Abstract
Purpose:
To evaluate the ocular and systemic pharmacokinetics of lotilaner ophthalmic solution, 0.25%, following bilateral topical ocular administration of single and repeated doses in rabbits.
Methods:
Dutch-belted rabbits (n = 34) were administered lotilaner ophthalmic solution, 0.25%, eye drops, either in a single bilateral dose (Group 1) or twice a day bilaterally for 7 days and once on Day 8 (Group 2). The pharmacokinetics and tissue distribution levels of lotilaner were assessed following the single dose in Group 1 and the last dose in Group 2. The drug levels were examined in various ocular tissues and whole blood. The maximal concentration of the drug (Cmax), time to maximal concentration, the terminal phase elimination half-life, the area under the concentration–time curve (AUC), and total clearance of the drug were determined.
Results:
In the eyelid margins, lotilaner exhibited the highest observed concentrations at 0.25 hour (h), presenting a mean Cmax of 14,600 ng/mL in Group 1 and 20,100 ng/mL in Group 2. The highest AUC was in the eyelid margin at 242,000 h×ng/mL in Group 1 and 535,000 h×ng/mL in Group 2. In the eyelid margin, the observed clearance rate (0.634 mL/h in single dose, 0.288 mL/h in repeat dose) was the slowest among all ocular tissues in both groups, with the longest half-life of 152 h (∼6.3 days) observed in the repeat dose group.
Conclusions:
Lotilaner ophthalmic solution, 0.25%, demonstrated rapid ocular tissue absorption into the eyelid margin tissue with a long half-life of almost a week. No adverse effects were observed following topical ocular administration in Dutch-belted rabbits.
Introduction
Demodex blepharitis, a chronic eyelid margin disease, is caused by an infestation of Demodex mites. It affects approximately 55–58% of the general US population1,2 and up to 60–70% of dry eye patients.2,3 Two species of Demodex mites, Demodex folliculorum and Demodex brevis, inhabit the human skin. 4 D. folliculorum lives in the eyelash follicle, whereas D. brevis burrows into the sebaceous and meibomian glands. The mites cause damage via mechanical, bacterial, and chemical mechanisms.5,6
If left untreated, Demodex infestation can cause various ocular problems beyond blepharitis, including keratitis, meibomian gland dysfunction, eyelash disorders, conjunctival inflammation, and corneal disorders.5,7 However, until recently, there were no FDA-approved treatments for D. blepharitis, and the available management strategies were found to be largely ineffective,8–13 or provided varying levels of temporary relief.4,13–18 In 2023, lotilaner ophthalmic solution, 0.25% (Xdemvy, Tarsus Pharmaceuticals Inc., Irvine, CA), was approved by the US FDA for the treatment of D. blepharitis in humans. The two pivotal trials, Saturn-1 and Saturn-2 that evaluated lotilaner ophthalmic solution, 0.25%, showed statistically significantly higher proportions of patients achieving grade 0 collarettes (0–2 collarettes/lid), mite eradication, and erythema cure in the study group compared with the control group.19,20 No serious safety concerns were observed, and the eye drop formulation was well tolerated by study participants.
Lotilaner is a member of the isoxazoline family of compounds. It has antiparasitic properties that selectively inhibit parasite-specific γ-aminobutyric acid chloride (GABA-Cl) channels in the invertebrate nervous system. 21 In the presence of lotilaner, GABA-Cls channels are unable to open in response to GABA, enduring spastic paralysis leading to the starvation and death of Demodex mites. 21 In addition, lotilaner is unaffected by dieldrin resistance mutations and has a low IC50 value for dieldrin-resistant GABA receptors (38.25 nM), indicating that the drug is potent even at low concentrations. 22 The activity of this drug class is specific to acari neuroreceptors; the lack of effect on the mammalian nervous system at clinically relevant doses has been confirmed via in vitro testing, demonstrating that lotilaner does not inhibit mammalian GABA-Cl channels at concentrations up to 30 µM. 22
The current study presents the ocular and systemic pharmacokinetics of lotilaner ophthalmic solution, 0.25%, following single and repeat bilateral topical ocular dosing in Dutch-Belted rabbits.
Methods and Materials
Materials
The formulation of the lotilaner used in this study is the same formulation studied in the pivotal (SATURN) clinical trials leading to the FDA approval of Xdemvy.19,20 It was formulated as a sterile ophthalmic solution at a concentration of 0.25% (weight/volume %). The product was a clear, colorless solution packaged in ready-to-use, white, opaque ophthalmic bottles. It was shipped and stored at room temperature, required no additional preparation, and was used as supplied in this study.
