Abstract
Cardiovascular disease (CVD), including hypertensive disorders of pregnancy (HDP) and peripartum cardiomyopathy, is a leading cause of pregnancy-related death in the United States. Women who are African American or American Indian/Alaskan Native, have HDP, are medically underserved, are older, or are obese have a major risk for the onset and/or progression of CVD during and after pregnancy. Paradoxically, women with no preexisting chronic conditions or risk factors also experience significant pregnancy-related cardiovascular (CV) complications. The question remains whether substantial physiologic stress on the CV system during pregnancy reflected in hemodynamic, hematological, and metabolic changes uncovers subclinical prepregnancy CVD in these otherwise healthy women. Equally important and similarly understudied is the concept that women's long-term CV health could be detrimentally affected by adverse pregnancy outcomes, such as preeclampsia, gestational hypertension, and diabetes, and preterm birth. Thus, a critical life span perspective in the assessment of women's CV risk factors is needed to help women and health care providers recognize and appreciate not only optimal CV health but also risk factors present before, during, and after pregnancy. In this review article, we highlight new advancements in understanding adverse, pregnancy-related CV conditions and will discuss promising strategies or interventions for their prevention, diagnosis, and treatment.
Introduction
The maternal mortality rate is rising in the United States even as rates decrease globally. Causes of maternal mortality are multifaceted, and in the United States, cardiovascular disease (CVD) is the primary cause of pregnancy-related mortality. 1 The question remains whether substantial physiological stress on the cardiovascular (CV) system during pregnancy—reflected in hemodynamic, hematological, and metabolic changes—uncovers subclinical prepregnancy CVD in otherwise healthy women. Equally important and similarly understudied is the concept that women's long-term CV health could be detrimentally affected by adverse pregnancy outcomes (APOs), such as preeclampsia (PE), gestational hypertension, and diabetes, and preterm birth. Thus, a critical life span perspective in assessment of women's CV risk factors is needed.
We highlight advances in understanding the emergence of CV conditions during pregnancy and postpartum (specifically hypertensive disorders of pregnancy [HDP] and peripartum cardiomyopathy [PPCM]) and their association with CV risk over the life course. We explore how risk prediction using biomarkers could potentially facilitate stratification of antenatal care and testing of potential preventative and therapeutic interventions.
We also discuss opportunities for counseling, planning, and interventions to optimize treatments for underlying medical conditions associated with increased CV risk to improve pregnancy outcomes and long-term CV health. We also review follow-up care models and recommend strategies for implementing these models into clinical practice.
Hypertensive Disorders of Pregnancy
Hypertension is the most common complication observed during pregnancy, affecting 8%–10% of all pregnancies in the United States. 2,3 HDP continue to be among the leading causes of pregnancy-related maternal mortality worldwide and contribute to 7%–12% of pregnancy-related maternal deaths in the United States annually. 4 –6 Importantly, the risk of developing CVD in women with a history of HDP is twice that seen in normotensive pregnancies. 7 HDP can be classified into four general categories: (1) PE and eclampsia, (2) chronic hypertension (of any cause), (3) chronic hypertension with superimposed PE, and (4) gestational hypertension. 8 Although we will reference other forms of HDP, we will predominantly focus on PE in this article.
