on BMD are inconsistent. 6,30,38 –40
Pregnancy, lactation, and fracture risk
The risk of fracture is generally low among premenopausal women 14 ; however, there are rare cases, estimated at a prevalence of 4–8 per 1 million pregnancies, of pregnancy- and lactation-associated osteoporosis. 41,42 Unlike most premenopausal women, this small group of patients are at increased immediate risk for fracture. As risk of fracture naturally increases with age, most studies correlating pregnancy and lactation with fracture risk do so in postmenopausal women. Data from large, prospective observational studies do not indicate that pregnancy, parity, lactation, or breastfeeding duration increases risk of fracture later in life (Table 1). 6,43 –47 Indeed, there is evidence to suggest that parity and breastfeeding may have a protective effect, lowering the risk for hip fracture in postmenopausal women. 43 –45
Listing of Large Prospective Studies Assessing the Effects of Pregnancy and Lactation on Fracture Risk
Excluded patients with a history of vertebral fracture at baseline or who had conditions or medications that influence BMD.
BMD, bone mineral density; CI, confidence interval; HR, hazard ratio.
Overall, the evidence indicates that pregnancy and lactation result in transient decreases in BMD that do not worsen risk of fracture later in life.
Effects of DMPA, GnRH Agonists, and GnRH Antagonists on Bone Health
Depot medroxyprogesterone acetate
DMPA is a progestin-only contraceptive with a subcutaneously administered form that is used in the management of endometriosis-associated pain. DMPA leads to greater suppression of estradiol compared with other progestin-only contraceptives, which results in a more pronounced effect on bone metabolism. 10 In premenopausal women aged 18 to 35 years who were first-time users of DMPA for contraception (n = 178), mean BMD decreases of 2.8% at the hip and 3.5% at the spine were reported after 1 year of treatment and decreases of 5.8%–7.7% at the hip and 5.7%–6.4% at the spine after 2 years of treatment. 48,49 Although recovery of BMD has been reported in some long-term studies, 10 bone loss may not be completely reversible. 49 –51 Because of its effects on BMD, prescribing information for DMPA recommends limiting its use to 2 years. 50 In a 2014 committee opinion (reaffirmed in 2017), the American College of Obstetricians and Gynecologists acknowledged BMD loss with DMPA and noted that the effect on fracture risk later in life remains an unanswered question; however, the committee concluded that these observations “should not prevent practitioners from prescribing DMPA or continuing use beyond 2 years.” 52
Multiple studies and meta-analyses have evaluated the influence of DMPA on fracture risk; however, analyses that include all skeletal fracture sites rather than those associated with fragility provide little insight into the potential for fracture related to low BMD. Among studies that focused on osteoporosis-related fractures, there was a signal for a small increase in fracture risk for DMPA users versus nonusers (Table 2). 53 –55 Notably, data were derived predominantly from younger populations, creating a skew toward premenopausal women. It remains to be determined what effect a history of DMPA use has on fracture risk in postmenopausal women.
Listing of Articles Assessing the Effects of Depot Medroxyprogesterone Acetate On Osteoporotic/Fragility Fracture Risk
Excluded women with conditions or history of medications known to affect bone metabolism.
DMPA, depot medroxyprogesterone acetate; IUD, intrauterine device; OCP, oral contraceptive pill.
The accumulated evidence suggests that decreases in BMD observed with DMPA may translate into increased risk of fracture in premenopausal women, a finding that necessitates further study. As fracture risk appears to be greatest in current or recent DMPA users and declines with discontinuation of therapy, exploration of fracture risk associated with past DMPA use in postmenopausal women is warranted.
