Abstract
Purpose:
To investigate the intraocular pressure (IOP)-lowering effects of omidenepag isopropyl (OMDI), a potent and highly selective prostanoid EP2 receptor agonist, as a potential first-line ocular hypotensive agent when combined with existing antiglaucoma agents in conscious ocular normotensive monkeys.
Methods:
Male cynomolgus monkeys were examined under conscious conditions. OMDI ophthalmic solution alone was topically applied to an eye or combined with other ophthalmic solutions at 5-min intervals. The contralateral eye was left untreated. IOP was measured before and at 2, 4, 6, and 8 h after instillation.
Results:
Topical application of OMDI to the eye resulted in statistically significant IOP reduction, which lasted for at least 6 h. The IOP-lowering effects of OMDI concomitantly administered with any of the tested antiglaucoma agents (timolol, brinzolamide, netarsudil, ripasudil, and brimonidine) were greater than those of OMDI alone. Furthermore, these enhanced IOP responses to their concomitant use were statistically significant compared with those of the tested antiglaucoma agents alone. Any combination of OMDI with the tested agents did not lead to serious abnormalities either systemically or locally in the eye.
Conclusions:
We demonstrated that OMDI has additive IOP-lowering effects when administered in combination with various antiglaucoma agents, namely, β-adrenergic antagonist, carbonic anhydrase inhibitor, Rho-associated coiled-coil containing protein kinase inhibitors, and α2-adrenergic agonist. These results suggest that OMDI provides additional clinical benefits because of its unique mechanisms of action when combination therapy is required.
Introduction
Omidenepag isopropyl (OMDI) is the first regulatory-approved prostaglandin E2 receptor EP2 subtype (EP2 receptor) agonist as an antiglaucoma drug worldwide, with potent ocular hypotensive effects in animals and humans.1–4 Since its approval by the Japanese regulatory agency in 2018, OMDI has been used as EYBELIS® ophthalmic solution 0.002% for the treatment of glaucoma and ocular hypertension. 5 OMDI is an isopropyl ester form of its parent compound omidenepag (OMD) and has improved corneal penetration. 6 OMD is structurally different from any existing prostaglandin analogs, including prostaglandin F (FP) receptor agonists, and possesses a novel and nonprostaglandin chemical structure with highly selective EP2 receptor agonistic activity.
Using ocular hypertensive monkeys, we previously demonstrated that OMDI stimulated aqueous humor outflow through the conventional and uveoscleral outflow pathways. 7 This mechanism of action is unique to OMDI because the FP receptor and other EP2 receptor agonists only stimulate the uveoscleral outflow pathway. 8 Furthermore, a double-blinded multicenter controlled phase 3 clinical study (AYAME Study [NCT02623738]) 3 provided evidence that OMDI is safe and tolerable with noninferior intraocular pressure (IOP)-lowering efficacy to that of the typical FP receptor agonist latanoprost. Notably, glaucoma treatment with FP receptor agonists is occasionally associated with prostaglandin-associated periorbitopathy (PAP), thereby limiting its clinical use.9–11 Unlike FP receptor agonists, our previous nonclinical studies suggest that OMDI does not cause PAP.12,13 Considering its IOP-lowering efficacy and novel mechanisms of action, OMDI may have potential as an alternative first-line therapeutical agent when FP receptor agonists are not used as the first choice.
Because only IOP is a validated risk factor for glaucoma,14,15 a principal cause of irreversible blindness worldwide, lowering IOP still remains an effective treatment method for glaucoma.16–19 In most cases, FP receptor agonists, including latanoprost, have been used as the first-line treatment to achieve targeted IOP and prevent further visual field loss.20,21 However, monotherapy with FP receptor agonists could not lower IOP sufficiently in ∼40% of patients who eventually required adjunctive therapy within 2 years to better control IOP. 22 Several classes of IOP-lowering agents, including Rho-associated coiled-coil containing protein kinase (ROCK) inhibitors, β-adrenergic antagonists, α2-adrenergic agonists, and carbonic anhydrase inhibitors (CAIs), can be used concomitantly with FP receptor agonists, depending on patient conditions and their mechanisms of action. Thus, to assess its potential as an alternative first-line IOP-lowering drug, it is essential to demonstrate that OMDI may provide additional IOP-lowering effects when used in combination with existing antiglaucoma agents.
Therefore, this study was designed to examine the IOP-lowering efficacy of OMDI alone and in combination with existing antiglaucoma agents in conscious ocular normotensive monkeys. They include the β-adrenergic antagonist timolol, CAI brinzolamide, 2 ROCK inhibitors netarsudil and ripasudil, or α2-adrenergic agonist brimonidine, which are often used as adjunctive treatment with FP receptor agonists.
