Abstract
Purpose:
To compare intraocular pressure (IOP), ocular surface disease (OSD) parameters, and safety in patients with open-angle glaucoma (OAG)/ocular hypertension (OH) and concurrent OSD treated with preservative-free latanoprost 0.005% cationic emulsion (PF-latanoprost-E) or travoprost-Z 0.004% ophthalmical solution containing a soft preservative system.
Methods:
Patients with OAG/OH and OSD were randomized to treatment with PF-latanoprost-E or travoprost-Z nightly for 3 months. Outcomes included mean diurnal IOP reduction; OSD endpoints, including symptom improvement, tear break-up time (TBUT), and corneal fluorescein staining (CFS) score; and safety after 1 and 3 months.
Results:
A total of 105 patients were randomized, 51 to PF-latanoprost-E and 54 to travoprost-Z. IOP reductions (LS mean differences) at 3 months were numerically greater in the PF-latanoprost-E than in the travoprost-Z group at 8AM (7.2 versus 6.0 mmHg), 10AM (6.7 versus 5.9 mmHg), and 4PM (6.0 versus 5.4 mmHg). LS mean changes in IOP from baseline in both groups at 1 and 3 months, however, were comparable. Mean ± SD CFS scores on the Ora scale at month 3 showed significantly greater reductions in the PF-latanoprost-E than in the travoprost-Z group (–1.07 ± 1.863 versus –0.16 ± 2.553 P = 0.0461). The mean TBUT at month 3 showed similar improvements in both groups (1.1 versus 1.0 s, P > 0.05). OSD symptoms improved but did not differ significantly in the two groups. Overall safety was comparable in both groups.
Conclusion:
PF-latanoprost-E effectively and safely lowered IOP and improved OSD parameters in patients with OAG/OH. These findings provide evidence for the beneficial effects of this new formulation of latanoprost in glaucoma patients with OSD.
Introduction
Glaucoma, characterized primarily by an increase in intraocular pressure (IOP), is the leading cause of irreversible blindness worldwide, affecting an estimated 76 million people in 2020. 1 The prevalence of glaucoma increases significantly with age, affecting an estimated 0.6% of individuals aged 40–49 years and an estimated 8.3% of individuals aged ≥80 years. 2 The goal of glaucoma therapy is the reduction of IOP, with topical ophthalmical treatments being the current standard of care for glaucoma. Although these treatments effectively reduce or prevent glaucoma progression, 3 30–70% of patients with glaucoma and ocular hypertension develop ocular surface disease (OSD) during treatment.4–7
OSD, including dry eye disease, is a spectrum of chronic, potentially debilitating anterior segment conditions. 8 OSD prevalence is affected by age, gender, systemic conditions, and topical and systemic medications. 8 OSD is difficult to diagnose due to the mismatch between objective signs and subjective symptoms.9,10 Damage to the ocular surface can range from mild to severe and is likely due to chronic inflammation associated with tear film instability and hyperosmolarity. 11 The high prevalence of OSD in patients using topical glaucoma therapy has been linked to preservatives in eyedrops, specifically benzalkonium chloride (BAK), which is present in approximately 70% of all ophthalmical formulations.12–14
OSD has been found to adversely affect the quality of life of patients with glaucoma,15,16 with medications containing BAK having greater adverse effects than BAK-free formulations. 17 The side effects of glaucoma medications can result in nonadherence to treatment,18,19 increasing the risk of glaucoma progression.20–23 Thus, eyedrops without preservatives or those containing preservatives other than BAK can prevent or ameliorate symptoms of OSD in patients with glaucoma or ocular hypertension.12,13
Studies in animal models have shown that preservative-free latanoprost 0.005% cationic emulsion (PF-latanoprost-E, Catiolanze®, Santen SAS) lowers IOP as effectively as preserved latanoprost 0.005% solution (P-latanoprost; Xalatan®; Pfizer), which contains 0.02% BAK.24–26 Compared with P-latanoprost, however, PF-latanoprost-E demonstrated better tolerance. PF-latanoprost-E consists of a proprietary cationic oil-in-water emulsion (Santen SAS) that improves the signs and symptoms of dry eye disease and serves as a drug delivery system for the prodrug latanoprost.27,28 This cationic emulsion is a patented eye drop formulation platform developed to optimize the interaction of the eye drop (i.e., the cationic emulsion) with the different layers of the tear film, mainly with the tear film lipid layer, and the ocular surface. 27 This multicenter, randomized phase II trial therefore compared the effects of PF-latanoprost-E 0.005% with those of a BAK-free travoprost 0.004% ophthalmical solution containing a soft preservative system (Travatan®Z, Alcon) on IOP reduction and on OSD parameters in patients with glaucoma or ocular hypertension and concurrent OSD.
Patients and Methods
This multicenter, randomized, investigator-masked, active-controlled phase II study was performed at 10 sites within the United States. The study protocol was approved by all relevant ethics committees, and the trial was registered at ClinicalTrials.gov (NCT01254370). The study was conducted in accordance with the tenets of the Declaration of Helsinki, and all subjects provided written informed consent to participate.
