Abstract
Dyspnea is one of the most common and distressing symptoms in patients with cancer and noncancer advanced diseases. The Japanese Society for Palliative Medicine revised previous guidelines for the management of respiratory symptoms in patients with cancer and newly developed clinical guidelines for managing dyspnea in patients with advanced disease, based on the result of systematic reviews for each clinical question and consensus among experts. We describe the recommendations of the guidelines as well as provide insights into the reasoning behind the recommendations and their development process. There has been a paucity of evidence regarding the interventions for dyspnea in patients with advanced disease. Thus, more clinical research that includes not only randomized controlled trials but also real-world observational studies is warranted.
Introduction
Dyspnea is a common symptom in patients with advanced diseases, with a reported prevalence of 50%–70% in patients with advanced cancer,1–4 and more frequent (up to 90%) in other advanced noncancer diseases, such as chronic obstructive pulmonary disease or congestive heart failure. 5 Moreover, dyspnea is related to poor quality of life (QOL) in patients with advanced diseases.6,7 Thus, managing dyspnea is one of the important roles of palliative care. The Japanese Society for Palliative Medicine (JSPM) initially published the Clinical Guidelines for Respiratory Symptoms in Cancer Patients in 2011 8 and revised them in 2016. 9 Although the previous guidelines addressed various respiratory symptoms, they specifically targeted patients with cancer. With the growing importance of palliative care for noncancer patients, the task group revising the guidelines broadened the scope to include noncancer patients. Simultaneously, to maintain focus and coherence, the revised guidelines concentrate solely on dyspnea.
This study presents the recommendations of the updated JSPM clinical guidelines for managing dyspnea in patients with advanced disease, providing insights into the reasoning behind the recommendations and their development process.
Development Process
The development of these clinical guidelines aimed to establish standard palliative interventions for dyspnea in patients with advanced disease. First, clinical questions (CQ) were developed based on a consensus meeting of taskforce members (six palliative care physicians, one nurse specialist, one pharmacist, and one epidemiologist). Six CQs (oxygen therapy, high-flow nasal cannula oxygen [HFNC], fan therapy, opioids, benzodiazepine, and corticosteroids) were selected and confirmed by the JSPM delegate members and two patient–family alliances (Japan Federation of Cancer Patients Groups and Non-Profit Organization (NPO) Consumer Organization for Medicine and Law [COML]). To expand the target population of the guidelines, nonpharmacological treatments (oxygen therapy, HFNC, and fan therapy) were included for any advanced diseases, whereas pharmacological treatments (opioids, benzodiazepine, and corticosteroids) were limited to patients with cancer. This distinction was made considering potential differences in treatment effects between diseases and the limited experience of palliative care for patients without cancer among specialists in both palliative care and specific advanced diseases because reimbursement for palliative care practice in Japan had historically been adopted only for patients with cancer and AIDS. The primary users of this guideline are all health care providers caring for patients with advanced diseases, including palliative care physicians, specialist physicians, primary care physicians, nurses, and pharmacists.
Systematic reviews
Systematic reviews (SRs) were conducted for each CQ by searching the Cochrane Central Register of Controlled Trials, MEDLINE, Embase, and Ichushi-web for articles published before September 23, 2019.
The primary screening involved reviewing titles and abstracts to select potentially relevant articles for each CQ. Secondary screening, which included reviewing the full texts of potentially relevant articles, was then conducted. After secondary screening, we selected relevant articles to provide evidence for each CQ. Additional articles were identified through hand searches of reference lists of selected articles and major review articles related to each CQ. Briefly, for CQs regarding nonpharmacological interventions, only randomized controlled trials (RCTs) of patients with all advanced diseases were evaluated. For CQs regarding pharmacological interventions, RCTs, non-RCTs, and observational studies with control groups were evaluated. The selection strategy for study design was as follows: (1) if there were at least two RCTs, inclusion was completed; (2) if there was no or only one RCT, non-RCTs or observational studies with control groups were included; (3) if there were no RCTs, non-RCTs, or observational studies with control groups, single-arm observational studies were included. Case reports and case series were excluded. The selection strategy for participants was as follows: (1) if there were studies that included only patients with cancer or performed subgroup analyses of only patients with cancer, inclusion was completed; (ii) if there were no studies that included only patients with cancer, studies with mixed populations in which at least 50% of the patients had cancer were included, and inclusion was completed; (3) if there were no studies that included only patients with cancer or studies in which at least 50% of the patients had cancer, studies with mixed populations in which <50% of the patients had cancer were included. The studies that only included patients without cancer were excluded. This selection strategy for participants was prioritized over the strategy for the study designs.
