Abstract
Abstract
Introduction:
As health care systems strive to meet the growing needs of seriously ill patients with high symptom burden and functional limitations, they need evidence about how best to deliver home-based palliative care (HBPC). We compare a standard HBPC model that includes routine home visits by nurses and prescribing clinicians with a tech-supported model that aims to promote timely interprofessional team coordination using video consultation with the prescribing clinician while the nurse is in the patient's home. We hypothesize that tech-supported HBPC will be no worse compared with standard HBPC.
Methods:
This study is a pragmatic, cluster randomized noninferiority trial conducted across 14 Kaiser Permanente sites in Southern California and the Pacific Northwest. Registered nurses (n = 102) were randomized to the two models so that approximately half of the participating patient-caregiver dyads will be in each study arm. Adult English or Spanish-speaking patients (estimate 10,000) with any serious illness and a survival prognosis of 1-2 years and their caregivers (estimate 4800) are being recruited to the HomePal study over ∼2.5 years. The primary patient outcomes are symptom improvement at one month and days spent at home. The primary caregiver outcome is perception of preparedness for caregiving.
Study Implementation—Challenges and Contributions:
During implementation we had to balance the rigors of conducting a clinical trial with pragmatic realities to ensure responsiveness to culture, structures, workforce, workflows of existing programs across multiple sites, and emerging policy and regulatory changes. We built close partnerships with stakeholders across multiple representative groups to define the comparators, prioritize and refine measures and study conduct, and optimize rigor in our analytical approaches. We have also incorporated extensive fidelity monitoring, mixed-method implementation evaluations, and early planning for dissemination to anticipate and address challenges longitudinally. Trial Registration: ClinicalTrials.gov: NCT#03694431.
Introduction
Background
Multiple evidence-based guidelines1-3 affirm the need to provide palliative care (PC) services across the care continuum to alleviate unnecessary suffering and improve quality of life for both patients and caregivers. For patients with a high symptom burden and significant functional limitations that make current clinic-based PC models unsuitable, 4 telephone-based support inadequate, 5 and hospice enrollment not yet tenable, home-based palliative care (HBPC) may be the only viable option. The decisional dilemma for health care systems and clinicians lies in how best to optimize delivery of high-impact HBPC services that meet the growing demand for such care while also remaining affordable for all payers including patients, caregivers, employers, public purchasers, and health systems.
To address this, our study compares a standard HBPC model that includes routine home visits by a nurse and prescribing clinicians (physicians and nurse practitioners) with a more efficient tech-supported HBPC model that aims to promote timely interprofessional team coordination through synchronous video consultation with the prescribing clinician while the nurse is in the patient's home. If the more efficient tech-supported model yields short-term reductions in symptom burden and helps patients spend more time at home that is comparable with standard HBPC, this could lead to redefining PC practice and policy for dramatically expanded, affordable access to HBPC. The purpose of this invited article was to describe the study protocol, stakeholder engagement efforts before and during study start-up, and early implementation challenges and learnings.
Objectives and hypotheses
Aim 1
Determine if tech-supported HBPC is noninferior to standard HBPC as measured by the co-primary patient-and-family-centered outcomes of symptom improvement (short term) and days at home in the last 180 days of life (long term). We will also measure several secondary outcomes: quality of life, care experience-satisfaction, acute care utilization, and survival.
Hypothesis
People with serious illness who receive tech-supported HBPC will have short-term, comparable improvements in symptoms from baseline to one month and will spend a similar number of days at home in their last six months of life compared with standard HBPC. Secondary outcomes are also expected to be similar between care models.
Aim 2
Compare the effects of tech-supported HBPC and standard HBPC on the primary outcome of caregiver perception of preparedness for caregiving and secondary outcomes of quality of life, anxiety-depression, caregiving burden, care experience-satisfaction, and health care utilization (for those caregivers who give permission to access their utilization data from the health system).
Hypothesis
Caregivers receiving standard and tech-supported HBPC will report similar perceptions of preparedness for caregiving.
Aim 3
Examine the effectiveness of HBPC across several subgroups, for example, cancer, organ failure, neurodegenerative diseases, multiple chronic conditions (three or more diseases/conditions), participation of a caregiver in the patient's care, racial/ethnic minorities, and low income.
Aim 4
Describe the longitudinal pattern of PC service exposure, intensity of acute health service use, and quality of end-of-life care in patients with serious illness who have been referred for HBPC services but did not enter the HBPC pragmatic trial (passive population cohort).
Aim 5
Describe barriers and facilitators to implementation of tech-supported HBPC.
Methods
Study design
Setting
Two Kaiser Permanente regions (Southern California [KPSC] and Oregon/Washington [KPNW]).
Timing
This five-year study started on January 1, 2018. The protocol, operating procedures, systems, tools, workflows, and trainings were completed in the first year. Participant enrollment commenced on January 7, 2019 with 14 sites launched in a staggered manner through February 4, 2019 and will extend through June 2021.
Population
English and Spanish-speaking patients (target enrollment 10,000) with a serious illness and prognosis of one to two years who have a high symptom burden and significant functional limitations that precludes clinic-based care and their caregivers (estimate 4800).
