Abstract
In 2009, the European Association for Palliative Care (EAPC) developed a framework on palliative sedation, acknowledging this practice as an important and ethically acceptable intervention of last resort for terminally ill patients experiencing refractory symptoms. Before and after that, other guidelines on palliative sedation have been developed in Europe with variations in terminology and concepts. As part of the Palliative Sedation project (Horizon 2020 Funding No. 825700), a revision of the EAPC framework is planned. The aim of this article is to analyze the most frequently used palliative sedation guidelines as reported by experts from eight European countries to inform the discussion of the new framework. The three most reported documents per country were identified through an online survey among 124 clinical experts in December 2019. Those meeting guideline criteria were selected. Their content was assessed against the EAPC framework on palliative sedation. The quality of their methodology was evaluated with the Appraisal Guideline Research and Evaluation (AGREE) II instrument. Nine guidelines were included. All recognize palliative sedation as a last-resort treatment for refractory symptoms, but the criterion of refractoriness remains a matter of debate. Most guidelines recognize psychological or existential distress as (part of) an indication and some make specific recommendations for such cases. All agree that the assessment should be multiprofessional, but they diverge on the expertise required by the attending physician/team. Regarding decisions on hydration and nutrition, it is proposed that these should be independent of those for palliative sedation, but there is no clear consensus on the decision-making process. Several weaknesses were highlighted, particularly in areas of rigor of development and applicability. The identified points of debate and methodological weaknesses should be considered in any update or revision of the guidelines analyzed to improve the quality of their content and the applicability of their recommendations.
Introduction
Palliative sedation is estimated to precede 10%–18% of deaths in Europe, although its use is known to vary considerably between countries due to different religious, cultural, and social norms.1–3 Its practice still raises various issues such as the assessment of refractoriness of suffering, especially when suffering is primarily existential,1,2,4–6 the determination of the time frame within which it can be administered,2,6 the potentially life-shortening effect of the withdrawal or withholding of artificial hydration,5,6 or the risk of confusion with “hidden euthanasia,” especially when it is administered continuously and deeply.2,5
In 2009, the European Association for Palliative Care (EAPC), recognizing palliative sedation as an important and acceptable intervention for terminally ill patients with refractory symptoms, developed a framework on palliative sedation 7 to guide policy and facilitate the development of high-quality local procedural guidelines. 8 Several other guidelines have also been developed in Europe, showing differences in terms and concepts. 3 All tend to be based on expert consensus due to the paucity of data from prospective clinical studies. As part of the Palliative Sedation project (https://palliativesedation.eu), we analyzed commonly used guidelines on palliative sedation in the eight participating European countries (Belgium, Germany, Hungary, Italy, the Netherlands, Romania, Spain, and United Kingdom) to provide an overview of the European guidelines and the quality of their development to inform the updating of the EAPC framework on palliative sedation. 7
Methods
Survey design
The most frequently used guidelines were identified through an online survey (C.C. and E.G.). For this purpose, the definition of clinical guideline as systematically developed statements to assist practitioner and patient decisions about appropriate health care for specific clinical circumstances8–11 was adopted.
The questions were based on previous questionnaires identified through a PubMed and Google Scholar search (in English, 2005–2020 period, search terms: [“palliative sedation” AND “survey”]. Questions were approved by the consortium partners. The survey was designed in Survey Monkey, 12 asking (1) Does your country have national published guidelines on the use of Palliative Sedation? and (2) If yes, “Which are the three most widely used Palliative Sedation Guidelines in your field?”
This survey was approved on December 12, 2019, by the Ethics Committee of the University of Navarra, Spain (nr: 2019.194).
Data sources
A purposive sampling strategy was used; each consortium partner was asked to identify ≥18 clinicians (one physician and one nurse from each category) working in palliative care (hospital, care home, hospice, and home care settings) and other fields of medicine (anesthesiology, intensive care, internal medicine, oncology, and primary care), and capable of identifying the most frequently used guidelines on palliative sedation. The intention and research effort were directed at recruiting a comprehensive sample of physicians and nurses in different categories, who could report on clinical guidelines. However, in several countries, it was not possible to find enough representatives in all of these categories.