New advancement in the pathogenesis of HDP
PE is classically defined as new-onset hypertension and proteinuria developing in the second half of pregnancy and resolving after delivery, yet it is multiorgan HDP that can present with systemic manifestations (e.g., elevated liver enzymes and low platelets) even before the development of proteinuria. 2 According to the most recent definition endorsed by the International Society for the Study of Hypertension in Pregnancy, PE is defined as new-onset hypertension (systolic >140 mmHg and diastolic >90 mmHg) accompanied by one or more other features: proteinuria, other maternal organ dysfunction (including the liver, kidney, and nervous system), or hematological involvement, and/or uteroplacental dysfunction, such as fetal growth restriction and/or abnormal Doppler ultrasound findings of uteroplacental blood flow. 9
Although our understanding of the pathophysiology of PE has increased dramatically in the past 30 years, the precise mechanisms underlying this condition are still emerging. It is thought that one of the initiating events in PE is placental ischemia as a result of uteroplacental malperfusion and defective remodeling of the uterine spiral arteries due to insufficient trophoblast invasion. 10 Studies in experimental models of PE have shown that placental ischemia is associated with oxidative stress and abnormal natural killer cells (NK cells) at the maternal–fetal interface. 11,12 These changes, along with other genetic and environmental factors, lead to increased release of proinflammatory cytokines (e.g., TNF-α and IL-6), exosomes (including microRNAs), antiangiogenic factors (e.g., soluble fms-like tyrosine kinase-1 [sFlt-1] and soluble endoglin), autoantibodies to the angiotensin II type 1 receptor, and cell-free fetal DNA, as well as lower levels of proangiogenic factors (e.g., placental growth factor [PlGF]) in the maternal circulation. 11,13,14 Imbalances in these pathways and differences in the maternal responses are thought to cause differential effects on maternal endothelial and metabolic dysfunction and, consequently, varying degrees in the severity of PE. 12,15 In addition, although clinical observations and experimental studies point to the importance of nitric oxide during gestation, further studies are needed to examine whether its deficiency in mitigating oxidative stress and inflammation, as well as metabolic and endothelial dysfunction, could be a possible mechanism for PE. 16,17 A few of the abovementioned pathways are illustrated in Figure 1.
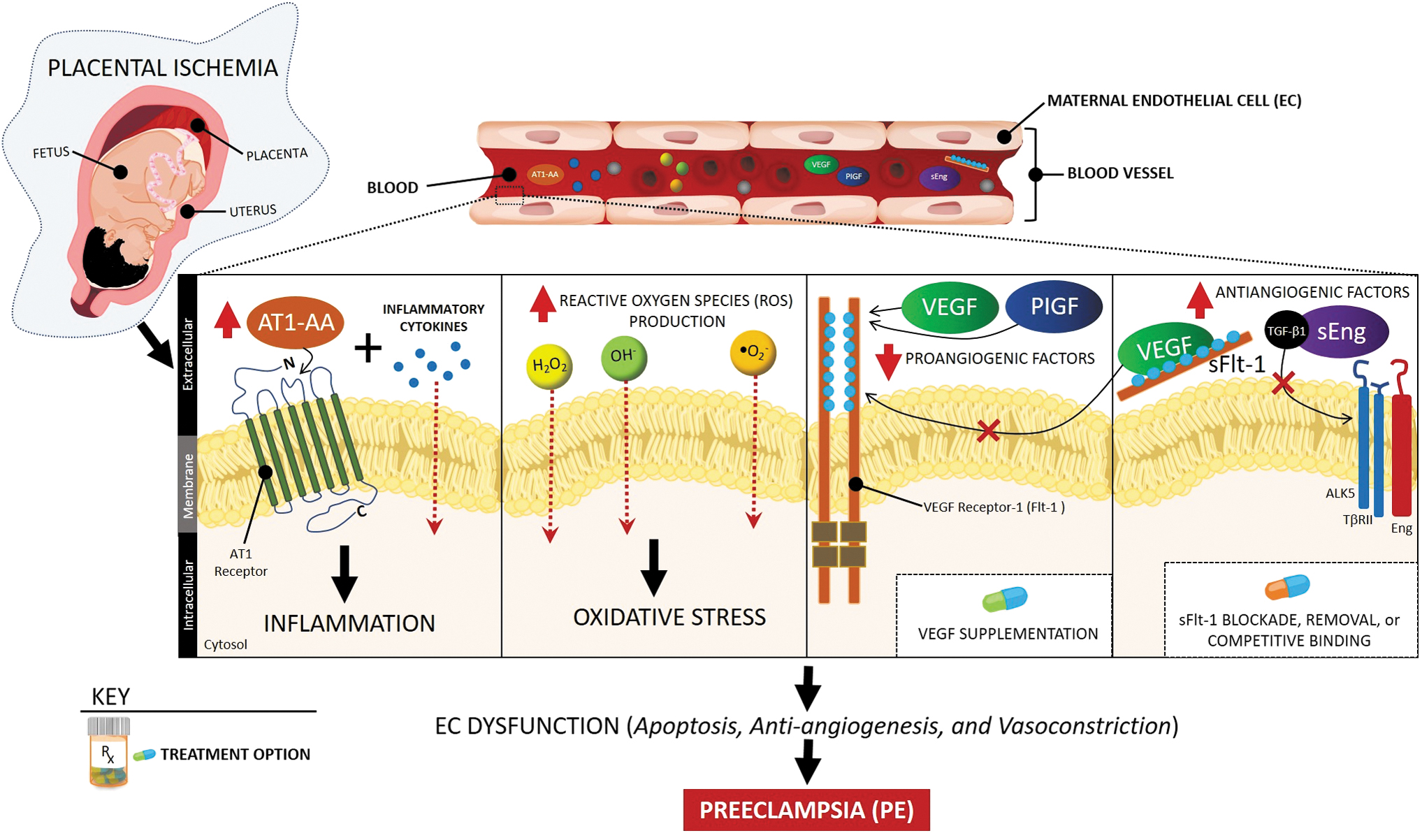
