Abstract
Despite ongoing awareness, educational campaigns, and advances in technology, breast cancer screening remains a complex topic for women and for the health care system. Lack of consensus among organizations developing screening guidelines has caused confusion for patients and providers. The psychosocial factors related to breast cancer screening are not well understood. The prevailing algorithm for screening results in significant rates of patient recall for further diagnostic imaging or procedures, the majority of which rule out breast cancer rather than confirming it. For women, the consequences of the status quo range from unnecessary stress to additional out-of-pocket expenses to indirect costs that are more difficult to quantify. A more thoughtful approach to breast cancer screening, coupled with a research agenda that recognizes the indirect and intangible costs that women bear, is needed to improve cost and quality outcomes in this area.
Introduction
Prevalence of breast cancer, relative risk of disease, and associated morbidity and mortality
W
Breast cancer incidence is affected by numerous factors. In the United States, rates of breast cancer vary by age, race/ethnicity, and geographic location. Breast cancer is age dependent; the chance that a woman will be diagnosed with the disease at age 40 is 1:227 compared with 1:26 at age 70. 2 White women have the highest incidence of breast cancer in the U.S., followed closely by African American women. Asian, Hispanic, and Native American women have a lower risk of developing and dying from breast cancer. 2,3
Genetic mutations can increase the risk of breast cancer; however, the majority of breast cancers (85%) occur in women without family history of the disease. In most cases, the disease appears to be associated with spontaneous mutations rather than heredity. 1
Since 1990, breast cancer mortality has decreased significantly. This decline is believed to be due to several factors including improvements in mammography technology leading to earlier detection, increased public awareness of the importance of screening, and improved treatment options. However, breast cancer remains the second leading cause of cancer death in women, exceeded only by lung cancer. The probability that a woman's death will be attributed to breast cancer is approximately 1 in 36 (approximately 3%). Clearly, breast cancer continues to be a significant public health concern.
Mammography is the only screening test for breast cancer that has been shown to reduce mortality; 2 specifically, it has been associated with a 19% reduction in breast cancer deaths (approximately 15% for women in their 40s and 32% for women in their 60s). 4 Overall, the sensitivity of mammography (i.e., likelihood that the test will detect existing disease) is 70%–90%; however, for women with denser breast tissue the sensitivity is lower (30%–48%). 5
The benefit of mammography in terms of early detection of cancer is balanced by certain limitations or “harms.” 4 One of these unintended consequences is the need for additional imaging or biopsy following the screening; in many cases, the results of the further evaluation are benign, and often referred to as “false positives.” The cumulative risk of a false positive result for a 40- or 50-year-old woman undergoing annual mammography for 10 years is approximately 61%. 4 For these women, the need for additional testing can increase fear, anxiety, and costs.
Current breast cancer screening guidelines
There are more clinical practice guidelines for breast cancer screening than for almost any other medical procedure. Contradictory recommendations from well-respected professional organizations have caused confusion for clinicians and patients alike (Table 1). Consider the case of a hypothetical 45-year-old woman. The American College of Obstetricians and Gynecologists (ACOG), the American Cancer Society (ACS), and the National Comprehensive Cancer Network (NCCN) recommend that she receive a mammogram annually and that she should do self-exams periodically as well. 6 The National Cancer Institute (NCI) recommends a mammogram every 1–2 years and advises against self-exams. The U.S. Preventive Services Task Force (USPSTF) advises against breast self-examination and does not recommend a mammogram for her until age 50, at which time it advises biennial screening. 7 Adding further confusion, preventive service coverage mandated by the Department of Health and Human Services through the Affordable Care Act (ACA) follows earlier USPSTF 2002 recommendations for breast cancer screening, providing coverage for mammographies beginning at age 40.
Adapted from American College of Obstetricians and Gynecologists (ACOG). ACOG Practice Bulletin: Clinical Management Guidelines for Obstetrician-Gynecologists. Obstet Gynecol 2011;118:372–382.
In a recent study, research analysts with the ACOG surveyed 224 obstetrician-gynecologists to assess how the various guidelines influence their practices. 8 Results indicated that 83.5% of these clinicians were influenced by two or more breast cancer screening guidelines. Although 42% reported making at least one change after the release of the USPSTF recommendations, 73% said that they were more influenced by the ACOG and ACS guidelines. The existence of conflicting guidelines complicates the use of any single guideline as the standard of practice in primary care and quality measures, as well as other settings.