All included articles were assessed for predefined outcomes and risk of bias following the Minds Manual for Guideline Development 2020 ver. 3.0. 10 Literature review was independently performed by two SR team members for each CQ, and the details of the process and results of each SR have been documented elsewhere.11–16
Drafting recommendations and Delphi method
Tentative draft recommendation statements with rationales were crafted by the taskforce member responsible for each CQ, relying on evidence summaries from SRs. Subsequently, the taskforce reviewed and confirmed the first draft recommendations, incorporating modifications through discussion. The modified Delphi method was then used to assess the validity and establish consensus for each draft recommendation. 17 Delphi rounds were conducted by six palliative care physicians, one nurse specialist, and one pharmacist from JSPM, along with representatives from eight related academic organizations, including the Japanese Respiratory Society (pulmonologist), Japanese Society of Medical Oncology (oncologist), Japan Primary Care Association (JPCA; primary care physician), Japanese Society of Cancer Nursing (oncology nurse), and Japanese Society for Pharmaceutical Palliative Care and Sciences (palliative care pharmacist), and one patient–family alliance (COML). After three Delphi rounds and modifications to the draft recommendations, a consensus was achieved. The external review involved seven reviewers, including three palliative care specialists (physician, nurse, and pharmacist from the JSPM), two oncologists (the Japanese Society of Clinical Oncology and the Japan Lung Cancer Society), one primary care physician (JPCA), and one respiratory therapist (the Japan Society for Respiratory Care and Rehabilitation). The final version of the recommendations received approval after this external review.
Evidence and recommendation levels
We used the Minds grading system 10 to specify the level of evidence and the strength of recommendations, adhering to the principles of the Grading of Recommendations Assessment, Development, and Evaluation system 18 (Table 1).
Recommendation Table
Recommendations
Assessment of dyspnea
When we see patients who complain of dyspnea, the intensity, pattern, and influence on the daily life of the dyspnea should be evaluated, first. In addition, a thorough evaluation regarding the etiology of dyspnea should be made. This assessment includes history, physical examinations, laboratory tests, and imaging studies. If there is room, treatments for the underlying etiology of dyspnea should be obtained before considering symptomatic treatments.
Oxygen therapy
CQ 1-1: Is supplemental oxygen useful for dyspnea in patients with advanced disease with hypoxemia at rest?
We found five RCTs on supplemental oxygen for dyspnea in patients with advanced disease with hypoxemia at rest.19–23 Although three of them evaluated the effect of supplemental oxygen at rest,21–23 the remaining two evaluated its effect on exertion.19,20 Although one study showed significant improvement in dyspnea, 22 four studies did not show a significant difference in dyspnea.19–21,23 Among two studies evaluating exercise tolerance,19,20 only one study 19 showed significant improvement in the supplemental oxygen group. Only one study 20 evaluated QOL, which did not differ significantly. We could not find any studies evaluating somnolence and discomfort.
Based on these results, we could not find concrete evidence on the efficacy of supplemental oxygen for dyspnea in patients with advanced disease with hypoxemia at rest. However, one study showed a significant improvement, and the remaining studies showed the tendency of superiority of supplemental oxygen. Moreover, the evidence that oxygen therapy contributes to prolonging survival in patients with hypoxemia has been established, and oxygen therapy is thought to be a well-established treatment for hypoxemia internationally. Thus, we concluded that supplemental oxygen is likely to be useful.
Recommendation 1-1: Supplemental oxygen is suggested for dyspnea in patients with advanced disease with hypoxemia at rest (2C).
CQ 1-2: Is supplemental oxygen useful for dyspnea in patients with advanced disease without hypoxemia at rest?
We found 39 RCTs on supplemental oxygen for dyspnea in patients with advanced disease without hypoxemia at rest.19,24–60 Based on the differences in the target population and administration method of oxygen therapy, these studies were categorized into four subgroups: (1) supplemental oxygen during exertion, (2) short-burst oxygen, (3) supplemental oxygen at rest, and (4) supplemental oxygen during rehabilitation.