Design
This is a pragmatic, noninferiority cluster randomized trial to compare tech-supported HBPC with standard HBPC at 14 sites in two Kaiser Permanente regions that have existing HBPC programs. Registered nurse (RN) case managers across these sites were randomized (stratified by site) to tech-supported or standard HBPC so that approximately half the patient-caregiver dyads receive routine in-home nurse and prescribing clinician visits (standard HBPC; patients: 5000/caregivers: 2400) and the other half receive nurse visits coupled with remote prescribing clinician consultation-supervision (tech-supported HBPC; patients: 5000/caregivers: 2400) (Fig. 1). Features of the study align with the pragmatic-explanatory continuum indicator summary (PRECIS) guidance for pragmatic trials (Table 1).6,7
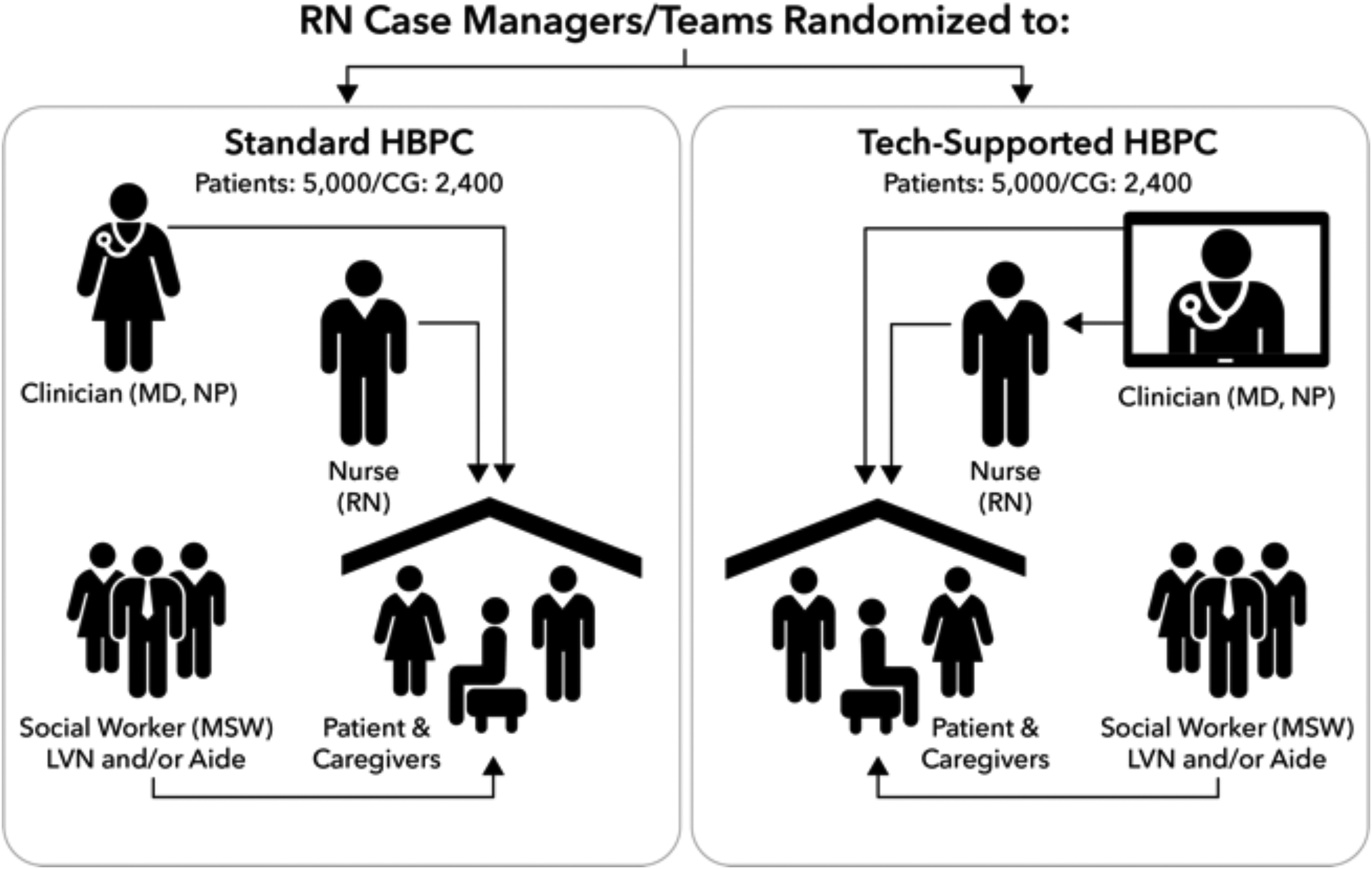
HomePal study design.
PRECIS Criteria for Pragmatic Trials
EMR, electronic medical record; HBPC, home-based palliative care; PC, palliative care.
We are also conducting a parallel observational cohort study to passively examine through electronic medical record (EMR) information, the patterns of PC service exposure, and overall health service use for all patients who have been referred for HBPC services but did not enter the trial.
Randomization
RN case managers (n = 102) who are full or part-time employees are the unit of randomization. The randomization scheme was stratified by geographical regions within the 14 clinical sites.
