Abstract
Dry eye disease (DED) is a multifactorial disorder characterized by disruption of tear film homeostasis, resulting in ocular surface inflammation and damage. Although several Food and Drug Administration-approved topical treatments are available, direct comparisons of their efficacy and safety are complicated by variability in study designs and corneal staining grading scales. This review systematically evaluates and compares the efficacy and safety of topical therapies approved in the United States, focusing on anti-inflammatory and semi-fluorinated alkane (SFA)-based therapies. A systematic literature review identified 12 randomized controlled trials involving a total of 6,984 patients with varying severity of DED eligible for inclusion, with 8 providing data suitable for quantitative meta-analysis and 5 for exploratory regression analysis. Meta-analysis indicated that cyclosporine 0.1%/SFA showed the most significant early improvement (within ≤4 weeks) in total corneal fluorescein staining, outperforming other treatments. Exploratory regression analysis further supported these findings, demonstrating that cyclosporine 0.1%/SFA had the fastest and most consistent reduction in corneal staining, with the steepest improvement slope and strong predictability (R2 = 0.871). Safety analyses highlighted improved local tolerability for SFA-based therapies compared with traditional anti-inflammatory treatments, notably lower instillation site discomfort for both cyclosporine 0.1%/SFA (2.5%–9.9%) and perfluorohexyloctane (≤1%) vs. other cyclosporine formulations. SFA-based therapies, especially cyclosporine 0.1%/SFA, demostrated robust efficacy in improving signs of DED with superior tolerability profiles compared to traditional anti-inflammatory treatments. These findings support the role in effectively managing ocular surface inflammation and optimizing treatment strategies in DED.
Introduction
Dry eye disease (DED) is a common and multifactorial ocular disorder that affects millions worldwide, with prevalence estimates ranging from 5% to 50% depending on geographic region. 1 In the United States alone, an estimated 16.7–50.2 million individuals are affected. 2 DED significantly impairs quality of life by disrupting daily activities such as work, driving, and reading. It can also negatively impact mental health due to chronic pain. 3 Despite its widespread prevalence and impact, DED often remains undiagnosed. Studies have revealed high rates of foreign body sensation, corneal staining, and abnormal osmolarity among asymptomatic individuals, underscoring the disconnect between clinical signs and symptoms and the challenges of accurate diagnosis. 4
DED is characterized by a loss of tear film homeostasis, involving tear hyperosmolarity, ocular surface inflammation, and neurosensory abnormalities. 5 As described in the TFOS DEWS II Pathophysiology Report, tear hyperosmolarity is a central factor in the pathogenesis of both aqueous-deficient and evaporative DED. 6 It triggers desiccating stress that damages the ocular surface both directly and indirectly by initiating inflammatory cascades. 7 These cascades involve the release of proinflammatory mediators such as cytokines and matrix metalloproteinases, which disrupt epithelial barrier integrity, induce goblet cell loss, and exacerbate tear film instability.6,8 Inflammation in DED acts as both a cause and a consequence, perpetuating a vicious cycle of damage. Chronic hyperosmolarity and mechanical stress activate innate and adaptive immune responses, further amplifying ocular surface inflammation and glandular dysfunction.6–9 This vicious cycle contributes to disease chronicity through meibomian gland dysfunction (MGD), persistent tear film dysregulation, and worsening ocular surface damage. The resulting pathological changes lead to clinical manifestations such as dryness, discomfort, and visual disturbances, significantly impairing quality of life and productivity, underscoring the need for timely diagnosis and intervention.6–9
The diagnosis of DED requires evaluating both clinical signs and patient-reported symptoms, as their correlation is often inconsistent. 10 Symptoms such as dryness, discomfort, and visual disturbances can vary widely in severity and often do not align with clinical findings, making diagnosis challenging. Corneal staining, a key indicator of ocular surface health, is widely used to assess DED in clinical trials and serves as a primary endpoint for regulatory agencies like the U.S. Food and Drug Administration (FDA). 11 The American Society of Cataract and Refractive Surgery highlights corneal staining as essential to normalize before vision-related surgeries.11–13 The Oxford Grading System uses a scale from 0 to 5 to evaluate corneal staining, while the National Eye Institute (NEI) Scale ranges from 0 to 15 across 5 corneal regions.14,15 However, grading methodologies and evaluation approaches often vary across treatments, making it challenging to directly compare therapeutic outcomes. 15 This review aims to assess the effects of different treatments on the signs and tolerability, particularly corneal staining.
Objectives
Despite the availability of various prescription topical treatments for DED, there remains a gap in comprehensive comparative analyses among commonly prescribed anti-inflammatory agents, such as cyclosporine (0.05%, 0.09%) and lifitegrast (5%), as well as newly approved semi-fluorinated alkane (SFA)-based therapies, including perfluorohexyloctane (PFHO) and cyclosporine 0.1%/SFA, which uniquely combines an anti-inflammatory agent within an SFA formulation. Given the variability in formulations, a systematic evaluation of their relative efficacy and safety is essential for informing clinical practice. The primary objective of this review is to evaluate the efficacy of these treatments in addressing the signs of DED across varying severities for chronic use. In addition, it seeks to assess the safety profiles of these prescription topical therapies available in the United States. By synthesizing data from selected clinical studies and critically analyzing outcomes, this review aims to enhance understanding of treatment effectiveness and provide evidence-based insights to inform clinical decision-making.
Furthermore, this review intends to identify gaps in existing knowledge, highlight the limitations of current treatments, and propose directions for future research. The ultimate goal is to optimize the management of DED and improve patient outcomes through a better understanding of the comparative effectiveness and safety of available therapies.
Methods
A comprehensive search was conducted using electronic databases, including PubMed (from 1946 to August 1, 2024) and Google Scholar (all years to August 1, 2024). The search aimed to identify randomized, controlled, double-masked trials evaluating interventions for DED. Search terms and keywords included combinations such as “Dry Eye Disease,” “DED,” “keratoconjunctivitis sicca,” “corneal fluorescein staining,” “anti-inflammatory treatments,” and “semi-fluorinated alkane.” The search was restricted to English-language publications, and only fully published articles were included. Abstracts, unpublished studies, and non-English-language studies were excluded.
