Abstract
Background:
For patients with serious illnesses, one aim of palliative care services is to reduce the frequency and severity of hospital-based episodes of care. Since hospital-alternative palliative care may consume costly resources, providers need to efficiently target high-intensity services toward those most at risk for such adverse episodes of care.
Objective:
Our objective was to investigate progressively more restrictive diagnosis-based indications of serious illness as used to prospectively identify patients with higher average rates of hospitalization.
Design/Setting:
We designed an observational cohort quality improvement study using historical Medicare claims records to evaluate diagnostic inclusion criteria for targeting palliative and serious illness care resources. We first isolated a Seriously Ill Population (SIP) and then defined More SIP and Most SIP subgroups.
Measurements:
Our primary outcome measure was the 2019 acute-care count of hospitalizations for patients in the SIP, More SIP, and Most SIP subgroups, respectively.
Results:
The More SIP and Most SIP subgroups exhibited higher hospitalization rates. However, they also excluded progressively more seriously ill patients who did experience hospitalizations. In addition, almost half of the Most SIP subgroup were not hospitalized at all, despite having an average hospitalization rate greater than one.
Conclusion:
Allocating resources (personnel and services) toward reducing hospitalizations when almost half of the targeted population never goes to the hospital could result in unnecessary expenditures and exclude patients that could potentially benefit. Engaging community-based services to detect changes in status could provide supplemental indications of when and for whom to target palliative care resources.
Key Message
Palliative care resources are scarce, requiring accurate targeting. We developed diagnostic criteria for cohorts with high hospitalization rates but found these criteria would exclude patients needing palliative care and include patients that may not have benefited. Our study suggests that diagnostic criteria are not sufficient for palliative care resource targeting.
Introduction
For patients with serious illnesses, one of the more onerous burdens involves the revolving door of emergency department (ED) visits, hospitalizations, and post-acute recovery services. For example, patients with cancer—despite groundbreaking treatment innovations that improve long-term prognoses—may end up in the hospital multiple times for concomitant problems such as uncontrolled pain, infections, dehydration, and gastrointestinal distress. Similarly, patients with chronic diseases of the heart, lung, liver, or kidney may experience episodic decompensation or exacerbation of underlying conditions, necessitating numerous visits to the ED and hospital. As serious illnesses progress, their complexity may be compounded by challenges involving behavioral health, changes in cognition, and memory decline. Hospital visits may become more frequent and require extended lengths of stay, accelerating deterioration in patients’ quality of life and making a return to a stable community-dwelling status increasingly difficult. Thus, an important aim of palliative care services for serious illness patients is to mobilize community-based resources that reduce the frequency and severity of these adverse hospital-based events.
As the US healthcare system embraces imperatives to move towards value-based payment models, reducing hospitalizations in the seriously ill population could help reduce spending overall. However, hospital-alternative palliative care resources may themselves be relatively high in intensity and cost. In addition, the health care sector is now experiencing resource limitations (e.g., workforce shortages). The deficit of experienced and skilled medical personnel who could provide palliative and serious illness care has created a need to consider how to allocate resources based on need. Thus, to reduce hospitalizations in seriously ill populations, there is a need for quality improvements that help providers efficiently target scarce resources toward those who stand to benefit the most.
As a starting point for the identification of patients with unmet palliative care needs, the Center to Advance Palliative Care (CAPC) and the National Committee for Quality Assurance (NCQA) have developed diagnostic criteria to proactively identify patients who could benefit from palliative and serious illness care resources. 1 However, while most patients who stand to directly benefit from high-intensity palliative care resources may be represented by these diagnostic criteria, the converse may not be true: not all patients who meet the listed diagnostic criteria will need high-intensity palliative care resources. More specifically, many who meet the CAPC/NCQA criteria may never go to the hospital. While palliative resources may offer other benefits, high-intensity resources targeted specifically toward reducing hospitalizations in patients who are never admitted could be misallocated. Thus, there is an ongoing need to investigate how to better target palliative care resources, particularly the role of more accurate and stringent enrollment criteria. 2
Objective
In previous studies, we investigated the challenges associated with applying progressively more restrictive criteria for prospectively enrolling heart failure patients into care coordination programs. 3 We specifically investigated two mechanisms: narrow cohort focus and utilization heterogeneity. In the narrow cohort focus mechanism, progressively more stringent enrollment criteria exclude patients who do go to the hospital—notwithstanding a lower overall probability of doing so. As a result, opportunities to reduce hospital utilization for excluded beneficiaries may be missed. In the utilization heterogeneity mechanism, overall hospitalization rates may be higher for the patients meeting more stringently defined enrollment criteria, but many—if not most—may still exhibit zero hospitalizations.
