Abstract
Abstract
Objective:
To report cancer presentations with a median survival of 6 months or less and the effect of treatment on survival.
Methods:
We searched the MEDLINE database to find studies on solid and hematologic cancers that reported presentations consistently shown to have a median survival of 6 months or less. Independent prognostic factors were combined if their combination resulted in greater than 50% 6-month-mortality. For each terminal presentation, we evaluated whether treatment improved survival.
Results:
The search identified 1500 potentially relevant articles, of which 650 were evaluated and 383 were included. Despite different cancer characteristics, a fairly universal picture of terminal disease included decreasing performance status, advancing age, weight loss, metastatic disease, disease recurrence, and laboratory abnormalities indicating extensive disease. Most of the prognostic indicators found were continuous, independent risk factors for mortality. We found little evidence that treatment improved survival at these terminal stages, with increased risk for toxicity.
Conclusion:
This systematic review summarizes prognostic factors in advanced cancer that are consistently associated with a median survival of 6 months or less. There is little evidence that treatment prolongs survival at this stage.
Introduction
This systematic review summarizes the data on common cancer presentations associated with a median survival of 6 months or less and the effect of treatment on survival. A similar study on terminal noncancer presentations has been published separately. 4
Methods
The MEDLINE database was searched comprehensively to identify studies in any language published between 1980 and November 2010 that evaluated cancer survival, using the terms neoplasm and prognosis, survival, survival analysis, or median survival. Prognostic factors were searched for using terms such as performance status, weight loss, or neoplasm metastasis. The search was augmented by scanning the Surveillance Epidemiology and End Results survival monograph 5 and references of identified reviews and articles.6,7
Potentially relevant studies were retrieved and evaluated if they included a cancer presentation with a median survival of 1 year or less. We report only those presentations that had a median survival of 6 months or less in all studies reviewed. We chose easily identifiable clinical presentations and readily available laboratory tests. We extracted data on trials published between 1980 and 2010 but only include those between 1999 and 2010, allowing us to perform an extensive literature review while providing the most recent data.
If specific cancer presentations had median survivals that were above and below 6 months, further subgroup analyses explored potential heterogeneity in the results. For multivariate analysis, independent risk factors were combined and included if their combination was associated with a 50% or greater 6-month mortality risk.
Survival estimates were reported for the best available care. Once the results for each cancer were obtained, cancers with similar terminal presentations were reported together. For each of the presentations, we performed an additional search of randomized trials to evaluate whether treatment improved survival. We report “no survival benefit” when there is trial evidence of no significant survival benefit, and “no evidence of survival benefit” when these presentations were excluded from the trials. Further information on the trial evidence can be found in the Supplementary Tables available online at www.liebertonline.com/jpm.
Results
The search identified 1500 potentially relevant articles, and 650 were retrieved and evaluated. We included 289 studies in the prognosis analysis, with 94 additional studies on treatment (Fig. 1). The results for cancers in general are shown in Table 1, and for specific cancers, organized by cancer site, in Tables 2 and 3 (Supplementary References are available online at www.liebertonline.com/jpm). We define advanced as locally advanced or metastatic disease, and terminal as a presentation with a median survival of 6 months or less. Other terms used, such as refractory disease or anemia, are clarified in the tables.
