Abstract
Persicaria minor is a shrub native to Southeast Asia that is commonly known as “kesum.” Its leaf extract is used medicinally and in dietary supplements. To add to the overall toxicological dataset, Biokesum® Polygonum minus (Persicaria minor) standardized extract was tested in a battery of guideline genotoxicity tests, namely the OECD Guideline 471 bacterial reverse mutation test, the OECD Guideline 473 in vitro mammalian cell chromosomal aberration test, and the OECD Guideline 487 in vitro micronucleus assay. The use of multiple standardized, validated assays increases the overall predictive value relative to individual assays and provides greater confidence in the test results. All three assays were negative, demonstrating that Biokesum® is not genotoxic.
INTRODUCTION
Polygonum minus, also known as Persicaria minor, is a shrub native to Southeast Asia. Its leaves are used in traditional medicine and are consumed raw or cooked as food. 1 In spite of its popularity, few conventional toxicology studies are available. This article presents the results of three genotoxicity assays on Biokesum® Polygonum minus (Persicaria minor) standardized extract, performed at Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC)-accredited laboratories according to OECD test guidelines and in compliance with OECD good laboratory practices (GLPs) [C(97) 186/Final and ENV/MC/CHEM(98)17]. Genotoxicity studies are essential in substantiating the safe use of substances as foods or dietary supplements and are required by numerous regulatory frameworks around the world. 2,3
MATERIALS AND METHODS
Biokesum® is a commercially available Polygonum minus (Persicaria minor) extract. It is prepared by aqueous extraction of the leaves and stems of Polygonum minus in a U.S. FDA-audited facility, Phytes Biotek (Shah Alam, Malaysia). Biokesum® is standardized to contain ≥0.45% quercetin-3-glucuronide and ≥100 mg/g total phenolic content, in units of mg gallic acid equivalents (GAE)/g dried extract (DE). Biokesum® Batch No. KE 220103 (expiry January 17, 2025) was used in all three genotoxicity studies.
BACTERIAL REVERSE MUTATION TEST
Biokesum® was evaluated for mutagenicity potential in Salmonella typhimurium test strains TA98, TA100, TA1535, and TA1537 and in Escherichia coli test strain WP2 uvrA (pKM101) according to OECD Guideline 471. 4,5 Bacterial strains (Molecular Toxicology, Inc., Boone, NC) were stored as stock cultures in Oxoid Nutrient Broth No. 2 (Thermo Fisher Scientific) at −80 ± 10°C or maintained on minimal glucose agar (MGA) without histidine-biotin (S. typhimurium) or tryptophan (E. coli) as master plates at 2–8°C for up to 3 months. Master plates were characterized for histidine (S. typhimurium) or tryptophan (E. coli) requirement, sensitivity to UV radiation, ampicillin resistance, and rate of spontaneous revertants and met the appropriate criteria. Bacteria were cultured in Oxoid Nutrient Broth No. 2 at 37 ± 1°C for 15 hrs to achieve late exponential or early stationary phase growth and adjusted to a density of 18 × 108 cells/mL. 4
The metabolic activation system was S9 homogenate isolated from male Wistar rats treated with sodium phenobarbitone (16 mg/mL) and β-naphthoflavone (20 mg/mL) by intraperitoneal injection 3 days prior to sacrifice. The sterility of each batch of S9 homogenate was assessed by streaking on a nutrient agar plate followed by incubation for 24 h at 37°C. Protein content was evaluated using a modified Lowry method. Metabolic capacity was evaluated by incubating S. typhimurium strain TA100 with 2-aminoanthracene and benzo(a)pyrene. The S9 homogenate was stored at −80 ± 10°C and thawed immediately prior to use. 4
Thawed S9 homogenate (1 mL) was mixed with 9 mL cofactor solution (4 mM nicotinamide adenine dinucleotide phosphate [NADP] disodium salt, 5 mM glucose-6-phosphate, 8 mM magnesium chloride, and 33 mM potassium chloride) in phosphate buffered saline (PBS), pH 7.34 and added to appropriate bacterial cultures. 4
Biokesum® solubility was evaluated in distilled water, acetone, ethanol, acetonitrile, N,N-dimethyl formamide, and dimethyl sulfoxide (DMSO). A preliminary precipitation test was performed by mixing Biokesum® in DMSO (0.00625–5 mg/plate) with soft agar, spreading onto master plates, and incubating for 2 h at 37 ± 1°C. Cytotoxicity was assessed by mixing freshly prepared cultures of S. typhimurium strain TA100 with Biokesum® (0.00625–5 mg/plate), with and without metabolic activation, with soft agar containing histidine and biotin in triplicate in a shaking water bath at 37 ± 1°C for 62 hrs. Cytotoxicity was scored using a code system that rated the appearance of the bacterial background lawn; possible codes ranged from 0 (complete lack of any bacterial lawn) to 4+ (thick, healthy background lawn). 4