Inclusion criteria
This study enrolled adults aged ≥18 years with ocular hypertension, open-angle glaucoma (with or without pseudoexfoliation or pigment dispersion), or chronic angle-closure glaucoma with a patent iridotomy requiring IOP-lowering therapy and able to wash out any current IOP-lowering therapy, defined as described, 29 as well as OSD. 8 The study eye was required to have a best-corrected visual acuity (BCVA) of 1.0 logMAR (20/200 Snellen equivalent) at screening and baseline, IOP ≥22 mmHg at hour 0 at the baseline visit, an ocular discomfort score on the Ora Ocular Symptomology Scale (0 least severe, 5 most severe) of ≥2 at screening and baseline, a tear break-up time ≤10 s at screening and baseline, and a corneal fluorescein staining (CFS) score ≥1 on the modified Oxford scale at screening.
Exclusion criteria
Patients were excluded if they had known sensitivity or poor tolerance to topical prostaglandin analogues, an IOP >34 mmHg at screening or baseline, a central corneal thickness >620 µm in either eye, any ocular abnormality precluding accurate Goldmann tonometry, a recent history of ocular trauma or laser or refractive eye surgery, a recent ocular infection or inflammation, or were pregnant or planning to become pregnant.
Definition of study populations
The full analysis set (FAS) population was defined as all randomized subjects, for whom at least one efficacy evaluation under treatment was available. The per-protocol (PP) population was defined as all subjects in the FAS who did not have any major protocol deviations. The safety population was defined as all randomized subjects for whom there was evidence of study medication use.
Study protocol
The study consisted of four visits over 12 weeks. The screening visit included symptom questionnaires; slit-lamp biomicroscopy, including assessments of bulbar conjunctival hyperemia and blepharitis; measurements of BCVA, IOP, tear film break-up time (TFBUT), and CFS; corneal ultrasound pachymetry; and dilated fundus examination. Eligible patients subsequently underwent washout of any topical IOP-lowering medications for 2–4 weeks. Each patient was given an artificial tear formulation and a dosing diary. Patients were instructed to use artificial tears only if needed for ocular discomfort throughout the remainder of the study and not within 30 min of taking the study medication.
The baseline visit occurred 2–5 weeks after screening and included symptom questionnaires; measurements of BCVA, diurnal IOP (at 8AM, 10AM, and 4PM), TFBUT, and CFS; and slit-lamp biomicroscopy. The patients were randomized 1:1 to treatment with PF-latanoprost-E or travoprost-Z, each dosed once daily in the evening. All patients were evaluated on treatment at 1 and 3 months, using the same assessments as at baseline, as well as by dilated funduscopic examination.
Analytic methods
BCVA was assessed using the Early Treatment of Diabetic Retinopathy Study chart. IOP was measured using Goldmann tonometry and the two-person operator/recorder approach, with two measurements at each time point, and a third if the first two differed by >2 mmHg. CFS was graded using the modified Oxford scale, ranging from 0 (“no staining”) to 5 (“severe staining”), and by the Ora scale (0–4), with 0, 1, 2, 3, and 4, indicating no staining, occasional staining, countable staining, uncountable but not confluent staining, and confluent staining, respectively. Bulbar conjunctival hyperemia was graded on the McMonnies scale (1–6). In evaluating blepharitis, plugging was graded from 0 (none) to 3 (severe). Lid margin redness was graded using the Ora scale from 0 (none) to 3 (severe”), and lid swelling was graded from 0 (none) to 4 (very severe). Ocular symptoms were graded using the Ora Ocular Symptomology Scale, which assesses ocular discomfort, burning, dryness, grittiness, and stinging over the previous week on scales of 0 (least severe) to 5 (most severe). OSD symptoms in study participants were recorded before the 8AM measurement of IOP.
Study objectives
The primary efficacy objectives were to compare the effects of PF-latanoprost-E and travoprost-Z on IOP reductions at each time point (8AM, 10AM, and 4PM) at 4 and 12 weeks and differences in OSD signs and symptoms (including global symptoms, as determined by Ocular Symptomology Scale scores, and TFBUT, CFS, and bulbar conjunctival hyperemia). The primary safety objectives were to compare BCVA, the results of dilated fundus examinations, slit-lamp biomicroscopy, and adverse events in the two groups.
Calculation of sample sizes
Based on estimates from a previous study (Santen, unpublished results), a sample size of 45 patients per group was calculated to have approximately 82% power to detect a true mean difference of 1.1 s, assuming a standard deviation of 1.8 s for TFBUT at the 0.05 significance level. For CFS, a sample size of 45 patients per group was calculated to have approximately 83% power to detect a true mean difference of 0.5 U (half a grade), assuming a standard deviation of 0.8. Allowing for a 10% premature withdrawal rate, it was planned to enroll 50 patients per group.
Other statistical analyses
If both eyes of a patient qualified, the worse eye, defined as the eye with the greater CFS score (modified Oxford scale) at baseline, was analyzed, using the last observation carried forward method. Changes from baseline were determined by analysis of covariance models with treatment and CFS strata (baseline CFS ≤1 versus >1) as fixed factors and baseline values as covariates. Least square means were calculated, and 95% confidence intervals were constructed around these estimates.
Results
Patient characteristics
Overall, 105 patients were randomized, 51 to PF-latanoprost-E and 54 to travoprost-Z. These patients constituted both the safety and FAS populations. Of these 105 patients, 103 completed the week 4 visit and 102 completed the week 12 visit. All three noncompleting patients were in the travoprost-Z group and discontinued due to adverse events; a fourth patient in this group used a disallowed medication. These four patients were excluded from the PP population, which consisted of 101 patients, 51 randomized to PF-latanoprost-E, and 50 to travoprost-Z.