Some countries failed to identify experts in palliative sedation guidelines in intensive care, internal medicine, and oncology. It is possible that in these countries. palliative sedation is mainly performed by palliative care professionals rather than by other specialists. Lack of knowledge of guidelines in these settings could also be due to a common practice of sedation outside clinical guidelines. As the study did not aim to collect representative opinions from each discipline on the practice of palliative sedation, but rather to identify and comparatively analyze the most relevant clinical guidelines in each country, the investigators recruited more representatives of the categories able to identify guidelines, thus seeking to ensure the objective of the study to identify the most commonly used guidelines (Table 1).
Profiles of Participants of the Study
BE, Belgium; DE, Germany; ES, Spain; GB, Great Britain/United Kingdom; HU, Hungary; IT, Italy; NL, Netherlands; RO, Romania.
Survey launch
The survey link was sent by January 30th, 2020, and data collection closed by March 15th, 2020.
Selection criteria
For each country, the three documents most frequently referenced by the experts were selected. Those that did not meet the guideline definition8–11 were excluded (Table 2).
Most Cited Documents Per Country
x = cited; Documents in italics are those that have been excluded because they did not meet the criteria of guidelines.
APCNG, adult palliative care network guidelines (book.pallcare.info); EAPC, European Association for Palliative Care framework; ESMO, European Society for Medical Oncology Clinical Practice Guidelines for the Management of refractory symptoms at the end of life and the use of palliative sedation; FPZV, Palliatieve Sedatie, pallialine.be, Flanders; IKNL, Palliatieve sedatie. Landelijke richtlijn, Versie: 2.0 KNL; KNMG, Guideline for Palliative Sedation Royal Dutch Medical Association; NPE, Palliative Sedierung Arbeitsgemeinschaft heimbetreuende (Palliativ-)Ärzte (AG HPA) des Netzwerkes Palliativmedizin Essen; OMC/SECPAL, Guía de Sedación Paliativa (OMC y SECPAL), 2011; PCF, Palliative Care Formulary v6 (by Robert Twycross et al.)22; PRCPEX, Guía Clínica de Sedación Paliativa del PRCPEX. Uso y recomendaciones. 2015; PSIB, Guía de sedación paliativa: recomendaciones para los profesionales de la salud de las Islas Baleares. Regional Ministry of health, Baleares, Spain; S3-Leitlinie: Erweiterte S3-Leitlinie. Palliativmedizin für Patienten mit einer nicht-heilbaren Krebserkrankung Langversion 2.0—August 2019; SICP, Raccomandazioni della SICP sulla Sedazione Terminale/Sedazione Palliativa.
Content evaluation
Based on the work of Abarshi et al.,8,10 the analysis included definitions and terminology of palliative sedation and compared the recommendations of each guideline against the EAPC framework on palliative sedation, 7 highlighting similarities and differences between the selected guidelines. (Supplementary Data S1: Data extraction template for guidelines 12 ; Table 3).
Ten Core Recommendations Enumerated in the European Association for Palliative Care Framework 7
X = given; / = not given.
EAPC, European Association for Palliative Care; EOLC, end-of-life care; ESMO, European Society for Medical Oncology; FPZV, Federatie Palliatieve Zorg Vlaanderen; IKNL, Integraal Kankercentrum Nederland; KNMG, Royal Dutch Medical Association; OMC/SECPAL, Organización Médica Colegial/Sociedad Espanola De Cuidados Paliativos; PRCPEX, Programa Regional de Cuidados Paliativos de Extremadura; PSIB, Profesionales de la Salud de las Islas Baleares; SICP, Società Italiana di Cure Palliative.
Methodological quality evaluation
The analysis assessed the overall methodological quality according to the Appraisal of Guidelines for Research and Evaluation (Appraisal Guideline Research and Evaluation [AGREE] II), 9 which is a validated international tool designed to assess guidelines developed by local, regional, national or international groups, or affiliated governmental organizations. 9 It consists of 23 key items organized in six domains: scope and purpose, stakeholder involvement, rigor of development, clarity of presentation, applicability, and editorial independence.8,9
The key items are followed by one global rating item about the general quality of the guideline (overall assessment). 9 All items are rated on a seven-point Likert scale ranging from “1” where the required information was absent to “7” where the criteria were fully satisfied. 8 The results are shown as a percentage score for each domain and as an overall score ranging from one to seven points.