Eligible studies were randomized, controlled, double-masked trials comparing interventions for DED with a control arm, such as saline or vehicle. Included studies evaluated FDA-approved topical treatments containing anti-inflammatory ingredients or SFA-based therapies currently available in the United States for the treatment of chronic DED. Interventions using short-term relief treatments were excluded. Participants included individuals diagnosed with DED of any severity, ranging from mild to severe. Studies were required to report on primary or secondary outcomes relevant to the review’s objectives. All phases of studies were included if they met above standards. Ad hoc analyses were considered in descriptive efficacy comparisons, and extension studies were included in safety evaluations. However, open-label studies and pooled data from multiple interventional studies were excluded from efficacy assessments to ensure methodological consistency.
The primary efficacy assessment was total corneal fluorescein staining (tCFS). Secondary outcomes included safety profiles and tolerability metrics, if applicable. Studies with any follow-up duration from the time of intervention were considered for inclusion.
Results
A total of 43 unique records were identified through the search process. Of these, the full texts of 28 records were retrieved and reviewed. Studies were excluded if they lacked a control arm, were not randomized controlled trials, were not double-masked, utilized off-market or investigational ingredients, employed nontopical treatments, or involved interventions that were not anti-inflammatory or vehicle-based. Ultimately, 12 studies fulfilled all inclusion criteria and were included in the descriptive synthesis. Among these 12 studies, 8 provided sufficiently comparable quantitative data suitable for inclusion in the meta-analysis, while 5 studies reported adequate data across multiple consistent time points allowing exploratory regression analysis. The study selection process is summarized in Fig. 1. Details of characteristics of included interventional studies are summarized in Table 1. Extended studies focusing on safety profile evaluations are described in subsequent sections. Across all included studies, a total of 6,984 subjects with DED were evaluated. Study sample sizes ranged from 206 to 877 participants. The mean age of participants across included studies ranged from 46.7 to 61.5 years.
Flow diagram of study selection process. Flow diagram illustrating the study selection process. From 50 records identified, 43 were screened after removing duplicates. Of these, 28 full-text articles were assessed for eligibility, with 15 excluded based on predefined criteria. Ultimately, 12 studies were included in the descriptive synthesis, of which 8 studies provided sufficiently comparable data for quantitative synthesis (meta-analysis).
Characteristics of Included Studies
This table summarizes the characteristics, design, and key outcomes of pivotal clinical trials evaluating currently available chronic DED treatments. The data include information on study design, interventions, patient population, endpoints, treatment duration, and safety profiles, highlighting the efficacy of these treatments in improving clinical signs and, where applicable, symptoms of DED.
AE, adverse event; cCFS, central corneal fluorescein staining; DED, dry eye disease; EDS, eye dryness score; ICSS, inferior corneal staining; LGCS, Lissamine Green Conjunctival Staining; MGD, meibomian gland dysfunction; NEI, National Eye Institute; OSDI, Ocular Surface Disease Index; SANDE, Symptom Assessment in Dry Eye; tCFS, total corneal fluorescein staining; TEAE, treatment-emergent adverse event; TBUT, Tear Breakup Time; VAS, Visual Analog Scale; VR-OSDI, Visual-related function subscale of the Ocular Surface Disease Index (questions 6–9).
Treatments with anti-inflammatory ingredients
Cyclosporine ophthalmical solution 0.09%, Sun Pharmaceutical Industries, Inc., Cranbury, NJ
Approved by the FDA in 2018, it increases tear production in patients with keratoconjunctivitis sicca due to presumed ocular inflammation and uses a nanomicellar formulation to improve cyclosporine delivery.16–19 Cyclosporine 0.09% was evaluated in 3 double-masked, vehicle-controlled trials involving patients with mild to moderate DED. Following a 14- to 20-day vehicle run-in period, patients were randomized to cyclosporine 0.09% or vehicle drops twice daily for 12 weeks.17–19
Efficacy was assessed using the extended NEI grading scale (0–20) and 0–4 scale for 5 regions of the cornea, including 0.5 increments on an individual basis.17–19 In the Goldberg et al. trial, cyclosporine 0.09% significantly improved tCFS, with reductions of −0.8 at week 4, −1.3 at week 8, and −1.4 at week 12, compared with −0.6, −0.9, and −1.2 in the vehicle group (P < 0.01 for each time point). 19 In the 12-week Schechter et al. study, cyclosporine 0.09% showed a numerically greater reduction in tCFS compared with vehicle at week 2; however, the difference was not statistically significant (P = 0.0557). No additional tCFS reduction data were reported for later time points. 18 Resolution of central corneal fluorescein staining (cCFS) improved from a baseline of 38.3% (cyclosporine 0.09%) and 37.5% (vehicle) to 54.1% vs. 47.3% at week 4 (P = 0.04), increasing to 65% vs. 56.9% by week 12 (P = 0.02).
The safety of cyclosporine 0.09% was evaluated across 3 trials with similar results.17–19 The most frequently reported ocular adverse event (AE) was instillation site pain, occurring in 24.2% of cyclosporine 0.09%-treated patients compared with 4.3% in the vehicle group in the Goldberg et al. trial. This is consistent with the other 2 trials, which showed an occurrence of 15.1% for the cyclosporine 0.09% group.17–19 Conjunctival hyperemia was observed in 8.1% of cyclosporine 0.09%-treated patients vs. 5.1% in the vehicle group. 19
Cyclosporine ophthalmical emulsion 0.05%, Allergan, Irvine, CA
Approved by the FDA in 2003, it was the first prescription treatment for DED and remains a key therapy for increasing tear production in patients with keratoconjunctivitis sicca.20–22 Due to its approval ∼2 decades ago, the availability of controlled studies for cyclosporine 0.05% is limited compared with newer therapies.