GnRH agonists
GnRH agonists are used in the treatment of multiple conditions affecting premenopausal women, including endometriosis, uterine fibroids, and adenomyosis, as well as for fertility preservation. 11 After an initial flare of pituitary gonadotrophs, GnRH agonist administration profoundly reduces luteinizing hormone and follicle-stimulating hormone levels, thereby suppressing estrogen and progesterone production. This mechanism of action induces maximum ovarian suppression, with associated side effects that include decreases in BMD. 11 Sauerbrun-Cutler and Alvero 11 reviewed the effects of GnRH agonists on bone loss and fracture in 2019. They cite bone loss with GnRH agonists of 2%–6% in total hip, lumbar spine, and proximal femur, with partial to full recovery observed 1 year after treatment cessation. Among GnRH agonists, differences have been observed in the degree of effect on BMD. For example, reductions in BMD during 6 months' treatment with buserelin (−3.7%) in a small study of women with endometriosis or uterine fibroids (N = 27) were less than that observed with leuprolide (−5.1%), a finding that is likely attributable to differences in the extent of sex hormone suppression. 56 To mitigate the hypoestrogenic effects of GnRH agonists, including reductions in BMD, they may be administered in conjunction with add-back therapy to restore estrogen to a level at which bone loss is prevented but endometrial tissue is not stimulated. 11 Data from meta-analyses 57,58 and recent studies 59,60 confirm that add-back therapy mitigates bone loss associated with GnRH agonist treatment.
GnRH antagonists
GnRH antagonists, which offer the potential for partial rather than complete suppression of the hypothalamic-pituitary-ovarian axis, may differ from GnRH agonists with regard to changes in BMD. The greatest body of evidence to date for a GnRH antagonist used in the treatment of gynecologic conditions in premenopausal women comes from studies of elagolix. Over the course of treatment for 12 weeks to 24 months in randomized clinical trials, modest (<1%–4%), dose-dependent reductions in BMD at the total hip, spine, and femoral neck have been observed with elagolix at doses of 150 mg once daily/75 mg twice daily to 600 mg once daily/300 mg twice daily without add-back therapy (Table 3). 61 –65 As expected based on the mechanism of action, the greatest reductions in BMD were observed with the higher dose regimens. Further reductions in BMD were observed in long-term extension studies; 66 however, BMD was generally similar to or improved with respect to on-treatment values in patients with post-treatment measurements. 61,62,66,67
Listing of Recent Articles Assessing the Effects of Gonadotropin-Releasing Hormone Antagonists on Bone Mineral Density in Premenopausal Women
p < 0.05 vs baseline; b p < 0.01, c p < 0.001 vs. placebo, d p < 0.05; e p < 0.001 vs leuprolide, f p ≤ 0.01 vs leuprolide, g p < 0.05 vs leuprolide.
The delayed add-back group received 12 weeks of relugolix monotherapy followed by 12 weeks of relugolix plus add-back therapy.
AB, add-back; BID, twice daily; E2, estradiol; EAP, endometriosis-associated pain; FN, femoral neck; GnRH, gonadotropin-releasing hormone; HMB, heavy menstrual bleeding; LS, lumbar spine; NETA, norethindrone acetate; PT, post-treatment; QD, once daily; RCT, randomized controlled trial; TH, total hip.
In head-to-head comparisons, changes in lumbar spine BMD were similar during 24 weeks of treatment with elagolix 150 mg once daily, elagolix 75 mg twice daily, or DMPA (104 mg/0.65 mL administered on weeks 1 and 12) 61 and greater during 12 weeks of treatment with monthly depot leuprolide acetate 3.75 mg compared with elagolix 150 or 250 mg once daily (Table 3). 68 The difference in BMD reduction with leuprolide was attributed to greater estradiol suppression; on-treatment estradiol concentrations were significantly lower with leuprolide versus elagolix (median, 5 and 32 mg, respectively; p < 0.001). In a multivariable regression analysis, on-treatment concentration of estradiol was significantly associated with changes in lumbar spine BMD (p = 0.019).
Add-back therapy has proved effective for mitigating hypoestrogenic effects associated with GnRH antagonists, including changes in BMD (Table 3). 64,65,67,69 At month 6 in 2 identical, phase 3, randomized, placebo-controlled clinical trials of elagolix in women with heavy menstrual bleeding and uterine fibroids, differences in BMD reduction at most measured sites were statistically significant for elagolix 300 mg twice daily alone compared with placebo (p < 0.05), whereas changes from baseline in mean BMD in patients receiving elagolix 300 mg twice daily plus add-back therapy (1.0 mg estradiol/0.5 mg norethindrone acetate) were similar to the placebo group. 65 Patients enrolled in these phase 3 trials had the option of continuing treatment with elagolix or elagolix plus add-back therapy in a 6-month extension study. 68 After a total of 12 months' treatment, lumbar spine BMD decreased by 4.8% (95% confidence interval [CI] −5.4 to −4.1) with elagolix and 1.5% (95% CI −1.9 to −1.0) with elagolix plus add-back therapy. Increases in BMD were observed 6 months post-treatment in both groups and were maintained or further improved at 12 months post-treatment in patients who had received elagolix without add-back therapy. An ongoing phase 3 clinical trial is assessing the use of add-back therapy in conjunction with elagolix in women with endometriosis-associated pain.
