Abstract
In this report, sex/gender research relevant to reproduction on Earth, in conjunction with the extant human and animal observations in space, was used to identify knowledge gaps and prioritize recommendations for future sex- and gender-specific surveillance and monitoring of male and female astronauts. With overall increased durations of contemporary space missions, a deeper understanding of sex/gender effects on reproduction-related responses and adaptations to the space environment is warranted to minimize risks and insure healthy aging of the men and women who travel into space.
Introduction
T
Through a sex and gender lens, major influences of the space environment (viz., radiation, microgravity, and stress) on reproduction and physiological systems central to reproductive processes (i.e., endocrine and urogenital systems) were considered. This report on reproductive health in relation to space adaptation provides an overarching perspective on current sex and gender evidence, identifies knowledge gaps, establishes a framework for future work, and outlines key recommendations to direct future translational research and infrastructure.
Major Influences of the Space Environment
Radiation
Radiation dose to crew is mission-specific and dependent on multiple factors such as mission destination and duration, vehicle and habitat design, and solar conditions. Typical ISS exposures for a 6-month mission in low-Earth orbit are on the order of 54 to 108 mSv with exposure levels varying with solar activity and ISS altitude (NASA–Johnson Space Center Radiation Health Office, personal communication with Janice Huff, 2013). 2 Exposures during interplanetary travel required to reach Mars and on the Mars surface are far greater, with annual exposures spanning 210 to 1,070 mSV and 80 to 330msV, respectively. Relative to other organ systems, the gonads are highly sensitive to radiation exposure. 3 In men and women, temporary infertility is associated with high-dose, acute radiation exposure—for men, 150 mSv acute x-rays , and for women, 650 to 1500 mSvV acute x-rays or gamma rays to the ovaries. 4,5 How these tissues respond to exposure scenarios relevant to the space environment with respect to dose, dose-rate, and radiation quality, as well as the combined effects of other spaceflight environmental stressors such as microgravity, has not been addressed. 6 –8
Cancer risk assessment requires evaluation of organ doses (measured in Sv) from dosimetry and physical considerations, which are combined with age- and gender-dependent risk coefficients to project mission risk. Women have a higher incidence of radiation-induced cancers, largely driven by lung, thyroid, breast, and ovarian cancers, and therefore reach maximal safe days in space earlier than men. 9 There are presently insufficient numbers of humans exposed to space-characteristic radiation to accurately assess differential effects on men and women. NASA studies have examined cancer risk in animals following exposure to space radiation (protons and high-linear energy transfer (LET) radiation); however, little attention has been paid to sex differences.
Duration of exposure
The impact of flight length on reproductive health will require a greater focus with the transition to far longer exposures to both microgravity and space radiation. During the space shuttle era, most exposures were up to ∼2 weeks in duration. In contrast, ISS missions are extending durations of exposure to 6–12 months. This change is significant with regard to the physiology of sperm production by the testis in the male and the estrous cycle in the ovary in the female. Longer duration missions, spanning multiple reproductive cycles in both men and women, raise a significant knowledge gap concerning the impact of long-term spaceflight on reproductive health.
Microgravity and stress
There are a number of reproductive health concerns for men and women in space potentially related to microgravity and/or stress (including sleep disruption). Spermatogenesis in men may be impaired beyond effects of radiation exposure. A 120-day Russian bed rest study found that sperm collected after 50–60 days and 100 days exhibited a reduction in live spermatozoa showing active mobility and an increase in the percentage of morphologically/structurally altered spermatozoa. 10 Basic research studies of male rats exposed to 6 weeks of simulated microgravity have reported severe testicular and epididymal degeneration including massive testicular apoptosis 6 months later. These effects have been postulated to occur due to (a) chronic testicular hyperthermia, (b) invasion of inflammatory cells, and/or (c) catastrophic apoptosis, leading to aspermatogenic dysfunction. 11 There has been no post-spaceflight research involving humans on this topic.