Supplemental oxygen during exertion
In total, 22 RCTs evaluated supplemental oxygen during exertion.19,20,24,25,28,30,33,34,37,39,43,45,46,49,51–56,58,61 Of those, 21 studies evaluated dyspnea during exercise tests.19,25,26,28,30,33,35,37,39,43,45,46, 49 ,51–56,58,61 Of the 21 studies, 13 showed a significant improvement in dyspnea in the supplemental oxygen group,19,28,30,34,39,43,45,49,52–54,58,61 whereas the remaining 8 did not.25,26,32,37,46,51,55,56 We integrated 16 studies,19,25,26,28,30,37,39,45,46,48,51,52,54,55,58,61 and the meta-analysis showed a significant improvement in dyspnea in the supplemental oxygen group, with a standard mean difference (SMD) of 0.57 (95% confidence intervals [CIs]; 0.38, 0.77). However, four studies evaluated the effect of dyspnea on exertion during daily activities.35,42,46,55 Of those, two studies showed a significant improvement in dyspnea in the supplemental oxygen group,43,46 whereas the other two did not.35,55 The meta-analysis of two studies35,55 that used the Chronic Respiratory Questionnaire (CRQ) dyspnea domain as the outcome did not show any significant differences, with a mean difference of 1.34 (95% CIs; −0.35, 3.04). There were 21 studies on exercise tolerance.19,25,26,30,33–35,37,39,43,45,46,49,51–56,58,61 A meta-analysis of 12 studies19,25,26,37,43,46,49,51,52,54,55,61 was conducted, evaluating walking distance during the walk test, with 5 studies30,33,39,45,58 evaluating the duration of exercise on the steady load test. Both groups showed a significant improvement in the supplemental oxygen group. Regarding QOL, four studies were found.35,43,46,55 A meta-analysis of two studies was conducted,35,43 showing a significant improvement in QOL in the supplemental oxygen group. Only one study 46 evaluated discomfort, which showed that 11 of 14 participants who evaluated supplemental oxygen as effective reported “unacceptable” or “intolerable.” We could not find any studies evaluating somnolence.
Based on these results, we concluded that supplemental oxygen may improve dyspnea, exercise tolerance, and QOL during exertion in patients with advanced disease without hypoxemia at rest. We also noticed a possibility of discomfort from supplemental oxygen.
Recommendation 1-2a: Supplemental oxygen during exertion is suggested for dyspnea in patients with advanced disease without hypoxemia at rest (2B).
Short-burst oxygen
Four RCTs assessed short-burst oxygen.32,38,50,59 No study demonstrated a significant difference in dyspnea intensity. Only one study, 32 which evaluated QOL, did not show any significant difference. No studies assessing exercise tolerance, somnolence, or discomfort were identified. Given these findings, there is insufficient concrete evidence to support the conclusion that short-burst oxygen improves dyspnea, exercise tolerance, and QOL in patients with advanced disease without hypoxemia at rest.
Recommendation 1-2b: Short-burst oxygen is not suggested for dyspnea in patients with advanced disease without hypoxemia at rest (2B).
Supplemental oxygen at rest or during sleep
Four RCTs examined supplemental oxygen at rest or during sleep.24,29,44,48 Only one study 48 demonstrated a significant improvement in dyspnea, whereas the remaining three studies24,29,44 showed no significant difference in dyspnea intensity. Concerning exercise tolerance, three studies have been identified.24,29,48 One study 48 reported a significant improvement in exercise tolerance in the supplemental oxygen group, whereas the other two studies29,44 did not show any significant difference. For QOL, three studies have been identified.24,29,44 All three studies found no significant difference, and a meta-analysis of two studies24,44 also did not show any significant difference. One study 24 evaluated somnolence and discomfort, revealing no significant difference in either of the outcomes.
Based on these results, concrete evidence supporting the effectiveness of supplemental oxygen at rest or during sleep in improving dyspnea in patients with advanced disease without hypoxemia at rest is lacking. Therefore, we cannot recommend supplemental oxygen in this context. However, it is noteworthy that supplemental oxygen did not increase adverse events, and no severe adverse events (SAEs) were reported in the supplemental oxygen group. There was also a tendency for less dyspnea in the supplemental oxygen group in most studies although not statistically significant. Consequently, we consider it acceptable to explore the use of supplemental oxygen at rest or during sleep in these cases.
Recommendation 1-2c: Supplemental oxygen at rest or during sleep is not suggested for dyspnea in patients with advanced disease without hypoxemia at rest (2B).