Analysis of the FAS showed that the baseline demographic and ocular characteristics of the PF-latanoprost-E and travoprost-Z groups were well balanced (Table 1). The 105 patients consisted of 71 (67.6%) women and 34 (32.4%) men of mean ± SD age 64.6 ± 9.1 years. Of these patients, 63 (60%) were white, 41 (39%) were African American, and one (1.0%) was a native Hawaiian or Pacific Islander. In addition, 69 (65.7%) had brown, 24 (22.9%) had blue, nine (8.6%) had hazel, and three (2.9%) had green irises. Moreover, 63 (60%) had baseline CFS scores ≤1, and 42 (40%) had baseline CFS scores >1. Similar results were observed in the PP population (data not shown).
PF-latanoprost-E, preservative-free latanoprost 0.005% cationic emulsion; travoprost-Z, travoprost-Z 0.004% ophthalmical solution containing a soft preservative system; CFS, corneal fluorescein staining; TFBUT, tear film break-up time.
IOP endpoints
Mean IOPs at 8AM, 10AM, and 4PM were lower than baseline at months 1 and 3 in both groups (Fig. 1). At 3 months, LS mean IOP reductions in the PF-latanoprost-E and travoprost-Z groups were 7.2 mmHg and 6.0 mmHg, respectively, at 8AM; 6.7 mmHg and 5.9 mmHg, respectively, at 10 AM; and 6.0 mmHg and 5.4 mmHg, respectively, at 4PM. The differences between these groups were not statistically significant, indicating that the IOP-lowering effect of PF-latanoprost-E was noninferior to that of travoprost-Z. Similar results were observed in the PP population (Supplementary Table S1).
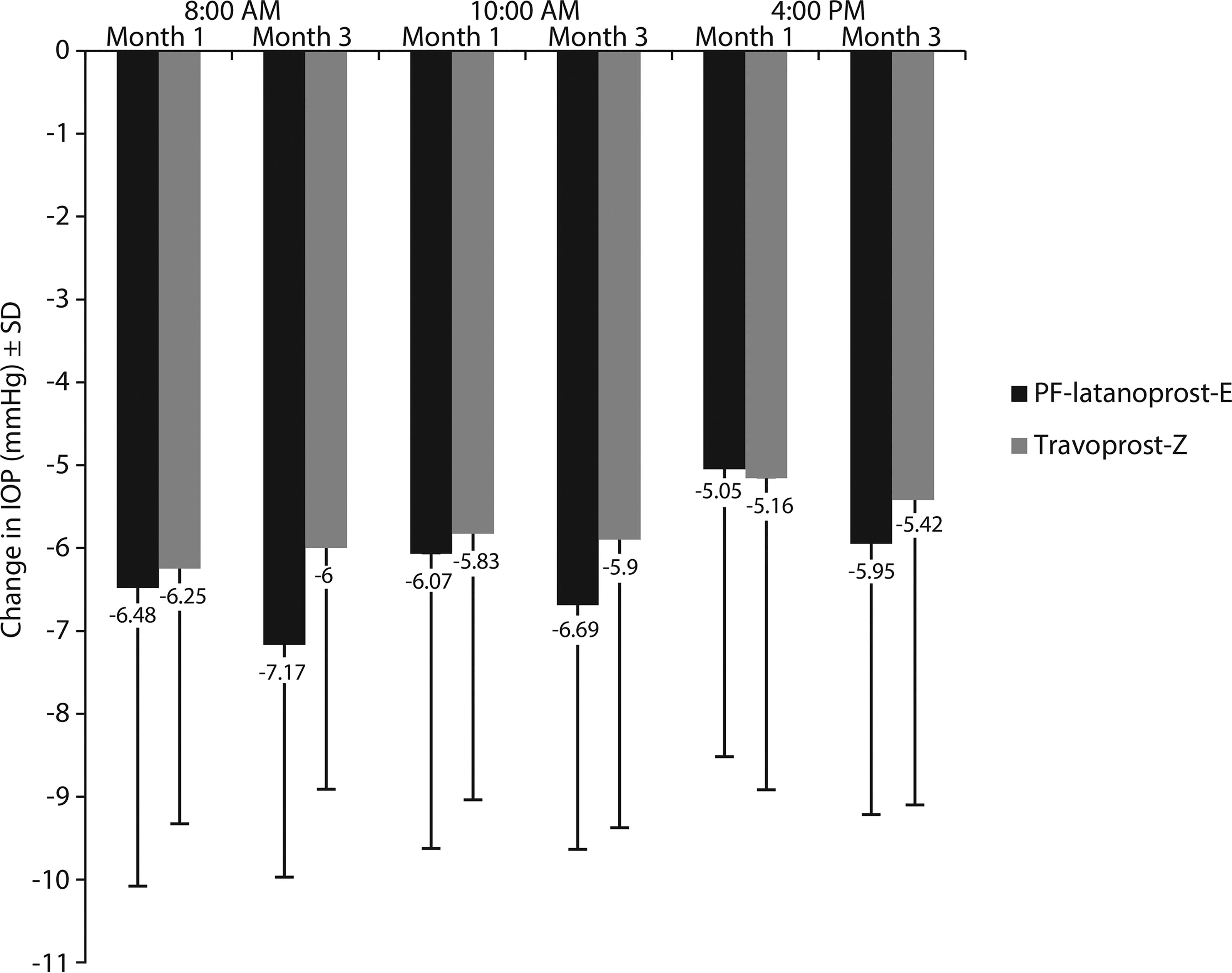
Changes in mean IOP in the worse eye at each diurnal time point from baseline to months 1 and 3 by treatment group (full analysis set with last observation carried forward). Abbreviations: PF-latanoprost-E, preservative-free latanoprost 0.005% cationic emulsion; travoprost-Z, travoprost-Z 0.004% ophthalmical solution containing a soft preservative system.