One publication each was identified for the GnRH antagonists ASP1707, linzagolix, and relugolix (Table 3). 69 –71 A phase 2, dose-ranging clinical trial assessed the efficacy and safety of ASP1707 3 to 15 mg once daily compared with placebo and monthly leuprolide acetate 3.75 mg in premenopausal women with endometriosis (N = 540). 70 Decreases in BMD at week 24 were observed in all active-treatment groups; however, the magnitude of decrease in total hip and spine BMD was significantly greater with leuprolide versus ASP1707 (−1.2% to −2.3% vs. −3.9%; p < 0.001). Notably, median serum estradiol concentrations were lower in the leuprolide treatment group than in any of the ASP1707 dose groups. A phase 2b, dose-ranging clinical trial evaluated linzagolix 50 to 200 mg/day in premenopausal women with endometriosis. 71 Modest, dose-dependent reductions in lumbar spine BMD (1%–3%) were observed during the 24-week treatment period in patients who received linzagolix doses of 75 mg/day or higher. Two phase 3, randomized, placebo-controlled clinical trials compared relugolix 40 mg once daily administered with add-back therapy (1.0 mg estradiol/0.5 mg norethindrone) at the start of treatment (immediate add-back) or initiated after 12 weeks of monotherapy (delayed add-back) in premenopausal women with heavy menstrual bleeding and uterine fibroids. 69 During the first 12 weeks of treatment, total hip and lumbar spine BMD decreased by 1% and 2%, respectively, in the delayed add-back therapy group, and minimal decreases in lumbar spine BMD were observed in the immediate add-back therapy group. Initiation of estradiol and norethindrone acetate halted further BMD loss in the delayed add-back therapy group but did not reverse prior BMD decreases. No data on the reversibility of changes in BMD were reported for ASP1707, linzagolix, or relugolix.
There is currently a lack of data on short- or long-term fracture risk associated with GnRH antagonist use in premenopausal women.
Clinical Perspective
Premenopausal women have a low baseline risk of fracture (Fig. 3). 14 Risk of fracture roughly doubles for every decade of life after age 50 years. 72 Therefore, when assessing the downstream sequelae of reductions in BMD due to life events or use of pharmacologic therapies, the premenopausal and postmenopausal epochs warrant separate evaluation. For premenopausal women, the available data indicate that although pregnancy and lactation stress the calcium economy and reduce BMD, 22 this appears to not translate into adverse skeletal consequences in their younger years, except in exceedingly rare cases (Table 4). Studies of DMPA users, however, suggest that fracture risk is slightly elevated among premenopausal women. 53,55 For GnRH agonists and GnRH antagonists, clinical trials have not shown an increase in fracture risk, but the sample size, duration of follow-up, and low baseline risk of the study populations limit the ability to detect rare fracture events. Moreover, clinical trials typically excluded women with T-scores or Z-scores less than −1.5 or who had conditions than can be associated with decreased BMD, thus omitting patients who may have a baseline elevated risk.