Methods
Drugs
OMDI was synthesized by Ube Industries, Ltd. (Yamaguchi, Japan). Ophthalmic solution of 0.0006% OMDI and their vehicle (citrate buffer containing polyoxyl 35 castor oil, edetate disodium, glycerin, pH adjuster, water, and benzalkonium chloride as preservative) were formulated by Santen Pharmaceutical Co., Ltd. (Osaka, Japan). Netarsudil mesylate was synthesized by Ark Pharm, Inc., (Arlington Heights, IL). An ophthalmic solution of 0.01% netarsudil and its vehicle (saline containing 1% polysorbate 80) were formulated by Santen Pharmaceutical Co., Ltd.
Timoptol® ophthalmic solution 0.5% (0.5% timolol) was obtained from Santen Pharmaceutical Co., Ltd., Azopt® ophthalmic suspension 1% (1% brinzolamide) was purchased from Novartis AG (Basel, Switzerland), Glanatec® ophthalmic solution 0.4% (0.4% ripasudil) was purchased from Kowa Company, Ltd. (Tokyo, Japan), and Alphagan® P 0.15% (0.15% brimonidine) was purchased from Allergan plc (Dublin, Ireland). Saline was used as the vehicle for 0.5% timolol, 1% brinzolamide, 0.4% ripasudil, and 0.15% brimonidine.
Animals
Adult male cynomolgus monkeys aged 5–16 years (Keari Co., Ltd., Osaka, Japan; Shin Nippon Biomedical Laboratories, Ltd., Tokyo, Japan; and Eve Bioscience Co., Ltd., Wakayama, Japan) were used in the study. The animals were individually housed, and a 12-h light–dark cycle was maintained.
All experimental procedures were performed in compliance with the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research, with approval of and monitoring by the Animal Care and Use Committee at Santen Pharmaceutical Co., Ltd.
IOP measurements in monkeys
Conscious ocular normotensive monkeys were used in all monkey studies. Before the study, all monkeys underwent training to be restrained in a monkey chair (CL-4535; Primate Products, Miami, FL) and undergo IOP measurements without the use of sedation or general anesthesia.
Two 20-μL ophthalmic solutions (0.0006% OMDI and existing antiglaucoma agents [0.5% timolol, 1% brinzolamide, 0.01% netarsudil, 0.4% ripasudil, or 0.15% brimonidine]) or their vehicles were topically administered at time 0, with a 5-min interval. The contralateral eye remained untreated. After instillation of each ophthalmic solution, the general behavior and ocular conditions of animals were examined. IOP measurements were performed on conscious animals just before and 2, 4, 6, and 8 h after drug administration. IOP was measured 3 times at each time point, and the mean IOP was calculated and used as the value for each time point.
The monkeys were restrained in the sitting position in the monkey chair for IOP measurement, and local corneal anesthesia was administered by topical application of 0.4% oxybuprocaine solution (Benoxil® ophthalmic solution 0.4%) to the monkey's eyes. Subsequently, IOP was measured using a pneumatonometer (Model 30 Classic; Reichert Technologies, Depew, NY). The mean IOP changes were calculated as the differences from the baseline values and expressed as mean ± standard error of the mean (SEM).
Statistical analysis
All statistical analyses were performed using SAS 9.4 (SAS Institute Inc., Cary, NC). IOP changes were compared using Tukey–Kramer test with the baseline as a covariate using the linear mixed-effects model. Differences were considered statistically significant (2-sided) when the P value was < 0.05.
Results
Table 1 summarizes the baselines and IOPs observed at all the time points after instillation of vehicle or each existing IOP-lowering drug tested onto the monkey eyes. No general behavioral changes were observed after the concomitant use of OMDI with any of the drugs tested. Combination of OMDI with any of the other tested drugs did not cause ocular abnormalities either, namely, corneal opacity, hyperemia in the iris and conjunctiva, edema in the conjunctiva, secretion, miosis, and mydriasis.
The Actual Values of Baselines and Intraocular Pressures after Topical Instillation of Omidenepag Isopropyl Alone and in Combination with Other Antiglaucoma Agents in Conscious Ocular Normotensive Monkeys
Two 20-μL ophthalmic solutions (0.0006% OMDI and existing antiglaucoma agents [0.5% timolol, 1% brinzolamide, 0.01% netarsudil, 0.4% ripasudil, or 0.15% brimonidine]) or their vehicles were topically administered at time 0, with a 5-min interval. The contralateral eye remained untreated. IOP was periodically measured before and after drug administration under conscious and restrained conditions, as described in the table. The data represent the mean ± SEM.
IOP, intraocular pressure; OMDI, omidenepag isopropyl; SEM, standard error of the mean.