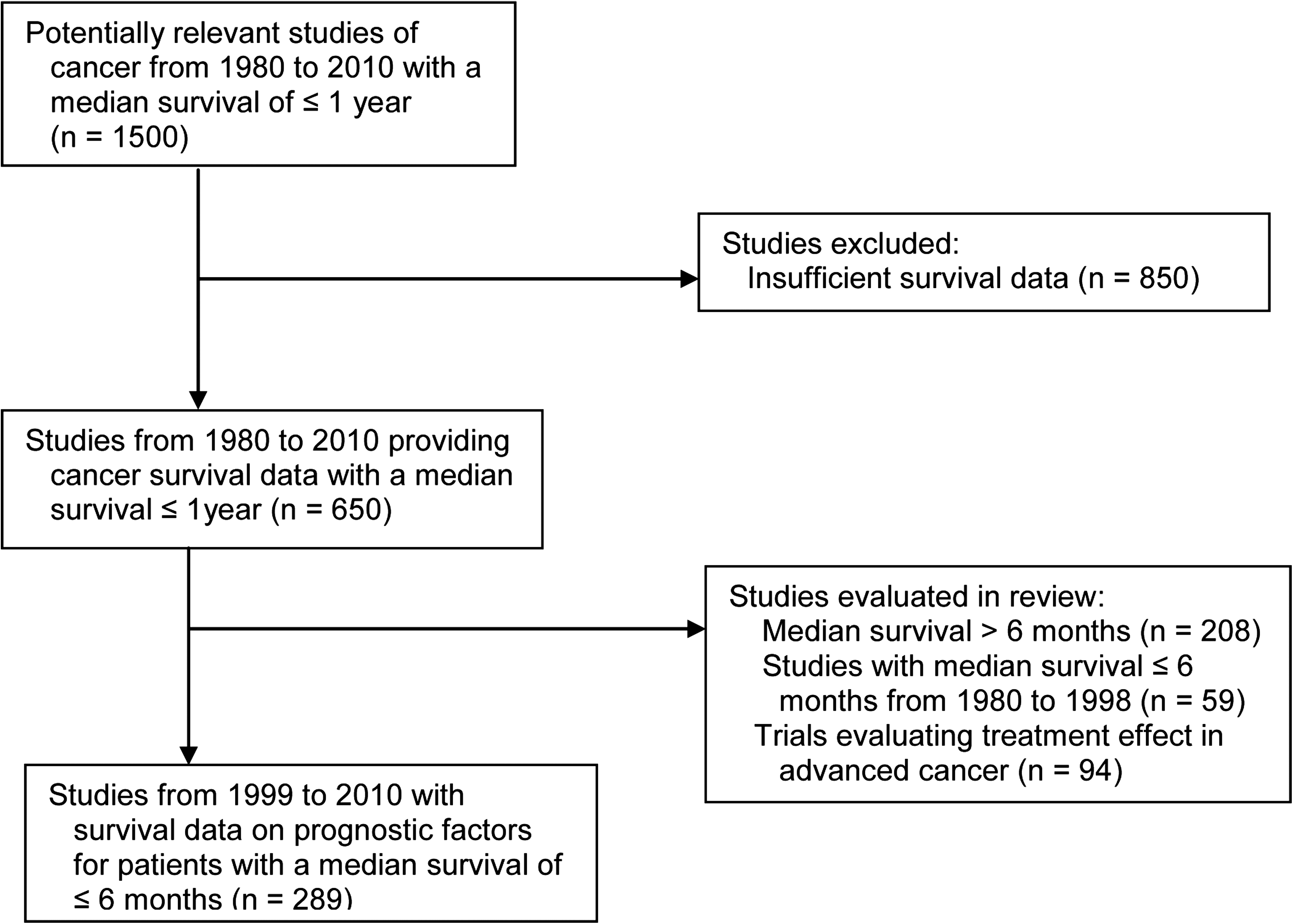
Flow chart of studies search.
Presentations with a median survival of 6 months or less, and survival benefit of treatment. Supplementary References are available online at www.liebertonline.com/jpm
Presentations associated with a median survival of 6 months or less, and survival benefit of treatment. Supplementary References are available online at www.liebertonline.com/jpm
KPS, Karnofsky Performance Status; ECOG, Eastern Cooperative Oncology Group.
Presentations associated with a median survival of 6 months or less, and survival benefit of treatment. Supplementary References are available online at www.liebertonline.com/jpm.
KPS, Karnofsky Performance Status; ECOG, Eastern Cooperative Oncology Group.
General solid cancer presentations
General terminal presentations of solid cancers
Survival in advanced cancers is strongly dependent on tumor-related factors, while in terminal disease patients generally enter a common final pathway, characterized by anorexia, weight loss, failing performance status, and systemic inflammation.6,8,9
Performance status is one of the most significant survival predictors in advanced cancer.10,11 The Karnofsky Performance Status (KPS)10,12,13 and the Eastern Cooperative Oncology Group (ECOG) scales, 14 described in Table 4, both have good reliability and validity. For any advanced solid cancer, a KPS less than 60 (ECOG status greater than 2) is associated with a median survival of 6 months or less (Supplementary Table 1 available online at www.liebertonline.com/jpm). There has been little change in survival over the past 30 years for those with poor functional status.10,15 Of note, most oncology trials require an ECOG performance status of 0 or 1 for inclusion, and trials enrolling patients with ECOG status greater than 2 are extremely rare. 16
Other terminal presentations associated with any advanced solid cancer include serum calcium greater than 11.2 mg/dL, acute venous thromboembolism, brain metastasis with KPS less than 70 or 2 or more brain metastases with extracranial metastases, spinal cord compression with decreased ability to walk, and malignant pericardial effusion (Supplementary Table 1 available online at www.liebertonline.com/jpm). There is no evidence that treatment of the cancer or the specific complication improves survival in these terminal presentations (Supplementary Table 1 available online at www.liebertonline.com/jpm).