OSD endpoints
Mean baseline Ora Ocular Symptomology Scale scores, TFBUT, bulbar conjunctival hyperemia scores, and CFS scores are shown in Table 1. Reductions in mean CFS score from baseline to month 3, as assessed using the Ora scale, were significantly greater in the PF-latanoprost-E than in the travoprost-Z group, both in the FAS population (P = 0.0461; Fig. 2) and in the PP population (P = 0.0379; Supplementary Table S2). Similar results were obtained using the modified Oxford scale, with the reduction in CFS score at month 3 being greater in the PF-latanoprost-E than in the travoprost-Z group in both the FAS (−0.36 vs. −0.14, P = 0.1001) and PP (−0.36 vs. −0.07, P = 0.0436) populations (Supplementary Table S3).
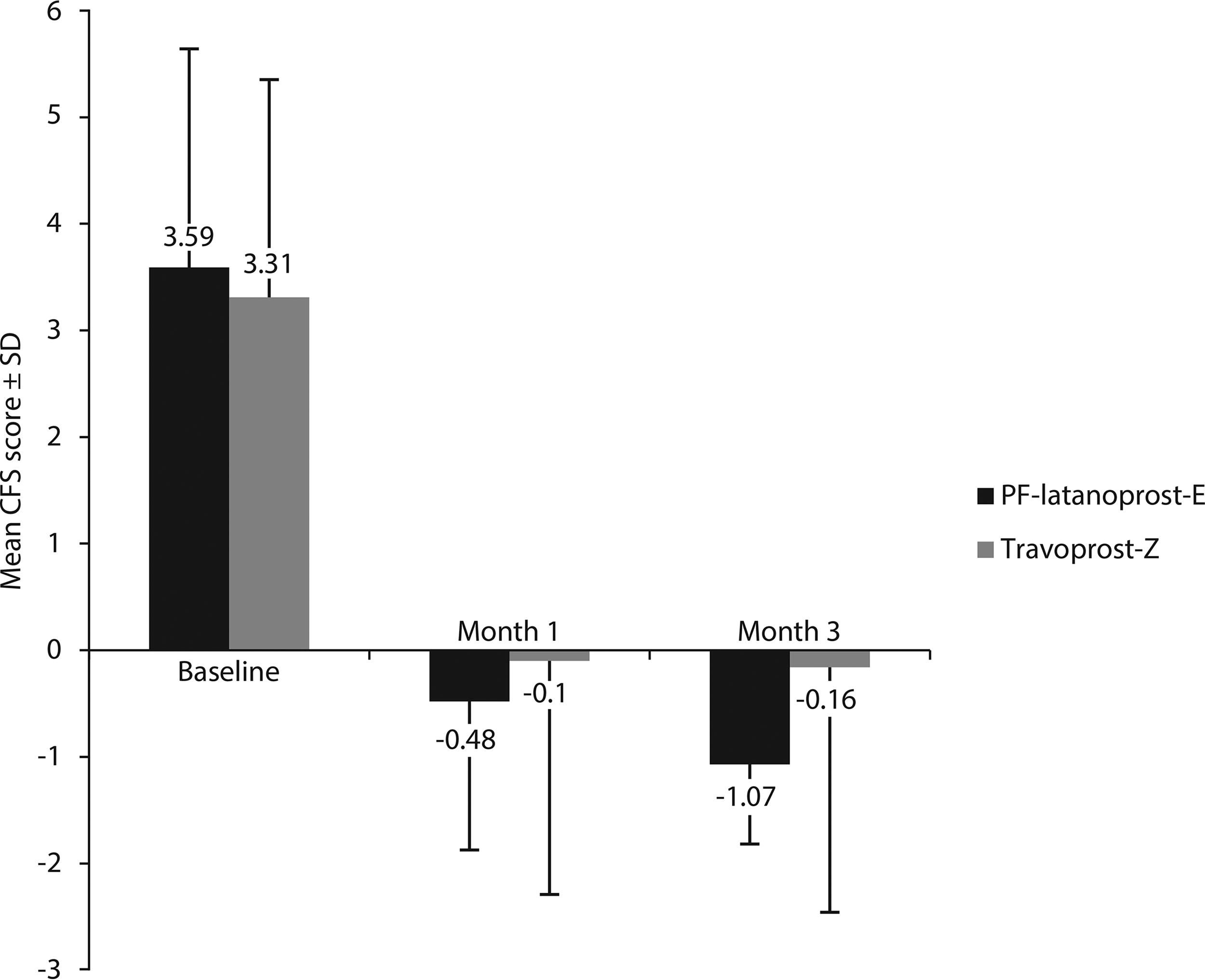
Mean CFS scores at baseline in the worse eye in the PF-latanoprost-E and travoprost-Z groups and changes from baseline in mean CFS at months 1 and 3 (Ora scale—full analysis set with last observation carried forward). Mean CFS showed a significantly greater improvement in the PF-latanoprost-E than in the travoprost-Z group at month 3 (P = 0.0461). Abbreviations: PF-latanoprost-E, preservative-free latanoprost 0.005% cationic emulsion; travoprost-Z, travoprost-Z 0.004% ophthalmical solution containing a soft preservative system; CFS, corneal fluorescein staining.