Signaling pathways, potential biomarkers, and therapeutic targets in PE. Placental ischemia-derived imbalance in proangiogenic (VEGF and PlGF) and antiangiogenic (sFlt-1 and sEng) factors, oxidative stress, and inflammation led to systemic vascular dysfunction reflected in hypertension and proteinuria in PE. Potential targeted interventions include blockade, removal, or competitive binding of sFlt-1 or supplementation of recombinant isoforms of proangiogenic factors. AT1-AA, AT1 receptor autoantibodies; PE, preeclampsia; PlGF, placental growth factor; sEng, soluble endoglin; sFlt-1, soluble fms-like tyrosine kinase-1; VEGF, vascular endothelial growth factor.
Potential biomarkers for prediction, early detection, and prognosis of HDP
Numerous studies have evaluated the role of biochemical and biophysical markers individually, in combination, and with the addition of clinical history in an attempt to improve risk prediction. 2,18,19 Overall, clinical, biochemical, and epidemiological findings suggest that PE is not a single disorder and that different pathways may converge on a common syndromic end point. 20 Identifying these disease subtypes may allow development of novel biomarkers and therapies.
Some of the most commonly studied biophysical markers include the mean arterial pressure and uterine artery pulsatility index for early detection of alterations in uteroplacental blood flow. 2 However, uterine artery Doppler studies alone have a low predictive value for the development of early-onset PE and an even lower value for late-onset PE. 2 Commonly studied biochemical markers include PlGF, sFlt-1, and soluble endoglin 2,18,19 and their ratios 21 as early indicators of abnormal cellular signaling pathways (Fig. 1).
Regardless of the parameters used, screening for PE in low-risk women is associated with very low positive predictive values ranging from 8% to 33%. 18 In general, biomarkers perform better in the prediction of early-onset PE (using first- and second-trimester parameters) than late-onset PE. Combinations of biomarkers fare better, with prediction rates of early-onset PE ranging from 30% to 100% in small studies. 22 Verification of these findings in large prospective studies has not yet occurred.
At present, further studies to identify and validate novel biomarkers with high predictive value are warranted. In addition, evidence that accurate prediction of early-onset PE can be followed by interventions that improve maternal or fetal outcomes is limited, and this area remains an important research opportunity.
Novel treatment strategies for HDP
Current recommendations for treating PE are focused on managing hypertension and preventing seizures and, if necessary, delivering the fetus. Few interventions specifically act on factors in the pathogenesis of PE, rather than simply manage the symptoms and adverse maternal and fetal outcomes. Novel treatment strategies for PE target imbalances in several potential pathways.