Animal study
All animal experiments were conducted in accordance with the tenets of the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research and approved by the Institutional Animal Care and Use Committee at Powered Research, Research Triangle Park, NC.
A total of 34 male Dutch-Belted rabbits, aged 4 to 6 months old and weighing between 1.7 and 1.9 kg, were purchased from Covance, Denver, PA. All animals underwent a pre-shipment ocular assessment by the vendor. The rabbits were acclimated to the study environment for 7 days and were singly housed in stainless steel cages with free access to water and standard dry pellet rabbit feed ad libitum. At the completion of the acclimation period, each animal was physically examined by a laboratory animal technician to determine suitability for study participation. Examinations included, but were not limited to, the skin and external ears, eyes, abdomen, neurological condition, behavior, and general body condition. An additional complete ocular examination using a slit lamp biomicroscope and indirect ophthalmoscope was performed by a veterinary ophthalmologist to evaluate ocular surface morphology, anterior segment inflammation, and posterior segment findings on all animals prior to dosing to serve as a baseline.
Veterinary care was available throughout the course of the study, and animals were examined by the veterinary staff as warranted by clinical signs or other changes. All veterinary examinations and recommended therapeutic treatments were documented in the study records.
Experimental groups
There were two experimental groups in this study. In the first group, a single bilateral (OU) dose of lotilaner ophthalmic solution, 0.25% (Group 1), was administered, and the pharmacokinetics and tissue distribution levels of lotilaner were assessed following the single dose. In the second group, lotilaner ophthalmic solution, 0.25% was administered twice daily bilaterally for 7 days and once on Day 8 (Group 2), and the pharmacokinetics and tissue distribution levels of lotilaner were assessed following the last dose (Table 1). The drug levels were examined in the conjunctiva, cornea, eyelid margin, iris-ciliary body, lacrimal gland, aqueous humor, lens, and whole blood.
Dosing Schedule of Lotilaner Ophthalmic Solution, 0.25% and Time-Points for Tissue Collections in Single-Dose and Repeat-Dose Groups
Allowable time windows for timed collations are as follows: <10 min, no window; 11–59 min, ±1 min; 1–6 h, ±5 min; 7–24 h, ±7 min; 24 h or greater (Example: 48 or 72 h), ±10 min.3.
Single-dose (first/last dose) comprised two 31 μL drops with a dosing interval of 30 s.
SID, once daily; BID, twice daily; OU, both eyes; OE, ocular examination; PK, pharmacokinetic.
Drug administration and time points
Eighteen (18) rabbits were used in Group 1 to determine the ocular and systemic pharmacokinetics and tissue distribution over a 14-day period. Whole blood samples were collected prior to dosing at baseline. For each animal, a total volume of 62 μL of lotilaner ophthalmic solution, 0.25% was administered to the superior limbal area of the globe of each eye as two 31 μL drops with a dosing interval of 30 s. Sample collection of ocular tissues for pharmacokinetics was performed after euthanizing two animals at each of the following time points post-dose: 0.25, 2, 6, 24, 48, 72, 120, 168, and 336 h. Sample collection of whole blood for pharmacokinetics was performed in all surviving animals (number of animals decreased by 2 at each consecutive time point) at the following time points post-dose: 0.25, 2, 6, 24, 48, 72, 120, 168, and 336 h.
In Group 2, an additional 16 rabbits were used to evaluate the pharmacokinetics and tissue distribution of lotilaner after topical administration. The animals received a volume of 31 μL of lotilaner ophthalmic solution, 0.25% to the superior limbal area of the globe of each eye twice a day, 12 h apart, on Days 1–7 and once on Day 8. Whole blood pharmacokinetic samples were collected from all animals at baseline, 6 h after the first dose on Days 1 and 4, and pre-dose on Day 8. Terminal sampling of the ocular surface tissues for drug concentrations was performed after euthanizing two animals at each of the following time points, following the last dose on Day 8: 0.25, 6, 24, 72, 168, 240, 336, and 672 h. Sample collection of whole blood for pharmacokinetics was performed in all surviving animals (number of animals decreased by 2 at each consecutive time point) at the following time points following the last dose on Day 8: 0.25, 6, 24, 72, 168, 240, 336, and 672 h.
During instillation, lotilaner ophthalmic solution, 0.25%, was administered via a calibrated pipette at a volume of 31 μL per instillation. Care was taken not to touch animals’ eyes with the pipette tip. A new pipette tip was used for each instillation. While the eye was gently held open, the dosing formulation was dispensed onto the globe of the eye (superior limbal area) and allowed to flow across the ocular surface, with excess material flowing into the fornix. The upper and lower eyelids were gently held open for approximately 5 s to prevent blinking. Instillation was then repeated on the contralateral eye. Upon completion of dosing, the animal was returned to its cage.