Reproductive changes during or post flight have not been systematically studied in female astronauts. In female adult mice that were cycling at launch (Space Transportation System [STS]-131, STS-133, and STS-135), spaceflight induced cessation of cycling, loss of corpora lutea, and significantly reduced estrogen receptor mRNA levels in the uterus (HM Holets, University of Kansas Medical Center, unpublished data). 12 Comparable evaluations in women are lacking.
Systematic data regarding reproductive history in men and women astronauts is lacking. Although many male astronauts have produced children following spaceflight, information on conceptions and birth outcomes upon returning from space are not available. The incidence of infertility following prolonged spaceflights in men and women and the incidence of pregnancy complications in women are not known. In female astronauts who have delayed pregnancy, it will be difficult to parse out the effects of space travel (microgravity, radiation exposure) from natural effects of aging. Female astronauts who experience hypertensive pregnancy disorders may require additional monitoring for cardiovascular disease, because on Earth these disorders are associated with increased risk of future adverse cardiovascular events such as stroke and myocardial infarction. 13
Physiological Systems Central to Reproductive Function
Endocrine system
The hypothalamic–pituitary–gonadal (HPG) and hypothalamic–pituitary–adrenal (HPA) axes cross-communicate through neuronal signaling and hormonal negative feedback. Several hypothalamic neurotransmitters are known to be altered by real or simulated microgravity or severe changes in gravity, namely histamine (increases), 14 serotonin (decreases), 15 oxytocin (decreases), 16 norepinephrine (decreases), 17 and to a lesser magnitude, glutamate (decreases). 18 These neurochemical disparities may be a direct result of the significant change in the gravitational environment on neuronal signaling or an indirect effect via the hormonal response from the downstream hormonal axes.
Alterations in HPG and HPA axes functioning have been reported following spaceflight. Most evaluations focused on the reduced levels of circulating testosterone in men 19 that appear to rebound after return to Earth. 20 This has been recapitulated in short- and long-duration spaceflight and long-duration head-down bed rest studies. 21 A recent report 22 indicated no decrement in serum total, free, and bioavailable testosterone during either long-duration spaceflight or bed rest, but a significant decrease on landing day for the long-duration flights and after short-duration spaceflight. The HPA axis, which exhibits both a circadian rhythm and enhanced output of circulating glucocorticoids (i.e., cortisol) in response to stressors, exhibits increased activation during spaceflight, simulated microgravity, and during long-duration stress studies. 23 –26 However, like the gonadal hormones, circulating concentrations of these hormones quickly recover upon return to Earth. 27
With respect to women, estrogen and gonadal function in space and in simulated microgravity (bed rest) are grossly understudied. Oxytocin, a hormone that acts to attenuate the HPA axis 28 and dampen stress responses in women, 29 exhibits long-lasting reductions following spaceflight. 14 Similarly, oral contraceptives (OCs) decrease the release of oxytocin 30 but do not increase stress responsiveness, as OCs reduce cortisol release in response to stressor exposure as well. 31 OC use is not mandatory during spaceflight; however, flight surgeons have recommended 30–35 mcg OCs to female astronauts to provide (a) better suppression of the ovary (less chance for cysts and breakthrough bleeding, depending upon OC formulation) and (b) reduced risk of bone loss. Circulating levels of testosterone, dihydroepiandrosterone, corticosteroid-binding globulin, prolactin, and sex hormone–binding globin are affected by OCs 32,33 and are involved in stress responses and the regulation of both the HPG and HPA axes. Unfortunately, with ever-changing formulations, periodic reevaluation of oral contraceptive cost–benefit is also required.
Genitourinary system
While reproductive functions have not been a direct concern during space travel, diseases of organs of the reproductive system have adversely affected flight duration. For example, a serious case of prostatitis in a cosmonaut occurred in 1985 that impacted the length of the mission. 34,35 Little is known about sexual activity in space, and infrequent ejaculation resulting in accumulation of prostate secretions can support bacterial growth. Hence, the risk of prostatitis may well need to be addressed again during prolonged exploratory travel.