Carcinoma of unknown primary
Metastatic adenocarcinomas and poorly differentiated carcinomas of unknown primary site have a median survival of 6 months or less when associated with KPS less than 80, malnutrition, disease recurrence, or metastatic disease in the liver, bone or adrenal glands (Supplementary Table 1 available online at www.liebertonline.com/jpm). No improvement in survival has been seen with any treatment.
Solid neoplasms by site
Breast cancer
The clinical presentations for metastatic breast cancer associated with a median survival of ≤6 months can be found in Table 2 (Supplementary References are available online at www.liebertonline.com/jpm). There is no evidence from randomized trials that treatment improves survival for the terminal presentations listed here. These prognostic factors also predict poorer response to treatment and greater risk for serious toxicity (Supplementary Table 2 available online at www.liebertonline.com/jpm).
Central nervous system cancers
Primary brain cancer
Median survival for glioblastomas is 6 months or less when associated with a KPS of less than 70, suboptimal surgical resection, refractory disease, or two or more of the following: KPS less than 90, age greater than 55 years, recurrent disease, a lesion in a critical brain region, large tumor volume, anemia, or thrombocytosis (Supplementary Table 2 available online at www.liebertonline.com/jpm). No survival benefit of treatment has been seen for the terminal presentations listed here.
Digestive system cancers
Colorectal cancer
Median survival for metastatic colorectal cancer is 6 months or less when associated with KPS less than 70, age greater than 75 years, or two or more of the following: KPS less than 90, age greater than 70 years, peritoneal carcinomatosis, brain metastases, more than 2 metastatic sites, malignant ascites, or refractory disease (Supplementary Table 2 available online at www.liebertonline.com/jpm). There is no evidence of a treatment survival benefit for those with the presentations listed here, who have reduced treatment response rates and increased toxicity.
Esophageal and gastric cancers
Median survival for advanced esophageal and gastric cancers is 6 months or less when associated with a KPS less than 80, a disease-free interval less than 6 months, or two or more of the following: KPS less than 90, liver or peritoneal metastases, serum alkaline phosphatase greater than 100 U/L, lactate dehydrogenase greater than 200 IU/L, or serum hemoglobin less than 1l g/L (Supplementary Table 2 available online at www.liebertonline.com/jpm). No survival benefit has been seen with treatment for these terminal presentations.
Hepatobiliary and pancreatic cancers
Median survival for locally advanced or metastatic pancreatic and biliary tract cancers is 6 months or less when associated with KPS less than 90, visceral or distant metastases, malignant ascites, biliary obstruction, recurrent or refractory disease, poorly differentiated or infiltrating disease, gross residual disease, malnutrition, serum lactate dehydrogenase greater than 500 IU/L, venous thromboembolism, or portal vein thrombosis (Supplementary Table 2 available online at www.liebertonline.com/jpm). There is no treatment survival benefit for patients with advanced pancreatic or biliary tract cancer and an ECOG performance status greater than 1, and no evidence of a survival benefit for the other terminal presentations listed here.
Locally advanced or metastatic hepatocellular carcinoma has a median survival of 6 months or less when associated with KPS less than 60, severe symptomatic cirrhosis, large tumor volume, any brain metastasis, or two or more of the following: KPS less than 90, extrahepatic metastases, moderate cirrhosis, alfa-fetoprotein level 400 ng/mL or greater, or portal vein thrombosis (Supplementary Table 2 available online at www.liebertonline.com/jpm). No survival benefit has been seen with treatment for those with large tumor size, jaundice or symptomatic cirrhosis.
Head and neck cancers
Squamous cell carcinomas of head and neck
Median survival for recurrent, refractory or metastatic squamous cell carcinoma of the head and neck is 6 months or less when associated with a KPS less than 90, significant weight loss, unresectable disease, cancer recurrence with any metastasis, pretreatment anemia, or hypercalcemia (Supplementary Table 2 available online at www.liebertonline.com/jpm). No survival advantage has been seen for any treatment of recurrent or metastatic head and neck cancer.
Anaplastic thyroid carcinoma
Anaplastic thyroid carcinoma is the most lethal of all cancers. 5 Most patients have evidence of extracapsular extension with incomplete resection at surgery or distant metastatic spread, and a median survival of 3–5 months (Supplementary Table 2 available online at www.liebertonline.com/jpm). No survival benefit has been seen with any treatment.