Clinical studies suggest that an imbalance in prostacyclin (platelet inhibitor and vasodilator) and thromboxane A2 (platelet activator and vasoconstrictor) is involved in the pathogenesis of PE. 23,24 Thus, recent clinical guidelines recommend low-dose aspirin to reduce the incidence of PE because low-dose aspirin can inhibit thromboxane A2 without altering secretion of endothelial prostacyclin. 25
In addition, imbalances of angiogenic and antiangiogenic factors are novel targets for PE prevention. In PE, elevated levels of sFlt-1 during pregnancy mediate hypertension, proteinuria, and glomerular endotheliosis by inhibiting downstream signaling of vascular endothelial growth factor (VEGF) and PlGF (Fig. 1). 26,27 In preclinical animal models of PE, administration of recombinant isoforms of these angiogenic factors or the interruption of sFlt-1 production with small interfering RNA (siRNA) molecules and small-molecule inhibitors—such as phosphodiesterase 5, proton pump, or sodium–potassium pump inhibitors—has shown promise in reducing blood pressure, abnormal urinary protein, and kidney damage. 26,27 Statins, such as pravastatin, may inhibit sFlt-1 production in animals models of PE and pregnant women with severe PE. 27,28 Other treatment strategies with the potential to lower hypertension include removing excess circulating sFlt-1 with antibodies and extracorporeal techniques, such as dextran sulfate apheresis or adsorption columns. 27
Although ongoing work on novel treatment strategies for HDP appears to be promising, further research is needed to establish effective, evidence-based treatment recommendations during pregnancy in women with HDP, which do not harm the expecting mother or unborn child.
Peripartum Cardiomyopathy
PPCM is a leading cause of maternal mortality. 29 PPCM is defined as systolic heart failure (left ventricular ejection fraction <45% and/or fractional shortening <30% on echocardiogram) presented in the last month of pregnancy or the first 5 months after delivery in otherwise healthy women. 30 Overall, the incidence of PPCM has been increasing in the United States, although estimates range from 1 in 1000 to 1 in 4000 live births, 31 with significant variation by geographic location. 32 The predisposing factors include advanced maternal age, PE, chronic hypertension, and multiple-gestation pregnancies. 33 Furthermore, the incidence of PPCM is higher in African American women, who also have worse outcomes and higher mortality. 34,35
Pathophysiology of PPCM
The cause of PPCM is largely unknown. Several genetic variations have been associated with PPCM. 36 –38 Notably, familial clustering of PPCM has been shown, 39 –41 and some women with PPCM have a truncated mutation in TTN, the gene encoding titin, a sarcomeric protein involved in the structural, mechanical, and regulatory functions of cardiac muscle. 38 However, not all women with these genetic variations develop PPCM, so other contributing and/or precipitating factors might be involved. Some postulated mechanisms involve hemodynamic stress of pregnancy, malnutrition, or myocarditis, but evidence is insufficient to support their major role.