In the Sall et al. trial, patients with moderate-to-severe DED were randomized 1 per 1 to receive cyclosporine 0.05% or vehicle twice daily for 6 months following a 2-week vehicle run-in period. CFS was assessed using the Oxford scale (0–5). Improvements in CFS were significantly greater in the cyclosporine 0.05% group compared with the vehicle group, at month 4 (P = 0.044) and month 6 (P = 0.008); however, the specific numeric reduction in staining scores was not reported. 22
The most frequently reported ocular AE was burning upon instillation, observed in 14.7% of cyclosporine 0.05%-treated patients compared with 6.5% in the vehicle group. Stinging eyes and eye discharge were reported in of cyclosporine 0.05%-treated patients, 3.4%, 3.1%, respectively. Discontinuation rates due to AEs were 6.5% in the cyclosporine 0.05% group vs. 4.5% in the vehicle group. 22
Cyclosporine ophthalmical solution 0.1%Harrow Inc, Nashville, TN
Approved by the FDA in 2023, it is a 0.1% cyclosporine ophthalmical solution formulated in an SFA-based vehicle. Its detailed clinical evaluation of efficacy and safety is in the next section.23–27
Lifitegrast ophthalmical solution 5%, Bausch & Lomb Americas Inc., Bridgewater, NJ
Approved by the FDA in 2016, lifitegrast treats signs and symptoms of DED by inhibiting T-cell-mediated inflammation through blocking the LFA-1/ICAM-1 interaction.28–37 Lifitegrast was evaluated in 3 phase 3 clinical trials involving patients with mild-to-moderate DED [Sheppard (OPUS-1)] and moderate-to-severe DED [Tauber (OPUS-2), Holland (OPUS-3)], randomized to lifitegrast 5% or placebo twice daily for 12 weeks.32–34
In Sheppard (OPUS-1), CFS was assessed using the Ora scale (0–4 points per region), with tCFS calculated as the sum of inferior, superior, and central region scores (0–12 scale). Lifitegrast significantly improved inferior corneal staining (ICSS, P = 0.0007) and tCFS (P = 0.0148) at week 12. 32 In Tauber (OPUS-2), a similar tCFS scoring (0–12 scale) was used, though the Ora scale was not explicitly referenced. This study found no significant differences in ICSS or tCFS at week 12. 33 In Holland (OPUS-3), the primary endpoint was improvement in eye dryness score (EDS), while corneal staining assessments focused primarily on ICSS (0–4 scale). An ad hoc intention-to-treat (ITT) analysis demonstrated significant improvement in ICSS at week 12 (treatment effect of 0.17, P = 0.0144), though no significant differences occurred at earlier time points (weeks 2 and 6). 34
Across all 3 studies, lifitegrast exhibited a consistent safety profile. The most frequent ocular treatment-emergent adverse events (TEAEs) were instillation site irritation or pain, observed in 24% [Sheppard (OPUS-1)], 15.9% [Tauber (OPUS-2)], and 18.2% [Holland (OPUS-3)] of patients, rates higher than placebo groups.32–34 Dysgeusia was the most common nonocular AE consistently reported in lifitegrast-treated patients (12.9%–16.2%). Most TEAEs were mild-to-moderate in severity.32–34
SFA-based treatments
SFAs are amphiphilic compounds composed of perfluorocarbon and hydrocarbon segments, valued for their nontoxic nature, ability to dissolve lipophilic drugs and unique properties such as low surface tension and high biocompatibility. SFAs, such as PFHO and perfluorobutylpentane (PFBP), have been investigated for their potential to stabilize the tear film, reduce evaporative loss, and enhance drug delivery. These properties address key pathophysiological mechanisms of DED.38–42
PFHO ophthalmical solution, Bausch & Lomb Americas Inc., Bridgewater, NJ
Approved by the FDA in 2023, it is indicated for the treatment of signs and symptoms of DED. Its formulation contains PFHO, an SFA, without anti-inflammatory components.43–47
PFHO was evaluated in 2 multicenter, randomized, double-masked, saline-controlled phase 3 trials [Tauber et al. (GOBI) and Sheppard et al. (MOJAVE)], to assess its efficacy and safety. Both trials enrolled adults with mild-to-moderate DED with MGD, randomized to PFHO or hypotonic saline (0.6%) 4 times daily for 8 weeks.43,44 In both studies, tCFS was measured using the NEI standard (0–15 scale). PFHO demonstrated statistically significant improvements in tCFS compared with saline. At week 8, the mean change from baseline in tCFS was −2.0 in the PFHO group vs. −1.0 in the saline group [Tauber et al. (GOBI), P < 0.001] and −2.3 vs. −1.1 [Sheppard et al. (MOJAVE), P < 0.001].43,44
The safety of PFHO was consistently demonstrated across Tauber et al. (GOBI), Sheppard et al. (MOJAVE), and an extension study. In Tauber et al. (GOBI) and Sheppard et al. (MOJAVE), PFHO exhibited good tolerability, with ocular AEs being mild and showing similar incidences between PFHO and saline groups [Tauber et al. (GOBI): 9.6% vs. 7.5%; Sheppard et al. (MOJAVE): 12.9% vs. 12.3%]. Instillation site pain occurred in less than 1% of patients in both trials, and no serious treatment-related events were reported.32,33 Across all trials, the most commonly reported AEs were blurred vision (1.3%–3.0%), blepharitis (1.6%), conjunctival hyperemia (1.3%), and instillation site pain (0.3%–1.0%), all of which were mild and transient. In the extension study, PFHO’s safety profile remained favorable, with a small proportion of patients discontinuing treatment due to AEs (2.4%).43,44