HBPC comparators
Consistent with standards from the National Consensus Project for Quality Palliative Care8,9 and models of interdisciplinary PC,4,5,10,11 HBPC services across the two comparators are encouraged to adhere to best practices addressing bio-psycho-social-spiritual needs and providing comprehensive PC support regardless of delivery method. The team may assume a co-management, primary, or mixed role. Patients remain on HBPC if the service continues to meet patients' needs. Both delivery models include biweekly interdisciplinary group (IDG) meetings to review current cases and discuss progress toward meeting patients' goals (Table 2).
Standard and Tech-Supported Home-Based Palliative Care Intervention Components
LVN, licensed vocational nursing; MD, medical doctor; NP, nurse practioner; RN, registered nurse; SW, social worker.
To ensure a level of standardization across HBPC teams regardless of treatment arm, the health system required all HBPC team members to complete the CSU San Marcos Palliative Care Institute online PC essentials training before study launch. The HBPC staff completed the modules independently or in a group setting, coupled with debriefing sessions. Prescribing clinicians were encouraged to complete a list of recommended modules available from the Center to Advance Palliative Care. Our workforce training efforts are consistent with recommendations for conduct of pragmatic trials6,7 to ensure sustainability and to minimize operational impact. All HBPC clinicians will be expected to complete life care planning training by the end of 2019.
Standard HBPC
The current standard HBPC12,13 approach across our clinical sites is to utilize the RN case managers to initiate the evaluation and lead the team-based care. The RNs conduct the initial start of care visit in the patient's home. The prescribing clinician generally conducts a separate home visit up to 14 days later. The frequency of subsequent home visits is determined based on the care plan and emerging needs of the patient/caregiver.
Tech-supported HBPC
Tech-supported HBPC is also led by an RN case manager who conducts the initial start of care visit in the patient's home. The prescribing clinician participates in the visit remotely through video with the patient/caregivers while the RN is in the patient's home. We expect that early access to the prescribing clinician for discussion and concurrence on the treatment plan builds trust and confidence with the patient/caregivers, potentially reducing the need for an in-home prescribing clinician visit. The care teams may deviate from using video visits if it is clinically warranted or if the patient/caregiver declines. The teams use video visits at their discretion during follow-up touchpoints to address acute changes in condition, goals of care discussions, medication changes and review, recertification review, evaluation for transition off HBPC, or other uses primarily driven by the nurse in the home. The tech-supported clinicians have tablets and smartphones that are equipped with enterprise-wide secure, video-conferencing applications. We provided targeted training on interprofessional communications coupled with role play on use of the video application for the four-way nurse-patient-caregiver-prescribing clinician interaction before study launch.
Outcomes measures
We based our selection and timing of outcome measures for patients and caregivers in the literature, considering the hypothesized short and longer term impact of HBPC, and iterative feedback on what matters most from our diverse stakeholders. We also considered measure responsiveness, burden, likelihood that measures will be used in routine care, and harmonization with other Patient-Centered Outcomes Research Institute (PCORI)-funded PC studies (see Table 3 for a full summary).
Summary of Data Collection Schedule
Process measures are captured from the EMR: HBPC services include in-home visits (provider, RN, SW, and other ancillary staff), virtual encounters (telephone or video), and advice calls during nonbusiness hours and weekends; outpatient PC services includes clinic visits and virtual encounters (telephone or video); inpatient PC services; hospice enrollment and length of stay; use of physician aid in dying.
Can be completed within seven days of HBPC admission visit if not completed previously.
Health care utilization includes: all-cause hospitalizations, observational stays, emergency department visits, skilled nursing facility (SNF) days, home health days, and use of outpatient services (primary and specialty care).
If patient is deceased, we will try to obtain these two measures from the caregiver.
DCC, obtained through telephone by the Data Coordinating Center; RN/SW, per routine care if available via EMR; RS, research staff (obtained by telephone).
Patients
We are measuring symptom burden with the total Edmonton Symptom Assessment Scale (ESAS) score.14,15 The ESAS measures physical and emotional symptoms and overall well-being using a 0 to 10 point scale; financial distress and spiritual pain are also measured but not included in the total score. Days at home is a co-primary measure.16,17
Caregivers
Caregivers have told us that feeling well prepared in caring for their loved one is what matters most and this measure closely relates to the education, skills training, and support they would receive from the HBPC team; thus we are using the eight-item Preparedness for Caregiving Scale.18,19
Recruitment and Study Procedures
Eligibility criteria
Patients
Trial cohort
Given the pragmatic real-world study design, the target patient population is broadly inclusive of all health plan members who are 18 years and older, of all race and ethnicities, English and Spanish speakers who are enrolled into HBPC including those with nonmalignant (e.g., heart failure, chronic obstructive pulmonary disease (COPD), end-stage kidney or liver disease, and neurodegenerative diseases) and malignant serious illness20-23 who have an expected survival of one to two years, and are homebound.
Caregivers
Trial cohort
We identify adult family or other caregivers (at least 18 years old) during the telephone screening/intake as someone “who helps you with your care.” We are excluding professional caregivers who are hourly wage employees; we estimate ∼80% of patients will have a caregiver and 60% of those caregivers will agree to participate.