One major PPCM research effort has been directed toward exploring the hormonal/vascular hypothesis (Fig. 2). It has been suggested that pregnant mice lacking antioxidative signal transducer and activator of transcription 3 (STAT3) developed cardiomyopathy due to the presence of a cathepsin D-cleaved 16-kDa prolactin product implicated in endothelial cell and cardiomyocyte apoptosis and dysfunction. Low STAT3 levels in the myocardium and the presence of circulating 16-kDa prolactin fragments also were detected in PPCM patients undergoing heart transplants. 42 In addition, the role of microRNA146a in the cross talk between endothelial and cardiomyocyte cells was implied in prolactin-induced PPCM. 43
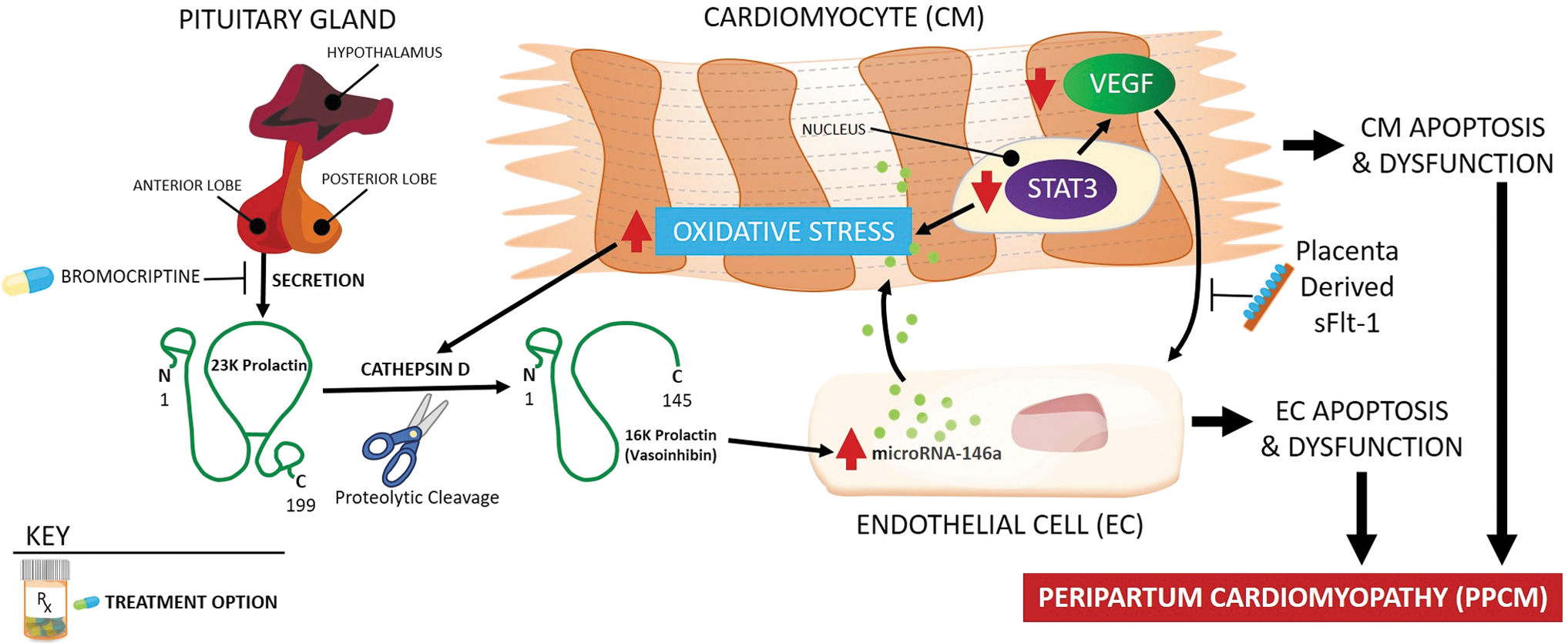
Pathophysiology of peripartum cardiomyopathy. In pathogenesis of peripartum cardiomyopathy, the role of cathepsin D-cleaved 16-kDa prolactin fragment, microRNA146a, and decreased VEGF signaling has been suggested. Suppression of prolactin production by bromocriptine had positive therapeutic effects in some preclinical and clinical studies. STAT3, signal transducer and activator of transcription 3.
On the other hand, reduced levels of VEGF were identified in PPCM patients, supporting the hypothesis that PPCM is a systemic vascular disease stemming from impaired VEGF signaling in endothelial cells. 44 Moreover, the late gestation increase in placenta-derived circulating antiangiogenic sFlt-1, which binds to and inhibits VEGF, may work in concert with an already present, toxic shorter form of prolactin. Both effects may converge into a potent CV insult that leads to PPCM. 45 Importantly, sFlt-1 is increased in PE 46 and multiple-gestation pregnancy, 47 and these women are also more prone to develop PPCM. However, sFlt-1 is elevated only in a subset of women with PPCM, 45 and the origin of sFlt-1 in women after delivery is unknown. It is important to note that an sFlt-1 increase can also be observed in nonpregnant patients with heart failure, 48 suggesting a common pathophysiological mechanism for PPCM and other forms of heart failure. Additional research is needed to verify the implication of sFlt-1 in advancing PPCM diagnosis and treatment.
Challenges in establishing diagnostic and prognostic biomarkers for PPCM
Unfortunately, PPCM is still a diagnosis of exclusion, and because it is a rare disease, establishing prognostic markers is a challenge. Further studies are needed to validate the routine use of microRNA146a, along with 16-kDa prolactin, and cathepsin D in diagnostic strategies (Fig. 2) and to establish whether genetic testing can identify women at risk.