Cyclosporine ophthalmical solution 0.1%, Harrow Inc., Nashville, TN
The efficacy of cyclosporine ophthalmical solution 0.1%/SFA was evaluated in 3 randomized, double-masked, vehicle-controlled phase 3 clinical studies [Sheppard et al. (ESSENCE-1), Akpek et al. (ESSENCE-2), and Peng et al.].23–27 These trials enrolled adults with moderate-to-severe DED randomized to receive cyclosporine 0.1%/SFA or vehicle (SFA only) twice daily for 12 weeks [Sheppard et al. (ESSENCE-1)] or 4 weeks [Akpek et al. (ESSENCE-2) and Peng et al.], with an optional 12-week extension in Peng et al. Corneal staining was assessed using the NEI grading scale (0–15 points). Cyclosporine 0.1%/SFA demonstrated significant improvements compared with vehicle in tCFS across all studies. In Sheppard et al. (ESSENCE-1), the mean reduction from baseline at week 4 was −0.8 points (P = 0.0002) compared with the vehicle. 23 In Akpek et al. (ESSENCE-2), the mean tCFS reduction at week 4 was −4.0 vs. −3.6 for vehicle (P = 0.03). 24 Similarly, in Peng et al., cyclosporine significantly reduced tCFS by −4.8 compared with −3.0 with vehicle at week 4 (mean difference: −1.8, P < 0.001), with sustained efficacy observed through the additional 12-week extension. 27
The safety of cyclosporine ophthalmical solution 0.1%/SFA was consistently demonstrated across all trials and their extension studies. Treatment-related ocular AEs were mild to moderate and showed comparable incidences between active treatment and vehicle [20.4% vs. 20.5% in Sheppard et al. (ESSENCE-1); 16.8% vs. 17.8% in Akpek et al. (ESSENCE-2); 14.6% vs. 10.7% in Peng et al.].23,24,27 Commonly reported AEs included instillation site pain (2.5%–9.9%), reduced visual acuity (1.7%–12.6%), and blurred vision (0.5%–2.9%).23,24,27 In the 52-week extension, cyclosporine ophthalmical solution 0.1%/SFA maintained a favorable safety profile, with low discontinuation rates due to AEs (1.5%). 25
Summary of efficacy and safety
To facilitate meaningful comparisons, tCFS scores from all included trials were normalized to the standardized NEI grading scale (0–15 points), enabling fair evaluation of topical treatment effects for DED. Treatments were categorized based on mechanisms of action and formulations into anti-inflammatory therapies (cyclosporine 0.09%, cyclosporine 0.05%, cyclosporine 0.1%/SFA, and lifitegrast 5%) and SFA-based therapies (PFHO and cyclosporine 0.1%/SFA) reflecting underlying differences in mechanisms of action and formulations. Due to variability in study endpoint durations (ranging from week 2 to week 12), analyses were conducted separately for early onset efficacy (defined as ≤4 weeks) and later endpoint efficacy (defined as weeks 8–12) for 3 treatment classes, anti-inflammatory therapies, SFA-based therapies, and all DED therapies, accounting for these timing differences. Four studies initially identified in the literature review were excluded from meta-analyses due to incomplete or incompatible endpoint data reporting, resulting in 8 studies included in the meta-analysis. Exploratory regression analyses complemented these findings by evaluating improvement trends over time, explicitly acknowledging that while linear regression was employed due to limited available data points, future analyses with additional data could more accurately characterize nonlinear relationships.
To evaluate early therapeutic effects, the earliest reported numerical efficacy data within the first 4 weeks of treatment from each included study were used, ensuring a meaningful and consistent comparison of early treatment response. Meta-analysis of early therapeutic effects (≤4 weeks) included 7 studies with reported data, incorporating these earliest available data points. The pooled mean difference significantly favored active treatments over controls (−0.51; 95% CI: −0.73, −0.29; P < 0.00001), with moderate heterogeneity observed (I2 = 58%, P = 0.01), reflecting variability in study designs, endpoints, and patient populations. Figure 2 illustrates the overall meta-analysis results for all DED therapies, clearly favoring active treatments. Subgroup analyses highlighted significant early reductions in corneal staining with anti-inflammatory therapies, particularly cyclosporine 0.1%/SFA (−0.50; 95% CI: −0.81, −0.20; P = 0.001), as shown in Fig. 3. Similarly, SFA-based therapies demonstrated significant early efficacy, with cyclosporine 0.1%/SFA notably exhibiting robust superiority (−0.67; 95% CI: −0.86, −0.48; P < 0.00001), depicted in Fig. 4.
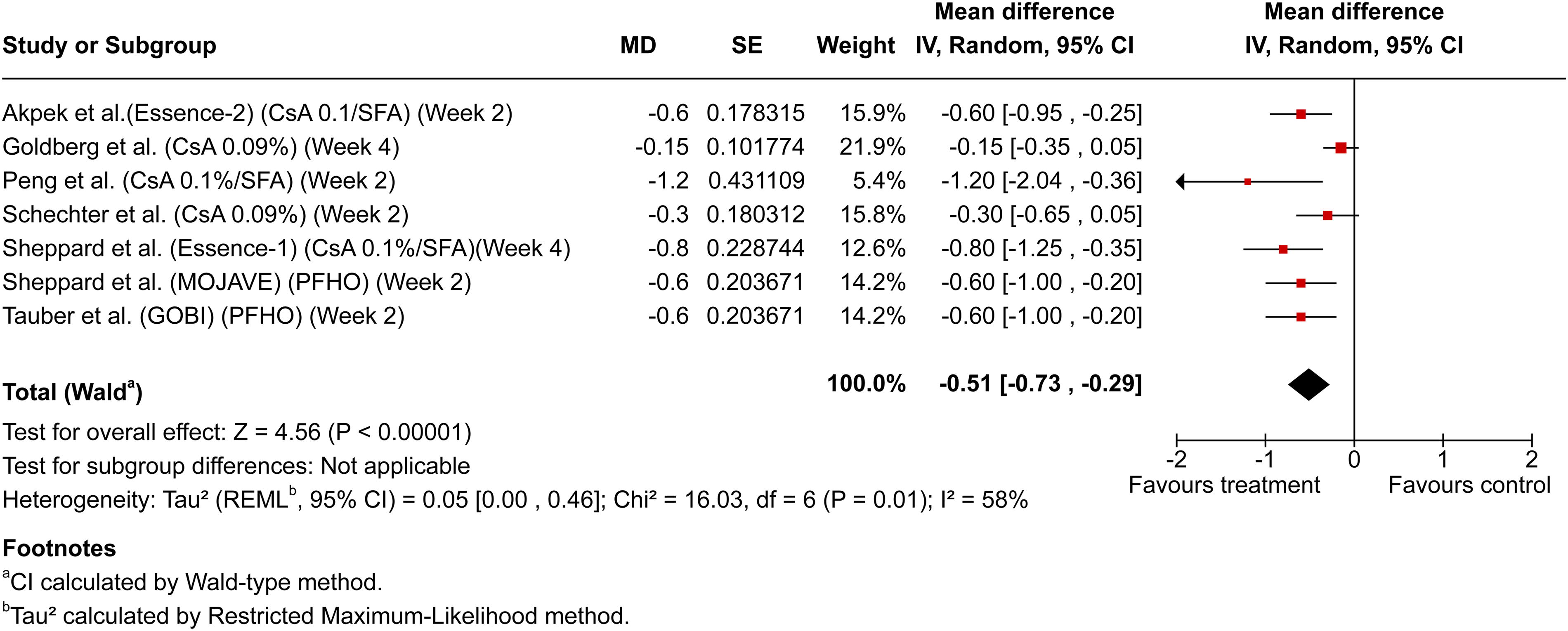
Forest plot summarizing meta-analysis results of total corneal fluorescein staining (tCFS) reduction (≤4 weeks). Forest plot illustrating meta-analysis results of tCFS reductions within the first 4 weeks across all dry eye disease therapies included in the review. The pooled mean difference favors active treatments compared with control arms. CsA 0.1%/SFA, cyclosporine 0.1% in SFA; CsA 0.09%, cyclosporine 0.09%; PFHO, perfluorohexyloctane.