Appraisers
Two of the guidelines7,13 were written in English and two14,15 had an official English translation. The Italian guideline 16 was translated into English by an Italian speaking researcher of the German team (H.B.). The Spanish17–19 and Flemish 20 guidelines were translated by researchers from the respective countries participating in the project (E.G. and M.V.d.E.). All information was entered into an Excel spreadsheet and evaluated independently by three researchers (S.M.S., B.J., and H.B.).
Results
One hundred twenty-four of the 208 clinicians asked completed the survey (response rate 60%) (Table 1), reporting between five and eight documents per country (44 different documents in total). Nine of the reported documents were eligible for analysis: two international,7,13 four national,14–17 and three regional.18–20 Figure 1 provides an overview of the study selection process and Table 2 an overview of the most cited documents per country. Some of the guidelines were cited by experts from several countries: The EAPC framework 7 by experts from five countries, the European Society for Medical oncology (ESMO) 13 and the Royal Dutch Medical Association (KNMG) 14 guidelines by experts from four countries, and the Integraal Kankercentrum Nederland (IKNL) 15 by experts from two countries, respectively. Four countries (Germany, Hungary, Romania, and United Kingdom) did not have national/regional guidelines and nominated one of the above-mentioned international guidelines.
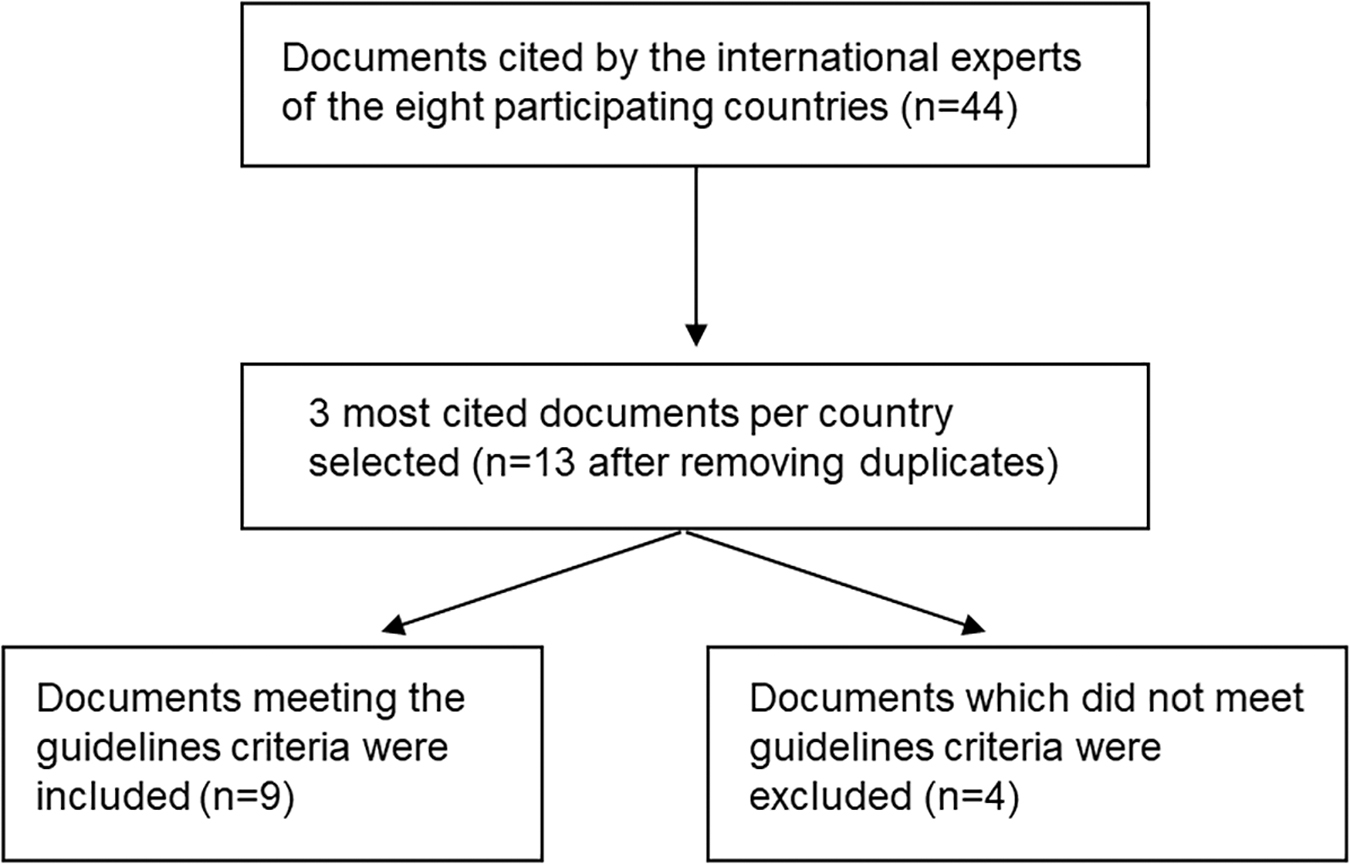
Flowchart of the study selection process.