Skin and melanin neoplasms
Melanoma
Locally advanced or metastatic melanoma has a median survival of 6 months or less when associated with a KPS less than 80, serum calcium greater than 11.2 mg/dL, serum lactate dehydrogenase greater than 400 IU/L, metastases to the brain or spine, or metastases to the liver plus one other site (Supplementary Table 2 available online at www.liebertonline.com/jpm). Chemotherapy does not significantly improve survival while increasing treatment-related toxicity.
Thoracic cancers
Non-small–cell lung carcinoma
The clinical presentations of locally advanced or metastatic non-small cell lung cancer that are associated with a median survival of 6 months or less are shown in Table 2, with Supplementary References available online at www.liebertonline.com/jpm. No survival benefit has been seen with treatment for patients with KPS less than 70, who have been shown to have decreased treatment response and increased toxicity (Supplementary Table 2 available online at www.liebertonline.com/jpm).
Urogenital cancers
Female genital cancers
Median survival for locally advanced or metastatic ovarian, endometrial and cervical cancers is 6 months or less when associated with KPS less than 60, hypercalcemia, brain metastases, intestinal or urinary tract obstruction, unresectable disease, or two or more of the following: KPS less than 80, recurrent or refractory disease, disease-free interval of less than 6 months, suboptimal resection, significant weight loss, or extra-abdominal metastases (Supplementary Table 2 available online at www.liebertonline.com/jpm). No survival benefit has been seen for treatment in these terminal presentations.
Male genital cancers
Once metastatic prostate cancer becomes refractory to hormonal androgen blockade, median survival is 6 months or less for those with KPS less than 60, hemoglobin less than 10 g/dL, serum hemoglobin less than 12 g/dL with KPS less than 80, or spinal cord compression with decreased ability to walk (Supplementary Table 2 available online at www.liebertonline.com/jpm). There is no evidence of a survival advantage of treatment in patients with the terminal presentations listed here.
Urologic neoplasms
Median survival for locally advanced or metastatic bladder and renal cell carcinomas is 6 months or less when associated with KPS less than 70, hypercalcemia, brain metastases, or three or more of the following: KPS less than 80, serum lactate dehydrogenase greater than 300 IU/L or alkaline phosphatase greater than 220 U/L, disease-free interval of less than 1 year, visceral metastases, or hemoglobin less than 11.5 g/dL (Supplementary Table 2 available online at www.liebertonline.com/jpm). There is no evidence of a survival benefit with treatment in patients with KPS less than 60, comorbid illness, organ dysfunction, or recurrent disease.
Hematologic neoplasms
Acute leukemias
Acute lymphoblastic and myeloid leukemias have a median survival of 6 months or less when associated with age greater than 70 years, disease refractory to two or more courses of chemotherapy, recurrence with extramedullary disease involving the central nervous system, recurrence after a disease-free interval of less than 14 months, or 2 or more of the following: age greater than 60 years, KPS less than 80, central nervous system involvement, evidence of hemorrhage or infection, white blood cell count greater than 25×109/L, or serum lactate dehydrogenase less than 500 IU/L. No treatment for acute leukemia has been shown to improve survival for elderly patients or those with these poor prognostic factors (Supplementary Table 3 available online at www.liebertonline.com/jpm).
Chronic leukemias
Median survival for chronic myeloid leukemia with progression to blast phase is 6 months or less when associated with greater than 50% blast cells in the peripheral blood, recurrent or refractory disease, or myeloid blast transformation associated with age greater than 50 years, white blood cell count greater than 50×109/L, platelet count less than 100×109/L, or hemoglobin less than 10 g/dL (Supplementary Table 3 available online at www.liebertonline.com/jpm). No survival advantage has been seen with any treatment for chronic leukemia.
Chronic lymphocytic leukemia has a median survival 6 months or less when disease is recurrent after twol or more courses of chemotherapy or refractory to treatment (Supplementary Table 3 available online at www.liebertonline.com/jpm). No treatment for has been shown to improve survival.
Non-Hodgkin's lymphoma and multiple myeloma
Median survival for aggressive non-Hodgkin's lymphoma and multiple myeloma is 6 months or less when associated with secondary extranodal or extramedullary involvement of the central nervous system, primary central nervous system lymphoma related to human immunodeficiency virus, or two or more of the following: KPS less than 70, age greater than 65 years, refractory disease, elevated serum lactate dehydrogenase, hypoalbuminemia, myeloma with platelet count less than 80×109/L, or myeloma with serum creatinine 2 mg/dL or greater (Supplementary Table 3 available online at www.liebertonline.com/jpm). There is no evidence of a survival benefit with treatment for these terminal presentations.