Although recovery is expected in most patients, 49 the presence of a TTN gene truncation 38 or higher levels of sFlt-150 correlated with worse cardiac function and adverse clinical events. An ejection fraction lower than 20% and larger heart size at presentation are prognostic for poor recovery. 49,51 On the other hand, a higher level of relaxin-2, with effects that are mediated in part by VEGF, was associated with improved 2-month recovery. 50 African American women with PPCM have worse outcomes, sometimes despite adequate therapy. 34,49,52,53 Socioeconomic factors, access to medical care, and genetic factors may contribute to these worse outcomes in women of African descent. 37,54
Treatment concepts for PPCM
Because PPCM has no specific treatment, its management is similar to other common forms of systolic heart failure (fluid management with diuretics, vasopressin, and inotropes, with the addition of beta-blockers, vasodilators, and aldosterone antagonists, and, in extreme cases, use of mechanical supportive devices), with attention to drugs contraindicated for pregnant and lactating women (such as ACE inhibitors/angiotensin II receptor blockers).
Several studies tested bromocriptine intervention in suppressing prolactin production in women with PPCM (Fig. 2). Nonrandomized studies in South Africa and Germany demonstrated improvement, but not a full recovery, of left ventricular function among women treated with bromocriptine. 55,56 In a subsequent, larger, prospective, nonplacebo-controlled, randomized clinical trial of patients with PPCM, the addition of bromocriptine to standard therapy for heart failure was associated with a high recovery rate and a very low rate of adverse outcomes up to 6 months postpartum. 57 Spontaneous recovery occurs in a large proportion of patients, thereby complicating interpretations of nonplacebo-controlled studies and trials, as well as future trials evaluating such treatments as bromocriptine. Predictors for recovery are not established.
Nevertheless, as promising as these studies are, caution also should be exercised because 5-year follow-up data of a German PPCM cohort showed a frequent persisting or de novo hypertension and arrhythmia. 44 These studies further underline an urgent need for additional validation of the current therapeutic approaches as well as exploration of novel pharmacological targets and drug repurposing that are safe for both mothers and babies.
Adverse Pregnancy Outcomes and Long-Term Risk for CVD
Several large cohort studies and meta-analyses have found links between APOs and long-term risk for CVD in women. 58 –65 These adverse outcomes include PE and other HDP, 58,60,64 gestational diabetes mellitus (GDM), 59,63 preterm birth, 60,62,66,67 pregnancy loss, 61,63 and small for gestational-age infants. 67
In studies assessing long-term (10–20 years after pregnancy) risk, women diagnosed with PE have an estimated two- to fourfold increased risk of future CV or cerebrovascular events compared with unaffected women. The risk for future CV events is similar for women diagnosed with gestational hypertension and increased even more when HDP are associated with other adverse outcomes such as iatrogenic preterm birth and stillbirth. 68,69 Several recent studies demonstrate short-term CV risk as well, 60,70 revealing an approximate 50% increase in the diagnosis of CVD within 5 years 70 and a two- to threefold increase in the diagnosis of hypertension within 2–7 years after pregnancy for affected women. 60
Researchers are now attempting to determine whether underlying pathology for CVD exists preconceptionally versus whether the APO acts directly on maternal cardiometabolic function to cause future CVD. HDP and subsequent CVD share common predisposing factors. Several studies suggest that pregnancy likely unmasks existing pathology and that APOs are not causative, although they may hasten and/or mediate the process toward CVD. 59,63,71 Regardless, controlling CV risk and aggressive treatment of APOs are important strategies to improve pregnancy and immediate and long-term CV outcomes in women.