Sample collection
For blood sampling at the predetermined sample collection time points, rabbits had at least 0.5 mL of whole blood drawn from the marginal ear vein. After collection, the tubes were gently mixed by inverting the tubes 5 to 8 times. Blood samples were stored on wet ice prior to freezing. Within 20 min of the blood collection time, the sample was snap-frozen and stored at −80°C until processed for analysis.
Approximately 10–15 min prior to the predetermined terminal ocular tissue sample collection time points, two animals were tranquilized with 50 mg/kg ketamine/10 mg/kg xylazine administered intramuscularly. The animals were euthanized at each specific time point with an overdose of sodium pentobarbital administered intravenously, followed by auscultation to ensure death. After euthanasia, conjunctivas (palpebral and bulbar) were collected, and both eyes were enucleated, followed by extraction/removal of superior and inferior eyelids, which were snap-frozen in liquid nitrogen. The maximal volume of aqueous humor from both eyes was removed via a 27- or 30-gauge syringe and snap-frozen by immersing in liquid nitrogen. The remaining eye was snap-frozen in liquid nitrogen. The eye tissues (cornea, conjunctiva [bulbar and palpebral], lens, iris ciliary body, and main lacrimal gland) were dissected from the frozen eye and refrozen in liquid nitrogen. All samples were placed into individual vials and weighed. Samples were stored at −80°C until concentration analysis.
The blood and tissue samples were assayed separately for each eye using a liquid chromatography-tandem mass spectrometry (LC/MS/MS) method on a Sciex API 4000 triple quadrupole mass spectrometer with a Turbo Ionspray source. Lotilaner was originally developed as a flea and tick medication for companion animals, and the bioanalytical assays developed to support the indication were developed in whole blood to best predict the effect on the target organism.
The method was linear from a lower limit of quantification of 1 ng/mL to an upper limit of quantification) of 5000 ng/mL. Chromatographic separation was conducted with a Kinetex 2.6 µm XB-C18, 50 × 2.1 mm column with mobile phases of 0.1% formic acid in water (A) and 0.1% formic acid in acetonitrile (B). The method utilized fluralaner as an internal standard, and the quantitative results were based on the lotilaner/fluralaner peak-area ratio. Sample extraction was based on a 20.0 µL sample or sample homogenate protein precipitation approach using 100 µL of acetonitrile containing 250 ng/mL of internal standard, vortexed and centrifuged at 4,000g for 10 min in a 4°C refrigerated centrifuge, 100 µL of supernatant was dried down under heated nitrogen and reconstitution with 100 µL of 40% mobile phase A, 60% mobile phase B. Concentration analyses were performed with separate standard curves for each matrix. The whole blood was analyzed without homogenization or dilution. Aqueous humor samples were analyzed without homogenization and quantitated against a phosphate-buffered saline standard curve. Ocular tissues were homogenized 10x with rabbit plasma prior to sample extraction. An additional 10x dilution with rabbit plasma was performed for 0.25–72 h post-dose for the superior palpebral conjunctiva and superior bulbar conjunctiva samples prior to the assay to ensure they were within the quantitative range.
Pharmacokinetic data analysis
Pharmacokinetic (PK) parameters of lotilaner were derived with a sparse sampling noncompartmental analysis (NCA) using Phoenix WinNonLin (Certara USA, Inc. Princeton, NJ) and the linear up/log down trapezoidal rule. The noncompartmental PK parameters reported were the time to maximal concentration (Tmax), the terminal phase elimination half-life (T½), the maximal concentration of the drug (Cmax), and the area under the concentration–time curve from the last administered dose to the last observed quantifiable concentration (AUClast). In addition, the apparent total clearance of the drug (CL/F) was also evaluated. T½ was reported if the percent extrapolated AUC was <20%, except for the iris-ciliary body, which was <25%. The R2 of the linear regression for T½ was between 0.576 and 1.00 for reported matrices.
Nominal time points (in hours) were used in the concentration–time data analysis. For NCA, drug concentrations below the limit of quantitation (BLQ) were set to zero. Time points occurring after the first BLQ sample were set to missing and not used in the calculations. Parameters are reported as the mean, with the standard error of the mean reported for Cmax and AUClast.
Results
The mean concentration–time profile of lotilaner in the blood, conjunctiva (regions combined), cornea, eyelid margins, aqueous humor, iris ciliary body, lens, and lacrimal gland is summarized in Table 2 and Figures 1 and 2. The inferior and superior eyelid margins are plotted separately for the single- and repeat-dose groups in Figure 3.