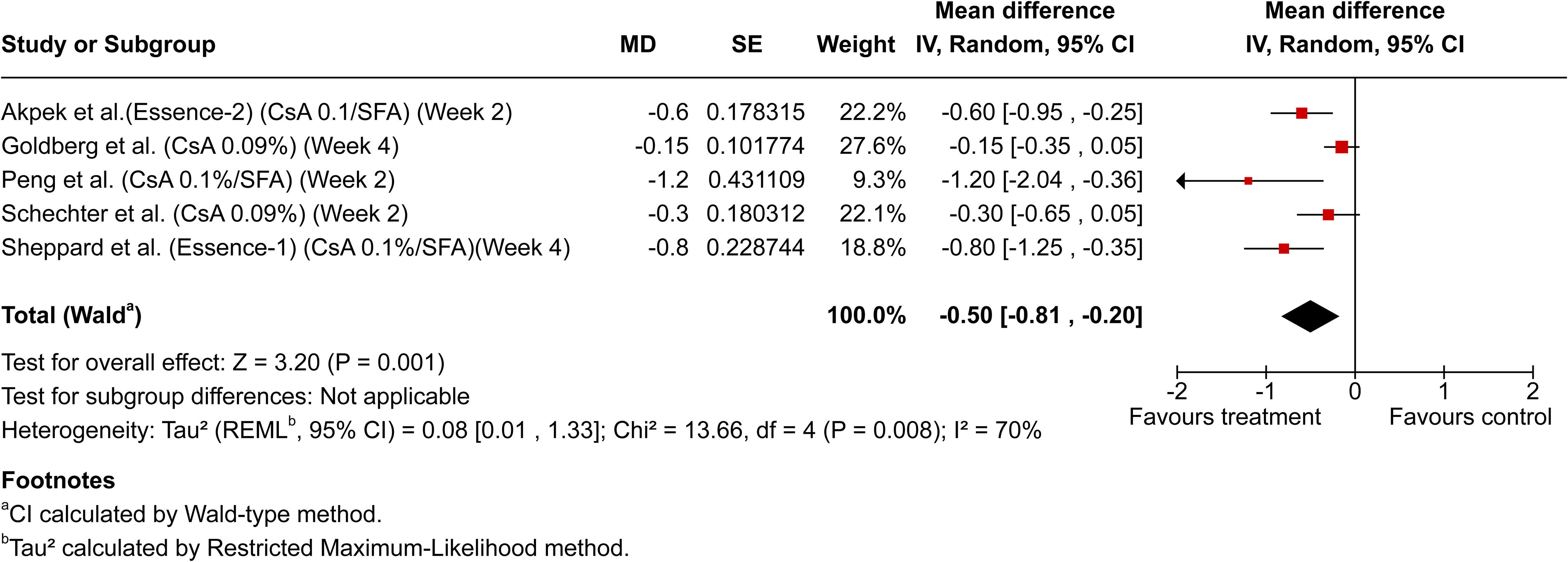
Forest plot of meta-analysis for early total corneal fluorescein staining (tCFS) reduction (≤4 weeks) in anti-inflammatory therapies. Forest plot demonstrating subgroup meta-analysis of anti-inflammatory therapies, specifically showing significant early reductions in tCFS compared with controls within ≤4 weeks of treatment initiation. CsA 0.1%/SFA, cyclosporine 0.1% in SFA; CsA 0.09%, cyclosporine 0.09%.