Patients
Observational cohort
We are tracking patients who are referred to HBPC but who are not immediately enrolled in the trial through their EMR-based data through the end of the study or until they enroll in the trial. The observational cohort also includes non-English and non-Spanish speakers.
Recruitment
Because of minimal risks expected with study activities, we have a waiver of written consent to administer surveys and a waiver of consent and HIPAA to passively collect EMR-based data for all patients unless patients explicitly request to opt out of the study. Patients/caregivers also receive information letter that describes the study activities and how to opt out in their HBPC program packet.
We are identifying patients from either routine physician-initiated referrals or a population-based proactive case finding process. As per routine workflows, HBPC referrals are routed to the respective clinical sites where administrative and clinical staff determine programmatic and insurance eligibility. After the administrative and clinical staff complete their review and are scheduling the start of care home visit, research staff initiate recruitment calls to obtain verbal consent from the patient and designated caregiver and to complete the baseline assessments. Clinical programs that are expanding to proactive outreach may additionally identify eligible patients from an existing health system risk stratification model that uses EMR data to flag frail patients with an expected prognosis of one to two years. 24 Patients and caregivers are considered enrolled in the trial once they provide consent, complete the baseline surveys, and the patient is admitted to HBPC.
Study procedures
Figure 2 summarizes the overall participant flow and Figures 3 and 4 are the CONSORT diagrams for patients and caregivers, respectively. Recognizing that survey completion can be challenging in this population, we have prioritized collecting the ESAS to capture patient symptom severity followed by the distress thermometer, Caregiver Preparedness Scale, and then other secondary surveys. Administering all surveys takes ∼30 to 40 minutes. Surveys can be completed on more than one call within a seven-day window of start of care. Within two to five days of the clinical referral, either a standard or a tech-supported RN makes a start of care home visit. If patients are not initially admitted to HBPC, they are not included in the trial cohort. However, they are eligible to join the trial if they are admitted to HBPC at a later point. Consistent with intent-to-treat (ITT) principles, we follow patients for at least 12 months regardless of service transitions (e.g., to hospice) or until death; research staff contact the patients/caregivers 1 and 6 months after the start of HBPC to administer the surveys as given in Table 3.
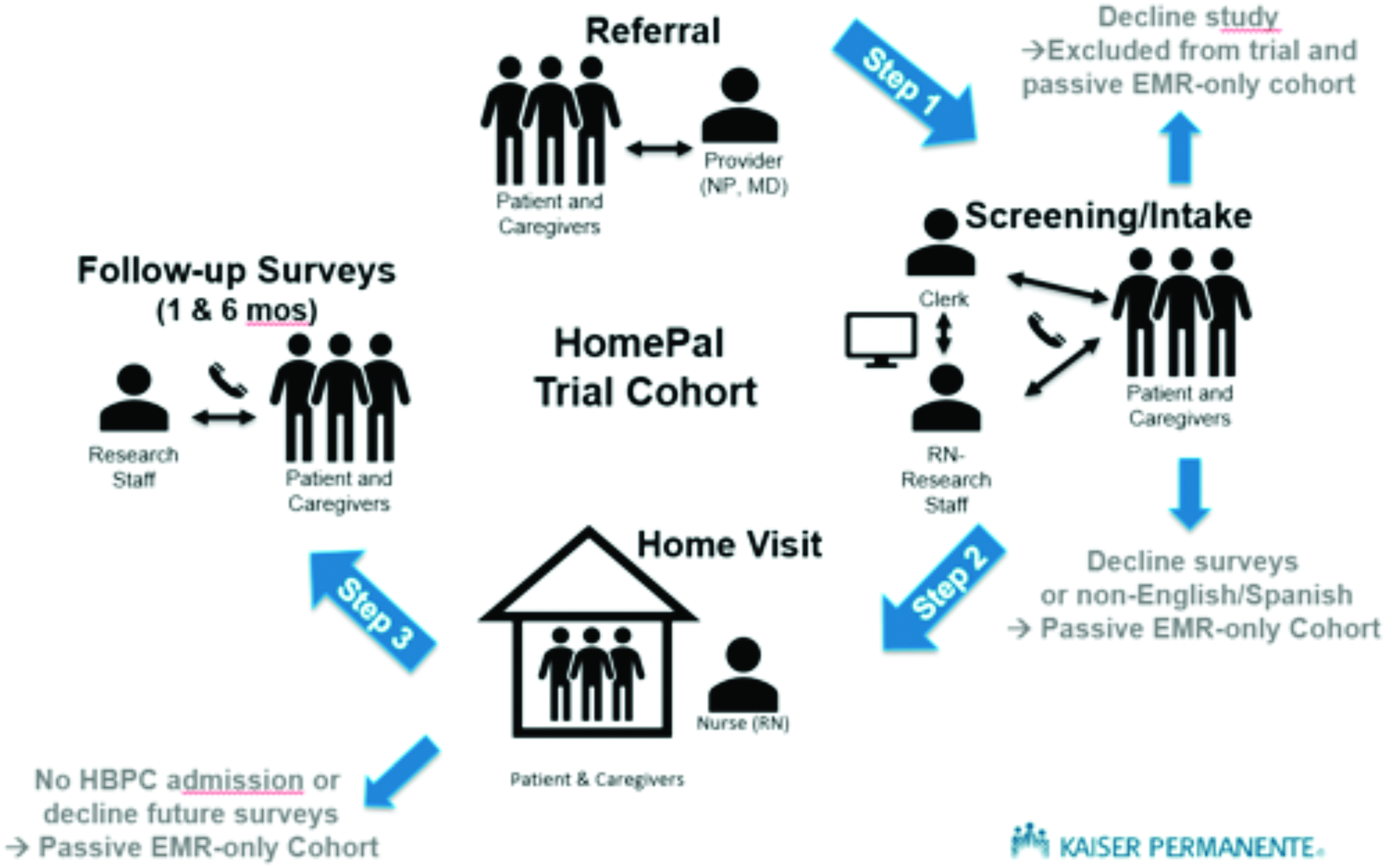
HomePal participant flow.