All OSD symptoms, including dryness, which were measured immediately before the 8AM measurements of IOP, showed improvements from baseline at 1 and 3 months in both study groups. Although the mean values of all OSD symptoms, except for grittiness, were numerically greater in the PF-latanoprost-E than in the travoprost-Z group at month 3, none of these differences was statistically significant in both the FAS (P > 0.05 each; Fig. 3) and PP (data not shown) populations.
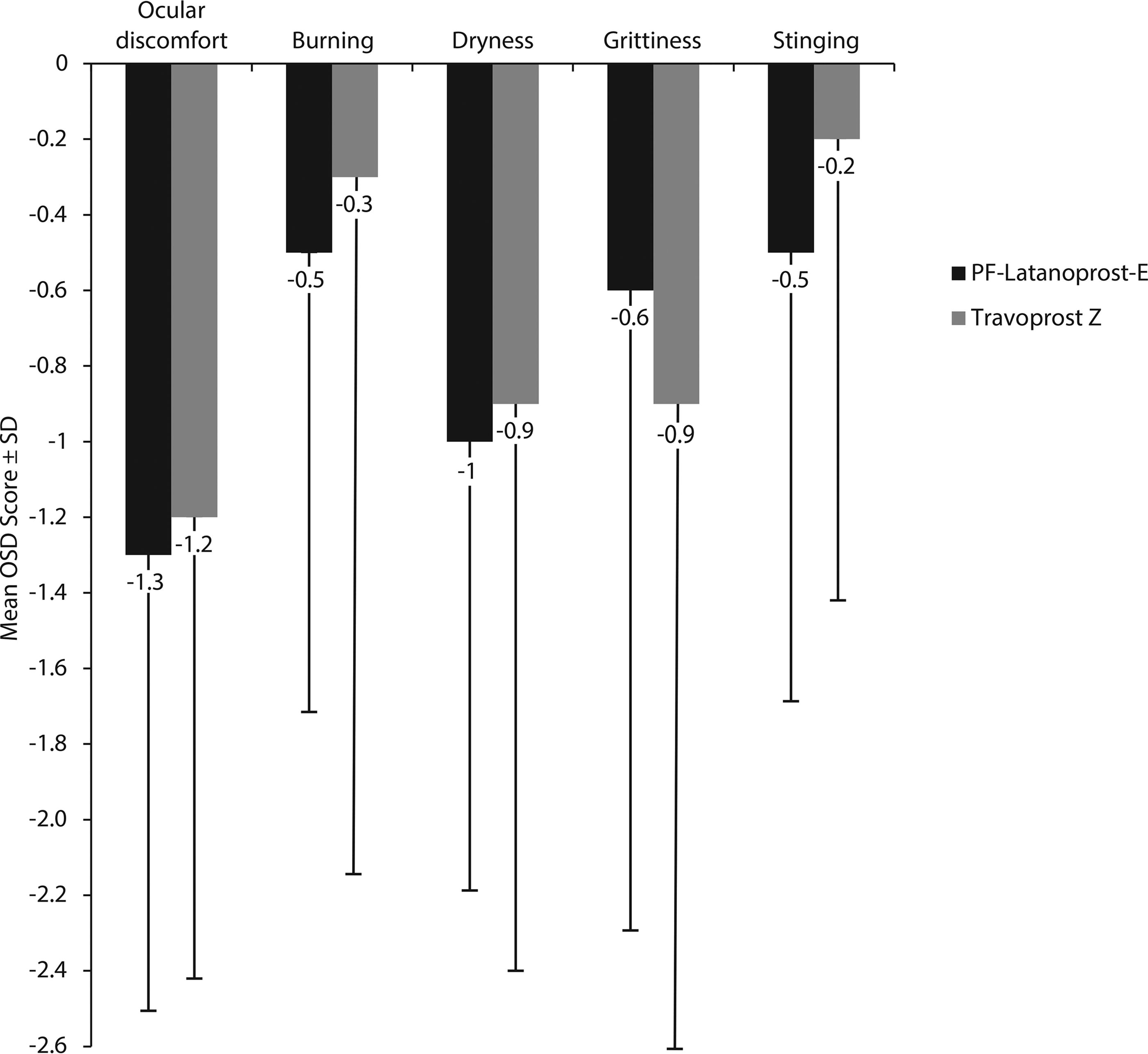
Mean changes from baseline to 3 months in OSD symptoms in the PF-latanoprost-E and travoprost-Z groups. Abbreviations: PF-latanoprost-E, preservative-free latanoprost 0.005% cationic emulsion; travoprost-Z, travoprost-Z 0.004% ophthalmical solution containing a soft preservative system; OSD, ocular surface disease.