Positive control substances, 2-nitrofluorine, 2-aminoanthracene, 9-aminoacridine, sodium azide, and 4-nitroquinoline-N-oxide, were obtained from Sigma Aldrich and prepared in DMSO. 4,5
On the day of use, fresh Biokesum® stock solutions were prepared and added to bacterial suspensions (18 × 108 cells/mL) in triplicate. Vehicle control and positive controls were evaluated in triplicate. 4
In the first trial, bacterial suspensions (100 µL) were mixed with the freshly prepared test item, vehicle, or the positive control (100 µL); the metabolic activation system or PBS (500 µL); and molten soft agar (2 mL) containing histidine-biotin (S. typhimurium) or tryptophan (E. coli) (Himedia) and poured onto MGA plates. Plates were incubated at 37 ± 1°C for 65 hrs and then inspected for evidence of cytotoxicity using the code system, and revertant colonies were counted manually. 4
In the second trial, bacterial suspensions (100 µL) were mixed with the metabolic activation system or PBS (500 µL) and the freshly prepared test item, vehicle, or positive control (100 µL) and incubated at 37 ± 1°C for 21–25 min with shaking. The incubated components were then mixed with molten soft agar (2 mL) and poured onto MGA plates. Plates were incubated at 37 ± 1°C for 65 hrs and were then inspected for evidence of cytotoxicity using the code system, and revertant colonies were counted manually. 4
For both trials, each test strain was diluted up to 10−7 in PBS and plated in triplicate on nutrient agar plates (Himedia) to obtain a viable count. Plates were incubated at 37 ± 1°C for 65 hrs, and then the number of colonies per plate was counted manually. For both trials, plates consisting of S9 homogenate, PBS, 5 mg/plate test item, soft agar, DMSO, and MGA were maintained at 37 ± 1°C for 65 hrs to assess potential contamination. 4
IN VITRO MICRONUCLEUS ASSAY
An in vitro micronucleus assay was performed on Biokesum® according to OECD Guideline 487 to assess structural and numerical chromosomal effects. 6,7 The study was conducted in accordance with Indian Council of Medical Research guidelines on biomedical research on human participants and was approved by the Institutional Ethics Committee.
Test substance solubility was evaluated in distilled water and DMSO (Sisco Research Laboratories or Finar). Stock solutions of the test substance were prepared at up to 200 mg/mL in DMSO and were diluted in culture medium to final concentrations up to 2 mg/mL. Dilutions were incubated at 37 ± 1°C, 5 ± 1% CO2 for 24 hrs and then evaluated for pH and inspected visually for precipitation. 6
Blood was collected from a healthy, nonsmoking 24-year-old male with no recent history of exposure to radiation or other genotoxic substances into a tube containing sodium heparin. In the initial cytotoxicity evaluation, duplicate aliquots of 0.5 mL whole blood were mixed with 4.4 mL culture media and 100 µL phytohemagglutinin (Gibco) and incubated for 44–48 hrs (37 ± 1°C, 5 ± 1% CO2). Culture media comprised RPMI 1640 (Gibco) with 10% fetal bovine serum (FBS, Gibco) and 1% penicillin-streptomycin (Sigma and Lonza, respectively). After incubation, cultures were centrifuged at 1500 rpm for 10 min, and the pellet was resuspended in fresh culture medium. Lymphocytes were then treated with the test substance or vehicle control (50 µL) with or without metabolic activation (0.5 mL) for 3 hrs or with the vehicle or test substance without metabolic activation, plus 6 µg/mL cytochalasin B (Sigma–Aldrich), for 24 hrs. The metabolic activation system was prepared as described above. After the 3-hr incubations, cells were centrifuged at 1500 rpm and resuspended in media containing 6 µg/mL cytochalasin B for an additional 20–24 hrs at 37 ± 1°C, 5 ± 1% CO2. Cells were then harvested via centrifugation, incubated with 0.56% potassium chloride for 10 min at room temperature, and mixed with 4 mL freshly prepared, cold fixative (acetic acid:methanol, 1:3, Finar; SDFCL and Finar, respectively) for 10 min at room temperature, followed by centrifugation at 2000 rpm for 10 min. The fixation incubation procedure was repeated two additional times. After the third fixation, cell suspensions were dropped onto chilled slides. Slides were air dried, stained for 5 min with acridine orange, and examined via light microscopy (40x) for frequencies of mononucleate, binucleate, and multinucleate cells. A total of 500 cells per culture were assessed. To assess cytotoxicity, the cytokinesis-block proliferation index (CBPI) was calculated using Equation 1.