Current screening technologies and approach to diagnosis
Several types of technology are used for breast cancer screening:
9
• • • • •
In the screening mammography setting with film screen mammography or FFDM, approximately 10%–14% of women are asked to return for further evaluation of a finding seen on the mammogram, although it may vary by radiologist and by site. 12,13 On further evaluation with diagnostic mammography and/or breast ultrasound, the majority of these findings are shown to be the summation of normal tissue, cysts, or other benign processes. 11 A small percentage of women—about 2%—are referred for breast biopsy. In some settings, more advanced imaging (e.g., MRI) is used for further assessment. The substantial cost and potential stress of the added imaging evaluations are a concern for health care providers, employers, public and commercial payers, family members, and individual patients.
Cancer risk and breast density
The term ‘breast density’ describes the relative amount of fatty and fibro-glandular breast tissue that is visible on a mammogram. Mammographic breast density can impact screening mammography in two ways. First, denser tissue may make it more difficult to detect breast cancer (masking). Second, dense breasts are an independent risk factor for breast cancer. There are four categories of breast density: • Almost entirely fatty, 10% of women; • Scattered areas of fibro-glandular, 40% of women; • Heterogeneously dense, 40% of women; and • Extremely dense, 10% of women.
The 50% of women in the heterogeneously dense and extremely dense categories are considered to have dense breasts. Compared with women who have average breast density, the risk of developing breast cancer for women with heterogeneously dense breasts is 1.2 times greater than average, and the risk for women with extremely dense breasts is 2 times greater. 14 A person's risk depends upon breast density plus other risk factors such as gene mutations, family history, and personal history. 15 Additional or alternate screening options for women with dense breasts include MRI (described above), ultrasound, and computerized tomography, which differs from breast tomosynthesis. Whole breast ultrasound, the most commonly used modality, detects approximately 1 additional cancer for every 234 screenings. However, ultrasound studies result in additional testing for many women, including biopsy, with most of the biopsies eventually found to be negative for cancer. 16
In 2014, the ACOG Committee on Gynecologic Practice issued an opinion to the effect that although women with dense breasts have a “modestly increased risk of breast cancer and experience reduced sensitivity of mammography to detect breast cancer,” there is insufficient evidence to advocate for additional testing. 17 The Committee recommended that health care providers comply with state laws requiring them to disclose to patients their breast density as indicated in a mammogram report.
Despite the ACOG Committee Opinion and a similar position statement from the American College of Radiology, 18 many patients appear to be undergoing additional follow-up testing, potentially increasing both cost and anxiety for these patients. In contrast, the introduction of digital breast tomosynthesis has been shown to lower patient recall rates by up to 30%, with the greatest reduction in younger women and those with denser breasts. 19