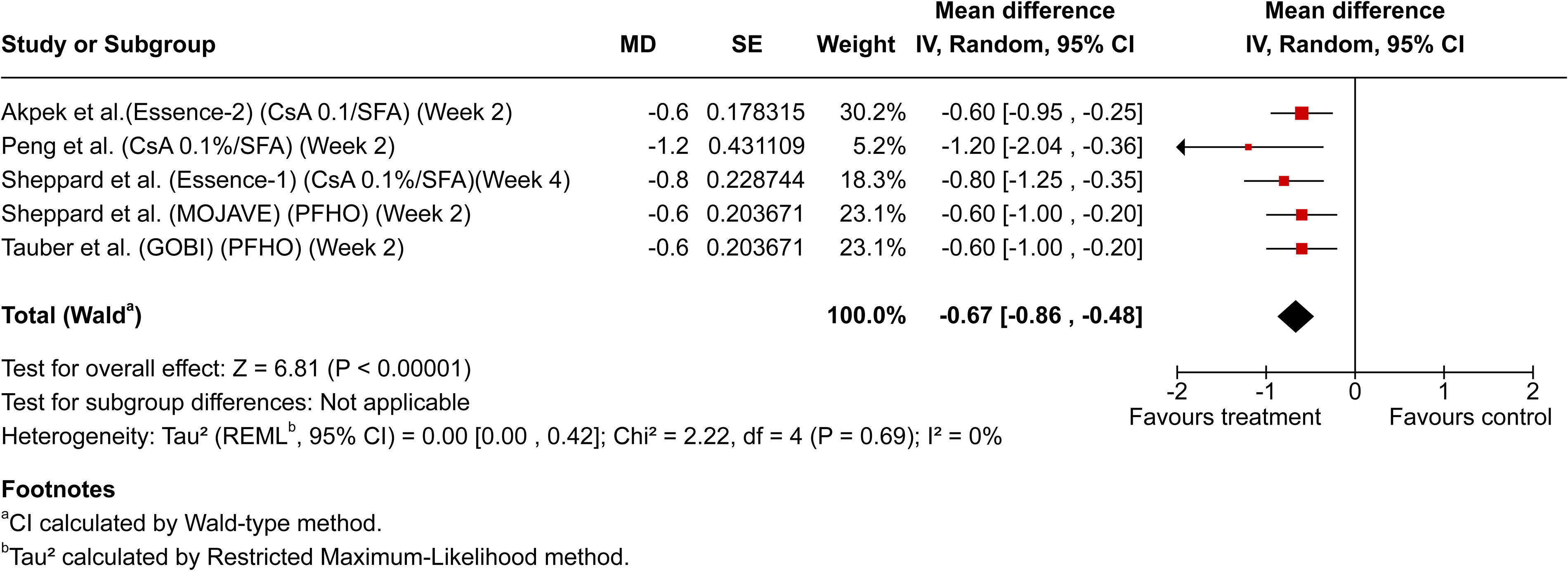
Forest plot of meta-analysis for early total corneal fluorescein staining (tCFS) reduction (≤4 weeks) in semi-fluorinated alkane (SFA)-based therapies. Forest plot depicting subgroup meta-analysis of SFA-based therapies, highlighting significant improvements in tCFS compared with control arms during the first 4 weeks of treatment. CsA 0.1%/SFA, cyclosporine 0.1% in SFA; PFHO, perfluorohexyloctane.
Analysis of later endpoints (≥8–12 weeks) involved 4 studies with reported data. Due to the limited number of studies, a subgroup of meta-analysis was not performed. Results favored active treatments with a pooled mean difference of −0.60 (95% CI: −1.13, −0.07; P = 0.03), despite significant heterogeneity (I2 = 89%, P < 0.0001, Fig. 5).
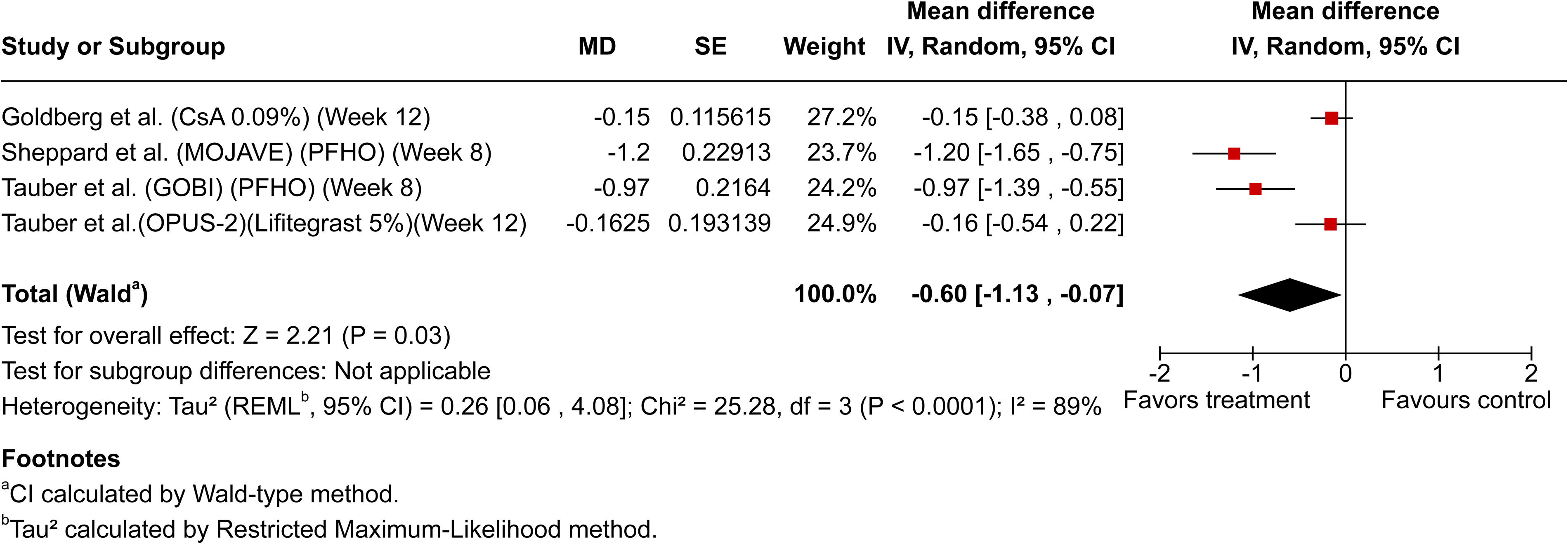
Forest plot summarizing meta-analysis of later endpoint efficacy (weeks 8–12). Forest plot summarizing meta-analysis findings of later endpoint efficacy (8–12 weeks) for semi-fluorinated alkane (SFA)-based therapies, demonstrating significant treatment-related reductions in total corneal fluorescein staining (tCFS) despite observed variability across studies. CsA 0.09%, cyclosporine 0.09%; PFHO, perfluorohexyloctane; Lifitegrast 5%, Lifitegrast ophthalmical solution 5%.