Supplemental oxygen during rehabilitation
Nine RCTs assessed supplemental oxygen during rehabilitation.27,31,36,40–42,47,57,60 Of these, six studies evaluated dyspnea as an outcome,27,31,41,47,57,60 and all reported no significant difference in dyspnea intensity. A meta-analysis of four studies,27,41,47,57 which used the modified Borg scale, also showed no significant difference (mean difference: 0.16, 95% CIs; −0.75, 1.06). Another meta-analysis of two studies,27,31 which used the CRQ dyspnea domain, revealed no significant difference (mean difference: 0.08, 95% CIs; −0.41, 0.57). Regarding exercise tolerance, nine studies were identified.27,31,36,40–42,47,57,60 Two studies31,36 showed a significant improvement, whereas the remaining seven studies reported no significant difference. Meta-analyses of three studies27,42,57 evaluating walk distance and two studies41,47 evaluating peak exercise capacity showed no significant difference. For QOL, six studies were found.27,31,40,42,47,57 All six studies reported no significant difference, and a meta-analysis of two studies27,57 also showed no significant difference. No studies evaluating somnolence were identified. Regarding discomfort, one study 31 reported that 69% of the supplemental oxygen group did not use oxygen outside training owing to the hassle of carrying or embarrassment.
Based on these results, there is no concrete evidence supporting the idea that supplemental oxygen during rehabilitation improves dyspnea, exercise tolerance, and QOL in patients with advanced disease without hypoxemia at rest.
Recommendation 1-2d: Supplemental oxygen during rehabilitation is not suggested for dyspnea in patients with advanced disease without hypoxemia at rest (2B).
High-flow nasal cannula oxygen therapy
CQ2: Is high-flow nasal cannula oxygen therapy useful for dyspnea in patients with advanced disease with hypoxemia?
We identified six RCTs on HFNC for dyspnea in advanced disease patients with hypoxemia.62–67 Two studies each evaluated the effect of short-term HFNC,64,67 long-term HFNC,62,66 and HFNC during exertion.63,65 Three of those studies62,64,66 showed a significant improvement in dyspnea with HFNC, whereas one study 67 demonstrated significant improvement in the usual oxygen group, and two studies62,65 did not show any significant difference in dyspnea intensity. Because of differences in intervention or insufficient outcome reporting, a meta-analysis was not feasible. Regarding QOL, two studies were found.62,66 Although a meta-analysis was not conducted, both studies reported a significant improvement in the HFNC group. For discomfort, four studies were identified.62,64,65,67 Although statistical analysis was not performed, all four studies indicated a tendency for more discomfort in the HFNC group. Regarding skin disorders, only one study 62 reported redness and ulcer development in 1 patient each out of 29 patients on HFNC.
Based on these results, we concluded that there is a potential benefit of HFNC for dyspnea in advanced disease patients with hypoxemia, as most included studies showed an improvement in dyspnea and QOL. However, HFNC was associated with discomfort in most studies, and there is a concern about the limited availability of HFNC.
Recommendation 2: High-flow nasal cannula oxygen therapy is suggested for dyspnea in patients with advanced disease with hypoxemia that is refractory to standard oxygen therapy (2C).
Fan therapy
CQ3: Is fan therapy useful for treating dyspnea in patients with advanced disease?
We identified 10 RCTs (one with 3 arms, divided into 2 RCTs) on fan therapy for dyspnea in advanced disease patients.68–76 Among these, six studies69,71–73,75,76 demonstrated a significant improvement in dyspnea with fan therapy, whereas the remaining four studies68,70,74 did not report any significant difference in dyspnea intensity or perform statistical analysis. A meta-analysis of five studies68,71,73,75,76 revealed a significant improvement in dyspnea in the fan therapy group, with an SMD of 1.43 (95% CIs; 0.17, 2.70). Regarding self-efficacy or sense of ease, four studies were found.68,71,73 One study 71 did not show any difference, and although the remaining three studies indicated a tendency toward more self-efficacy or a sense of ease in the fan therapy group, statistical analysis was not performed. For discomfort, three studies were identified.68,70,71 Although individual studies did not conduct statistical analysis, the meta-analysis of these results showed no significant difference.
Based on these findings, we concluded that fan therapy improves dyspnea in patients with advanced disease. Furthermore, discomfort or other adverse events did not increase with fan therapy compared to placebo.
Recommendation 3: Fan therapy is recommended for dyspnea in patients with advanced disease (1B).
Opioids
CQ4-1: Is systemic morphine useful for treating dyspnea in patients with advanced cancer?