6,7
Percent cytostasis was calculated using Equation 2:
CBIPT is the CBPI of treated cells, and CBIPC is the CBPI of the corresponding vehicle control. 6,7
The main study was performed in the same manner, except that positive controls were included. Positive control substances were cyclophosphamide monohydrate (Sigma–Aldrich), mitomycin C (MP Biomedicals), and colchicine (Himedia). 6,7
Three main study slides per concentration were evaluated for micronucleus formation; cytotoxicity was evaluated on one slide per treatment. Binucleate cells were only included in the analysis if the cytoplasm remained essentially intact and if the daughter nuclei were of approximately equal size. Micronuclei were identified using the following criteria:
6
Micronuclei should have the same staining characteristics and a similar morphology to the main nuclei; Micronuclei are separate in the cytoplasm or only just touching a main nucleus; Micronuclei are smooth-edged and smaller than approximately one-third the diameter of the main nuclei.
Data were statistically evaluated using SPSS software, version 27, using ANOVA followed by Dunnett’s t-test at a 95% level of confidence (i.e., P < 0.05).
IN VITRO CHROMOSOMAL ABERRATION TEST
Biokesum® was evaluated for its ability to produce structural chromosomal aberrations in an OECD Guideline 473 assay. 8,9
Blood was obtained from healthy adult donors, two males and one female, with no history of smoking and no known illnesses or recent vaccinations or exposures to potentially genotoxic agents, such as radiation. Heparinized whole blood (0.4 mL) was cultured in 9.6 mL RPMI 1640 media containing L-glutamine (MP Biomedical), 10% fetal bovine serum (Himedia), and 1% penicillin-streptomycin solution (Gibco). Lymphocyte mitosis was stimulated with phytohemagglutinin-M (10 µL/mL) followed by a 48-hr incubation at 37 ± 1°C, 5% carbon dioxide, and 90–100% humidity. 8,9
Test substance solubility was evaluated in analytical-grade water and DMSO. Dilutions of a DMSO stock solution (505 mg/mL) were added to 10 mL culture media to assess precipitation visually. For the cytotoxicity test, Biokesum® (39.0625–625 µg/mL) was added to cultured lymphocytes for 3 and 24 hrs without metabolic activation and for 3 hrs with metabolic activation. Cytotoxicity was assessed based on percent reduction in the mitotic index relative to the solvent control group. Mitotic index was calculated using Equation 3:
8
Percent reduction in the mitotic index compared to the vehicle control was calculated using Equation 4:
For the main study, duplicate cultures of proliferating lymphocyte cultures were treated with Biokesum® (0.15625, 0.3125, or 0.625 mg/mL) or with the vehicle or positive control substance (0.1 mL); for the cultures with metabolic activation, S9 mixture (0.5 mL, prepared and characterized as previously described) was also added. On the day of use, S9 homogenate (1 mL) was mixed with sterile water, 0.2 M phosphate buffer, pH 7.4, 0.1 M NADP, 1.0 M glucose-6-phosphate, 0.4 M magnesium chloride, and 1.65 M potassium chloride. 8
The positive controls for this study were methyl methanesulphonate (Sigma–Aldrich) and cyclophosphamide monohydrate (MP Biomedicals) and were formulated as stock solutions in analytical-grade water. 8,9
Cultures were incubated (37 ± 1°C, 5 ± 1% CO2) for 3 or 24 hrs without metabolic activation or for 3 hrs with activation. At the completion of the 3-hr incubation periods, cultures were centrifuged at 1000 rpm, washed to remove the test substance, and resuspended in 10 mL RPMI-1640 medium. Three-hour cultures were incubated for an additional approximately 18 hrs, and then colchicine (5 µg/mL) was added for a final 3-hr incubation. The 24-hr cultures were not rinsed and resuspended; colchicine was added for the final 3 hrs of the 24-hr exposure period. 8,9