Renal and urinary tract problems are a recurrent issue with space travel. Microgravity and privacy concerns can lead to infrequent and incomplete bladder emptying, increasing the risks for urinary tract infections. 33 In addition, medications for motion sickness reduce bladder emptying. Formation of stones becomes more likely when hydration is compromised, and microgravity increases the amount of calcium excreted in the urine. Inadequately treated stones and urinary tract infections could eventually impair renal function.
On Earth, urinary tract stones are more common in Caucasian men, but struvite stones are more common in women. 36 Urinary tract infections are more common in women 37 and prostatitis occurs only in men. The long-held belief that the urinary tract is sterile is being called into question with emerging work on the urinary microbiome. 38 There are no data regarding sex differences in spontaneous or unprovoked urinary retention in the age range of the astronauts. However, data regarding provoked urinary retention after surgery, with nearly equal numbers of men and women of similar age to the astronauts, do not show a sex difference. 39,40
Cumulative experiences in space travel suggest there is no sex difference in the incidence of urinary tract stones during space travel or after returning to Earth, however women have experienced a higher incidence of urinary tract infections in space than men. 41 Possible explanations for this sex difference include predisposition from women's urethral anatomy, adjustment to voiding in microgravity, a higher incidence of urinary retention, and a higher incidence of catheterization. 42,43
Interactions of Reproduction with Other Aspects of Health
Estrogen receptor signaling
Parallels between spaceflight effects on physiology and aging have been known since the 1960s. 44 Ground-based studies have identified age-related alterations in estrogen receptor (ER) signaling in many major organ systems. Several of these organ systems have shown changes in experimental animals during spaceflight. Altered ER signaling in these estrogen-regulated systems, shown in Figure 1 are also known to accelerate certain aging processes.
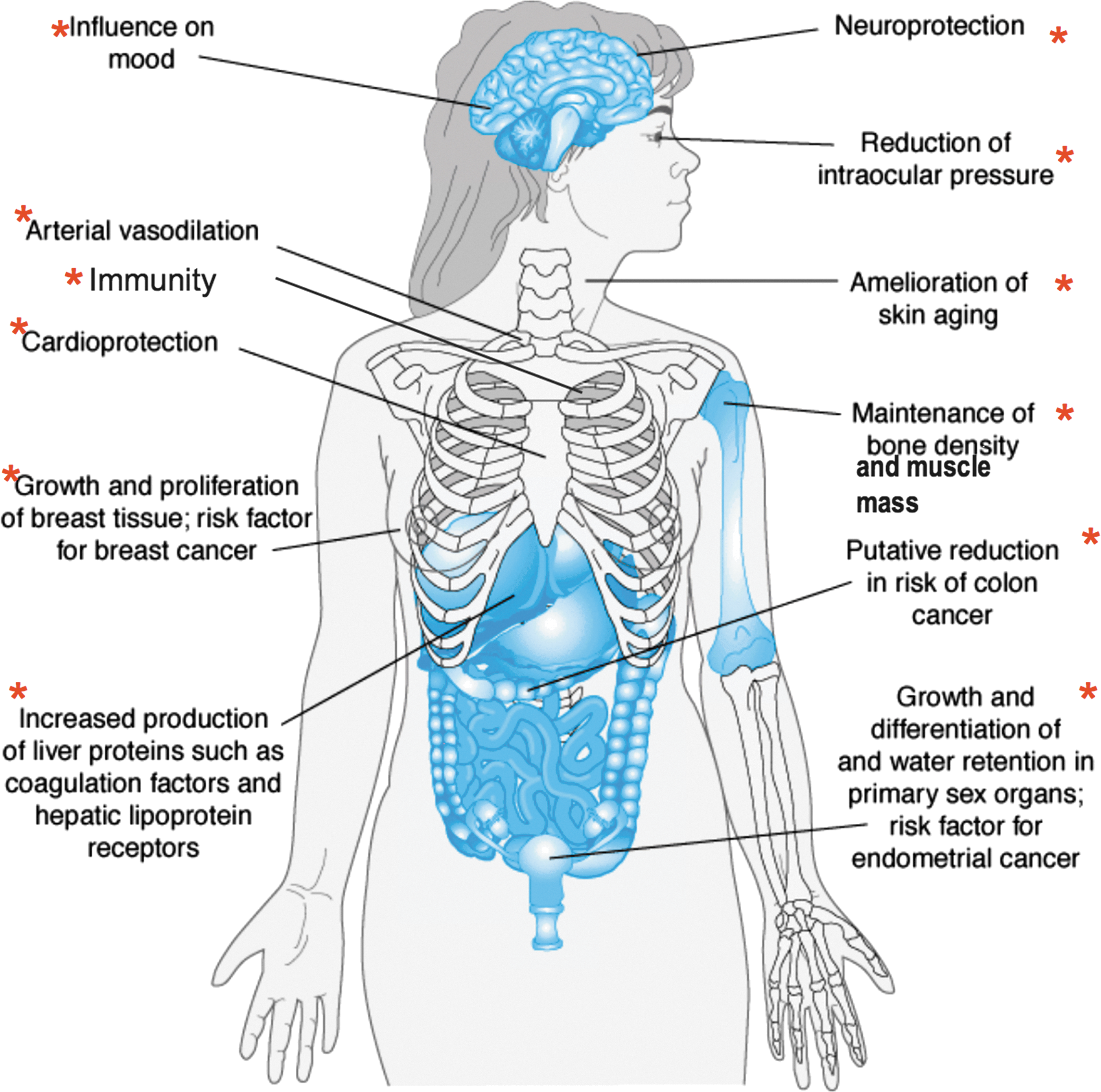
Estrogen-regulated systems known to be affected by both spaceflight and aging. Adapted from Molina PE. Endocrine Physiology, Third Edition.
The spaceflight-induced decline in multiple physiological systems may involve a common mechanism in dysregulation of reproductive steroid receptor–dependent signaling pathways. If this is true, then spaceflight is likely to reduce gonadal function in both men and women. A central crosscutting question for space- and ground-based study is whether there are sex differences in altered estrogen signaling in the spaceflight-affected and aging systems. Although the role of ER signaling in maintaining function in these systems, as well as the changes in these systems associated with aging populations have been reported on Earth, a Medline search using the terms “spaceflight” and “estrogen receptors” shows no published research in this area. Thus, there are major knowledge gaps regarding whether changes in these