Main characteristics of the guidelines
Published between 2007 and 2015, they were issued from European umbrella organizations in various fields in medicine, palliative and hospice care,7,13 national medical associations involved in palliative care,14–17,20 and governmental organizations.18,19 Two guidelines7,18 were developed using a systematic literature review and a consensus method and four13,15,16,19 used a consensus-based method within the group of authors, while another 14 used a nonsystematic literature review and comments from individuals and organizations. In two of them,17,20 the method was not reported. Only one 7 mentioned an external review (Table 4).
Characteristics of the Guidelines
Terminology
Palliative sedation
Most guidelines use the term “palliative sedation.” The EAPC framework 7 uses both terms “palliative sedation” and “therapeutic sedation.” The Italian guideline 16 is the only one to use the combination of terms “terminal sedation/palliative sedation,” stressing that the term terminal refers to extremely limited prognosis. Its 16 content, as well as the content of the Balearics guidelines, 19 is limited to continuous palliative sedation.
Palliative sedation is defined as an intervention aiming at alleviation of intolerable suffering, resulting from one or a combination of symptoms. It is an intervention that is monitored7,13 and deliberate,14–18,20 differentiating it from situations where sedation is a side effect of treatment. It can take place at the patient's home,7,13–19 in a hospital,7,13–19 in an inpatient hospice,7,14–16,18 or in a nursing home.14–16,19
Palliative sedation can be light or deep, as well as intermittent or continuous. Some guidelines14,15 define continuous sedation more narrowly as continuous sedation until death. Most guidelines7,13–18,20 insist that medication must be administered proportionally. Therefore, the level of sedation should be as low as necessary to provide as much needed relief from suffering. In most definitions,13–16,18–20 the criterion of closeness to death is included, while in the EAPC framework, 7 this is only relevant for cases of continuous deep sedation.7,21
Some guidelines7,13–16,18 stress that the aim of palliative sedation is not to shorten the patient's life and thus to clearly distinguish it from euthanasia, particularly in terms of aim, means, and timing, with continuous deep palliative sedation being reserved for the terminal phase of life.14,16
Refractory symptom
Refractoriness of a symptom is characterized by the absence of conventional modes of treatment, which are sufficiently effective or timely enough, and/or these modes of treatment are accompanied by unacceptable side effects.7,13–16,20 The most frequently mentioned indications include agitated delirium, dyspnea, pain, and convulsions. 7 Five guidelines7,13,14,16,17 specify, among the refractory symptoms, the special case of emergency sedation to provide rapid relief of overwhelming symptoms in dying patients.
Two guidelines14,15 define refractoriness as being applicable not only to a single symptom but also to a cluster of symptoms, whose combination leads to a condition that the patient finds intolerable.
The ESMO guideline 13 describes that the physician alone can determine the refractoriness of the symptom. The Dutch guidelines14,15 explicitly mention the patient's input of intolerability and emphasize that the physician should determine whether a symptom is treatable or not, the patient's intolerability of its intensity, and the physician and the patient should jointly decide whether the symptom is refractory.