We identified seven RCTs on systemic morphine for dyspnea in patients with advanced cancer.77–83 Two studies77,78 were placebo controlled, two compared with other opioids, one with nebulized morphine, and the remaining two compared with benzodiazepine. Both placebo-controlled studies demonstrated a significant improvement in dyspnea with systemic morphine, whereas studies with active controls and those with different administration routes did not. A meta-analysis of two placebo-controlled studies77,78 indicated a significant improvement in dyspnea with systemic morphine (SMD: 0.78, 95% CIs; 0.10, 1.45); however, the meta-analysis of two studies comparing other opioids82,83 did not show any significant difference (SMD: 0.48, 95% CIs; −0.23, 1.19). No studies evaluating QOL were found. Regarding somnolence, four RCTs were identified.78,80–82 The meta-analysis of these studies did not show any significant difference (risk ratio [RR]: 1.11, 95% CIs; 0.60, 2.03). For SAEs, two studies were found.80,81 The meta-analysis did not show any significant difference in SAEs between systemic morphine and controls (RR: 1.25, 95% CIs; 0.64, 2.45).
Based on these results, we concluded that systemic morphine improves dyspnea in patients with advanced cancer compared with placebo. In addition, there was no significant difference in adverse events between systemic morphine and controls. However, we could not conclude that morphine is superior to other opioids at this time.
Recommendation 4-1: Systemic morphine is recommended for dyspnea in patients with advanced cancer (1B).
CQ4-2: Is systemic oxycodone useful for treating dyspnea in patients with advanced cancer?
We identified one RCT on the use of systemic oxycodone for dyspnea in patients with advanced cancer. 82 This study compared systemic oxycodone with morphine but was prematurely terminated because of poor accrual. Although dyspnea was significantly improved at 60 minutes in both groups, no superiority or noninferiority in dyspnea intensity was observed. No studies evaluating QOL were found. Regarding somnolence, one RCT 82 and four observational studies without a control group84–87 were identified. In the RCT, two patients (22.2%) in the morphine group developed moderate-to-severe somnolence, whereas no patients in the oxycodone group developed somnolence. Integrated data from four observational studies showed that 19.7% (15/76) of patients with cancer who received systemic oxycodone for dyspnea developed somnolence. For SAEs, one observational study reported that 2 of 24 patients with cancer who received systemic oxycodone died within two days.
Based on these results, concrete evidence supporting the improvement of dyspnea in patients with advanced cancer treated with systemic oxycodone is lacking. However, in the only included RCT, a significant improvement in dyspnea from baseline was observed in the systemic oxycodone group, and the reduction in dyspnea intensity from baseline was similar to that in the morphine group, which is considered the first-line treatment, although it was not statistically significant. Moreover, no SAEs associated with systemic oxycodone administration for dyspnea in patients with advanced cancer were identified. Thus, we concluded that systemic oxycodone could be an alternative to systemic morphine.
Recommendation 4-2: Systemic oxycodone is suggested for treating dyspnea in patients with advanced cancer (2C).
CQ4-3: Is systemic hydromorphone useful for treating dyspnea in patients with advanced cancer?
We identified one RCT on the use of systemic hydromorphone for treating dyspnea in patients with advanced cancer. 88 This study compared systemic hydromorphone for incidental dyspnea with nebulized saline and did not show any significant difference in the change of dyspnea intensity. No studies evaluating QOL, somnolence, or SAEs were found.
Based on these results, concrete evidence regarding the efficacy of systemic hydromorphone for dyspnea in patients with advanced cancer is lacking. However, the only included study showed a significant improvement in dyspnea from baseline in the systemic hydromorphone group. This study targeted incidental dyspnea, which might be expected to improve spontaneously over time, representing a significant limitation. Consequently, the evidence is insufficient to conclude whether systemic hydromorphone may be beneficial for dyspnea in patients with advanced cancer.
Recommendation 4-3: No recommendation can be made for the use of systemic hydromorphone for dyspnea in patients with advanced cancer (-C).
CQ4-4: Is systemic fentanyl useful for treating dyspnea in patients with advanced cancer?