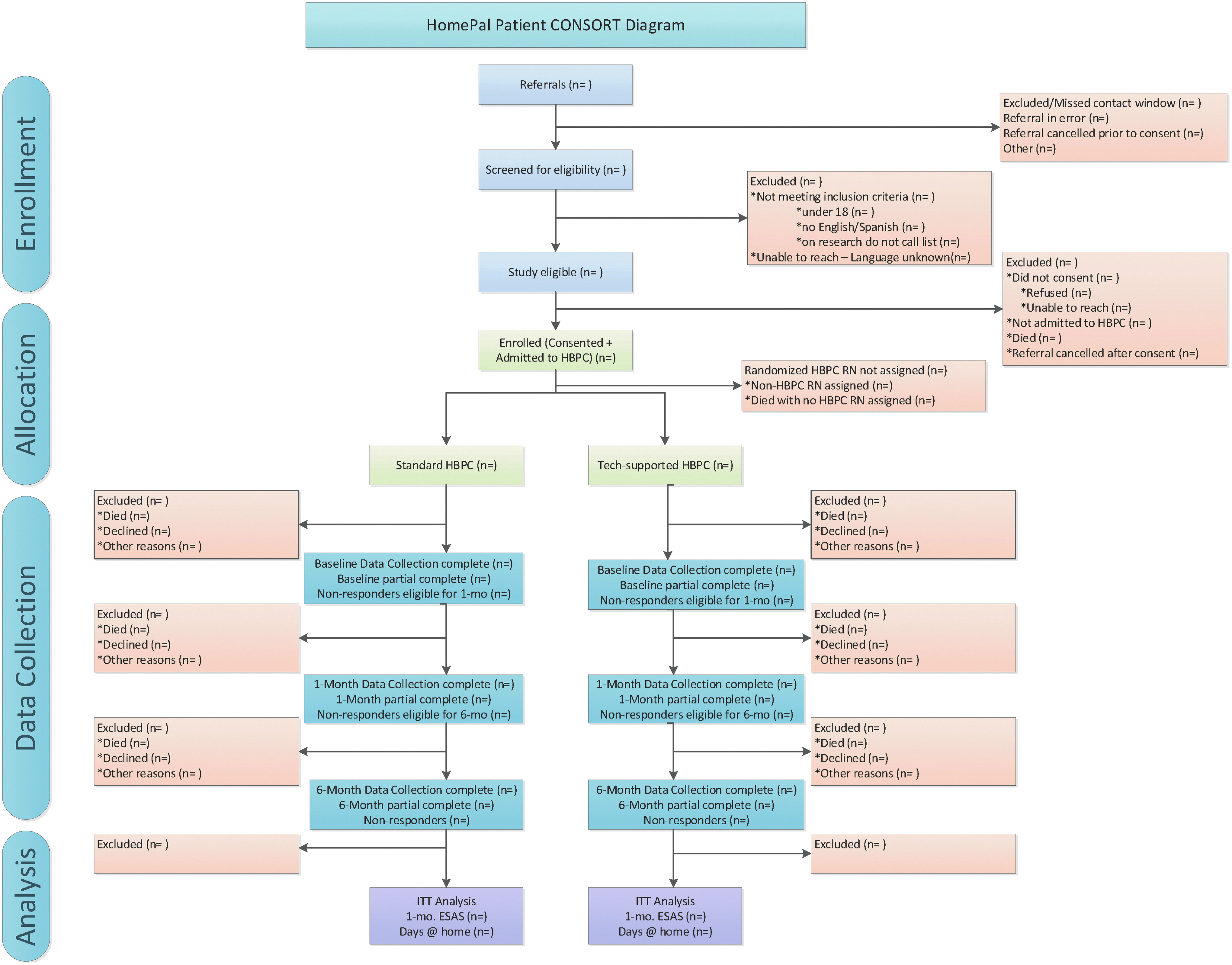
HomePal patient CONSORT diagram.