Additive IOP-lowering effects of OMDI with aqueous humor production inhibitors (a β-adrenergic antagonist or a CAI) in monkeys
First, we evaluated the IOP-lowering effects of OMDI combined with either timolol (Fig. 1) or brinzolamide (Fig. 2), both of which suppress aqueous humor production through blockade of β-adrenoceptors and inhibition of carbonic anhydrases, respectively. Two independent experiments were performed to examine these combinations separately. The average baseline IOP was 17.5–18.1 mmHg (n = 8) for the OMDI–timolol experiment and 17.9–19.3 mmHg (n = 5–6) for the OMDI–brinzolamide experiment (Table 1).
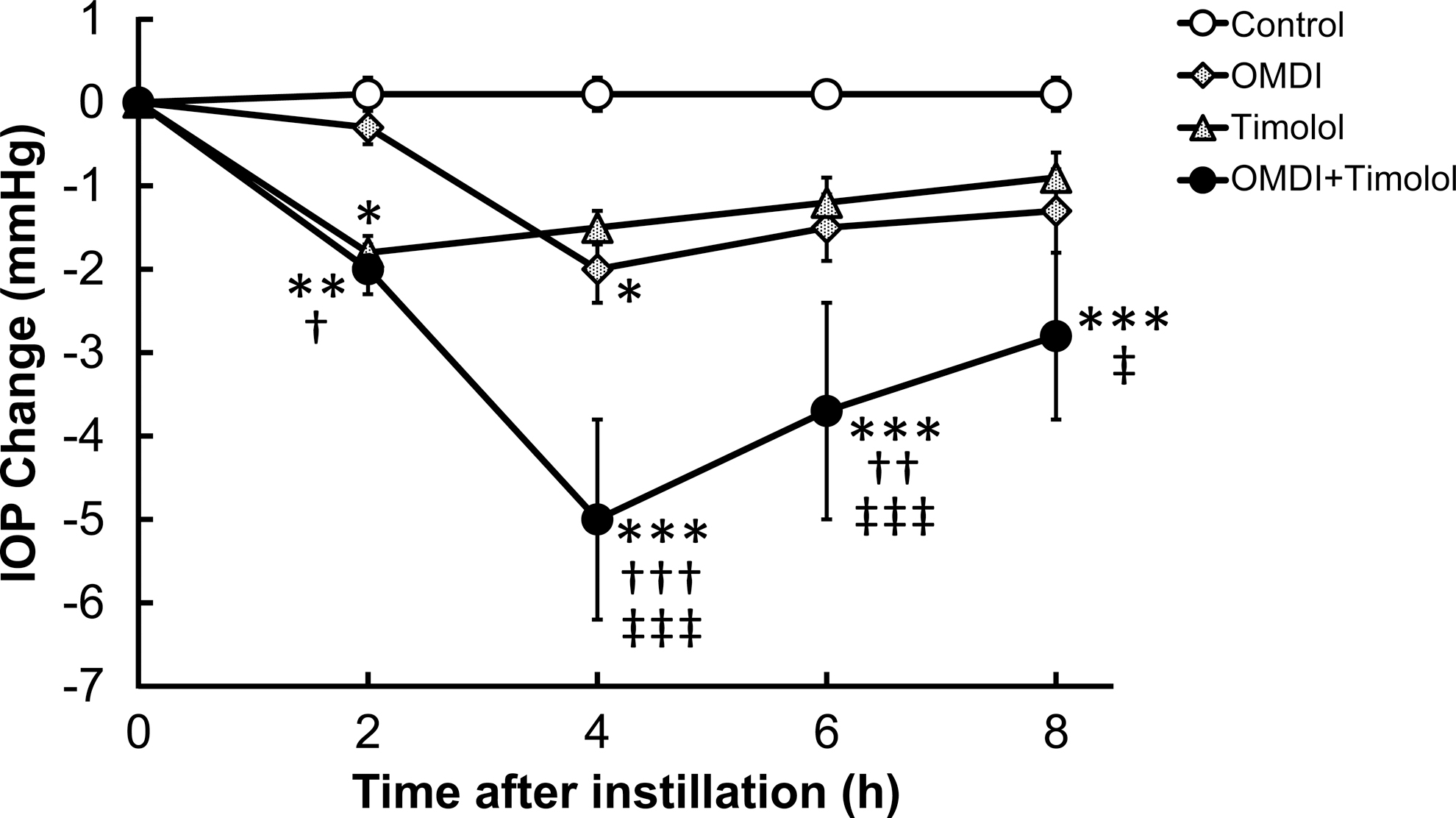
Additive IOP-lowering effect of OMDI ophthalmic solution with β-adrenergic antagonist Timoptol® 0.5% (0.5% timolol) after topical administration to the eye of conscious ocular normotensive monkeys. Two drugs (0.0006% OMDI and 0.5% timolol) or their vehicles were topically administered to the eye in each monkey at time 0, with a 5-min interval. The contralateral eye remained untreated. The IOP changes were calculated as the differences from the baseline values. The data represent the mean ± SEM of 8 eyes. *P < 0.05, **P < 0.01, and ***P < 0.001 compared with control using the Tukey–Kramer test; †P < 0.05, ††P < 0.01, and †††P < 0.001 compared with OMDI alone using the Tukey–Kramer test; ‡P < 0.05 and ‡‡‡P < 0.001 compared with timolol alone using the Tukey–Kramer test. IOP, intraocular pressure; OMDI, omidenepag isopropyl.