Patient Perspectives on Breast Cancer Screening
From the patient perspective, breast cancer screenings, and especially recalls, are often regarded as burdensome in terms of anxiety, fear, confusion, and cost. The cycle of procedures following an abnormal result can be particularly taxing on women (Fig. 1). Misperceptions about screening processes, lack of consensus opinion among professional organizations, and additional costs associated with false positive results (e.g., out-of-pocket, co-pay, deductible) all contribute to the potential burden imposed on patients. Although some of these factors are well documented, others are harder to elucidate and can only be assumed based on the qualitative research.
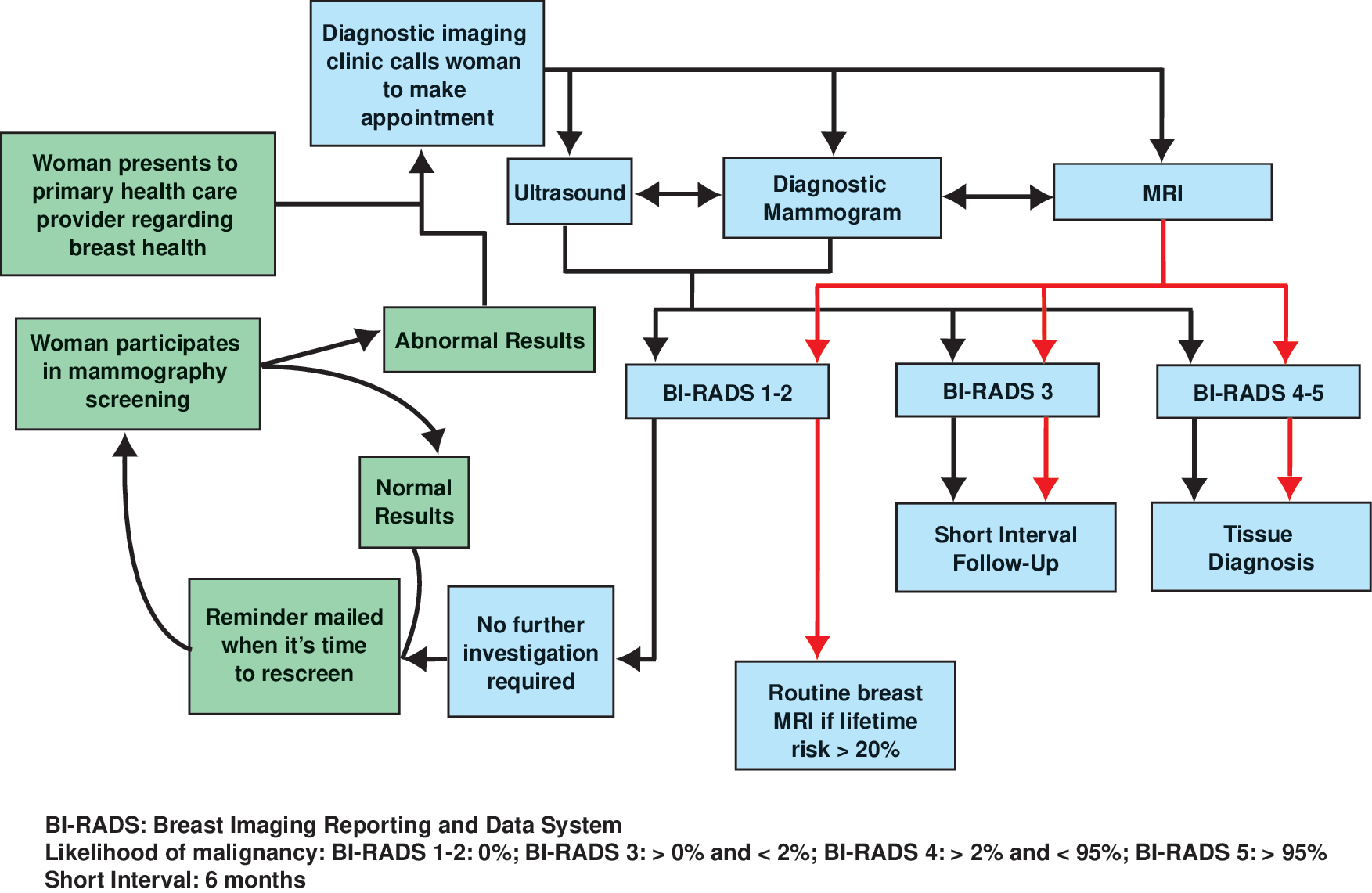
The typical mammography screening cycle (left side, in green), and the sequence of possible events following an abnormal screening result (right side, in blue). Two-way arrows between types of diagnostic imaging represent the potential for multiple imaging procedures occurring prior to Breast Imaging Reporting and Data System (BI-RADS) categorization and subsequent management. Management after MRI (red arrows) may differ from other imaging modalities depending upon BI-RADS categorization.
Fear of breast cancer screening
There has been a vast amount of research showing that emotions, such as fear, can have a major impact on behavior and that this reaction is central to patterns of patient healthcare utilization or avoidance. 20 Currently, it is unclear whether fear is a barrier or facilitator of breast cancer screenings. 20 Also, it is important to recognize that some behaviors and emotional responses to breast cancer screening may vary by ethnicity and socioeconomic status. One study found that compared with women of different backgrounds, African American females displayed a generalized fear of the health care system and a sense of fatalism (fear of imminent death upon diagnosis of breast cancer). 21 These factors may contribute to the fact that African American women present with breast cancer at later stages of the disease. 3
Psychosocial impact of false positives
The psychosocial implications of having an inconclusive screening mammogram vary significantly, and women experience a range of emotional responses. 22,23 For example, in a Swedish study with a small sample size, 13 women interviewed after being recalled reported experiencing a “roller-coaster” of emotions; some felt that the worst case scenario was imminent, whereas others felt that being supported by family members evoked feelings of strength and hope. For almost all women, the uncertainty about a possible abnormality resulted in an increase in anxiety. 22 The stress experienced by women after an abnormal screening has been shown to be transient in nature 24 and is not likely to cause long-term symptoms of depression or a deterioration of overall mental health. 25 However, one study has shown that the emotional impact of additional views or biopsies can have a powerful adverse short-term effect. 26 Additionally, an individual patient who has multiple recalls from screening may begin to question the accuracy and value of the examination.