Regarding refractory psychological symptoms and existential distress, three guidelines14,15,20 mention them as part of an indication, pointing out that they usually occur in context of physical decline and are therefore never isolated. Four guidelines7,13,16,18 mention them as a possible indication in themselves. Two17,19 of the three Spanish guidelines recognize severe anxiety as an indication, although do not mention specifically existential suffering.
Some guidelines7,13,16 recognize that it is much more difficult to assess the refractoriness of psychological or existential distress because of its nature, its dynamic and idiosyncratic course, the paucity of standard assessment methods, and its possible onset at earlier stages of the disease and place this indication under a special rubric, making specific recommendations7,13 such as starting with respite sedation for 6–24 hours and considering continuous deep sedation only after repeated trials of respite sedation. Most of the guidelines7,13–16 recommend that psychosocial and spiritual expertise, as well as the participation of relatives and carers, be sought during the assessment.
Life expectancy
There are significant differences between guidelines regarding references to life expectancy. For some,13,17 this is vaguely described as “end of life,” while others provide ranges from a few hours to a few days7,16,19,20 or two weeks before death14,15,18 as precondition for continuous deep palliative sedation.
The Dutch guidelines14,15 are the only ones to provide guidance for the sensitive situation in which a patient with refractory symptom(s), but with a life expectancy of more than two weeks, decides on their own to stop taking fluids or having them administered. They state this may lead to a decision to initiate deep and continuous sedation provided that the patient's decision to stop drinking or discontinue artificial hydration precedes the decision to apply palliative sedation, and that a time interval between the two be present to judge the consistency of the patient's wish. 14
Intermittent sedation may be used earlier to provide, for example, transient relief, while awaiting the benefits of another therapeutic approach.7,13,14
Preemptive discussion
The three Spanish guidelines17–19 do not mention the need for a preemptive discussion about the potential role of sedation in end-of-life care and contingency planning. The guidelines7,13–16,20 that do recommend it, state that this discussion should be offered early enough in the palliative course of the disease7,13–16,20 and repeatedly,7,13,16 thus allowing patients to express their anxieties 16 and to discuss their end-of-life care preferences and the measures to be taken in the event of severe distress.7,13–16,20
When the patient agrees, 13 the presence of significant family members during these discussions is recommended.7,13,16 This may help to reduce the patient's anxiety16,20 and that of their family (carers), and may possibly facilitate future grieving. 16
Evaluation and consultation procedures
All guidelines stress that, except in cases of acute and unpredictable distress, 15 the medical rationale for sedation and the decision-making process should be based on input from a multidisciplinary team, the patient, and family members, rather than by the attending physician alone.
They differ regarding the expertise of the person(s) responsible for assessment and decision making. Four7,13,18,19 describe that responsibility falls to a physician or health care team with sufficient experience and expertise in palliative care. Four other guidelines15–17,20 strongly recommend the consultation of a palliative care team. This contrasts with the position of the KNMG guideline, 14 which mainly recommends the recourse to an expert if the attending physician has doubts about their own expertise or experiences difficulties in deciding to initiate palliative sedation due to the complexity of a situation.
Informed consent
All guidelines refer to informed consent as a requirement for palliative sedation. The health care team should therefore actively discuss the option of palliative sedation while the patient still has mental capacity, and thus in advance of the stage when it remains the only available option. 14
Guidelines emphasize the need to involve the family members in this discussion to help them accept the patient's wishes, to understand the reasons why the health care team proposes this therapeutic approach, and to inform them both about the procedure and the consequences, such as partial or complete loss of communication possibilities with the patient.
When the patient lacks decision-making capacity, advance directives should be consulted. In the absence of advance directive covering the use of palliative sedation, the consent of the patient's legal representative must be sought.7,13–20 The nature of this consent varies. In two guidelines,7,16 the decision must be made by the attending physician using the indications given by the patient's representative, based on the patient's presumed preferences. In three guidelines,13–15 it is the patient's representative who makes the decision on behalf of the patient, but they may elect to leave the decision to the physician.