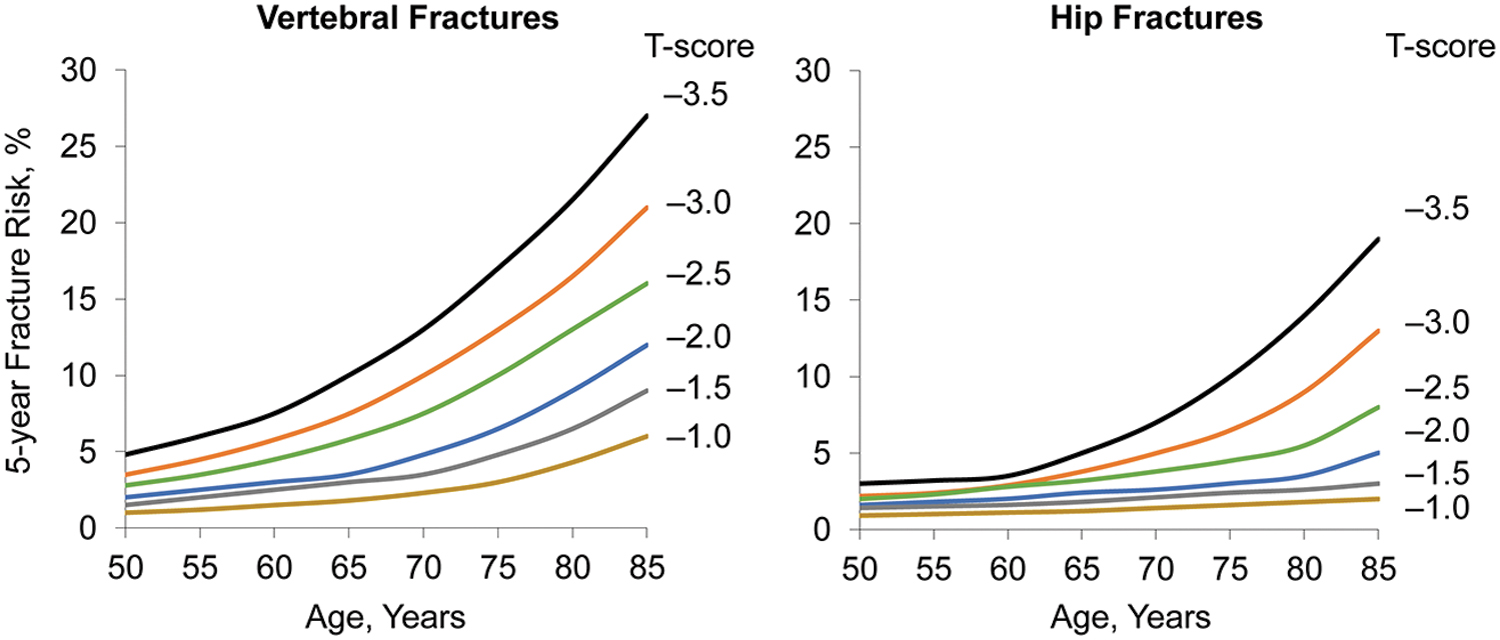
Influence of age on fracture risk. Five-year vertebral (left panel) and hip (right panel) fracture risk with age and T-score based on data from the Study of Osteoporotic Fractures. Reproduced with permission from Cummings et al. 14
Summary of Effects on Bone Mineral Density and Fracture Risk
In postmenopausal women, data indicate no increased fracture risk associated with prior pregnancy or lactation and are insufficient for DMPA and GnRH agonists or antagonists. To assess the potential for later life fracture risk associated with GnRH antagonist use, a recent modeling study used data from phase 3 clinical trials of elagolix to simulate treatment effects in women ages 50 to 79 years of age. 73 Results of this modeling analysis revealed only a minimal effect of treatment on the 10-year risk of major osteoporotic fracture or hip fracture and a slight increase in the projected proportion of women reaching risk-based thresholds for antiosteoporotic treatment. Based on these findings, we would not anticipate that modest on-treatment changes in BMD warrant concern regarding later life fracture risk. Notably, the studies on which the fracture risk model was based did not include add-back therapy. With the mitigating effects of add-back therapy on BMD reductions, the combination of GnRH agonists or antagonists with add-back therapy would be expected to have an even smaller effect on fracture risk.