Discussion
This systematic review reports on presentations that have been consistently shown to have a median survival of 6 months or less. The course of most cancers eventuates in a fairly universal clinical picture, with certain indicators of terminal disease, including decreasing performance status, advancing age, weight loss, metastases to the brain, spine, or liver, disease, recurrence and laboratory abnormalities indicative of inflammation or extensive disease. Most of these prognostic indicators are continuous, independent risk factors for mortality. Cancers with relatively good prognosis and treatment options, such as breast cancer, become terminal when the patient manifests KPS less than 60 or at least three prognostic factors, while cancers with poor prognosis, such as biliary cancers, become terminal with KPS less than 90 or 1 prognostic factor. Our review of studies from 1980 to 1998 showed that survival for these presentations has not changed significantly over the past 30 years, despite many treatment advances.
We found little evidence that treatment prolongs survival in terminal disease. Patients with poor performance status, bulky disease, advanced age, or organ dysfunction generally fail to respond to treatment while experiencing increased toxicity.17–25 Aggressive treatment for patients with poor prognostic factors may even worsen survival compared with no treatment, as is seen with pancreatic cancer, acute leukemia, and non-Hodgkin's lymphoma.19,21,22,26 However, for many terminal cancers little evidence is available, because randomized treatment data from this population is lacking. Over 90% of trial participants have ECOG performance status 0 or 1, no comorbid illness, and no evidence of organ dysfunction. 16 Where they have been included, subgroup analysis has shown that treatment does not reduce mortality.19,23,26–28 Future randomized trials in patients with terminal disease would be necessary to clarify the efficacy of treatment.
This review has several limitations. This is a qualitative study that provides an overview of common terminal cancer presentations found in published studies; quantitative validation studies are still needed. Although we have included data on terminal presentations of the most common cancers and the effect of numerous treatments, this is not an exhaustive list. We can still search for prognostic factors that could identify those individuals with these presentations who may have an extended prognosis. By definition, half of the patients with a median survival of 6 months will live longer than this, so clinicians must be careful in communicating this uncertainty in honest prognostic discussions with patients, while continuing to search for potentially effective treatments that may provide a survival benefit.
Another limitation of this review is that different types of studies and different populations of cancer patients were combined in the analysis. In this case, it is possible that publication bias or selection bias could have altered the results. However, indicators of mortality were remarkably similar across studies, and we only included those clinical presentations that were consistently shown to have a median survival of 6 months or less in all studies. Finally, we only report on survival in this analysis, and do not provide information on intermediate treatment outcomes such as progression-free survival or quality-of-life measures. Palliative treatments without a known survival benefit could still be offered if they are thought to improve other clinically relevant outcomes.
The “terminal illness” criterion for Medicare hospice eligibility requires enrollees to have a “life expectancy of 6 months or less, assuming the disease runs its normal course.” 29 Because prediction of individual prognosis is inexact, with a tendency to overestimate survival, hospice referrals are often delayed until death is imminent.6,30 Median survival in hospice across the United States is less than 3 weeks; over one third of hospice patients die within 7 days after enrollment, many receiving chemotherapy shortly before death.2,31
Prognostic discussions are hampered, in part, by physicians' reluctance to broach the subject.32,33 However, surveys indicate that the majority of patients want prognostic information, which allows them to make informed treatment decisions and prepare for the future.34,35 In one study, patients with metastatic cancer who had an accurate understanding of their prognosis were less likely to choose aggressive treatments, but had similar survival, compared with patients who overestimated their chances of survival. 36
Evidence is accumulating that earlier transition to palliative care may actually prolong survival in some cases. 37 One trial randomized patients with metastatic lung cancer to standard oncologic care with or without early palliative care and found that those randomized to palliative care received less chemotherapy and had a median survival that was 2.7 months longer than those receiving standard care. 38
In conclusion, this systematic review summarizes prognostic factors common to terminal cancer presentations, and reveals a lack of evidence that treatment prolongs survival at these stages. This could serve as a resource to help physicians provide guidance to cancer patients. Future prognostic studies and randomized trials aimed at this population are needed to validate these findings.
Footnotes
Author Disclosure Statement
No competing financial interests exist.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