Prepregnancy characteristics underlying associations
Women with chronic conditions (e.g., hypertension, diabetes, and renal, lung, and autoimmune diseases 72 –75 ), obese women, 3,59,60 and smokers 76 are at higher risk for pregnancy complications; these same factors also are associated with CVD. 58 Some nonmodifiable factors—such as race, education, geography, and age—are also associated with elevated risk for APOs and future CVD (Fig. 3). 60,77 –80
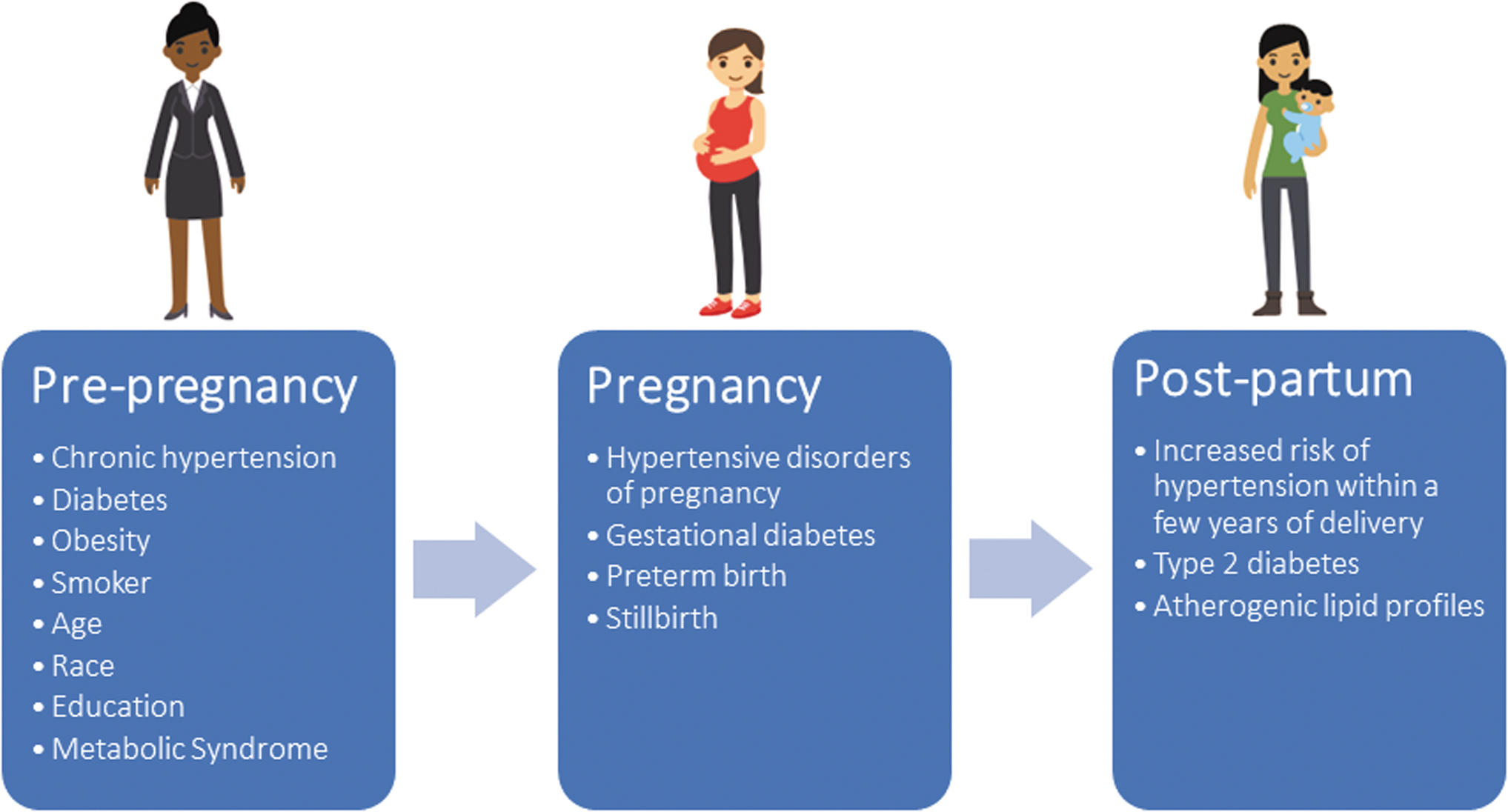
Accumulation of cardiovascular risk factors from preconception through pregnancy and postpartum periods. Demographic, clinical, behavioral, social, and environmental risk factors for cardiovascular disease may contribute to adverse pregnancy outcomes. These may persist throughout pregnancy and postpartum periods. Additional cardiovascular risk may be acquired or uncovered during pregnancy, including maternal conditions (hypertensive disorders of pregnancy and gestational diabetes) and fetal conditions (small for gestational age). Preconception and pregnancy risks may persist during the postpartum period and/or emergence of additional cardiovascular risk factors may occur, including maternal conditions (hypertension, diabetes, and dyslipidemia).