After the 3-hr colchicine incubation, each culture was centrifuged at 1000 rpm for 5 min, and the supernatant was discarded. Cultures were subject to hypotonic lysis (0.56% potassium chloride) for 20 min at room temperature. Lysates were centrifuged at 1200 rpm for 5 min, the supernatant discarded, and the pellet resuspended in 5 mL fixative (cold, freshly prepared methanol: glacial acetic acid [3:1, v/v)]. Pellets were maintained in the fixative for 10 min at room temperature. This fixative sequence was repeated two additional times. After the third change of fixative, pellets were resuspended in 2 mL of fixative, and the resulting solution was dropped onto clean, chilled glass microscope slides. Chromosome preparations were heat fixed, stained with 5% Giemsa for 10 min, air dried, mounted with distyrene/dibutyl phthalate/xylene, coded, and examined for metaphases by transmission light microscopy (oil immersion objective, 100X magnification). At least 300 metaphases were evaluated for each culture and were evenly divided between the replicates. Aberrations were categorized as chromatid type, chromosome type, or other. Gaps, breaks, fragments, and exchanges were recorded and scored for chromatid and chromosome type aberrations. Other types of abnormalities, such as multiple chromatid breaks, pulverizations, deletions, and polyploidy, were also scored. For aberration frequency calculations, “gaps” were not considered structural chromosome abnormalities. 8,9
To confirm the sterility of the test system, nutrient agar plates were placed in a biosafety cabinet for approximately 30 min and then incubated for 24 hrs (37 ± 1°C, 5 ± 1% CO2). 8
Data were statistically evaluated using the chi-squared test (2 × 2 contingency tests) by comparing treatment groups to Poisson-based 95% confidence limits for the distribution of the historical negative control data. A value of P < 0.05 was considered statistically significant. 8
RESULTS
Bacterial reverse mutation test
The results of the solubility test identified DMSO as the appropriate vehicle. Preliminary studies identified minimal Biokesum® precipitation at 5 mg/plate and no precipitation at lower concentrations. In addition, Biokesum® was not cytotoxic to S. typhiumurim strain TA100 at 5 mg/plate based on a lawn intensity score of 4+ (normal thick background lawn). Biokesum® concentrations selected for the main studies were 0.05, 0.16, 0.5, 1.6, and 5 mg/plate. 4
In both trials, there was no evidence of cytotoxicity at any Biokesum® concentration in any test strain (Tables 1 and 2). The mean number of revertant colonies per plate was comparable to that of the vehicle control plates for both trials. In both trials, the positive controls produced ≥3.6-fold increases in the number of revertant colonies per plate, compared to the vehicle control plates. The viable count of each test strain in each trial met acceptability criteria, ranging from 1 × 109 to 2 × 109 CFU/mL. Quality control plates for each trial revealed no evidence of contamination. 4,5
Summary of Colony Counts—Trial-I—Plate Incorporation Method
Table 1. Bacterial colony counts for the plate incorporation method. Values of revertants are mean±SD.
Lawn intensity.
4+ = Thick lawn: Distinguished by a healthy (normal) background lawn compared to vehicle control plates.
Positive controls.
With S9.
For S. typhimurium TA98, TA100, TA1535, and TA1537 = 4 μg/plate 2-aminoanthracene.
For E.coli uvrA pKM 101 = 30 μg/plate 2-aminoanthracene.
Without S9.
For TA98: 2 μg/plate 2-nitrofluorene.
For TA100 and TA1535: 1 μg/plate sodium azide.
For TA1537: 50 μg/plate 9-aminoacridine.
For E. coli uvrA pKM 101: 5 μg/plate 4-nitroquinoline N-oxide.
Summary of Colony Counts—Trial-II—Preincubation Method
Table 2. Bacterial colony counts for the preincubation method. Values of revertants are mean±SD.
Lawn intensity.
4+ = Thick lawn: Distinguished by a healthy (normal) background lawn compared to vehicle control plates.
Positive controls: With S9.
For S. typhimurium TA98, TA100, TA1535, and TA1537 = 4 μg/plate 2-aminoanthracene.
For E.coli uvrA pKM 101 = 30 μg/plate 2-aminoanthracene.
Without S9.
For TA98: 2 μg/plate 2-nitrofluorene.
For TA100 and TA1535: 1 μg/plate sodium azide.
For TA1537: 50 μg/plate 9-aminoacridine.
For E.coli uvrA pKM 101: 5 μg/plate 4-nitroquinoline N-oxide.