In the present study, we hypothesize that these same mechanisms may apply to prospective diagnostic criteria used to identify seriously ill populations that may require high-intensity palliative care resources to reduce hospitalizations. Our objective is to specifically investigate quality improvements that use progressively more restrictive diagnosis-based criteria to commensurately isolate patients with more advanced illness. We begin by using the initial starting point of the list of CAPC/NCQA diagnosis codes to define a comparison group. We then progressively limit diagnostic inclusion criteria by including only the more advanced condition diagnosis codes that are associated with higher average rates of hospitalization.
In the next section, we describe the design, setting, measurements, and results involved in investigating the two mechanisms indicated above. The ramifications of our hypothesis—if supported by evidence—suggest that prospectively targeting high-intensity palliative care resources using more stringent criteria may not be sufficient, notwithstanding that the resulting smaller cohorts exhibit higher overall hospitalization rates.
Design
We designed an observational cohort quality improvement study using historical medical claims records to evaluate diagnostic inclusion criteria for targeting high-intensity palliative and serious illness care resources. It is important to note our study was not designed to investigate the effects of such resources themselves. Rather, the present study was designed only to investigate the targeting effectiveness, namely, the degree to which various levels of stringency in diagnostic criteria can proactively identify patients likely to be hospitalized. We did not attempt to exclude those patients who may have been exposed to community-based palliative care.
Setting
Our study was based on a five percent sample of Medicare claims data contained in the year 2019. We used Limited Data Set (LDS) files obtained from the Research Data Assistance Center (RESDAC). 4 The study population included beneficiaries of all ages enrolled in Medicare Part A and Part B. We included beneficiaries with state buy-in, also known as dual-eligibles. We excluded Medicare Advantage (Part C) enrollees because the LDS files do not contain utilization claims for Part C.
From this initial population, we isolated a Seriously Ill Population (SIP) using the CAPC/NCQA diagnosis criteria, comprising a total of 985 diagnosis codes drawn from the International Classification of Diseases (ICD-10), as shown in Supplementary Appendix S1. The SIP ICD-10 codes were obtained from inpatient, outpatient, and carrier claims records in the LDS dataset. This SIP study population served as our comparison group.
We then defined two subgroups of the SIP population, characterized by progressively more stringent and severe subsets of ICD-10 diagnostic codes. In both cases, as shown in Figure 1, the more stringent subsets of ICD-10 diagnostic codes were contained within the original CAPC/NCQA SIP codes. We specifically designated a “More SIP” subgroup and a “Most SIP” subgroup with ICD-10 codes provided in Supplementary Appendix S1and summarized here:

The original CAPC/NCQA SIP ICD-10 codes with designated “More SIP” and “Most SIP” subgroups whose ICD-10 codes are contained within the comparison group. The “Most SIP” ICD-10 codes are similarly contained within the “More SIP” subgroup. CAPC, Center to Advance Palliative Care; ICD, International Classification of Diseases; NCQA, the National Committee for Quality Assurance; SIP, Seriously Ill Population.
More SIP: Representing about a third (318) of the more serious ICD-10 codes contained in the original 985 SIP diagnoses
Most SIP: Representing about a third (100) of the most serious ICD-10 codes contained in the 318 More SIP diagnoses
As indicated in Supplementary Appendix S1, some of the original SIP ICD-10 codes in a given condition, such as heart disease, were included in the More SIP and/or Most SIP subsets.
Measurements
Our primary outcome measure was the 2019 count of hospitalizations from inpatient claim records. We aggregated counts of the number of hospitalizations for the patients included in the SIP, More SIP, and Most SIP subgroups, respectively. For each of these subgroups, we represented aggregate hospital utilization in tabular format as well as in the form of histograms of hospital counts. We also calculated the average hospitalization rate in each of the subgroups. Within each subgroup, we calculated the percentage of members having zero hospitalizations.