systems during spaceflight may have a common mechanism due to alterations in ER as well as androgen receptor signaling in women and men. Importantly, the emphasis on ER signaling and relative lack of attention to androgens in both sexes could create false impressions that estrogen is more affected by aging and spaceflight than androgens and that women are more likely than men to have health problems in space, which is not supported by the literature. Because bone density is maintained by estrogen in both men and women, declining testosterone and the decrease in subsequent aromatization to estrogen would also impact bone health in men.
Bone and cardiovascular health
There are sex differences in response to stressors that affect the reproductive axis, and these can have long-term consequences for bone and cardiovascular health. Although contraceptive use is common among female astronauts, those that suppress ovarian function for extended periods [e.g., Depo-Provera or other contraceptives containing depot medroxyprogesterone acetate (DMPA), and Noristerat, Norigest, Doryxas, or others containing norethisterone enantate (NET-EN)] have been shown to adversely affect women's bone density on Earth. 45,46 Thus, if used in space, certain OC formulations may compound bone loss resulting from microgravity in female astronauts. This raises the question of whether total estrogen exposure would be reduced for female astronauts using those contraceptives on long-duration missions that, in turn, could impact bone health, muscle strength, and cardiovascular risks. (This argument is predicated on the assumption that female astronauts continue to cycle during spaceflight.) Thus, in women, increased risk for cardiovascular disease may be both directly and indirectly related to disruption of ovarian function, perhaps through binding of the adrenal cortical hormones to progesterone receptors and membrane surface receptors that also bind estrogen. 47,48
Brain–behavior relationships
Sleep
Flight-induced changes in gonadal hormone release may be the source of a nonreproductive functional outcome, namely altered sleep patterns. 49 Sleep–wake cycles are also disrupted in Earth-based, long-duration microgravity simulations. 50 Sleep quality is affected by some stressor exposures and is correlated with circulating testosterone and estradiol levels. 51 –55 Conversely, sleep deprivation disrupts normal gonadal hormone secretion 56 and increases glucocorticoid release, 57 thus potentially causing a positive feedback loop. Gonadal hormones affect the activities of brain monoamines, acetylcholine, and orexin, 58 –61 all neurotransmitters involved in maintaining wakeful state, sleep state, or transitions between sleep states. 62
Reproduction and rearing
Despite the consideration that in the distant future, human reproduction is likely to occur in space, 63 the current literature base is insufficient, limiting speculation about the possibility that intricate and complex phases of reproduction in mammals—including mating, fertilization, implantation, placentation, embryogenesis, organogenesis, prenatal and postnatal development, birth, lactation, and suckling—can occur in space.