Confusion due to mixed messaging
Variation in breast cancer screening guidelines is one source of confusion for patients. As noted above, the ACOG, ACS, NCCN, NCI, and USPSTF are not aligned in their recommendations about when to initiate mammograms (age 40 or 50), the frequency of mammograms (annual vs. biennial) or the advisability of breast self-exams. Media messages also affect the public perception of the utility of cancer screening. 27 In a 2004 U.S. survey of 500 adults, 87% said that they thought cancer screening was almost always a good idea, with most indicating that they would overrule a physician who recommended against a screening. 27 The extent to which patients are aware of available options for breast screening is also unclear. For instance, recent studies have demonstrated that 3-D mammography can improve cancer detection 11 as well as reduce recall rates from 15%–63%. 19,28 –31 Targeted education would help raise awareness of this option for patients seeking to gain earlier and more accurate detection of breast cancer as well as to avoid being recalled from screening. Further discussions could enable women to make informed decisions regarding various mammography options (e.g., preference for an insurance carrier to provide coverage, or electing a test with a possible out-of-pocket [OOP] cost but better accuracy in detecting cancer and lower chance of recall for findings that are normal on further evaluation).
Patient cost burden
There are a variety of ways that breast cancer screening—and particularly false positive results—can be a financial burden to a patient. In addition to the direct costs of imaging studies (e.g., OOP costs, deductibles, co-pays), there are indirect costs such as missed time from work (or use of paid time off), transportation, and caregiver costs (e.g., cost of childcare or for elder care). Some studies have attempted to quantify these costs; 32,33 however, the results must be cautiously interpreted. In a Norwegian study of breast cancer screening costs, travel and lost productivity costs were projected using governmental estimates for travel reimbursement and average pretax monthly salaries. These non-claims based costs were shown to constitute 21% of the total OOP costs to patients who were recalled, and 36% of the total OOP cost to all patients who were screened. 32 Although rough estimates, these findings shed light on the impact OOP costs may have on U.S. patients who are recalled due to false positive screening results. Although these studies address costs to women with health insurance, it is important to note that women with low income and/or women who are uninsured bear a disproportionate cost burden.
When examining many patient-centric issues, government and commercial payer claims data are insufficient; non-claims cost data must be collected to properly measure the extent to which indirect costs impact patients recalled due to inconclusive mammograms.
Influence of Other Stakeholders
Payer-related issues
Public and private insurers play a major role in women's access to breast cancer screening and thus the stage at which cancer is detected. Review of the current literature reveals a wide gap in care between commercially insured women and women who are publicly insured or uninsured. 34 In a large 2007 U.S. study, researchers found that uninsured patients and those covered by Medicaid were at considerably higher risk of being diagnosed with later stages of breast cancer compared with women who had commercial insurance. 34 In addition to a low compliance with initial breast screenings, another contributing factor to this disparity was that underinsured patients were less likely to receive timely follow-up of abnormal mammograms and were more likely to experience delays in treatment—both of which lead to increased risk of cancer-related morbidity and mortality and can magnify the cost burden for publicly funded programs.
Concerns about the cost of false positive recalls and increased utilization costs impact all payers who provide coverage for breast cancer screening. In a study of commercially insured women, the mean avoidable cost to health plans per recalled patient was reported as $1,238 compared with $0 for nonrecalled patients. 35 The follow-up diagnostic services that accounted for a majority of these costs included MRI, guided (large core needle) biopsy and open (diagnostic excisional) biopsy, ultrasound, and additional mammography. Biopsies alone accounted for two-thirds of recall expenditures. The costs of additional procedures required for the assessment of ultimately false positive results fall largely on health care insurers. Furthermore, a retrospective review in Connecticut using global Medicare reimbursement rates for whole breast ultrasound, follow-up examinations, biopsy, and aspiration found the additional costs of screening ultrasound for women with dense breasts was $180,802, or approximately $60,267 per cancer diagnosed. 36 For employers who self-insure, costs associated with false positive mammography pose a particular concern.
Newer technologies are emerging with the potential to improve quality and decrease costs associated with breast cancer screening on both an individual and population basis. As the evidence mounts regarding the ultimate value of these modalities, commercial health plans and public payers should give serious consideration to covering technologies that improve quality and cost.
Health policy and the role of government
Government involvement in patient safety and quality as they relate to mammography screening can be traced back to 1992, with the passage of the Mammography Quality Standards Act. 37 Subsequent legislation has provided grants for related research and funding for breast cancer screening surveillance.
Similarly, since 1991, the Centers for Disease Control and Prevention–funded National Breast and Cervical Cancer Detection Program (NBCCDEP) has provided free or very low-cost breast cancer screenings to women who are uninsured, who meet criteria for low income status, or who are members of racial or ethnic minorities. Limited funding for NBCCDEP enables the program to screen less than 1 in 8 eligible women aged 40 to 64 nationwide. Unfortunately, funding is not likely to become available for the follow-up imaging required for approximately 10% of screenings that are interpreted as abnormal.