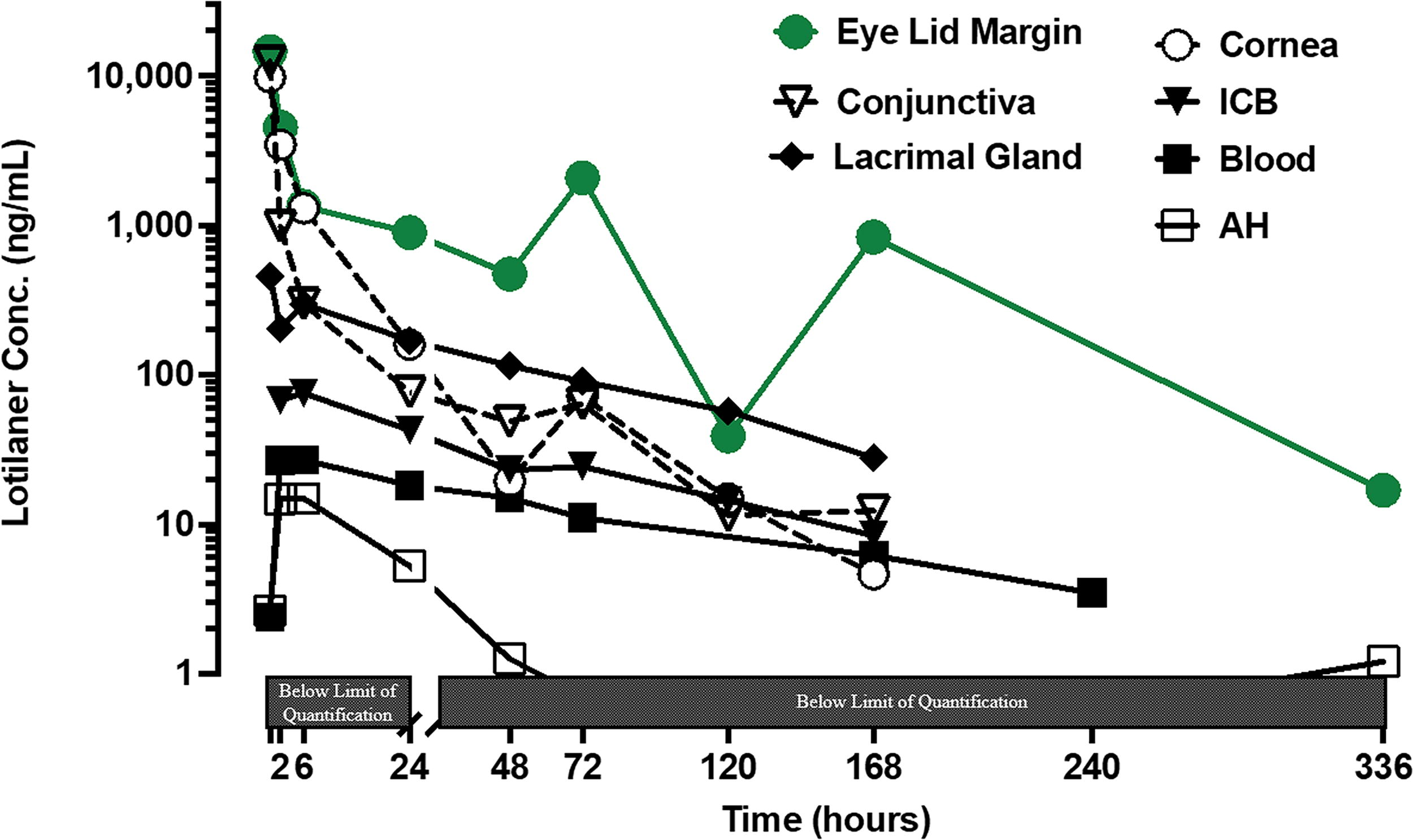
Pharmacokinetic profile of lotilaner ophthalmic solution, 0.25%, in ocular tissues and blood after administration of a single dose (62 µL). Concentrations were measured in the conjunctivas, eyelid margins, cornea, iris ciliary body (ICB), lacrimal gland, aqueous humor (AH), and blood. Concentration profiles for ocular tissue were generated from 2 rabbits (4 eyes) per time point. Data are shown as the mean. Data below the limit of quantification (<1 ng/mL) is not shown. The scale of the X-axis [Time (hours)] was split at the 24 h time point: The first part of the X-axis is set for 0 to 24 h and is 15% of the axis, and the second part of the X-axis is 24 to 336 h.