Exploratory regression analyses utilized data from 5 studies providing multiple time points (≥3) suitable for assessing consistent temporal trends in corneal staining improvement. Linear regression was chosen for this exploratory analysis due to its simplicity, clarity of interpretation, and utility in illustrating general treatment trends over time. However, it is important to acknowledge that therapeutic effects on corneal staining may not strictly adhere to a linear progression; therefore, this model primarily serves as an illustrative trend. Higher R2 values indicate that a greater proportion of outcome variability is explained by the model, reflecting more robust and reliable predictions.48,49 Notably, cyclosporine 0.1%/SFA exhibited the steepest slope and high R2 value (0.871), indicative of a faster and more consistent reduction in corneal staining compared with other treatments (Fig. 6).
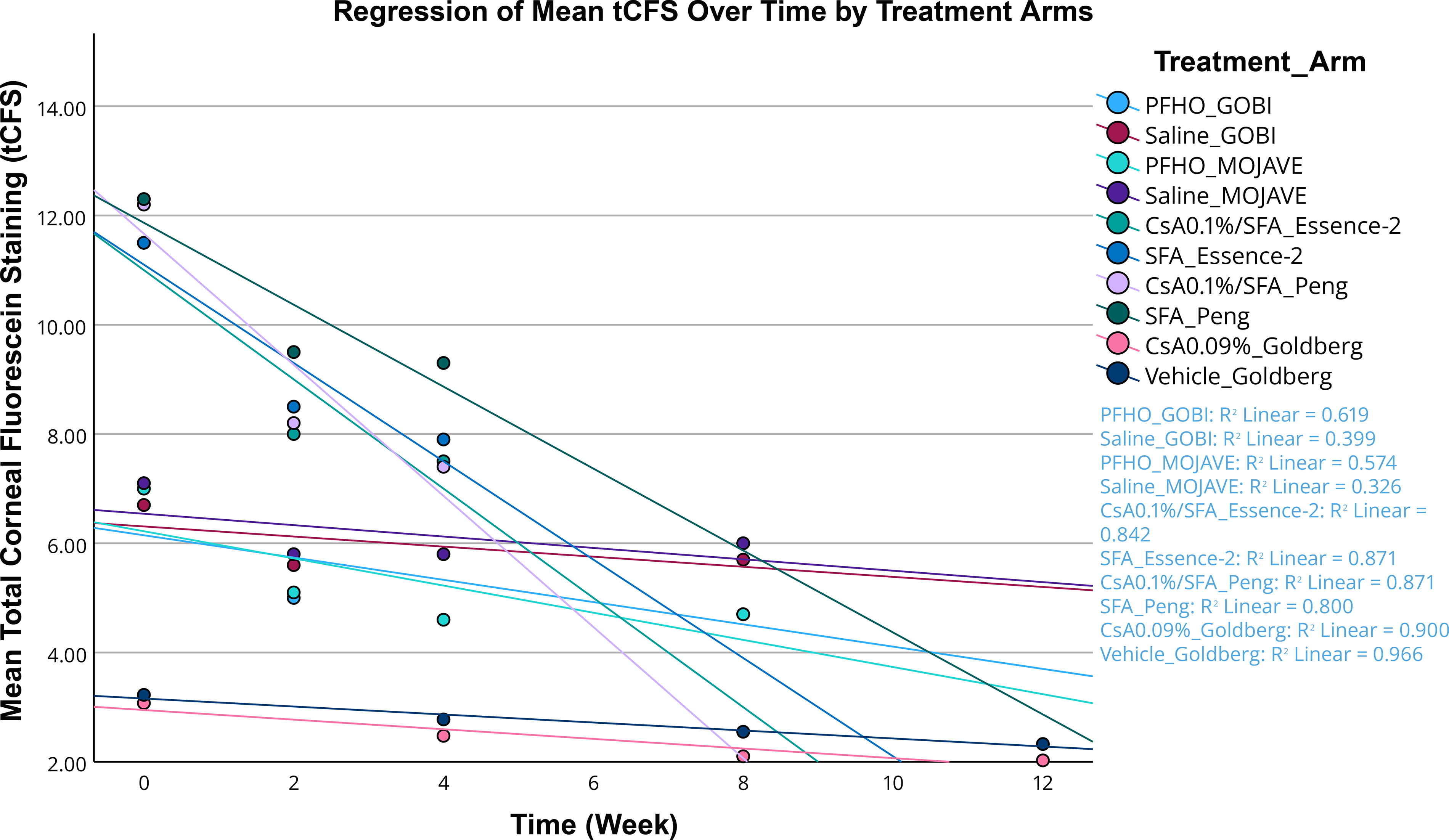
Regression analysis of total corneal fluorescein staining (tCFS) over time by treatment arms. Scatter plot with linear regression lines illustrating changes in mean tCFS scores over treatment duration across included studies. R² values indicate the strength and predictability of tCFS reduction, with cyclosporine 0.1%/SFA showing the steepest slope and highest R², reflecting greater efficacy and consistency compared with other treatments. CsA 0.1%/SFA, cyclosporine 0.1% in SFA; CsA 0.09%, cyclosporine 0.09%; PFHO, perfluorohexyloctane.
The safety profiles of cyclosporine 0.09%, lifitegrast 5%, cyclosporine 0.05%, cyclosporine 0.1%/SFA, and PFHO demonstrated notable differences in tolerability. Instillation site pain was common with cyclosporine 0.09% (15.1%–24.2%)17–19 and lifitegrast 5% (15.9%–24%),32–34 while cyclosporine 0.05% primarily reported instillation site burning (14.7%) and stinging (3.4%). 22 Lifitegrast 5% also commonly caused dysgeusia (12.9%–16.2%).32–34 In contrast, SFA-based therapies exhibited significantly better local tolerability, with considerably lower instillation site pain rates for cyclosporine 0.1%/SFA (2.5%–9.9%)23,24,27 and especially PFHO (∼1%).43,44 Overall, quantitative comparison clearly highlights the superior local tolerability of SFA-based treatments compared with traditional anti-inflammatory therapies.