The Flemish guideline 20 recommends that a consensus be reached between the health care team and the legal representative. The three Spanish guidelines17–19 do not describe the nature of the consent. In the absence of advance directives and a legal representative, five guidelines7,13–16 recommend that the health care team act according to the best interest of the patient and one guideline 19 that the assessment should be carried out by two different physicians.
Selection of the sedation method
Palliative sedation should be applied proportionally.7,13–18,20 Intermittent or light sedation should be generally attempted first.7,13–16,18 This is even more relevant when death is not expected in the near future. 14 Deeper sedation should be adopted when light sedation has been ineffective,7,13,16 or as a first-line approach when the suffering is intense and definitely refractory, death is expected within hours or a few days and the patient wishes it explicitly, or in an end-of-life catastrophic event.7,13,16
Dose titration
Two guidelines14,15 recommend a stepwise approach with a gradual increase in doses.
All guidelines recognize midazolam as the medication of choice for palliative sedation because of its short half-life. In case of midazolam failure, the addition of a neuroleptic (preferably levomepromazine) should be considered as a second step.14,15,17
Levomepromazine has been recommended as a first-line indication only in situations of refractory delirium,14,15,17,19 history of alcoholism, 19 and/or drug dependency. 19 In rare cases where the desired effect cannot be achieved, the administration of midazolam and levomepromazine should be discontinued. A third step with propofol is recommended,14,15,17–19 which should only be performed in a hospital environment and under the supervision of a person experienced in its use.14–17,20
The use of phenobarbital for this third step is controversial, while recommended by some guidelines,7,13,16–19 others14,15 have removed it from their recommendations because of the difficulty in obtaining it and the difficulty in dissolving it for parenteral administration.
Other benzodiazepines (diazepam,14–16 lorazepam,7,13–16 clonazepam,14–16 and flunitrazepam7,13,16) and neuroleptics (chlorpromazine,7,13,16–18 clotiapine, 20 and promethazine 16 ) have been mentioned as potential alternatives to midazolam and levomepromazine.
Finally, three guidelines14–16 consider the use of morphine as a sedative to be bad practice. Morphine should be administered or continued (alongside sedatives) solely for the relief of pain or dyspnea.
Patient monitoring and care
Four guidelines7,14,15,19 emphasize the importance for the physician to be present during the initiation of palliative sedation.
The patient should be evaluated at least every 20 minutes during the initiation phase until adequate sedation is achieved.7,16 Recommendations for assessment intervals thereafter vary from once14,15 to thrice per day.7,16
The evaluation must consider the following:
The degree of suffering,7,13–15,18,19 using an appropriate tool such as the Critical-Care Pain Observation Tool (CCPOT).
7
The level of consciousness with the purpose to alert if the sedation is too superficial or too deep.14,15 Different scales are proposed: RASS-PAL-scale,
7
Sedation scoring system,14,15 Rudkin scale,
16
and Ramsay scale.17–19
The possible adverse effects such as agitation, , or aspiration.7,14 The occurrence of new symptoms or complications such as bed sores or urinary retention.14,15
As for the parameters to be monitored, all guidelines describing the monitoring7,13–16 distinguish between intermittent and continuous sedation. For the former, the aim being to restore the previous level of consciousness later, all efforts should be made to preserve physiological stability. The occurrence of respiratory depression should lead to a reduction in medication doses and in the event of a life-threatening situation, careful administration of flumazenil as a benzodiazepine antagonist may be indicated. 7
As the purpose of continuous sedation is to ensure the comfort of the dying patient, the only parameters to be evaluated are those related to their comfort.7,13,15 Respiratory rate may be assessed to ensure the absence of tachypnea or respiratory distress, bearing in mind that in a dying patient, respiratory deterioration is to be expected and should not necessarily lead to a decrease in sedation. 7
Hydration and nutrition
Five guidelines7,13–15,20 provide guidance for decisions regarding hydration and nutrition. All agree that these decisions must be independent of the sedation decision, but there is some divergence in their approaches.