Results
In 2019, there were 62.5 million Medicare beneficiaries, 5 of which 39.9 million were not enrolled in Medicare Advantage plans. From our five percent subsample of Part A and Part B beneficiaries, we isolated the SIP population using 985 ICD-10 codes (Supplementary Appendix S1), resulting in 726,313 beneficiaries. This SIP population represented 36% of our original five percent subsample of beneficiaries in Medicare Parts A and B. As indicated previously, these 726,313 beneficiaries in the CAPC/NCQA population served as our comparison group. From this comparison group, we isolated the More SIP subpopulation using 318 ICD-10 codes (Supplementary Appendix S1), resulting in 447,903 beneficiaries, representing 22% of the initial Parts A and B population. Finally, we isolated the Most SIP subpopulation using 100 ICD-10 codes (Supplementary Appendix S1), resulting in 288,362 beneficiaries, representing 14% of the initial Parts A and B population.
Our primary outcomes of 2019 hospitalization count distributions are shown in Figure 2 using histogram plots to represent aggregate counts by percentage of each of the groups. These results are also represented in Table 1, along with the calculated percentages of members having zero hospitalizations and the counts of overall hospitalizations in each of the groups.
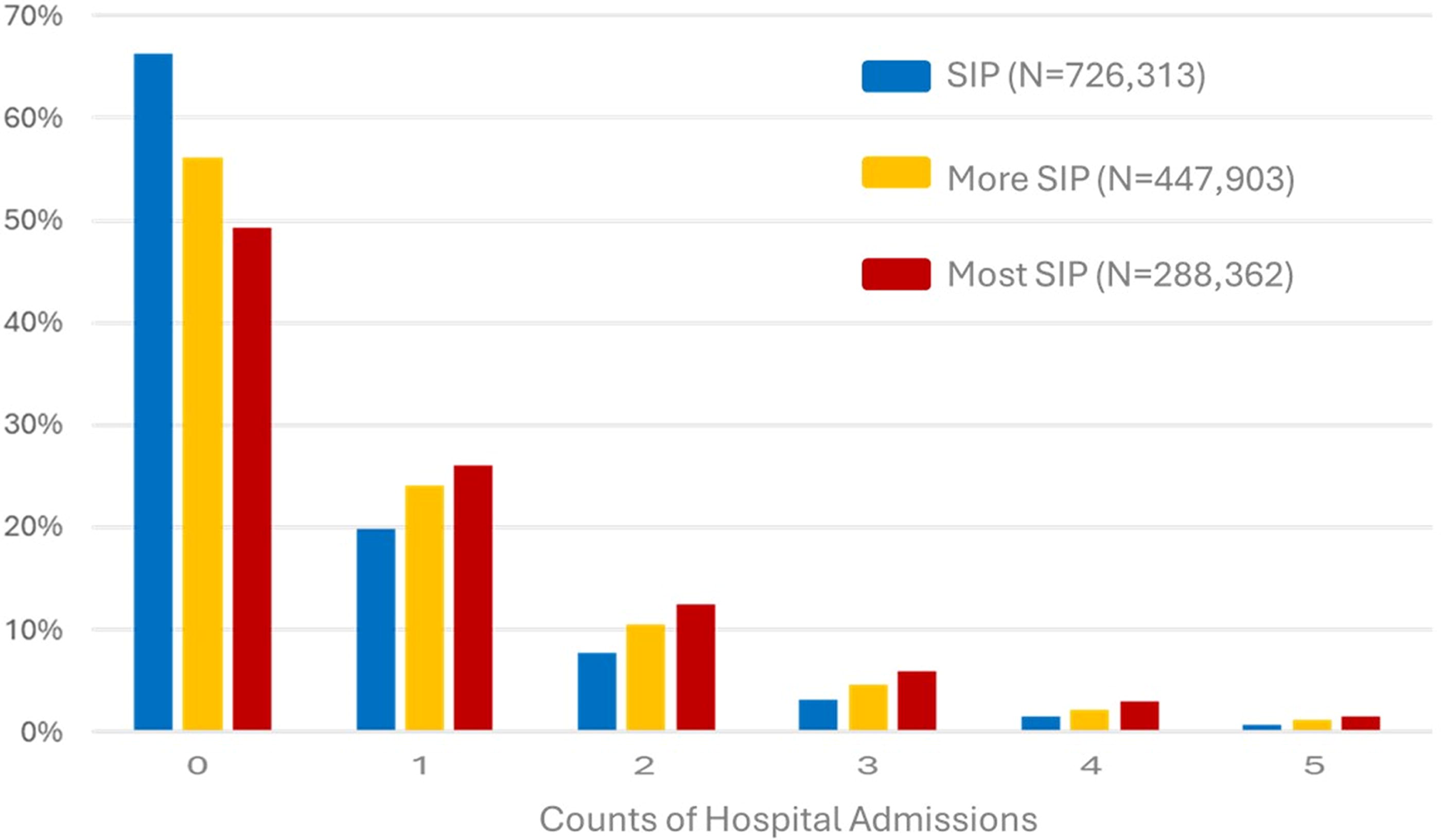
Histograms of 2019 counts of hospitalizations by percentages of the SIP, More SIP and Most SIP subgroups. SIP, Seriously Ill Population.