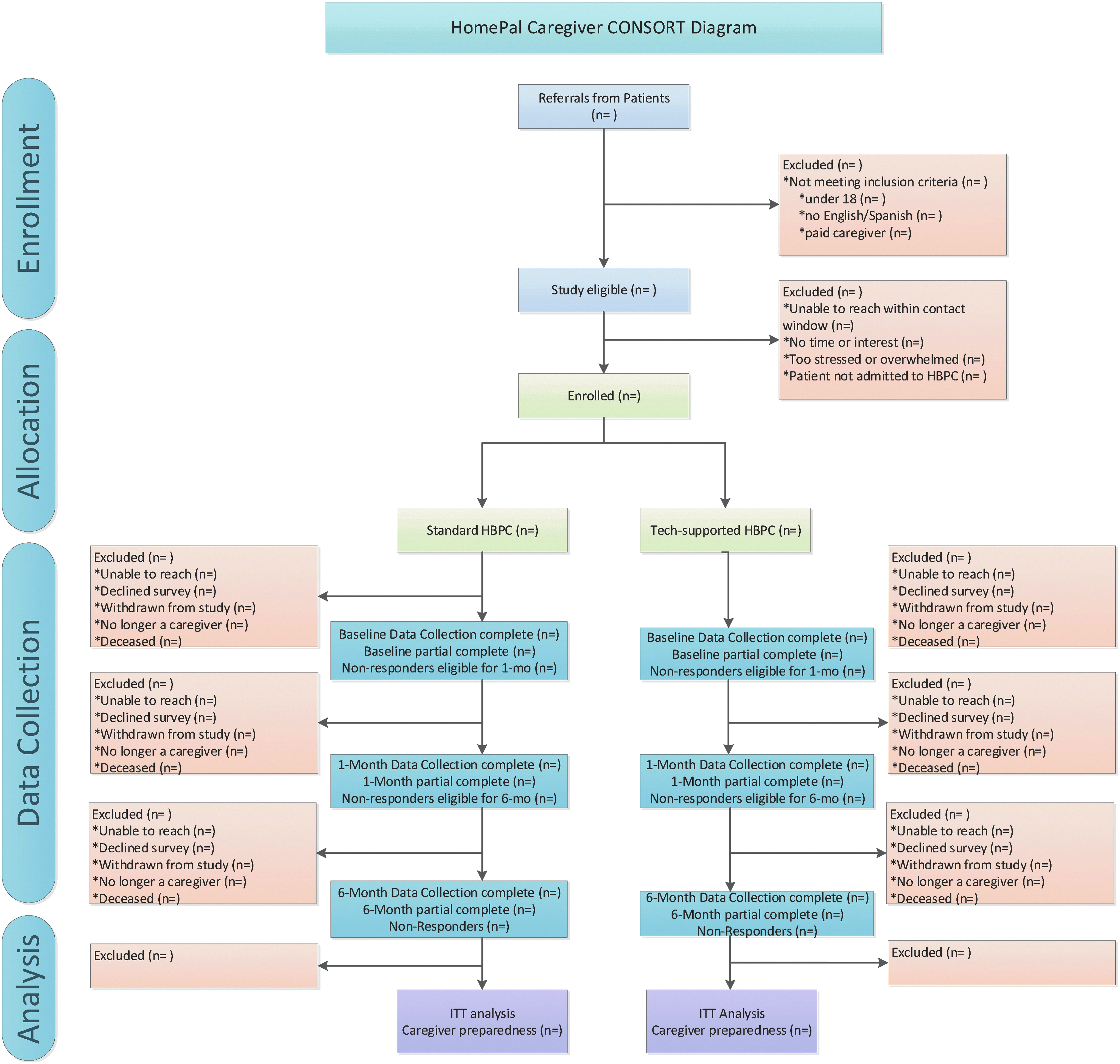
HomePal caregiver CONSORT diagram.
Qualitative assessment of HBPC implementation
To obtain formative evaluations and help with midcourse corrections, we are seeking feedback from tech-supported patients/caregivers and HBPC team members. We are inviting a small randomly selected sample of patients/caregivers to participate in 15-minute telephone interviews about their video visit experience after completion of their one- and six-month assessments. We will invite clinicians to complete annual web-based surveys of their experiences in implementing HBPC services. 25 We are also gathering data from embedded research staff participating in IDG meetings and through weekly feedback from multiple clinical stakeholders during operational meetings.
Data Management and Analysis Plan
Data management, quality control, and monitoring
The KP Northwest Center for Health Research Data Coordinating Center (DCC) is responsible for central coordination and management of study data. The DCC developed and maintains a central database integrating EMR and survey data from both regions and supports a centralized web-based electronic data capture system that tracks all aspects of participant flow and provides for remote data entry and management. The DCC uses rigorous quality control procedures to protect participant safety and confidentiality.
The data safety monitoring plan includes monitoring recruitment procedures and progress and potential adverse events, although minimal risks are expected with study activities. The data safety and monitoring board (DSMB) comprises a biostatistician and three subject matter experts and are meeting semi-annually to monitor, review, and assess study progress and safety data.