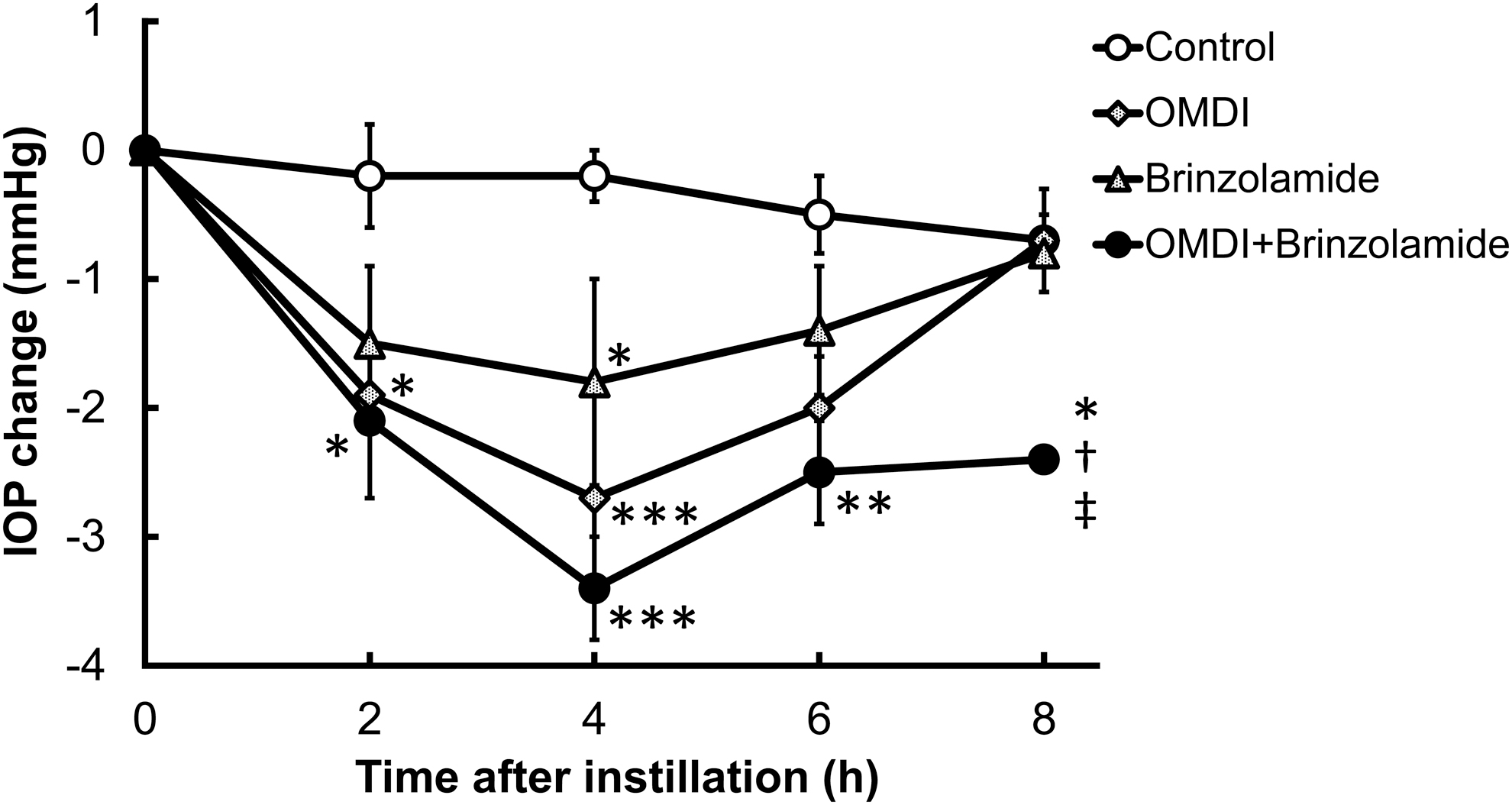
Additive IOP-lowering effect of OMDI ophthalmic solution with Azopt® (1% brinzolamide) topically administered to the eye in conscious ocular normotensive monkeys. Two drugs (0.0006% OMDI and 1% brinzolamide) or their vehicles were topically administered to the eye in each monkey at time 0, with a 5-min interval. The contralateral eye remained untreated. The IOP changes were calculated as the differences from the baseline values. The data represent the mean ± SEM of 5 eyes for OMDI alone and concomitant use of OMDI with brinzolamide and of 6 eyes for the control and brinzolamide alone. *P < 0.05, **P < 0.01, and ***P < 0.001 compared with the control using the Tukey–Kramer test. †P < 0.05 compared with OMDI alone using the Tukey–Kramer test; ‡P < 0.05 compared with brinzolamide alone using the Tukey–Kramer test.