Identification and prevention strategies for women at risk for APO-associated, future maternal CVD
Identifying a woman's risk before or early in pregnancy provides additional time to implement strategies to prevent APOs and perhaps slow progression to CVD. There is growing recognition that the occurrence of pregnancy complications reliably identifies women with underlying, often unrecognized CV risk factors, who may benefit from screening and preventive actions, such as preconception counseling to address risk and prepregnancy control of blood pressure, blood glucose, lipids, and weight. Women with a previous APO are at a higher risk in a subsequent pregnancy, a variable that can be used to identify risk preconceptionally. Biomarkers may be useful, particularly for nulliparous women who have no apparent risk factors. Established (e.g., hsCRP, 60,62,81 triglycerides, 82 lipids, 59,82 and glucose 59,60,81 ) and novel CV risk biomarkers (e.g., sFlt-1, PlGF, VEGF, and endoglin) 83 have been found to be altered in early pregnancy in women who later have an APO. However, more work is needed to identify which biomarkers or combinations of biomarkers (and their timing) are most predictive of risk.
Early intervention has the potential to reduce both APOs for women who become pregnant again and their lifetime risk of CVD. 84 –86 Medications and nonpharmacological strategies may improve pregnancy outcomes and risk factors for future CVD. 84,86 Low-dose aspirin is recommended for at-risk pregnancies with PE, diabetes, multifetal gestations, and renal disease, as well as, more recently, to reduce the risk of preterm birth. 23,24 Statins have shown promise to address potentially abnormal vascular function seen in PE and antiphospholipid syndrome. 87,88
CV assessment and follow-up at 3 months postpartum for women with HDP, GDM, fetal growth restriction, and preterm birth are recommended. 89 This screening should include a medical/pregnancy history, physical examination, biochemical testing, and nutrition assessment. Women should be counseled with regard to their individual identified risks and opportunities for prevention.
Importantly, women with healthier lifestyle profiles across all reproductive stages (i.e., preconception, pregnancy, and postpartum) have lower risks of APOs as well as lower risks for future CVD. 59,63 The American Heart Association's Life's Simple 7 defines ideal CV health based on behavioral (smoking status, physical activity, and diet) and clinical (weight, blood glucose, cholesterol, and blood pressure) risk factors that can be improved through lifestyle changes. 90
Women should be encouraged to adhere to a healthy diet, 91,92 get regular physical activity to decrease risks for APOs, address sleep-disordered breathing, 93 and breastfeed, which lowers risks for CVD (including hypertension and hyperlipidemia), type 2 diabetes, and cancer. 59,92,94 –96 Maintaining a normal weight before, during, and after pregnancy is important. Obese women and those with excessive weight gain during pregnancy and postpartum weight retention are at higher risk for poorer outcomes. 3,59,60 In addition, pregnant women with increased leisure-time physical activity early in pregnancy had low rates of GDM, similar to women with higher patterns of activity, suggesting that increased activity early in pregnancy may improve pregnancy outcomes. 97 Importantly, physical activity moderately attenuated the association of GDM with myocardial infarction and stroke. 63
Conclusions
Further research is needed to establish effective, evidence-based treatment recommendations during pregnancy, which do not harm the mother or unborn child. Work on novel treatment strategies for peripartum CV-related conditions appears promising. However, pregnant and lactating women are usually excluded from participating in drug trials, a conundrum that the Task Force on Research Specific to Pregnant and Lactating Women is tackling now. 98 Carefully planned prospective studies to address these research opportunities in pregnant and lactating women are needed. After pregnancy, comprehensive CV evaluations beyond the immediate postpartum period may provide the opportunity for counseling and interventions to mitigate the underlying risk factors to improve future pregnancy outcomes and lifelong CV health.
Footnotes
Acknowledgment
The authors thank Cheryl Kassed for her input and critical review of this article.
Disclaimer
The views expressed in this article are those of the authors and do not necessarily represent the views of the National Heart, Lung, and Blood Institute; the National Institute of Diabetes and Digestive and Kidney Diseases; the National Institutes of Health; or the U.S. Department of Health and Human Services.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No specific funding was received for this work.