Power calculation
We relied on published minimal clinically important difference (MCID) criteria for the ESAS 26 and data on days at home in the last 180 days of life from a cohort of patients who received HBPC in KP to inform our power calculation for the primary study hypothesis of noninferiority between standard and tech-supported HBPC. For the primary short-term outcome of improvement in symptoms (ESAS score) from baseline to one month, assuming a noninferiority MCID margin of 3 points for a difference-in-differences (DID) comparison of change in total ESAS and within-patient change standard deviation (SD) of 11.6, and a conservative power of 0.90, type I error of 0.025, and intraclass correlation coefficients ranging from 0 to 0.05, we should be able to detect a noninferior absolute DID as large as 2.2 and as small as 1.3 with anticipated data from ∼8500 patients (15% attrition at one month).27,28 Similarly, for the primary long-term outcome of days at home in the last 180 days of life for patients who are followed for at least 180 days after starting HBPC, based on our historical data (mean = 165 days, SD = 24) we should be able to detect a noninferior absolute difference as large as 18.8 and as small as 3.2 for margins ranging from 7 to 21 days, expecting ∼50% (n = 5000) of patients across both groups to die over the study period.
Analytical plan
The analyses will follow an ITT strategy. Our analysis for the short-term outcome of improvement in symptom burden (ESAS) from baseline to one month will be an ITT DID model, which will compare the average within-patient change in symptom burden between the two HBPC groups in patients surviving for at least one month. We will include as covariate adjustments in a generalized linear mixed-effects regression model any baseline covariates for which the standardized difference between HBPC groups is >0.1, as well as relevant RN and site characteristics. To accommodate such covariates and possible clustering of patients-within-RNs-within-sites, we will analyze the DID by a linear mixed-effects regression model with RN- and possibly RN-within-site-level random intercepts. We will test for both levels of clustering and will also test whether the DID cluster in the same manner. To declare noninferiority, we would need the lower 95% confidence limit for the regression parameter corresponding to tech-supported versus standard HBPC to be above our margin of −3.
For our long-term outcome of days at home in the last 180 days of life, we will use linear mixed effects to model counts of days as a function of relevant patient characteristics. The outcome of days at home will be aggregated at the patient level, but similarly allows for patient-within-RN-within-site clustering. To declare noninferiority we would require the lower 95% confidence limit on the parameter comparing tech-supported versus standard HBPC to be above our margin of −7.
We will adapt the generalized linear mixed-effects modeling framework described previously, using different link functions to address the different types of secondary outcomes (continuous, dichotomous, or counts). We will determine whether the intervention effects differ by prespecified subgroups using the same models described previously and will include an interaction term. To describe the different longitudinal patterns of PC service exposure, intensity of acute care use, and quality of end-of-life care for the observational, passive cohort, we will use latent class regression29,30 to identify clusters of individuals following similar trajectories.
We are assessing barriers and facilitators to tech-supported HBPC implementation through quantitative and qualitative methods. We are tracking video visit uptake, refusal rates and reasons, crossovers with prescribing clinician visits and reasons, and patient/caregiver complaints. Data from qualitative interviews of patients/caregivers, site visits, and clinician surveys will be analyzed using standard procedures for thematic analysis.31,32
Ethics Determination
The study was approved by the KPSC and KPNW Institutional Review Boards.
Study Implementation: Challenges and Contributions
Stakeholder engagement
Our Stakeholder Advisory Committee (SAC), comprising patients, family and other caregivers, professional subject matter experts, and health systems partners, actively collaborates with the research team by providing input on the study design and methods, conduct (recruitment and retention, data collection and analysis, and other activities), and dissemination efforts. Table 4 provides a detailed summary of how the SAC engagement informed the design and approach during the study start-up.
Stakeholder Influence on Study Design and Approach
SAC, Stakeholder Advisory Committee.
Fidelity monitoring
We are assessing fidelity on a biweekly (year 1) to monthly basis through extraction of key process measures, for example, prescribing clinician visits (home visits for standard HBPC and telephone/video for tech-supported HBPC). In addition, the research RNs attend IDGs to elicit and listen for technical, logistical, and staffing issues and assist the teams with troubleshooting, as well, providing context for deviations that might be captured on the fidelity reports. We are providing site and individual feedback reports to the RNs and prescribing clinicians and the leadership at each site to inform implementation and reinforce the study protocol. Tech-supported HBPC teams may not feel comfortable with remote prescribing clinician supervision and consultation for certain patients and thus can choose to deviate from the protocol if the clinical situation calls for it. These crossovers could potentially contribute to artificially minimizing differences between groups. Our fidelity monitoring tracks variation in the timing and use of video visits and overall adherence to study arm allocation.
Implementation challenges and mitigation strategies
As much as the study team aims to plan in advance and apply rigor to the study conduct, pragmatic trials embedded within health systems must contend with the complex, dynamic, and evolving micro- and macroenvironments in which care is delivered. This is especially true for care of patients with serious illness.6,33-35 Implementation challenges for the study fall under four domains: (1) clinician readiness and openness to integrating video visits in their practice, (2) participant enrollment and data collection, (3) HBPC program staffing and resources, and (4) the evolving telehealth landscape. These challenges and corresponding mitigation strategies are described briefly here and summarized in Table 5.
Implementation Challenges and Mitigation Strategies
CMS, Centers for Medicare and Medicaid Services; IDG, interdisciplinary group.