Mean TFBUT at month 3 showed similar improvements from baseline in the PF-latanoprost-E and travoprost-Z groups, although the between group differences were not statistically significant, in both the FAS (1.1 vs. 0.98 s, P > 0.05; Fig. 4) and PP (1.1 vs. 0.78 s, P > 0.05; data not shown) populations. As determined using the McMonnies scale, a higher percentage of patients in the travoprost-Z than in the PF-latanoprost-E group in the FAS population (43% vs. 26%, P = 0.0650) showed worsening of conjunctival hyperemia by at least one grade; similar results were observed in the PP population. Blepharitis assessments did not differ in the two groups at any time point. Based on patient self-reporting in the safety population, artificial tear use decreased from 1.1 ± 1.2 drops per day at baseline to 0.7 ± 1.0 drops per day at 3 months in the PF-latanoprost-E group and from 1.1 ± 1.3 to 1.0 ± 1.3 drops per day in the travoprost-Z group, but the difference was not statistically significant (Fig. 5).
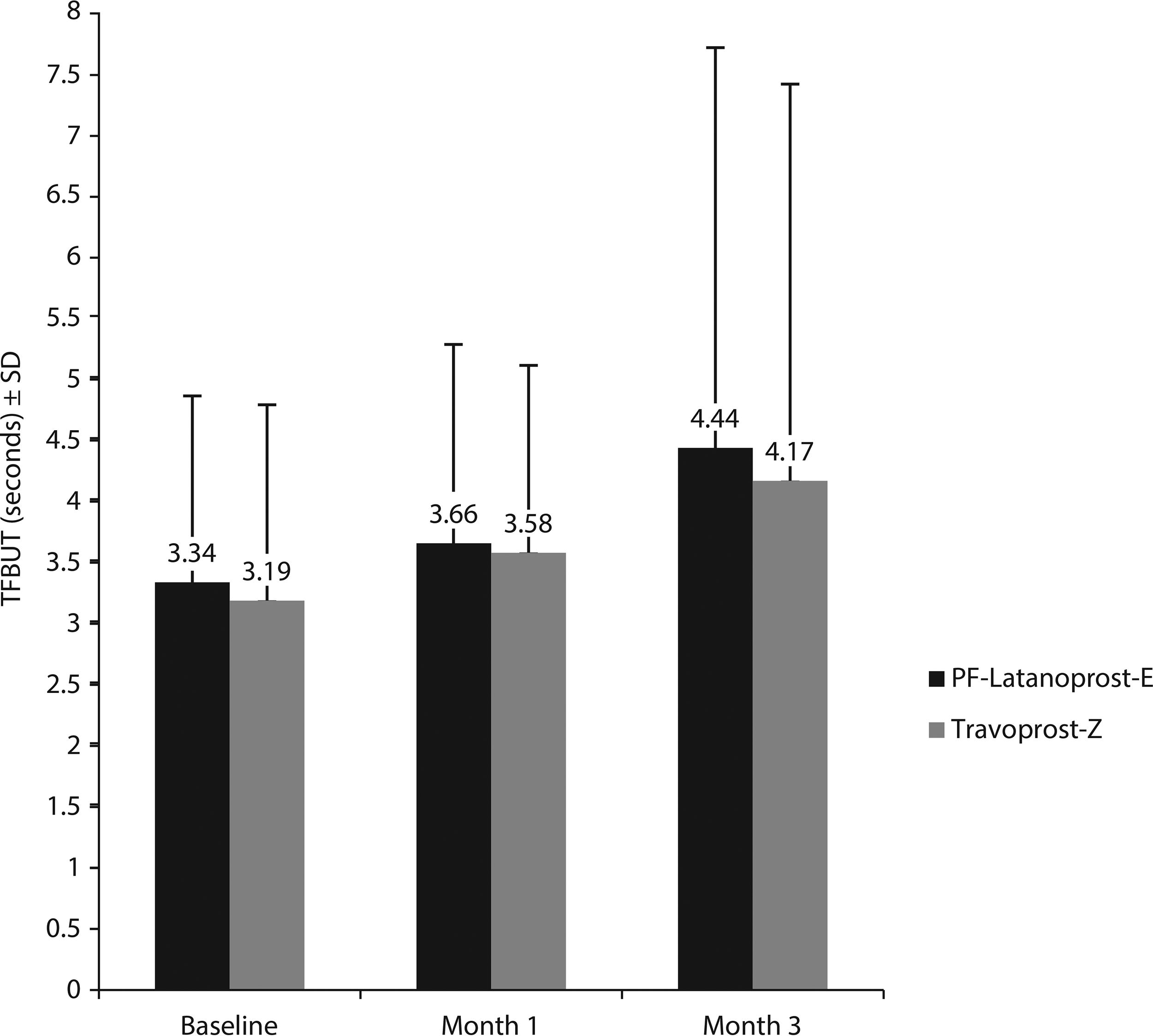
Mean tear film break-up time (TFBUT) at each visit by the treatment group. No statistically significant differences between the PF-latanoprost-E and travoprost-Z groups were observed at any time point. Abbreviations: PF-latanoprost-E, preservative-free latanoprost 0.005% cationic emulsion; travoprost-Z, travoprost-Z 0.004% ophthalmical solution containing a soft preservative system.