A single topical instillation of 0.0006% OMDI alone onto the eye led to statistically significant IOP-lowering effects that peaked at 4 h after instillation (−2.0 ± 0.4 and −2.7 ± 0.8 mmHg shown in Figs. 1 and 2, respectively). Topical administration of 0.5% timolol or 1% brinzolamide alone statistically significantly lowered IOP and their peak levels were observed at 2 h (−1.8 ± 0.2 mmHg, Fig. 1) and 4 h (−1.8 ± 0.8 mmHg, Fig. 2) after instillation, respectively. When administered concomitantly with either with timolol (Fig. 1) or brinzolamide (Fig. 2), OMDI produced remarkable and sustainable IOP-lowering effects: IOP reduction after concomitant administration of OMDI with either timolol or brinzolamide was statistically significant compared with the control group at all evaluated time points, and significantly greater than that achieved with OMDI, timolol, or brinzolamide alone (Figs. 1, 2). Thus, OMDI demonstrated additive IOP-lowering effects when used concomitantly with the drugs suppressing aqueous humor production.
Additive IOP-lowering effects of OMDI with ROCK inhibitors in monkeys
Next, 2 independent experiments were performed to evaluate the additive IOP-lowering effects of OMDI with either ROCK inhibitor netarsudil or ripasudil that stimulates the trabecular outflow pathway (Fig. 3). The average baseline IOP was 19.5–20.0 mmHg in the OMDI–netarsudil experiment (Table 1, n = 8) and 17.2–17.9 mmHg in the OMDI–ripasudil experiment (Table 1, n = 7).
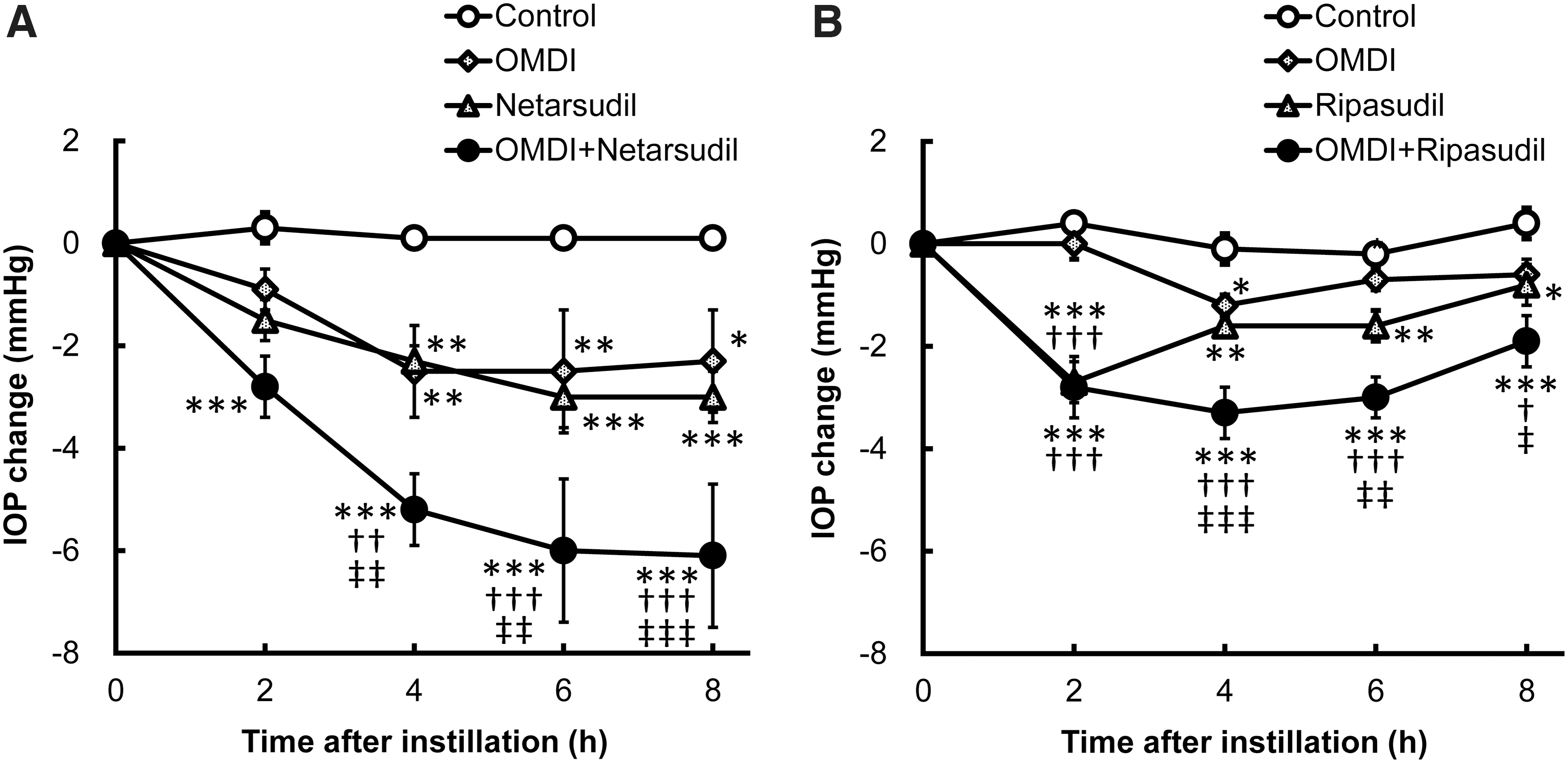
Additive IOP-lowering effects of OMDI with ROCK inhibitors (netarsudil and Glanatec® [0.4% ripasudil]) in conscious ocular normotensive monkeys.