We identified five RCTs on systemic fentanyl for dyspnea in patients with advanced cancer.83,89–92 Four of these studies89–92 were placebo controlled, and the remaining one compared fentanyl with morphine. All five studies included exertional or episodic dyspnea. In the four placebo-controlled RCTs, two studies89,92 did not show any significant difference in dyspnea intensity, and the other two90,91 did not perform statistical comparisons between groups. The meta-analysis of postintervention dyspnea intensity in these four placebo-controlled RCTs did not show any significant difference, with an SMD of 0.38 (95% CIs; 0.09, 0.86). The study comparing fentanyl with morphine also did not show any significant difference. No studies evaluating QOL were found. Regarding somnolence, two RCTs were identified.89,90 The meta-analysis of these studies did not show any significant difference, with a RR of 0.18 (95% CIs; 0.03, 1.20). For SAEs, one RCT was found. 92 This study did not show any significant differences.
Based on these results, it cannot be concluded that systemic fentanyl is superior to placebo or other opioids for dyspnea in patients with advanced cancer. However, no excessive adverse events from systemic fentanyl were found in the current evidence.
Recommendation 4-4: Systemic fentanyl is not suggested for dyspnea in patients with advanced cancer (2C).
CQ4-5: Is nebulized morphine useful for treating dyspnea in patients with advanced cancer?
We identified one RCT regarding the use of nebulized morphine for dyspnea in patients with advanced cancer. 79 This study compared nebulized morphine with subcutaneous morphine; however, it was prematurely terminated because of poor accrual. No significant difference in postintervention dyspnea intensity was observed between the two groups. No studies evaluating QOL were found. Regarding somnolence, one RCT 79 did not show any significant difference. For SAEs, one RCT and two observational studies93,94 without a control group were identified. In the RCT, no patient in the nebulized morphine group developed SAEs. Integrated data from two observational studies showed that 7% (2/27) of patients with cancer who received nebulized morphine for dyspnea developed SAEs.
Based on these results, concrete evidence supporting the efficacy of nebulized morphine for dyspnea in patients with advanced cancer is lacking. Moreover, although rare, cases of SAEs after receiving nebulized morphine for dyspnea have been reported in observational studies. Thus, there is no strong reason to recommend nebulized morphine.
Recommendation 4–5: Nebulized morphine is not suggested for dyspnea in patients with advanced cancer (2C).
Benzodiazepines
CQ5-1: Is benzodiazepine alone useful for dyspnea in patients with advanced cancer?
We identified two RCTs regarding benzodiazepine alone for dyspnea in patients with advanced cancer.80,81 Both studies compared benzodiazepine alone with systemic opioids. Although one study 81 did not perform statistical analysis on the proportion of improvement, dyspnea intensity was significantly lower in the benzodiazepine group. In contrast, another study 80 did not show any significant difference in either the proportion of improvement or dyspnea intensity. The meta-analysis of the proportion of improvement in the two studies did not show a significant difference, with an RR of 0.95 (95% CIs; 0.47, 1.89). Regarding relief of anxiety, one RCT 80 was found, but the amount of change in anxiety was not reported. For somnolence, two RCTs were identified.80,81 The meta-analysis of these studies did not show any significant difference, with an RR of 0.66 (95% CIs; 0.34, 1.30). For SAEs, two RCTs were found.80,81 The meta-analysis did not show any significant difference, with an RR of 0.78 (95% CIs; 0.41, 1.51).
Based on these results, concrete evidence supporting the efficacy of benzodiazepine alone for dyspnea in patients with advanced cancer is lacking. Thus, there is no strong reason to recommend benzodiazepine alone. However, no excessive adverse events from benzodiazepine alone were found in comparison with opioids.
Recommendation 5-1: Benzodiazepines alone are not suggested for dyspnea in patients with advanced cancer (2C).
CQ5-2: Is benzodiazepine in addition to systemic opioids useful for dyspnea in patients with advanced cancer?
We identified one RCT regarding the use of benzodiazepine in addition to systemic opioids for dyspnea in patients with advanced cancer. 80 This study compared benzodiazepine in addition to systemic opioids (the combination group) with systemic opioids alone and showed that a significantly higher proportion of patients improved dyspnea in the combination group. Regarding relief of anxiety, one RCT 80 was found, but the amount of change in anxiety was not reported. For somnolence, the same RCT 80 reported that 21.3% of the combination group and 31.4% of the opioid-alone group developed somnolence without statistical analysis. For SAEs, the same RCT 80 reported that 30.3% of the combination group and 37.1% of the opioid-alone group developed SAEs without statistical analysis.