Three guidelines14,15,20 point out that since proximity to death (within two weeks) is a precondition for the use of continuous palliative sedation, administration of fluids is considered medically futile in such cases.14,15 Besides, they state that continuous palliative sedation is administered mostly in patients who are no longer able or willing to take in fluids because they are dying of the underlying disease. Consequently, the life-shortening effect of discontinuing artificial hydration is said to be virtually nil. They argue that stopping artificial hydration does not imply “additional suffering,” as the patient's suffering is removed by sedation, and maintaining artificial hydration would prolong suffering and exacerbate it by increasing edema, ascites, bronchial secretions, urine production, and incontinence.
In contrast, EAPC 7 and ESMO 13 request health care providers, patients, and families to reach a consensus based on the best interests of the patient.
Concomitant medications
Medications and nursing procedures (e.g., mouth care) used for symptom control before sedation should be continued during sedation unless they are ineffective or produce distressing side effects.7,13–17,19 Nursing procedures and medications that are either incompatible with or unrelated to the patient's comfort goal should be generally discontinued during continuous sedation.7,14,15 The mode of administration of the medications, particularly oral, should be modified if necessary.14–16
The care and informational needs of the patient's family
The role of the family at the end of life of a patient is extremely difficult and complex. In addition to their role as relatives, they acquire the role of informal caregivers, observers, and informants, and the status of patient's representative, once the patient has been rendered incompetent as a result of the sedation.14,15 All guidelines agree that caring for the family is an integral part of the act of care during palliative sedation.
Care for the health care professionals
As the decision to apply palliative sedation is significant and potentially burdening, six guidelines7,13–16,20 highlight the importance of caring for the professional team during this procedure.
Methodological quality evaluation using the AGREE II instrument
Altogether, the EAPC framework 7 and the KNMG guidelines 14 received the best scores (four points out of the seven maximum attainable points) (Table 5). The domain scope and purpose received the highest ratings, with all guidelines scoring above 50%, four7,14,16,19 of them scoring above 80%. Stakeholder involvement had low scores, ranging from 24% to 67%. Clarity of presentation scored more than 50% for 5/9 guidelines and Applicability ranged from 0% to 25%, while editorial independence scored 0% since the potential influence of any funding body or competing interests were not discussed by any of the guidelines.
Quality Evaluation Using the Appraisal Guideline Research and Evaluation II Instrument
1. 
2. Overall score (on a scale of 1–7; 1 = lowest and 7 = highest) corresponds to the overall assessment given by the appraisers on the quality of guidelines, taking into account the appraisal items considered in the assessment process.
Most of the guidelines had low scores for rigor of development (range 0–40%) because of the absence of a systematic research method or its description, the lack of description of the criteria for selecting the evidence, the scarcity of clinical studies in the field of palliative sedation, and the lack of an external review.
Discussion
Main results of the study
The global content of these nine European guidelines is broadly similar and consistent with the EAPC framework. 7 However, some issues will need to be discussed when updating the framework. 7
The criterion of refractoriness remains complex due to the subjective nature of suffering and the absence of objective assessment scales. This is particularly true when the suffering is primarily psychological and/or existential. Although most guidelines recognize these as (part of) an indication, most make specific recommendations regarding their assessment and the mode of administration of sedation.
The guidelines differ as to the role of the patient in determining the refractoriness of the symptom(s), with some14,15 placing greater emphasis on the patient's role than others.
The requirement for palliative care expertise of the physician and/or the health care team determining refractoriness, or their consultation is stressed in most guidelines to avoid the administration of palliative sedation, while there are therapeutic alternatives. This raises the issue of patient access to palliative sedation in areas where there is a possible shortage of physicians and/or care teams specialized in palliative care.
Furthermore, there is a broad consensus on the need for a multidisciplinary team approach, which suggests, as Schildmann and Schildmann 21 have already pointed out, that the decision on palliative sedation is more than a medical decision and has ethical and societal dimensions. Nevertheless, there is still a lack of clarity on how the team and relatives should be involved in the decision-making process.
All guidelines mention a more or less precisely defined criterion of life expectancy for the administration of continuous palliative sedation, but the Dutch guidelines14,15 are the only ones to provide guidance for situations in which patients with refractory symptoms and a life expectancy of more than two weeks decide to limit their life expectancy by voluntarily stopping eating and drinking.