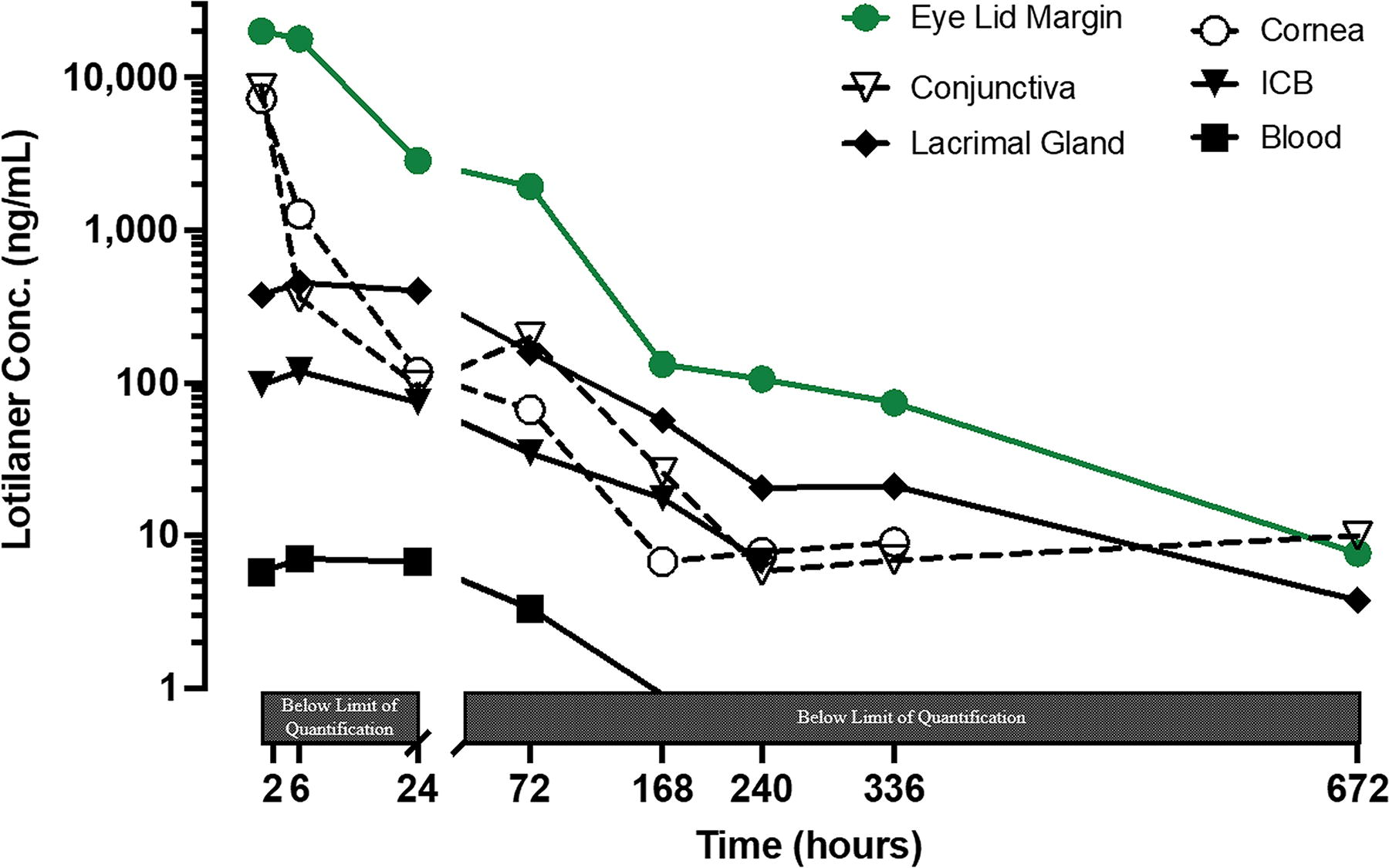
Pharmacokinetic profile of lotilaner ophthalmic solution, 0.25%, in ocular tissues and blood after a 7-day twice daily administration (31 µL per dose) and a single dose Day 8. Concentration was measured in conjunctivas, eyelid margins, cornea, iris ciliary body (ICB), lacrimal gland, and blood. Concentration profiles for ocular tissue were generated from 2 rabbits (4 eyes) per time point. Data are shown as the mean. Data below the limit of quantification (<1 ng/ml) is not shown. The scale of the X-axis [Time (hours)] was split at the 24 h time point: The first part of the X-axis is set for 0 to 24 h and is 15% of the axis, and the second part of the X-axis is 24 to 672 h.