Based on these results, we concluded that there is a potential benefit of benzodiazepine in combination with systemic opioids for dyspnea in patients with advanced cancer. In addition, no excessive adverse events from benzodiazepine in combination with systemic opioids were found in comparison with opioids alone. However, the included study only involved patients with a life expectancy of less than one week, and only one study was available, limiting the quality of evidence.
Recommendation 5-2: Benzodiazepines in addition to systemic opioids are suggested for dyspnea in patients with advanced cancer (2C).
Corticosteroids
CQ6: Are systemic corticosteroids useful for treating dyspnea in patients with advanced cancer?
We found two RCTs regarding systemic corticosteroids for dyspnea in patients with advanced cancer.95,96 One study included patients with lung involvement of cancer, and the other included patients who had three or more symptoms, evaluating dyspnea as one of the secondary outcomes. Although both studies showed a tendency of superior dyspnea improvement in the corticosteroid group, it was not statistically significant. The meta-analysis of these two studies, however, demonstrated a significant improvement of dyspnea in the corticosteroid group with a mean difference of 0.71 (95% CIs; 0.03, 1.40). Regarding QOL, one RCT 95 evaluated the Edmonton Symptom Assessment System and EORTC-QLQ-C30 as QOL parameters, showing significant improvement of somnolence in the corticosteroid group. Regarding delirium, two RCTs95,96 and one observational study 97 without a control group were found. In both RCTs, no cases of delirium were reported. In the observational study, the incidence of delirium was 8.1% (6/74). For SAEs, the meta-analysis of the two RCTs95,96 did not show any significant difference, with an RR of 0.96 (95% CIs; 0.19, 4.93).
Based on these results, we concluded that systemic corticosteroids might improve dyspnea in patients with advanced cancer. However, their use should be targeted at specific etiologies, such as lymphangitis carcinomatosis, major airway obstruction, superior vena cava syndrome, or multiple lung metastases, where the anti-inflammatory effect or reduction of peri-tumoral edema from systemic corticosteroids may provide palliation. Consequently, the routine use of systemic corticosteroids for dyspnea in patients with advanced cancer, regardless of its etiology, is not recommended. Regarding the duration of the corticosteroid treatment, one to two weeks was the treatment duration in the two RCTs.95,96 Considering the risk of adverse effects related to long-term corticosteroid use, we suggest that treatment duration should not exceed four weeks.
Recommendation 6: Systemic corticosteroids are suggested for dyspnea in specific situations of lung involvement in patients with advanced cancer (2C).
Discussion