In adult animals flown in space, fertilization difficulties could potentially occur due to disruption of hypothalamic neurochemistry important for proceptivity and receptivity components of copulation. Several hypothalamic transmitters are affected by physical factors involved during spaceflight. Oxytocin is both responsive to HPG axis functioning and attenuates HPA axis activation during stressor exposure in women. 26,27 Hypothalamic oxytocin is also critically involved in female sexual receptivity. 64 Unlike many other hormones, hypothalamic oxytocin remains reduced at 18 weeks following spaceflight in rodents. 14 This effect may be more significant in women, as OCs reduce circulating oxytocin levels. 28 Given the influence of spaceflight and OC use on oxytocin release, further research is necessary to determine whether this combination could have long-term effects on reproductive health.
Enduring changes in oxytocin following spaceflight 14 might be critical in postpartum behaviors, as oxytocin is implicated in social bonding. Thus, the implications of long-duration spaceflight for the viability of the immediate offspring as well as the subsequent generation of offspring are an unknown, possibly ever-lasting effect within the families of those chosen to participate in such missions.
Sex and Gender Considerations in Astronaut Reproductive Health
Gender roles and responsibilities can impact health and health outcomes. Although reproduction is a biological process, societal roles for parents including that of women functioning as the primary caregiver often require women to take extended family medical leave and, for some, a hiatus from their career. While paternity leave is more common now than in past decades, the majority of U.S. fathers do not take more than one week of leave after the birth of a child. 65 As compared with male astronauts, female astronauts delay reproduction (Table 1), which may be based, in part, on gender role requirements. Further, the biological processes that ensure successful reproduction are impacted by aging, a factor that can lead to infertility and pregnancy complications in those who delay childbearing. 66 Thus, childbearing and childrearing events that occur along a continuum are examples where the variables of sex and gender collide.
Data extracted from National Aeronautics and Space Administration medical records, current as of 2014.
Data extracted from the Lifetime Surveillance of Astronaut Health Lifestyle Questionnaire, which was self-reported and collected at various points from February 22, 1995 through April 30, 2004.
p<0.0001.
p=0.0349.
p=0.0001; N=159 due to missing data.
Table 2 shows that in this astronaut cohort, the age of first mission and percent married is significantly different between men and women. Table 2 shows that male astronauts have a higher average number of children than female astronauts, and even with the low numbers of female astronauts, this finding was statistically significant. An important hypothesis to consider is that the gender component impacts this finding. Because women are more likely to function as primary caregivers for their children and to take a hiatus from their career due to this parental role, we could speculate that some women choose to delay reproduction due to the extensive time required for preparation and training for space travel, which may be discordant with societal roles and responsibilities of parenting. This is an example of the complexity of the interplay of both sex and gender on reproductive health.
p=0.0005.
p<0.0001.