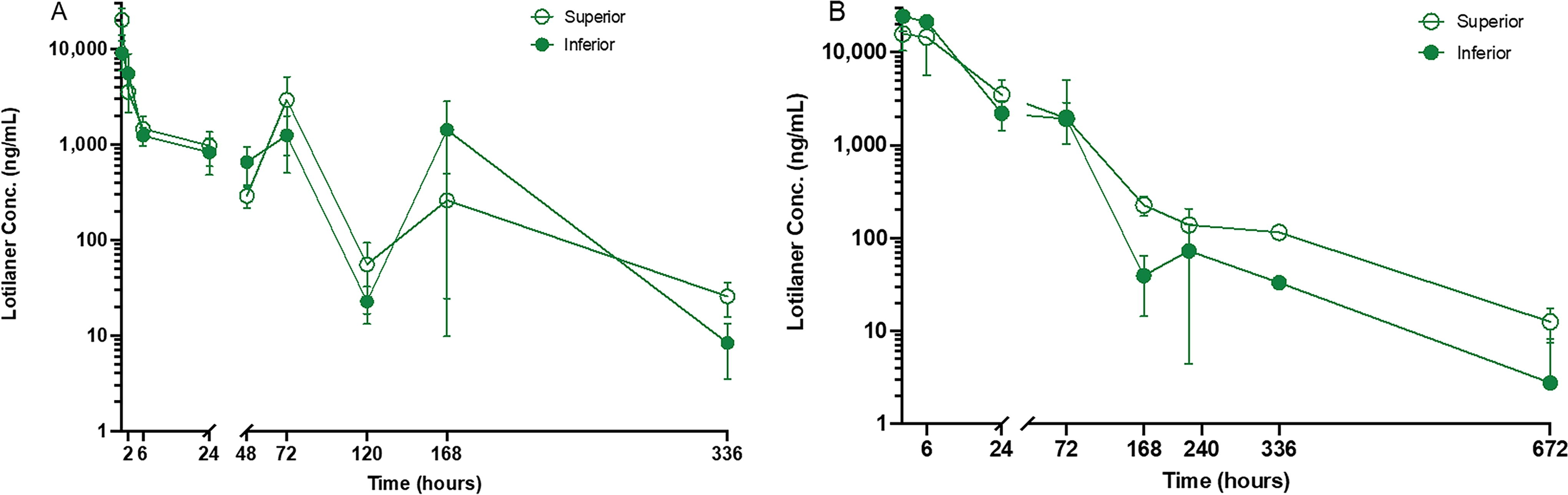
Pharmacokinetic profile of lotilaner ophthalmic solution, 0.25%, in eyelid margin after
Mean ± SEM Pharmacokinetic Paramseters of Lotilaner Ophthalmic Solution, 0.25% in Dutch-Belted Rabbits after Either Single-Dose or Repeated Dose Administration
AUClast, area under the concentration–time curve from the last administered dose to the last observed quantifiable concentration; CL/F, total clearance of the drug; Cmax, maximal concentration of the drug; Tmax, time to maximal concentration; SEM, standard error of the mean; —, no data; NR. not reported (does not fit accepted criteria).
Lotilaner concentration in the eyelid margins, conjunctivas, cornea, lacrimal gland, and iris-ciliary body
In the eyelid margins and conjunctivas, lotilaner exhibited the highest observed concentrations as early as 0.25 h (the first observed time point), presenting a mean Cmax of 14,600 ng/mL and 12,500 ng/mL, respectively, in the single-dose Group 1 and Cmax of 20,100 ng/mL and 9,630 ng/mL, respectively, in the repeat-dose Group 2 (Table 2 and Figs. 1 and 2). Observed concentrations of lotilaner were less abundant in the cornea and were substantially lower in the lacrimal gland and iris-ciliary body. A biphasic decline in exposure to lotilaner in ocular tissues was observed in both experimental groups (Figs. 1 and 2). In the target tissue of the eyelid margin, the observed clearance rate was the slowest among all ocular tissues in both groups (Table 2). Correspondingly, lotilaner remained quantifiable up to 336 h post-dose in the single-dose group and up to 672 h post-dose in the repeat-dose group (the last time points sampled in each group). There was no substantial difference in exposure of lotilaner between the inferior and superior eyelid margins (Fig. 3). The half-life of lotilaner in the eyelid margin was 152 h (∼6.3 days) and had a high degree of accuracy since the extrapolated area was 0.623% and the R2 was 0.999 (data not tabulated).
In the single-dose group, the total exposure in ocular tissues based on AUClast was highest in the eyelid margin (242,000 h×ng/mL) and lowest in the aqueous humor (757 h×ng/mL). The rank order of exposure was eyelid margin>cornea>conjunctiva>lacrimal gland>iris-ciliary body>aqueous humor. The rank order of clearance was reversed to AUClast. In the repeat-dose group, the total exposure in ocular tissues based on AUClast was highest in the eyelid margin (535,000 h×ng/mL) and lowest in the iris-ciliary body (8660 h×ng/mL) since aqueous humor was not analyzed. The rank order of exposure was slightly different from the single dose at eyelid margin>conjunctiva>cornea>lacrimalgland>iris-ciliary body. The rank order of clearance was reversed to AUClast.