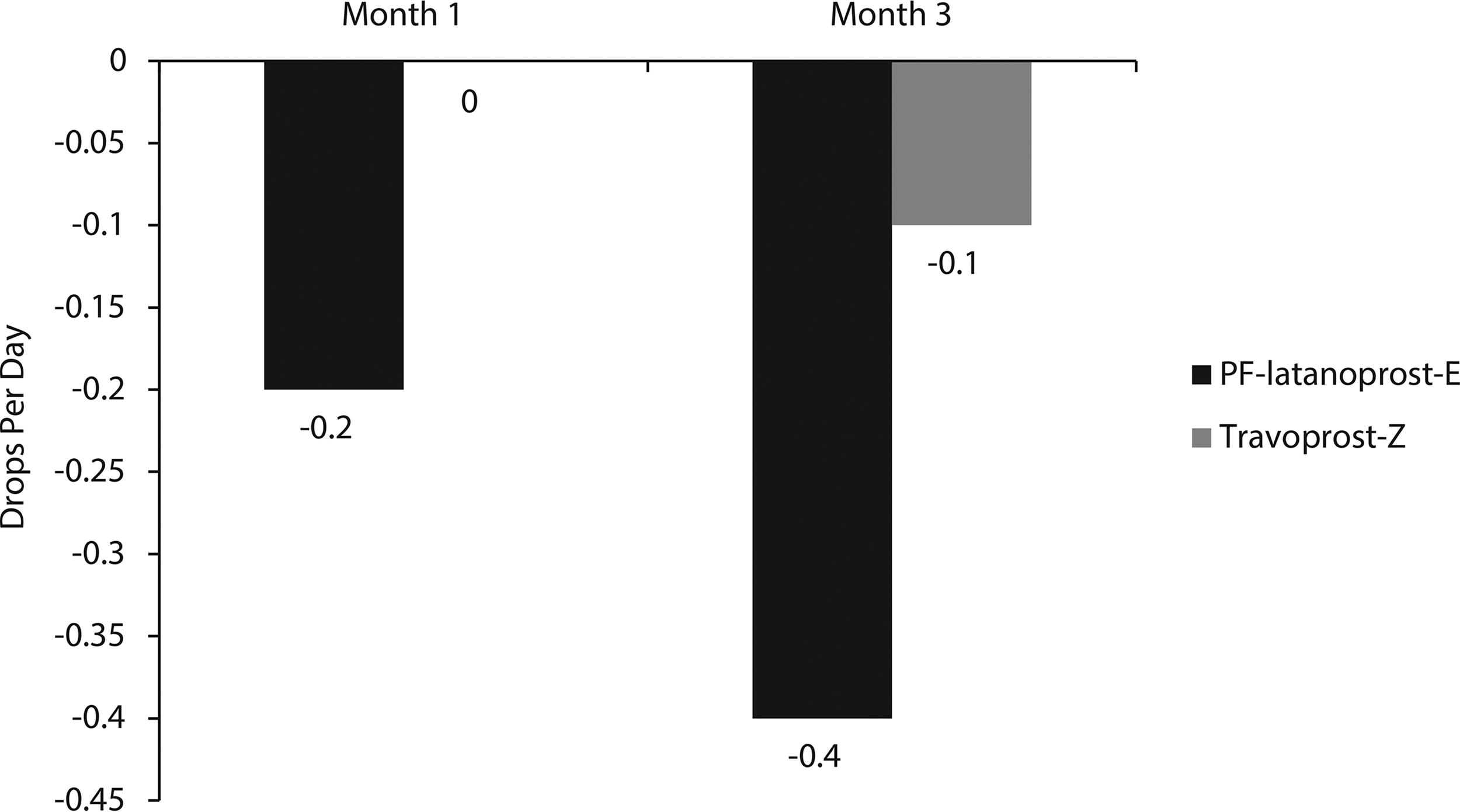
Change from baseline in mean artificial tear use per person, per day in the worse eye at months 1 and 3 in the PF-latanoprost-E and travoprost-Z groups. Abbreviations: PF-latanoprost-E, preservative-free latanoprost 0.005% cationic emulsion; travoprost-Z, travoprost-Z 0.004% ophthalmical solution containing a soft preservative system.
Safety
Eleven patients in the PF-latanoprost-E group and 13 in the travoprost-Z group reported treatment-emergent adverse events (Table 2). Treatment-emergent ocular adverse events (n = 6) occurred in three patients in each group, with all being resolved by the end of the study. These treatment-emergent ocular adverse events included ocular hyperemia (n = 1 per group), allergic conjunctivitis and eye irritation (n = 1 each in the travoprost-Z group), and meibomitis and punctate keratitis (n = 1 each in the PF-latanoprost-E group). One nonocular treatment-emergent adverse event was considered a serious adverse event, with one subject in the travoprost-Z group being diagnosed with and undergoing drainage of an intestinal abscess. Three patients in the travoprost-Z group discontinued treatment due to AEs, intestinal abscess, allergic conjunctivitis in response to the study drug, and ocular hyperemia (n = 1 each).
PF-latanoprost-E, preservative-free latanoprost 0.005% cationic emulsion; travoprost-Z, travoprost-Z 0.004% ophthalmical solution containing a soft preservative system; AE, adverse event; TEAE, treatment emergent adverse event.
Discussion
This prospective, multicenter phase 2 study demonstrated that PF-latanoprost-E 0.005% cationic emulsion was noninferior to BAK-free travoprost-Z 0.004% ophthalmical solution containing a soft preservative system in lowering IOP in patients with glaucoma or ocular hypertension plus OSD. PF-latanoprost-E produced numerically greater improvements in the majority of the signs and symptoms of OSD, greater reductions in artificial tear use, and statistically greater improvements in mean CSF score than travoprost-Z.