Review of the sex and gender interaction within each environmental stressor in space travel is beyond the scope of this particular work, but expanding gender impact beyond reproduction is worth exploring. Consider sleep deprivation, a well-known stressor in space travel, discussed above. Research on Earth indicates that sleep disruption impacts men and women differently and women experience sleep disruption at higher rates than men. 69 Women subjectively report lower sleep quality as compared to men. 70 In addition sleep disruption increases with aging and for women is most prominent in middle age through menopause. 68 The median age for U.S. astronauts (first transit) is 38.3 years and for all transits, 40.9 years (versus 41 and 43.4 years, respectively, for men). Women have more difficulty resuming normal sleep patterns after sleep disruption. Terrestrial, sex-based research indicates that sleep disruption in women can increase levels of inflammatory markers including hs-CRP and coagulation factors such as von Willebrand factor that, in theory, would increase risk of cardiometabolic disease and cardiovascular events. 71
It is well known that women present more frequently with “atypical” symptoms of cardiovascular disease such as fatigue, heartburn, and dyspnea and are less likely to receive a workup for cardiovascular disease. In addition, women have sex-specific risk factors such as polycystic ovarian disease, gestational diabetes, and preeclampsia. 67 Given that cardiovascular disease is the number one killer for men and women regardless of occupation, 68 it will benefit both male and female astronauts for future NASA research platforms to include both variables: sex and gender.
Knowledge gaps in health differences of male and female space travelers remain due to lack of consideration and inclusion of gender and sex as unique variables in research platforms. Yet, biologically, cessation of reproductive capacity is more readily identifiable in women and a direct function of aging, again highlighting delayed reproduction, potential acceleration of aging, and the impact of gonadal radiation exposure related to space travel. Gender and sex have significant interplay, carrying highly distinctive roles for men and women within the health care system.
Infrastructure
There is a major need to improve the research and healthcare reporting infrastructure for the NASA Human Research Program. Low numbers of study participants, variable astronaut age, and OC usage all limit statistical robustness required to determine if there are significant effects of spaceflight on reproductive health. There is a crucial need for a coordinated effort to augment the infrastructure for basic research studies, with priorities established to include male and female subjects, human and animal, for the purpose of advancing understanding of sex and gender factors in relation to spaceflight.
Research Recommendations
Understanding of reproductive changes in men and women (pre-, during, and post-flight) that extend into the health and rearing of their offspring is limited. There is a critical lack of information about the effects of spaceflight on gonadal function and bone loss, as well as about effects of cosmic radiation on women's health. Basic information is needed regarding effects of microgravity and circadian disruption on gonadotropin release and on concentrations of sex hormones in both women and men. Examination of the effects of OC usage on HPA axis tone pre-, during, and post-flight is warranted. Basic studies of the causes of the prolonged oxytocin reduction, the functional significance of such reductions, and whether flight duration has a role in the magnitude or duration of the reduction are needed. Development and use of mammalian models to ascertain the detrimental effects of spaceflight stressors on male and female physiology and behavior on prolonged flights, and effects on offspring are warranted.
Active prevention of urinary tract stones and voiding difficulties will be essential for extended missions. Studies of urinary tract manipulation in a low-gravity environment are needed. Better understanding of the risk factors for urinary tract infection in space is warranted, including changes in the urinary microbiome and bacterial behavior in space as well as stability of antibiotic formulations throughout long mission durations. Gonadal hormones are not sex-specific. Astronaut and post-flight studies need to be expanded to examine estrogen signaling, including regulation of ER in men and testosterone in women, particularly in relation to bone and cardiovascular function. Potential options for assisted reproductive technologies for male and female astronauts should be considered, e.g., sperm and oocyte cryopreservation and informed consent.
Footnotes
Acknowledgments
Jessica Garcia and Mary Wear, Johnson Space Center, provided key epidemiological data for this report from the Lifetime Surveillance of Astronaut Health (LSAH). David Dinges, Namni Goel, Janice Huff, Richard Jennings, and Virginia Wotring provided critical comments on an earlier draft of this manuscript.
Author Disclosure Statement
No competing financial interests exist.