There was an approximately 2-fold increase in exposure from the single-dose to the repeat-dose groups for lotilaner in the eyelid margin and a slightly more than twofold increase in the lacrimal gland. There was not much difference in exposure (based on AUClast) between the groups for the cornea.
Lotilaner concentration in aqueous humor and lens
Concentration data for lotilaner in aqueous humor and lens were collected in Group 1 animals and were summarized in Table 2 and Figure 1. Following a single instillation of lotilaner ophthalmic solution, 0.25%, the concentration was relatively low in the aqueous humor, with a mean peak concentration of Cmax 14.9 ng/mL at the 6 h time point. In the lens, lotilaner concentration was not detectable. Therefore, the concentration of lotilaner in the lens and aqueous humor were not studied in Group 2.
Lotilaner concentration in whole blood
Following a single administration of 62 µL dose in Group 1 animals, lotilaner was detected at the first time point (0.25 h) and was systemically absorbed with Tmax occurring at 6 h post-dose and a Cmax of 26.9 ng/mL (Table 2). Lotilaner was detectable in the blood for at least 168 h post-dose and was BLQ at 336 h (14 days) post-dose.
In Group 2, lotilaner was not detected in the blood of 6 animals (50% of the tested population) 6 h post dose on Day 1 and was present in the blood of all animals at other time points thereafter through 72 h post last dose. At 168 h post last dose, 5/8 rabbits had reportable lotilaner blood concentration, and by 240 h, all blood samples were below the limit of quantitation. The concentration of lotilaner in blood at 6 h post-dose on Days 1, 4, and 8 was 0.903 ng/mL, 5.84 ng/mL, and 7.06 ng/mL, respectively. Day 8 pre-dose mean lotilaner concentration was 6.22 ng/mL, similar to the concentration 6 h post-dose on Days 4 and 8. Following topical dosing on Day 8, the concentration of lotilaner in the blood slightly increased through 6 h post-dose with a Cmax of 7.06 ng/mL at 6 h before declining. Lotilaner concentration was undetectable in the blood samples 240 h (10 days) after the last dose.
In both experimental groups, a linear mono-exponential decline in concentration occurred through 168 h (7 days) after the last drug administration was observed. The mean calculated lotilaner half-life in blood was 57.7 h in Group 1 animals and 57.3 h in Group 2 animals. The accuracy of the half-life was high with a linear regression R2 of 1.00 and 0.995 for the single- and repeat-dose groups; both groups had a percent extrapolated area of 13.2%.
Safety and tolerability
All animals remained bright, alert, and responsive, and no veterinary care or therapeutic treatments were required for the duration of the study. No other adverse events were recorded. No morbidity or mortality was noted for any animal in any group during this study. Animals were humanely euthanized at the sample collection time points described in Table 1 (n = 2 animals per time point).
Discussion
Lotilaner is an isoxazoline compound that underwent human clinical trials for topical treatment of D. blepharitis.19,20,23–25 In the present study, the pharmacokinetic profile of lotilaner ophthalmic solution, 0.25% was studied to understand the drug’s tissue and blood distribution. In this study, we observed that a single-dose administration of lotilaner ophthalmic solution, 0.25%, led to high concentrations of the drug in the eyelid margins, conjunctiva, and cornea as early as 0.25 h post-administration and persisted for 168 to 336 h. Likewise, the repeat dose of one drop twice a day, 12 h apart for 7 days and a single dose on Day 8, had a similar trend post-last dose, with lotilaner detectable to at least 336 h.
Ocular administration of lotilaner resulted in the distribution of the drug into ocular tissues of interest and a relatively low concentration in the blood. Lotilaner has a high log P and is, therefore, more soluble in lipophilic/organic media than in aqueous. These physiochemical properties suggest that lotilaner is more likely to be distributed into and remains longer in ocular tissues that are more lipophilic, such as the eyelid margin (e.g., eyelash follicle and meibomian gland) where there is more fat and muscle, than into other ocular tissues. In vitro development studies have demonstrated that lotilaner is bound to rat, dog, and human plasma at >99.9%. 26
Study data suggest that among different eye tissues, the eyelid margin had both the largest exposure and one of the longest half-lives after repeat administration. The exposure of lotilaner measured as the AUClast in the eyelid margin in Group 1 (single dose) and Group 2 (repeat dose) was 242,000 h×ng/mL and 535,000 h×ng/mL, respectively, which represents approximately 5–10 times more concentration of the drug in the eyelid margin than other ocular surface tissues (cornea and conjunctiva). These results indicate the drug preferentially accumulates in the target tissue of the eyelid follicle where Demodex mites reside.