In both the experiments (Figs. 3A, B), a topical application of 0.0006% OMDI alone statistically significantly lowered IOP and its peak levels were observed at 4 h after instillation (−2.5 ± 0.9 and −1.2 ± 0.2 mmHg, Figs. 3A, B, respectively). Statistically significant IOP lowering was also observed after topical administration of 0.01% netarsudil and 0.4% ripasudil alone, peaking at 6 h (−3.0 ± 0.6 mmHg, Fig. 3A) and 2 h (−2.7 ± 0.4 mmHg, Fig. 3B) after instillation, respectively. The concomitant use of 0.0006% OMDI with 0.01% netarsudil (Fig. 3A) or 0.4% ripasudil (Fig. 3B) led to enhanced IOP responses, which were statistically significant compared with the control group at all evaluated time points. Both concomitant-use groups showed significantly greater IOP-lowering effects than those of any of the monotherapy groups. These results indicate that when used concomitantly, OMDI additively lowers IOP with ROCK inhibitors that stimulate the trabecular outflow pathway.
Additive IOP-lowering effects of OMDI with an α2-adrenergic agonist in monkeys
Finally, the IOP-lowering effects of OMDI were evaluated when used concomitantly with the α2-adrenergic agonist brimonidine that modulates aqueous humor production and the uveoscleral outflow pathway (Fig. 4). The average baseline IOP was 18.3–18.4 mmHg (Table 1, n = 6).
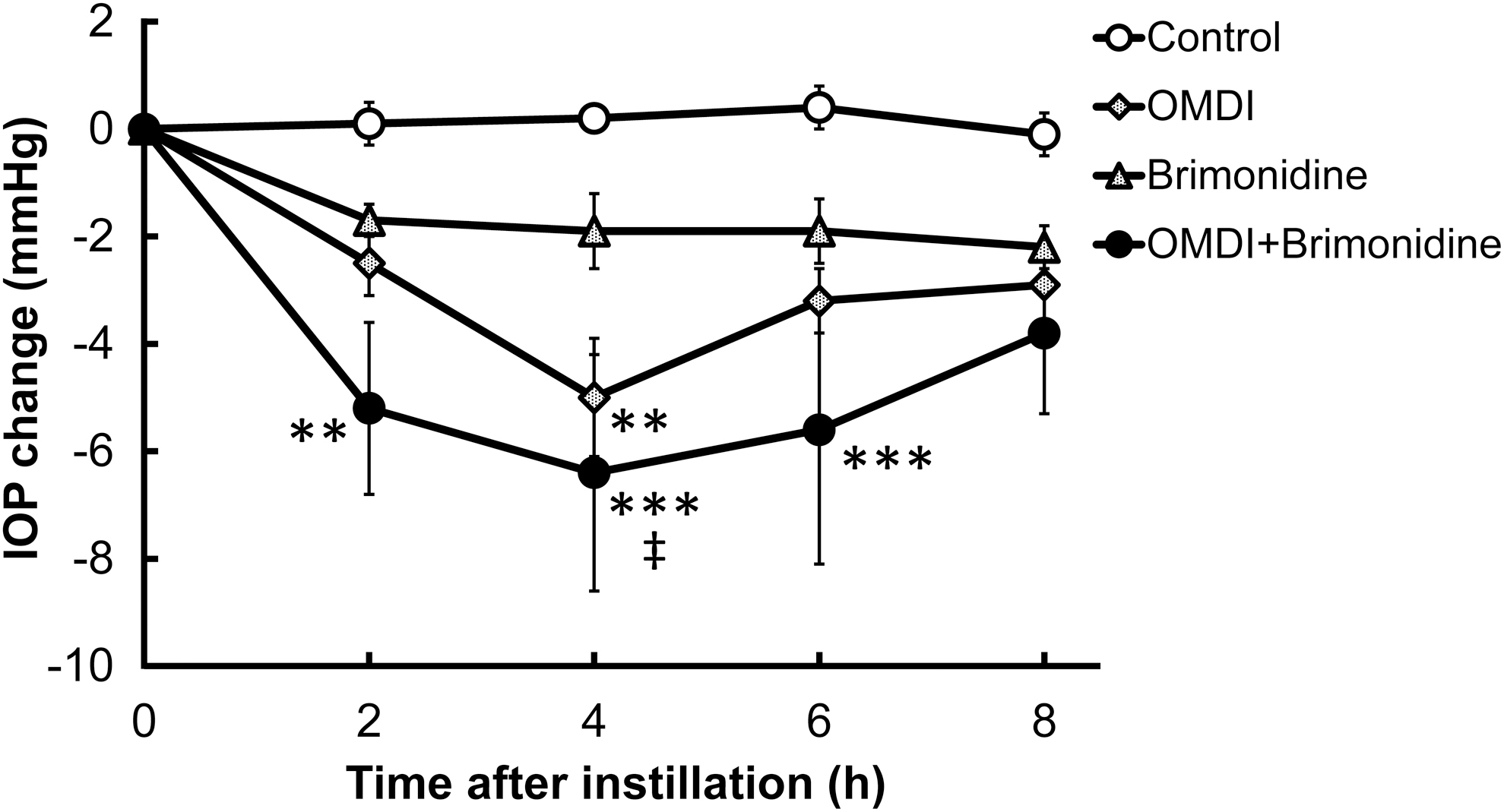
Additive IOP-lowering effect of OMDI ophthalmic solution with Alphagan® P (0.15% brimonidine) topically administered to the eye in conscious ocular normotensive monkeys. Two drugs (0.0006% OMDI and 0.15% brimonidine) or their vehicles were topically administered at time 0, with a 5-min interval. The contralateral eye remained untreated. The IOP changes were calculated as the differences from the baseline values. The data represent the mean ± SEM of 6 eyes. **P < 0.01 and ***P < 0.001 compared with control using the Tukey–Kramer test. ‡P < 0.05 compared with brimonidine alone using the Tukey–Kramer test.