All guidelines agree that the decision to continue or discontinue hydration and nutrition should be independent of the decision to use palliative sedation. However, there is no clear consensus on the decision-making process.
The three Spanish guidelines17–19 do not mention the need for preemptive discussion on the potential role of sedation in end-of-life care, thus highlighting a possible reluctance to address this topic early in the palliative course of the disease. The revision of the EAPC framework 7 should consider to what extent and in what way these cultural differences should be reflected.
In 2017, Abarshi et al. 8 analyzed 13 guidelines on palliative sedation from European and non-European countries. As our study was limited to European guidelines, only four13,14,16,18 of these are included in our study; our survey also identified four other European guidelines.15,17,19,20 Our AGREE II analysis confirms the evaluation of Abarshi et al., 8 highlighting methodological weaknesses and urging the need for improvement in four of the domains (stakeholder involvement, rigor of development, clarity of presentation, and applicability), which should be considered when updating the EAPC framework. 7
Strengths and limitations
This guideline analysis has some limitations. One of these is the limited number of European countries in which experts were asked. We are aware that relevant guidelines may be missing for a complete overview of European guidelines. However, the analysis provided strong indications for areas of improvement of the EAPC guideline, which is planned to be updated and rated in a Delphi consensus procedure with the participation of experts from a larger number of European countries. The selection of experts was also not representative of health care workers in the countries, and therefore generalizability is not possible.
Furthermore, some information, for example on editorial independence, potential influence of the funding body, or competing interests, may have been omitted because it was rendered self-evident by the authors of the guidelines. Finally, palliative sedation with its medical, ethical, and societal dimensions is complex and the AGREE II instrument may not be completely suitable for this kind of analysis.
Conclusion
This review highlights points of agreement and persistent debates across European countries regarding palliative sedation, such as how refractory symptoms should be determined, ethical issues related to the decision-making process regarding hydration, or the expertise required by the attending physician/health care team. These highlighted points will inform the discussion on the planned revision of the EAPC framework 7 on palliative sedation.
This review also underlines methodological weaknesses, particularly in terms of rigor of development and applicability. For any update or revision of the analyzed guidelines, the weaknesses mentioned above should be considered to improve the quality of their content and the applicability of their recommendations.
Footnotes
Authors' Contributions
Conception and design of the review: S.M.S., H.B., C.C., E.G., B.J., and L.R. Translation and data extraction: S.M.S., H.B., E.G., M.R., and M.V.d.E. Data analysis: S.M.S., H.B., and B.J., and S.M.S. led the writing of the script. All authors provided feedback on one or more versions of the review and contributed to the development of the article. All authors revised and approved the final article.
Acknowledgments
We acknowledge all H2020 PalliativeSedation (SEP-210506914) consortium members for their input: KU Leuven (Belgium), University of Pécs (Hungary), La Maddalena Cancer Center (Italy), Radboud University Medical Center, Nijmegen (Netherlands), Hospice Casa Sperantei (Romania), University of Navarra (Spain), Lancaster University (United Kingdom), European Cancer Patient Coalition (ECPC), and European Association of Palliative Care (EAPC) (![]() ).
).
Poster review in “Abstracts from the 12th World Research Congress of the European Association for Palliative Care”: Surges S, Garralda E, Jaspers B, Brunsch H, Rijpstra M, Hasselaar J, Van der Elst M, Menten J, Csikós Á, Mercadante S, Mosoiu D, Payne S, Centeno C, and Radbruch L. Review of European Guidelines on Palliative Sedation: A Working Base for the Updating of the EAPC Framework. Abstracts from the 12th World Research Congress of the European Association for Palliative Care. Palliative Medicine 2022; 36(1):3–122; (p. 85; abstract ID: P03:58), article first published online: May 10, 2022; Issue published: July 1, 2022; DOI:10.1177/02692163221093145
Data Management and Sharing
![]() (study protocol
12
) are publicly deposited.
(study protocol
12
) are publicly deposited.
Funding Information
This project received funding from the European Union's Horizon 2020 research and innovation program under Grant Agreement No. 825700.
Author Disclosure Statement
No competing financial interests exist.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