Despite the broad organizational commitment and support of system and clinical leaders to the study design of allowing half of care teams to continue standard practice while innovating in the tech-supported arm, it still takes substantial effort to engage clinicians who are being asked to alter their usual patterns of care delivery or to hold back while others are innovating. There is notable variation within and across sites in clinician readiness and openness for integrating video visits into their practice. Change management strategies such as reassurance that use of video visits with individual patients is ultimately based on the clinical team's judgment, peer-to-peer sharing of positive experiences by early adopters, and ongoing technical training and coaching have been positively received.
Second, participant enrollment and data collection efforts must allow for inherent programmatic variations and integrate with existing workflows to improve value for the system, to avoid disruptions to current operations, and to minimize the burden on patients/caregivers and clinicians. Health system adoption of the ESAS as a standardized symptom measure across all PC services will enrich the study's data collection efforts. We also need to have realistic expectations for patient enrollment because some sites have limited capacity to enroll new patients. A cross-site engagement workgroup provides a forum for sharing implementation learnings across sites.
Third, with the dynamic and evolving demands on the HBPC program staffing and resources, care team structures will continue to pose challenges. We developed study-wide and local site manuals of operations to provide anticipatory guidance on issues such as handling attrition or reassignment of the randomized RNs. Other related challenges include reassignment of prescribing clinicians and RNs depending on clinical need, reorganization of teams and greater integration of licensed vocational nurses and shifting of staffing and resources from HBPC to hospice programs. Research staff RNs are attending IDG meetings as ethnographers, learning the culture of the teams, and participating in the care delivery discussions. Change happens at the speed of trust and through this mechanism, the research and clinical teams are collaboratively troubleshooting unfolding video visit implementation challenges.
Finally, the telehealth landscape will continue to evolve over the course of the study in tandem with programmatic changes described previously. Our priority is to maintain standardized procedures for collecting data across sites and encourage organizational commitment to rigorous evaluation of the two HBPC delivery modalities even as clinical teams further innovate. The cross-site coordination from the DCC also anticipates and has proactively established practices for monitoring and engaging the stakeholder advisory groups, cross-site research staff, and DSMB in activities such as monitoring and accounting for the impact of system differences and regulatory and policy changes. Maintaining effective collaborations with system leaders and clinicians by providing ongoing audit and feedback, sharing new information, and supporting processes introduced by the study is intended to help the HBPC program achieve greater efficiency, access, and improved care experience.
Plans for dissemination
A key facilitator to dissemination and implementation of the more efficient tech-supported HBPC model is our involvement of key stakeholders throughout the process of conceptualizing, developing, and evaluating this intervention. We have and are continuing to incorporate the perspectives of patients, caregivers, clinicians, administrators, high-level operational and executive leadership, payers, advocacy groups, and policy makers throughout the study to ensure the tech-supported model is acceptable to this diverse group of stakeholders.
Our dissemination plan and activities are guided by an emerging framework (derived from ongoing PCORI and non-PCORI studies) outlining a systematic, comprehensive approach for planning and conducting patient-centered outcomes research dissemination and implementation. This framework includes distinct phases of activity to (1) assess, in advance and continually, potential policy and practice implications of the study; (2) identify stakeholders likely to be affected by—and interested in—study results; (3) strategically involve these stakeholders in identifying potential implications and stakeholder groups' likely responses to findings; (4) proactively and systematically work to minimize any adverse reactions and maximize beneficial reactions and steps to benefit from the study; and (5) plan, design, and carry out specific communication, dissemination, and implementation activities to facilitate and encourage desired stakeholder response to the study. The approach includes numerous activities conducted well before study findings are available, in contrast to many dissemination approaches that begin only upon the availability of findings.
Conclusions
The HomePal study is highly responsive to the imperative from collaborating stakeholders to move beyond the four clinic walls and provide PC in patients' homes, especially when high symptom burden is coupled with significant functional limitations, making clinic visits too burdensome for both patients and caregivers. It also addresses gaps in the current state of the science in PC 36 and HBPC in particular,37-44 with regard to rigorous evaluations of telehealth integration. In close partnership with stakeholders across multiple representative groups, we refined the comparators, prioritized, and refined measures and study conduct, and optimized rigor and statistical approaches. Our fidelity monitoring protocol, mixed-methods evaluations, and early planning for dissemination incorporated anticipated challenges and mitigation strategies. The comparison between standard HBPC and a potentially more efficient, tech-supported HBPC model will provide much-needed evidence and guidance to policy and practice leaders interested in providing effective, scalable, and sustainable seamless high-quality supportive care services.
Footnotes
Acknowledgments
The authors are greatly appreciative of the HomePal study participants, the entire HomePal Research Group, which includes investigators, clinical partners, and staff from Kaiser Permanente Southern California and Kaiser Permanente Northwest, consultants, SAC members, and members of the data and safety monitoring board. Kaiser Permanente Southern California, Pasadena, CA:
Author Disclosure Statement
None of the authors have a conflict of interest with the content of this paper.
All statements in this report, including its findings and conclusions, are solely those of the authors and do not necessarily represent the views of the PCORI, its Board of Governors, or Methodology Committee.