As seen in the other experiments, topical administration of 0.0006% OMDI alone statistically significantly lowered IOP with the peak level at 4 h after instillation (−5.0 ± 1.1 mmHg, Fig. 4). In contrast, 0.15% brimonidine alone did not show statistically significant IOP reduction, although IOP was numerically lower than that in the control group at all evaluated time points. The concomitant use of 0.0006% OMDI with 0.15% brimonidine led to statistically significant IOP reduction compared with the control group, which was greater than that in any of the monotherapy groups. Collectively, these results suggest that OMDI has the additive IOP-lowering effects when used concomitantly with any of the tested IOP-lowering agents including brimonidine, regardless of their mechanisms of action.
Discussion
In this study, OMDI demonstrated additive IOP-lowering effects when administered concomitantly with clinically used antiglaucoma drugs in conscious ocular normotensive monkeys, namely, β-adrenergic antagonist (timolol, Fig. 1), CAI (brinzolamide, Fig. 2), ROCK inhibitors (netarsudil and ripasudil, Fig. 3), and α2-adrenergic agonist (brimonidine, Fig. 4). More importantly, combinations of OMDI with any of these drugs did not cause noticeable systemic or local abnormalities. Given that OMDI lowers IOP through the different mechanisms of action of the abovementioned drugs, 7 OMDI may provide additional clinical benefits without causing serious side effects when combined with any of these existing drugs.
In this study, to evaluate the IOP-lowering effects of the combination of OMDI and existing antiglaucoma agents in monkeys, we used regulatory-approved products, except for OMDI and netarsudil. We preliminarily tested the IOP-lowering effects of regulatory-approved OMDI ophthalmic solution (available in Japan as EYBELIS® ophthalmic solution 0.002%) and netarsudil ophthalmic solution (Rhopressa® 0.02%); however, we observed extremely low IOP after administration of either product in monkeys. Accordingly, we performed dose–response studies of both agents and found the optimal dose for further evaluation, namely, 0.0006% and 0.01% for OMDI and netarsudil. We also confirmed that saline (vehicle for timolol, brinzolamide, ripasudil, and brimonidine), saline containing 1% polysorbate 80 (for netarsudil), and the vehicle for OMDI had no effects on IOP in separate experiments. Thus, we believe that the study design, including the statistical analysis procedures, was appropriate in detecting differences between IOP in vehicle- and drug-treated groups.