Characterization of ICD-10s and Numbers of Beneficiaries in SIP Comparison, More SIP and Most SIP Subgroups
Number of beneficiaries.
CAPC, Center to Advance Palliative Care; ICD, International Classification of Diseases; NCQA, the National Committee for Quality Assurance; SIP, Seriously Ill Population.
Discussion
As shown in Figure 2, all three utilization histograms exhibit skewed utilization patterns with prominent peaks at zero hospitalizations. These peaks indicate that among all possible counts of hospitalizations, zero is the most likely outcome in all three subgroups. The higher average group-level hospitalization rates in each of the subgroups are associated with progressively more patients experiencing multiple hospitalizations, represented by the longer right-hand tails of the histograms shown in Figure 2. More specifically, as indicated in Table 1, the More SIP and Most SIP subgroups exhibited higher hospitalization averages: .84 and 1.03, respectively, compared with .61 for the original CAPC/NCQA SIP comparison group. However, notwithstanding the higher overall hospitalization rates, the proportion of patients having zero hospitalizations did not drop as dramatically. In particular, almost half of the Most SIP subgroup were not hospitalized at all, despite having an average hospitalization rate greater than 1. Allocating resources (personnel and services) directed toward reducing hospitalizations when almost half of the targeted population never goes to the hospital could result in costly misallocation. We also note in Table 1 that with progressively more stringent diagnostic criteria, more patients who do have hospitalizations from the original CAPC/NCQA SIP comparison group end up being excluded in the More SIP and Most SIP subgroups. Specifically, in the More SIP subgroup, 66,812 of the comparison group hospitalizations are excluded, while in the Most SIP subgroup, 146,038 of the hospitalizations from the SIP comparison group are excluded. Thus, the narrowing of cohorts that exclude hospitalization counts of patients who could have potentially benefited from palliative care resources could represent a further misallocation of resources. In summary, both components of our hypothesis are supported by evidence: using more stringent enrollment criteria could leave out more who could benefit (narrow cohort focus) while providing hospital-avoidance resources to those who would otherwise not be admitted (utilization heterogeneity).
While our hypothesis is supported by evidence, it is important to acknowledge that our study provides limited new knowledge in terms of definitively indicating which individuals with serious illnesses will experience hospitalizations and/or would benefit from palliative care resources. It is possible that such individual-level indications could be obtained from additional criteria such as prior utilization (e.g., prior emergency department visits or hospitalizations), functional status (e.g., activities of daily living and instrumental activities of daily living) as well as social determinants of health. 6 However, it is also possible that the challenges identified using ICD codes are representative of a more pervasive problem—one common to all prospective approaches. As stated by the American Academy of Actuaries Committee on Risk Classification, 7 "Determining average experience for a particular class of risk is not the same as predicting the experience for an individual risk in the class. It is both impossible and unnecessary to predict experience for individual risks. If the occurrence, timing, and magnitude of an event were known in advance, there would be no economic uncertainty and therefore no reason for insurance."
As such, our own study findings may reflect the intrinsic limitations of prospective risk analysis. Thus, more stringent prospective criteria of any type may continue to exclude individual patients who do go to the hospital. And while average hospital utilization may be higher in the resulting subgroups, many of those included individuals may never go to the hospital. Stated differently, even within the more stringently defined subgroups, it may remain impossible to prospectively isolate exactly which individuals within such subgroups will go to the hospital.