The efficacy of PF-latanoprost-E can be attributed partly to the formulation. PF-latanoprost-E is a topical medication formulated as a cationic emulsion for the treatment of glaucoma. The cationic oil-in-water emulsion interacts with the layers of the tear film, mainly with the tear film lipid layer, resulting in its stabilization. 27 Homogeneous spreading of the emulsion prolongs its time of residence on ocular surface cells, thus maximizing the effect. 27 Recently, another type of cationic emulsion, Cationorm® (Santen), used for the treatment of dry eye, has been shown to stabilize the tear film and aqueous lipid interface and to thicken the tear film lipid layer.30,31
PF-latanoprost-E has demonstrated benefits in glaucoma management, due to its dual activity of reducing IOP and treating existing or treatment-related OSD. 28 Chronic exposure to BAK due to glaucoma treatments sets up a recurring circle by causing OSD symptoms, resulting in nonadherence to treatment,18,19 and leading to both elevated IOP and glaucoma progression.20–23 The occurrence of elevated IOP and glaucoma progression requires additional IOP-lowering medications, which further increase the BAK load, potentially worsening OSD symptoms. Optimizing the ocular surface using PF-latanoprost-E improves IOP control in patients with glaucoma.32,33 Previous modeling studies in France, Germany, and Italy have also indicated that PF-latanoprost-E could be a potential first-line option in patients with glaucoma and OSD.34–36
The safety profile of PF-latanoprost-E was also comparable with that of travoprost-Z. The incidence of conjunctival hyperemia was lower in PF-latanoprost-E treated patients, possibly due to the absence of preservatives. Preclinical studies of PF-latanoprost-E showed that it was well tolerated on the ocular surface and did not exhibit the ocular surface cytotoxicity associated with BAK.24,26 In addition, PF-latanoprost-E, which did not include BAK, was found to promote corneal wound healing, which may also contribute to improved safety. 25
The present study compared PF-latanoprost-E with BAK-free travoprost containing another preservative. This active comparator with a soft preservative system eliminated the effects of BAK on study outcomes. Although BAK prevents bacterial contamination and has been shown to increase corneal permeability and enhance ocular penetrance of active compounds,12,37,38 BAK removal was found to have no measurable effect on IOP reduction in patients with glaucoma.39–44 Moreover, although ocular toxicities were generally lower with BAK-free than with BAK-containing formulations, these findings were inconsistent and/or of insufficient magnitude to change prescribing patterns.13,17,45–48 In addition, the concentration of latanoprost-free acid in ocular tissues was found to be similar in rabbits dosed with PF-latanoprost-E and P-latanoprost, indicating that BAK did not influence the bioavailability of the drug. 24 In the present study, PF-latanoprost-E and BAK-free travoprost with a soft preservative system showed comparable efficacy in reducing IOP, with the former resulting in reduced damage to the corneal epithelium. These findings indicated that the absence of BAK did not affect the efficacy of these products on IOP, further indicating that the cationic emulsion contributes to the beneficial effects of preservative-free latanoprost at the ocular surface.
Inflammation plays an important role in the development of both glaucoma and OSD. 47 It has been hypothesized that inflammation starting at the ocular surface diffuses toward deeper ocular tissues, such as the trabecular meshwork after the long-term use of IOP-lowering medications containing preservatives, ultimately interfering with the efficacy of eyedrops. Although BAK triggers inflammation and is deleterious to the ocular surface,12,13 BAK removal alone is insufficient. In addition to being BAK-free, PF-latanoprost-E possesses all of the properties of both latanoprost and a cationic emulsion, with latanoprost reducing IOP and the cationic emulsion reducing OSD.27,28
The strengths of this study include its randomized controlled design and the masking of investigators, thereby minimizing bias. In addition, the study population, consisting of patients with concurrent glaucoma/ocular hypertension and OSD, enabled the comparison of treatments in the presence of both of these inter-dependent–related conditions.
The present study also had limitations. This study was an exploratory phase 2 trial that included a relatively small number of patients. Thus, these results require further confirmation in large phase 3 trials, one of which has recently been completed. 49
Conclusion
PF-latanoprost-E 0.005% was found to effectively lower IOP and improve the manifestations of OSD in patients with glaucoma in this study population. Although both PF-latanoprost-E and travoprost-Z significantly improved OSD symptoms relative to baseline at 1 and 3 months, the between-group differences were not significant. PF-latanoprost-E may represent an alternative first-line therapy for patients with concurrent glaucoma and OSD.
Footnotes
Acknowledgment
The clinical trial and the medical writing related to the article were sponsored by Santen Pharmaceuticals.
Author Disclosure Statement
J.B. has received research funding and consulting fees from Santen and Alcon. E.B.M. has performed research funded by AbbVie, Aerie, Inc., Allergan, Nicox Ophthalmics Inc., Ocular Therapeutix, ReGenTree, LLC, Perrigo, and Santen, Inc. S.S. has nothing to disclose. A.M. and D.I. were employees of Santen Pharmaceuticals at the time of this study. J-S.G. is an employee of Santen Pharmaceuticals. W.J.F. has nothing to disclose.
Funding Information
No funding was received for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