Our previous study demonstrated that OMDI facilitates aqueous humor outflow through the trabecular and uveoscleral pathways, leading to IOP reduction in ocular hypertensive monkeys. 7 In contrast, the β-adrenergic antagonist (timolol) and CAI (brinzolamide) reduce IOP through suppression of aqueous humor production. 23 In this study, combinations of OMDI with either agent result in statistically significant and additive IOP reduction. Notably, the additive IOP-lowering effects of the concomitant use of OMDI with timolol have been clinically confirmed (RENGE study [NCT02822729]). Therefore, the combination therapy of OMDI with drugs acting on aqueous humor production seems to be a reasonable choice because these combinations regulate aqueous humor dynamics at all possible locations, namely, both aqueous inflow and outflow through the trabecular and uveoscleral pathways.
Similarly, brimonidine, an α2-adrenergic agonist, lowers IOP by inhibiting aqueous humor production, 24 so it provides additive IOP-lowering effects when combined with OMDI. Given that brimonidine also stimulates the uveoscleral outflow pathway, 24 the combination of OMDI with brimonidine may have some overlapping effects on this outflow pathway.
Surprisingly, OMDI also demonstrated the additive IOP-lowering effects when combined with ROCK inhibitors that stimulate the trabecular outflow pathway as seen with OMDI.25,26 ROCK inhibitors promote cytoskeletal and morphological changes in trabecular meshwork cells, stimulating aqueous outflow. 27 Although the molecular mechanism of action of OMDI has not been fully elucidated, OMDI may increase the trabecular outflow pathway through different mechanisms of ROCK inhibitors. Thus, because of its unique mechanistic profile in the regulation of aqueous outflow pathways, OMDI can provide additive IOP-lowering effect when combined with the abovementioned IOP-lowering agents, regardless of their site of action in regulating aqueous humor dynamics.
Although discussing the detailed molecular mechanisms is not the scope of this study, it is reasonable to assume that OMDI may act through stimulation of EP receptors in the cells consisting of aqueous outflow pathways because it is a potent and highly selective EP2 receptor agonist. EP2 receptor agonism stimulates intracellular cyclic AMP (cAMP) production, 28 resulting in smooth muscle relaxation. 29 EP2 receptor stimulation also leads to matrix metalloproteinase (MMP) protein synthesis and/or MMP activation.30,31 Therefore, the functional and structural changes in trabecular meshwork and ciliary muscle tissues through muscular relaxation and MMP activation may be responsible for increased aqueous outflow promoted by OMDI.
β-adrenergic antagonists and CAIs suppress aqueous humor production by reducing water transport in ciliary epithelial cells that are mediated by decreased β-adrenergic receptor activity and intracellular cAMP levels and inhibition of carbonic anhydrase enzymatic activities, respectively. 23 α2-adrenergic agonists act through decreased cAMP levels and/or increased intracellular calcium levels after α2-adrenergic receptor stimulation in cells regulating aqueous inflow and uveoscleral outflow. 24 As described above, ROCK inhibitors cause cellular cytoskeletal and morphological changes accompanied by increased trabecular outflow.
Taken together, the site of action and molecular mechanisms for OMDI seem to be different from any of those for the existing IOP-lowering agents and there may be no crosstalk between OMDI-stimulated EP2 receptor signaling and those by other drugs used in this experiment. Further studies are underway to elucidate more detailed molecular mechanism underlying the IOP-lowering effect of OMDI.
In conclusion, we demonstrate that OMDI has additive IOP-lowering effects when combined with existing IOP-lowering agents, namely, the β-adrenergic antagonist timolol, the CAI brinzolamide, the 2 ROCK inhibitors netarsudil and ripasudil, or the α2-adrenergic agonist brimonidine, in conscious ocular normotensive monkeys. Thus, our study provides evidence demonstrating that OMDI may be used as monotherapy and in combination with the existing antiglaucoma agents tested as an effective and flexible treatment option for glaucoma in a clinical setting.
Footnotes
Acknowledgments
The authors thank Ms. Naoko Yamashita, Mr. Masafumi Mieda, Mr. Isao Matsuoka, and Mr. Masashi Kumon for their excellent technical assistance and Mr. Hiroshi Inoue for statistical analysis. We are also grateful to Dr. Masaaki Kageyama for valuable comments and thoughtful discussions.
Author Disclosure Statement
M.F., A.S., N.O.K., T.K., T.T., M.K., and K.M. are employees of Santen Pharmaceutical Co., Ltd., and N.K.S. is an employee of Santen, Inc. R.I. and K.Y. are employees of Ube Industries, Ltd.
Funding Information
The authors received no specific funding for this research.