Since its passage in 2010, the ACA has made a substantial impact on health care services and choices; in particular, it has made health insurance available to more women and mandated coverage for preventive services including screening mammograms. The launch and continuing expansion of value-based initiatives has enabled individual patients and provider organizations alike to set priorities and make decisions based on quality as well as cost. For example, health insurance exchanges and consumer-directed health plans enable individuals to make value-based choices, and accountable care organizations and other risk-bearing health care provider entities assume responsibility for both cost and quality health outcomes of their patient populations. Future research is warranted to assess the impact of the ACA on access to breast cancer screening services and how this will translate into earlier detection of breast cancer for new enrollees in health insurance exchanges and Medicare and Medicaid beneficiaries.
To date, 18 states have passed “dense breast” legislation requiring physicians to inform patients about their breast tissue composition. The language and provisions of these laws vary by state. Combined with conflicting guidance on screening interval, these laws have caused a great deal of confusion among women, their physicians, and radiologists and, in some cases, have led to clinical recommendations that are not congruent with position statements by the American College of Radiology and the American College of Obstetricians and Gynecologists, resulting in further confusion.
Individual Patient Risk and Needs Assessment
Multiple factors, such as age and breast density (as previously discussed), predispose women to varying risks for breast cancer. This is critical to current and future discussions pertaining to screening technology. Individual risk assessment and tailored approaches to screening have the potential to improve the value of the services that patients receive. It has been shown that risk factors affect the sensitivity and accuracy of mammography and ultimately impact the rate of false positive tests and the overall cost burden on the health care system. 38 Although many have been reported, the most significant risk factors associated with uncertain outcomes and higher rates of recall are breast density and age. Breast density is affected by a multitude of elements including hormone replacement therapy, menstrual cycle phase, parity, body mass index, and familial or genetic tendency. 38 The National Cancer Institute has developed a Breast Cancer Risk Assessment tool designed to calculate a 5-year and lifetime risk of breast cancer. The model uses a woman's personal medical history, reproductive history, and history of breast cancer among her first-degree relatives to estimate her risk of developing invasive breast cancer. 39 In theory, this information could be used by patients and their providers to inform decisions pertaining to screening options and advanced diagnostic technologies.
A fundamental shift toward tailored screening and a patient-centered approach must be accompanied by consensus around consistent recommendations, updated protocols, and revised algorithms designed to optimize value, eliminate disparities, and create access to the most appropriate technology.
Conclusions
Despite the vast amount of literature published about breast cancer screening, significant gaps remain. In particular, little is known about the indirect costs to patients resulting from recalls for further testing following an inconclusive mammogram. Although useful for measuring direct costs, claims data are limited in scope and cannot address a range of related issues that patients may face. The psychosocial impact of patient recalls is well documented; however, it is unclear how this might translate into additional costs for women, healthcare providers, employers, and payers.
The introduction of advanced breast screening technology (e.g., digital breast tomosynthesis) may be helpful in addressing some of the issues discussed above. Improved invasive cancer detection rates, and reduced false positive interpretations, have the potential to positively impact individual patients, the health care system as a whole, and health care delivery organizations in terms of meeting or exceeding quality benchmarks.
Optimizing the value of any new technology requires a focused effort and targeted education for all stakeholders (including patients) to ensure appropriate use and cost effectiveness. From a population health perspective, categorizing women by risk for breast cancer, and targeting effective interventions to those specific risk populations is likely to have a global positive effect. The concern is that our current risk models are underdeveloped: they emphasize family history when most breast cancers arise from spontaneous mutations; they use breast density when the assessment is subjective; and they use biopsy to prove a high-risk lesion when not all are related to proliferative changes and subsequent risk. Ultimately, continued research and innovation will enable us to better define risk and employ appropriate screening technologies to reduce the frequency of patient recalls and further improve breast cancer detection programs.
Footnotes
Acknowledgments
The authors gratefully acknowledge the contributions of Matthew Alcusky and Elizabeth A. Battaglino, who reviewed and provided valuable comments, guidance, and direction in the development of this article.
Disclosure Statement
Dr. Skoufalos, Dr. Vegesna, and Ms. Clarke are employed by The Jefferson School of Population Health. The school received financial support from Mary Ann Liebert, Inc., publishers, to review the literature, consult with designated experts, and develop and author this article. Dr. Harvey and Dr. Mass received an honorarium from Mary Ann Liebert, Inc., publishers for their participation in this article.