Notwithstanding the above challenges, our investigation may provide indications on quality improvements that better allocate costly (and increasingly rare) palliative care resources. Specifically, rather than relying on narrowing prospective enrollment criteria in a way that could exclude patients who need palliative and serious illness care, enrollment criteria could be initially broad, using, for example, the original CAPC/NCQA enrollment criteria. However, given that so many of such a broad group of patients do not go to the hospital, these initial enrollment criteria need not necessarily trigger automatic allocation of high-intensity palliative care resources. Instead, initial inclusion criteria could begin with lower-intensity care planning and surveillance, with more intensive resources being allocated on an adaptive basis when emerging indications suggest a need for such escalation of care. Agile service intensity is a specific strategy recommended by CAPC for high need seriously ill populations, as well as proactive identification. 8 The present findings may suggest a benefit to maintaining a broad, inclusive approach to proactive engagement, coupled with an agile, flexible continuum of care services. 9 Related conclusions have been inferred from similar studies using Medicare claims data for high-cost patients.10,11
Home and community engagement may represent an underdeveloped mechanism to support agile service intensity. Specifically, organizations that support housing, transportation, and meals may have more consistent contact with high-risk patients, compared with intermittent clinical contact available from medical providers. Already, collaborative palliative care models layered on top of other services such as local area agencies and individual clinicians are recognized as enhancing the quality of life, lowering costs, and providing care that better matches patient needs. Our findings from the present study suggest a potential reciprocal relationship, where community-based services may themselves be enlisted to provide early indications of changes in patient status. Such indications could in turn be used to adaptively target when and for whom those higher-intensity forms of palliative care services should be activated or escalated. Exploring how palliative care delivery models could incorporate such community-based engagement would be a natural next body of quality improvement investigations, given the intrinsic limitations to prospective enrollment that have been identified in our present research.
Limitations
Our study data were limited to traditional Medicare (Part A and B) beneficiaries; we believe further study would be needed to analyze similar mechanisms in Medicare Advantage (Part C) and private insurance plans. Specifically, Part C and private plans may provide supplemental benefits that could include more extensive community-based palliative care services that are not widely available to traditional Medicare beneficiaries, which could in turn reduce observed hospitalizations. An additional limitation was the use of 2019 data, which may be outdated. However, because 2019 provides the last year of data reflective of the pre-COVID-19 era, it may better represent utilization patterns following the public health emergency. In addition, we evaluated hospital utilization in the same year from which the diagnostic inclusion criteria were gathered. In operational practice, prospective criteria may be based on historical diagnoses gathered before program enrollment. Although our study did not make a distinction between diagnostic enrollment and delivery periods, we believe the same general phenomena would be observed.
As indicated earlier, our investigation was limited to characterizing candidate groups and subgroups for high-intensity palliative and serious illness care resources. We did not investigate the effects of the resources themselves, in terms of lowering hospitalization counts or severities.
Also, we did not investigate whether some patients exhibiting zero hospitalizations may have already received community-based palliative or serious illness care services. As a result, our study may have overestimated the number of patients without hospitalizations in the absence of palliative care.
Finally, it is important to note that our study is limited in the sense that it associates a lack of hospitalizations with a lack of need. Patients in need of palliative care may be underrepresented by this perspective. For example, patients may decline to visit the hospital for a variety of reasons, including perceived safety issues, patient and caregiver burnout, or unfamiliarity with hospital-based palliative care options. These patients, despite exhibiting zero hospitalizations, stand to benefit greatly from palliative care service models including reduction of symptom burden, relief of suffering, and goal-aligned decision-making about disease treatments. The imperative to find such patients and to match critical resources reinforces the findings of the present study. Specifically, challenges in prospective enrollment for palliative care services suggest a need to maintain a broad, inclusive approach of initial engagement, coupled with an agile continuum of palliative care services delivered in accordance to patient needs—not just in hospital settings, but in the homes and communities where seriously ill patients reside.
Conclusions
To reduce hospitalizations in seriously ill populations, there is a need to efficiently target high-intensity palliative care resources toward those who stand to benefit the most. We began with a CAPC/NCQA comparison group and embarked on a quality improvement investigation of the effects of more stringent diagnostic inclusion criteria. After limiting the comparison group to More SIP and Most SIP diagnosis categories, we found higher average counts of hospitalizations in the associated subgroups. However, as we applied more stringent inclusion criteria, we also ended up excluding more and more patients who did go to the hospital, and many—if not most—of the patients who remained in the subgroups continued to exhibit zero hospitalizations.
Our investigation may indicate a potential need for palliative care resource deployment models that use relatively broad proactive identification criteria, while augmenting such criteria with specific provisions in plans of care that provide active surveillance for changes in status to better determine when and for whom more intensive resources should be allocated. Such active surveillance coupled with agile service delivery may represent an underdeveloped but valuable bridge between community-based patient engagement and serious illness care.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