Discussion
Inflammation is a fundamental driver of DED, leading to tear film instability, epithelial damage, and ocular surface dysfunction. Anti-inflammatory therapies, such as cyclosporine 0.09%, cyclosporine 0.05%, cyclosporine 0.1%/SFA, and lifitegrast 5%, target different inflammatory pathways, primarily through suppressing T-cell activation and cytokine production.16–37,50 PFHO, an SFA-based therapy, also demonstrated efficacy by stabilizing tear film.23,24,27,38–47 While significant reductions in corneal staining were observed across these treatments, the magnitude and timing of improvements varied, reflecting inherent differences in formulations, mechanisms of action, and study designs.
Corneal staining remains a critical endpoint for assessing DED severity and treatment effectiveness, given its ability to directly reflect corneal surface integrity and epithelial health.12–15 However, differences in corneal staining grading scales among studies further complicate direct comparisons of treatment efficacy. 15 In addition, variability in comparator, the control arms across studies further complicates comparative efficacy assessments. For example, cyclosporine 0.1%/SFA was compared with its own SFA-based vehicle, while PFHO was compared with saline.23,24,27,43,44 Such differences in comparator treatments may confound direct comparative conclusions.
Patient population heterogeneity also presented challenges in comparing results. Studies of cyclosporine 0.1%/SFA primarily enrolled moderate-to-severe DED patients,23,24,27 while PFHO targeted mild-to-moderate cases,43,44 and cyclosporine 0.09% included a broad range from mild to severe.17–19 Lifitegrast studies focused more broadly on symptom improvement, with inconsistent corneal staining outcomes.32–34 Thus, additional research addressing these patient population gaps would strengthen clinical applicability across the full spectrum of DED severity.
Onset of action and sustained efficacy are critical factors in DED management. Timeframe analyses highlighted important distinctions between therapies. We acknowledge that the timeframe of assessment is a critical factor influencing observed results in tCFS. Combining studies without appropriately accounting for differences in evaluation timeframes would not provide a fair or accurate comparison of therapeutic efficacy. Some treatments, notably cyclosporine 0.1%/SFA and PFHO, demonstrated rapid onset of efficacy within the first 4 weeks,23,24,27,43,44 while other therapies, including cyclosporine 0.09% and lifitegrast, emphasized later endpoints at weeks 8–12.17–19,32–34 However, efficacy beyond 12 weeks remains uncertain, although some treatments demonstrated continued benefits in extension studies lasting up to 52 weeks.25,45 Substantial heterogeneity among studies remains a limitation, driven by diverse methodologies and patient populations.
Exploratory linear regression analyses provided insights into general treatment trends, serving as a simplified yet illustrative approximation of corneal staining improvements over time. Although this method does not fully capture the complexity of therapeutic effects, it highlights important temporal dynamics.
Safety and tolerability profiles are crucial for long-term adherence and treatment success. PFHO showed excellent tolerability comparable with saline while cyclosporine 0.1%/SFA presented a notably lower rate of instillation site discomfort than traditional anti-inflammatory therapies, such as cyclosporine 0.09% and cyclosporine 0.05%.17–19,22–24,27,43,44 Conversely, lifitegrast exhibited relatively high rates of instillation-related AEs.32–34 These tolerability differences emphasize the need for balancing efficacy with patient comfort, especially for chronic therapeutic management.51–53
The introduction of various treatment options has significantly expanded the therapeutic landscape for DED, providing clinicians with tools to tailor management strategies based on the underlying etiology and severity of the condition. While these therapies show promising outcomes, notable evidence gaps remain, including the absence of robust head-to-head comparative studies and standardized evaluation methodologies. Future research should prioritize standardized evaluation methods, robust comparative data, and tailored therapeutic approaches to better address the multifactorial nature of DED.
Conclusion
This review systematically evaluated the comparative efficacy and safety profiles of topical therapies approved for DED, focusing on anti-inflammatory treatments and SFA-based therapies. Anti-inflammatory treatments, including cyclosporine 0.09%, cyclosporine 0.05%, cyclosporine 0.1%/SFA, and lifitegrast 5%, showed significant reductions in corneal staining. However, the magnitude and timeframe of these improvements varied across therapies, reflecting differences in formulations and study designs. These findings confirm their effectiveness in managing ocular surface inflammation associated with DED. SFA-based treatments, notably cyclosporine 0.1%/SFA and PFHO, demonstrated both substantial efficacy in reducing corneal staining and superior local tolerability compared with traditional anti-inflammatory therapies, marked by significantly lower rates of instillation site discomfort. However, the interpretation of these findings is constrained by variability in trial methodologies, patient populations, and corneal staining grading scales, as well as the absence of direct head-to-head comparisons among treatments. Future studies employing standardized evaluation methods and real-world data are necessary to delineate optimal therapeutic strategies further, thereby enhancing personalized patient management and outcomes in DED.
Footnotes
Authors’ Contributions
The authors, A.F., M.A., and S.P., equally contributed to the conceptualization, writing, reviewing, and editing of this article. All authors provided critical insights into the interpretation of the data and approved the final version of the article. No external writing or editorial assistance was utilized during its preparation.
Compliance with Ethics Guidelines
This review is based on previously published studies and does not involve any new studies with human participants or animals performed by any of the authors. All sources were appropriately cited, and no ethical approval was required for this work.
Disclaimer
During the preparation of this article, all content was carefully reviewed and edited by the authors to ensure accuracy, clarity, and originality. The authors take full responsibility for the content of the published article.
Author Disclosure Statement
A.S. and F.T. are employees of Harrow. The remaining authors declare no conflicts of interest, including consulting agreements, stock ownership, honoraria, paid expert testimony, patent applications, or funding from Harrow.
Funding Information
The journal fees for this review article were funded by Harrow, Inc.