Here, we present the recommendations of the JSPM guidelines for the management of dyspnea in advanced disease patients, with Figure 1 illustrating the treatment algorithm based on these recommendations. These guidelines are derived from a combination of the best available evidence and expert consensus. However, it is crucial to acknowledge that the evidence levels for most recommendations remain low, primarily owing to the challenges of conducting high-quality clinical research in vulnerable advanced disease patients, including ethical conflicts. When making treatment decisions for dyspnea in a clinical setting, it is essential to consider pharmaco-economic outcomes and the availability of devices and medications. Most important, we strongly discourage the adoption of individual recommendations in isolation without considering the patient’s preferences.
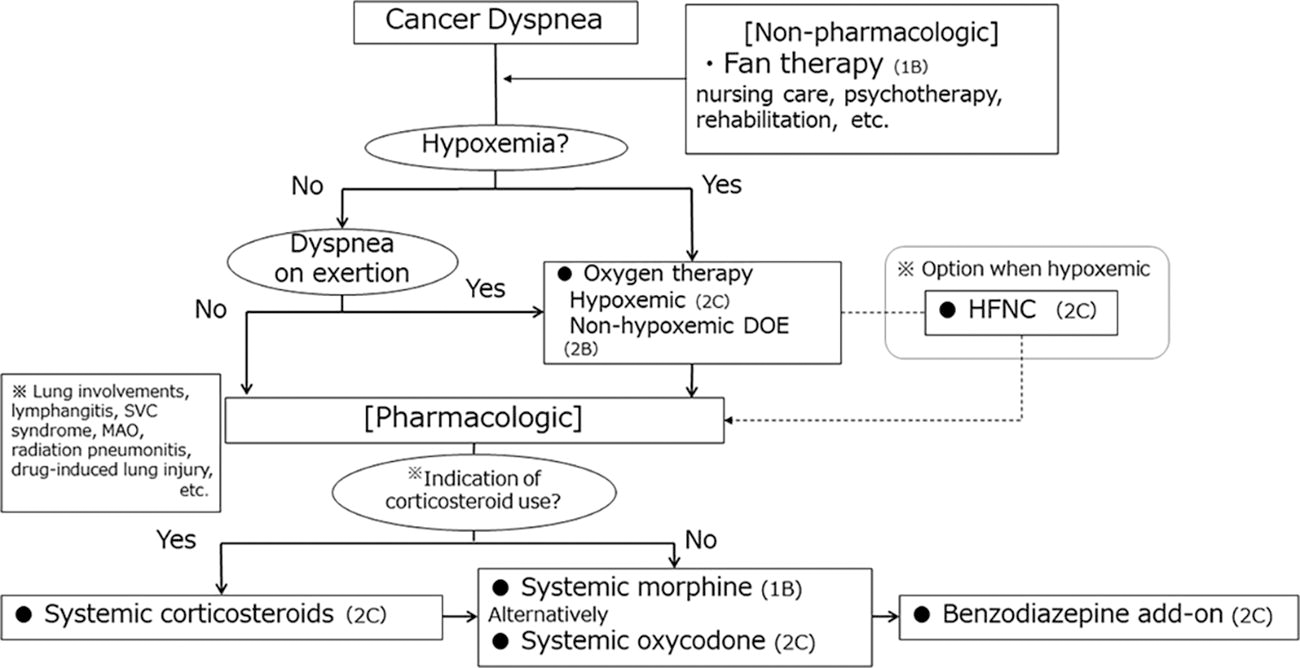
Overview of the recommendations.
To enhance the certainty of the evidence, we recommend increased efforts to conduct RCTs in this field. The use of real-world observational studies is also encouraged because RCTs often exclude patients with organ dysfunction or poor performance status, which are commonly found in palliative care settings. Through these future research endeavors, we aim to enhance these guidelines, ultimately contributing to improved clinical practice, alleviation of dyspnea in advanced disease patients, and enhancement of their QOL.
Footnotes
Acknowledgments
The authors thank the Japanese Respiratory Society, Japanese Society of Clinical Oncology, JPCA, the Japan Lung Cancer Society, the Japanese Society of Cancer Nursing, and the Japanese Society for Pharmaceutical Palliative Care and Sciences for their cooperation and advice in establishing recommendation statements.
Author Disclosure Statement
T.Y. has received lecture fees from Shionogi&Co., Ltd., Hisamitsu Pharmaceutical Co., Inc., Fujimoto Pharmaceutical Corporation, and Daiichi Sankyo Co., Ltd. Y.M. has received lecture fees from Hisamitsu Pharmaceutical Co., Inc., Fujimoto Pharmaceutical Corporation, Takeda Pharmaceutical Co., Ltd., Nippon Boehringer Ingelheim Co., Ltd., Chugai Pharmaceutical Co., Ltd., AstraZeneca Co., Ltd., and Daiichi Sankyo Co., Ltd. Y.K. has received lecture fees from Shionogi&Co., Ltd., Hisamitsu Pharmaceutical Co., Inc., Terumo Corporation, and Daiichi Sankyo Co., Ltd. S.G. has received lecture fees from Chugai Pharmaceutical Co., Ltd., Taiho Pharmaceutical Co., Ltd., Hisamitsu Pharmaceutical Co., Inc., Kyorin Pharmaceutical Co., Ltd., Nippon Boehringer Ingelheim Co., Ltd., and Astra Zeneca Co., Ltd. H.K. has received lecture fees from Daiichi Sankyo Co., Ltd. M.M. has received lecture fees from Shionogi&Co., Ltd., Hisamitsu Pharmaceutical Co., Inc., Daiichi Sankyo Co., Ltd., Takeda Pharmaceutical Co. Ltd., Ono Pharmaceutical, Chugai Pharmaceutical Co., Ltd., Kyowa-Kirin Co., Ltd., UCB Japan Co., Ltd., Eisai Co., Ltd., and Nihon Medi-Physics Co., Ltd. T.N. received research grants from I&H Co., Ltd., Cocokarafine Co., Ltd., and Konica Minolta Inc. and honoraria from Pfizer Japan, Janssen Pharmaceutical K.K., Boehringer Ingelheim, Eli Lilly Japan K.K., Mitsubishi Tanabe Pharma, Dentsu, and Otsuka Pharmaceutical. Other authors declare that there is no conflict of interest.
Funding Information
This work was funded by the Japanese Society for Palliative Medicine.