A biphasic distribution and clearance in the ocular tissues was observed in both experimental groups. A particularly pronounced biphasic distribution was observed in the less lipophilic conjunctiva and cornea tissues that presented high initial concentrations at 0.25 h and quickly dropped thereafter, following multiple administrations of lotilaner. The exposure of lotilaner to peri/intraocular tissue, including the lacrimal gland, aqueous humor, lens, and iris-ciliary body, was substantially lower.
The clearance rate of lotilaner was slowest in the eyelid margin (0.634 mL/h in the single-dose group, 0.288 mL/h in the repeat-dose group). This slower clearance may lead to a higher accumulation in the eyelid margin upon repeat dosing than in the other ocular tissues. Correspondingly, the T½ of lotilaner in the eyelid margin was one of the longest among all ocular tissues. As such, lotilaner remained quantifiable in eyelid margin tissue at a much higher concentration (up to 336 h post-dose in the single-dose group and up to 672 h post-last dose in the repeat-dose group) than other ocular tissues (Figs. 1 and 2). These concentration data indicate that lotilaner may have a higher preference for the eyelid margin tissue compared with the other ocular tissues.
Following repeat administration of lotilaner ophthalmic solution, 0.25%, twice daily over 7 days, it was not completely eliminated between doses, demonstrating a drug-accumulating effect following multiple administrations. However, the T½ of lotilaner in blood was short (57.7 h in the single dose and 57.3 h in the repeat-dose group [versus approximately 2.5 days to the 6.3 days in the eyelid margin]), suggesting a quick and consistent clearance of lotilaner from blood in both treatment groups. The concentration of lotilaner in the blood was detectable up to 7 days after the last dose in both groups and was not detectable at the next time point of measurement (at 14 days in the single-dose group and 10 days in the repeat-dose group). These results indicated that the drug excreted from blood after 7 days for both single- and repeat-dose groups while it still remained in the eyelid margin.
Since eye ointments are much more viscous than eye drops, eye ointments are believed to have higher residence time, producing high concentrations in the target tissue. Eye ointment is the preferred mode of drug administration for lid margin disease. However, ointments can cause blurry vision or a sticky sensation, negatively affecting their acceptability and compliance among patients. Given that lotilaner has a high log P and is more soluble in lipophilic/organic media,21,27 it was expected to reach fat-rich ocular tissues, like the eyelid margin and meibomian glands, irrespective of its mode of administration as drops or ointment. The present study demonstrates that lotilaner 0.25%, administered as an ophthalmic solution, was readily absorbed into the eyelid margin as early as 15 min after dosing (Tmax of 0.25 h). The concentration of the drug almost doubled in the eyelid margin with the repeat dosing and was detectable up to 28 days even after treatment cessation.
Conclusion
Lotilaner, as an ophthalmic solution, has unique pharmacokinetic properties that allow it to accumulate in the target tissue and thus provide greater exposure in disease treatment while maintaining lower exposure in surrounding ocular tissues. Since sampling human ocular tissues is not feasible, rabbit ocular PK data provides pharmacokinetic evidence that lotilaner is present at high concentrations in the eyelid margin where Demodex mites are located. The concentrations were substantial after a single dose through 14 days and after repeating dosing through the last observed time point at 28 days after treatment cessation.
Footnotes
Acknowledgments
Editorial assistance in the preparation of this article was provided by Jan Beiting (Wordsmith Consulting, Cary, NC) and Raman Bedi, MD (IrisARC-Analytics, Research & Consulting, Chandigarh, India) and funded by Tarsus Pharmaceuticals, Inc., Irvine, CA, USA.
Authors’ Contributions
C.S.C.: Conceptualization (equal); data curation (lead); formal analysis (lead); investigation (lead); methodology (equal); project administration (equal); resources (lead); software (lead); supervision (equal); validation (lead); visualization (lead); writing—original draft (lead); writing—review and editing (equal). E.Y.: Supervision (equal); visualization (supporting); writing—original draft (supporting); writing—review and editing (equal). S.M.: Supervision (equal); visualization (supporting); writing—original draft (supporting); writing—review and editing (equal). S.N.: Conceptualization (equal); data curation (supporting); formal analysis (supporting); methodology (equal); project administration (equal); supervision (equal); validation (supporting); visualization (supporting); writing—review and editing (equal). All authors approved the final version of the submitted article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
Author Disclosure Statement
C.C. and S.M. are consultants to
Funding Information
This study was funded by