Given the observations described in this review, is BMD monitoring necessary in premenopausal women who have experienced pregnancy or lactation or used medications associated with bone loss? Professional societies do not currently recommend BMD testing for premenopausal women without risk factors. 15,74,75 As a population, the risk of fracture is low in premenopausal women, even in subgroups with risk factors such as medication use. 53,55 Moreover, percent changes from baseline in BMD observed with DMPA and GnRH antagonist use are modest, particularly during short-term treatment, and may not be detectable at the individual level due to the innate variability of densitometry (Table 4). Post-treatment group level data also indicate at least partial BMD recovery after treatment cessation. In addition, the amount of drug-induced bone loss that is tolerable without reaching treatment thresholds later in life varies at the individual level due to differences in starting bone mass (i.e., the impact of bone loss will be less for patients with higher peak bone mass). 13
Osteoporosis screening with BMD measurement is recommended in the United States for postmenopausal women, beginning at age 65 years, 3,15,74 as BMD measurement has its greatest prognostic value for short-term osteoporotic fracture risk in older women. Earlier BMD assessment (at menopause or perimenopause) is suggested for women with risk factors such as family history of osteoporosis, low body mass, prior fracture, current smoking, excessive alcohol use, loss of height, and use of high-risk medications associated with reductions in BMD (e.g., glucocorticoids [risk by dose and duration discussed by Leib et al. 76 ], antiepileptic drugs, aromatase inhibitor therapy, chronic heparin). Women found to be at high risk of fracture due to a BMD T-score of −2.5 or less, a BMD T-score of −1 to −2.5 with a calculated 10-year risk of ≥3% for hip fracture or ≥20% for major osteoporosis-related fracture, or a recent fracture thought to be related to osteoporosis are candidates for pharmacologic therapy to reduce fracture risk. 77 –79 Repeat DXA for monitoring would be indicated for women who are nearing an intervention threshold but is probably not necessary for those with BMD that is average or well above average.
Conclusions
Pregnancy, lactation, and exposure to agents that may affect bone (e.g., contraceptives, glucocorticoids, antidiabetic medications, antiepileptic drugs) are a common experience among premenopausal women. The reductions in BMD associated with pregnancy, lactation, or medications, including GnRH agonists or GnRH antagonists, are modest, transient, and unlikely to increase fracture risk in premenopausal women. However, modest increases in fracture risk with current or past DMPA use have been observed in premenopausal women. As fracture risk in premenopausal women is low, the absolute risk remains small. At the individual level, monitoring BMD via DXA would generally not be informative because the magnitude of change is within the range of measurement variability and there is no established threshold for defining a BMD decrease that signals elevated fracture risk in premenopausal women. Further studies are needed to determine the effect of premenopausal changes in BMD on fracture risk later in life.
Footnotes
Authors' Contributions
All of the authors were involved in the conception and design of the article and in the interpretation of the data. The authors reviewed and revised article drafts, approved the article version that was submitted for publication, and take responsibility for the accuracy and integrity of the work.
Author Disclosure Statement
N.B.W. served as a consultant for AbbVie and ObsEva and is a speaker for Amgen and Radius. A.A. has received research support from the National Institutes of Health and has served as a consultant for AbbVie, Myovant, Bayer, ObsEva, and Novartis. N.B. has received research support from Radius and GE Healthcare and has served as a consultant for Amgen. C.D.O. and M.S. are employees of AbbVie. E.E.P. has received research support from AbbVie, Bayer, and ObsEva and has served as a consultant for Femasys. J.A.S. has received research support from AbbVie, Bayer Healthcare LLC., Myovant, ObsEva SA, Sebele Pharmaceuticals, Inc., and Viveve Medical. He has served as a consultant for Bayer HealthCare Pharmaceuticals, Inc., Camargo Pharmaceutical Services, LLC, Covance, Inc., Dare' Bioscience, DEKA M.E.L.A S.r.l., Femasys, Inc., KaNDy/NeRRe Therapeutics Ltd., Madorra Pty Ltd., Mitsubishi Tanabe Pharma Development America, Inc., Sebela Pharmaceuticals, Inc., and Sprout Pharmaceuticals, Inc. He has also served on speakers' bureaus for TherapeuticsMD and is a stockholder in Sermonix Pharmaceuticals. W.D.S. has received research support from AbbVie and has served as a consultant for AbbVie, including the Uterine Disorders National Steering Committee.
Funding Information
Medical writing support for development of this article was funded by AbbVie, Inc., and was provided by Crystal Murcia, PhD, and Kersten Reich, MPH, CMPP, of JB Ashtin.