In vitro micronucleus assay
The results of the solubility test identified DMSO as the appropriate solvent. When DMSO stock solutions were diluted with culture media, mild precipitation occurred at 0.25 mg/mL, and there was no visible precipitation at lower concentrations. The test substance did not affect culture pH. Therefore, the initial cytotoxicity test was performed using 0.015625–0.25 mg/mL test substance. Percent cytotoxicity ranged from 1.59% to 42.86%, and the percent reduction in CBPI was 57.14–60.32% at 0.25 mg/mL. The concentrations selected for the main study were 0.0625, 0.125, and 0.25 mg/mL. 6
Micronuclei frequency in binucleate cells was not significantly increased at any test substance concentration, compared to the vehicle control (Fig. 1, Supplementary Tables S1, S2, and S3). Each positive control substance elicited a statistically significant (P < 0.05) increase in micronuclei frequency. Cytotoxicity ranged from 39.34% to 42.62% in the 0.25 mg/mL main study cultures. 6,7
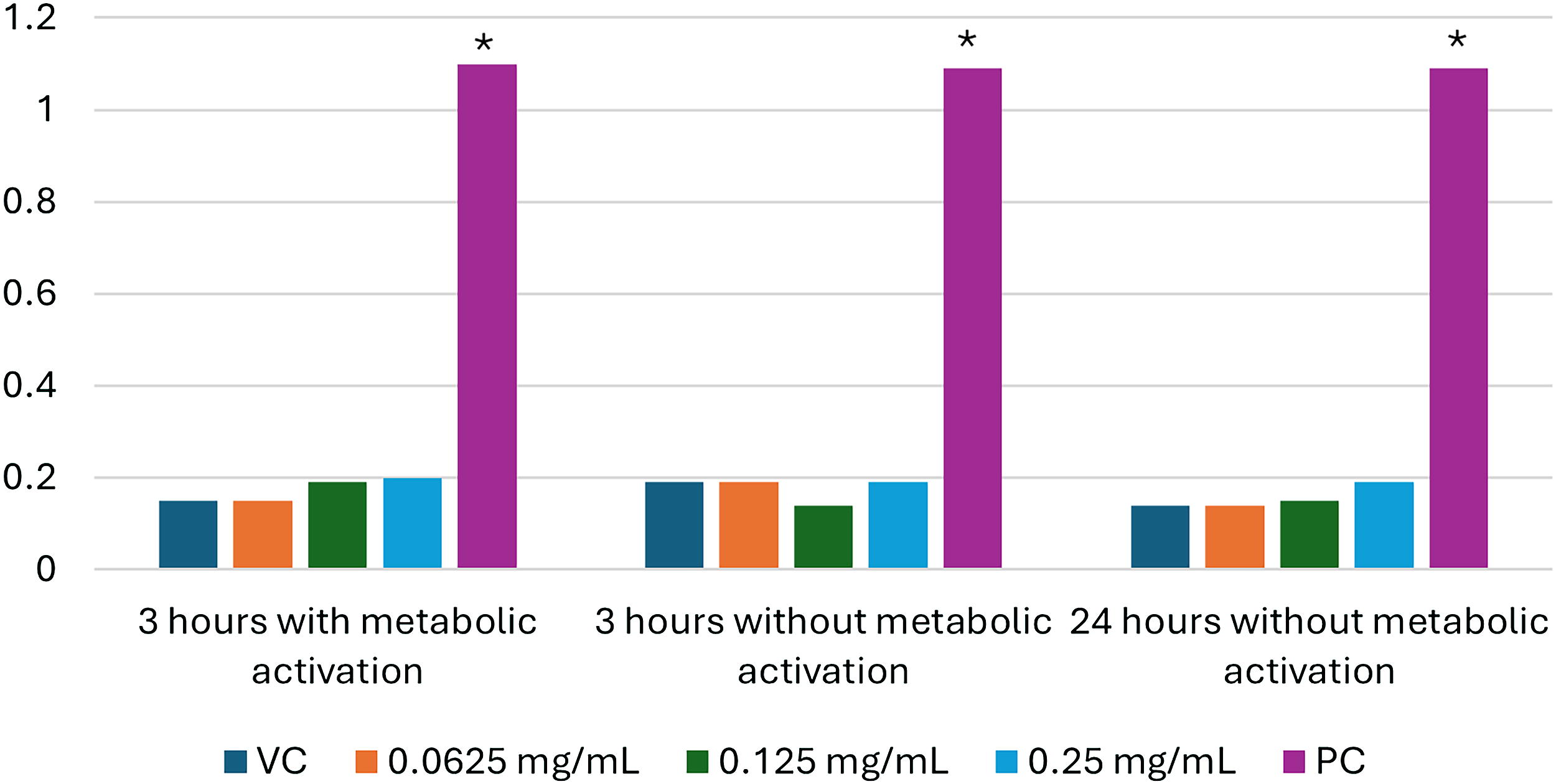
Percentage of human lymphocytes with micronuclei. *Statistically significant (P < 0.05). VC—vehicle control (DMSO); PC—positive control (cyclophosphamide monohydrate, 10 µg/mL, 3 hrs with metabolic activation; mitomycin C, 0.03 µg/mL, 3 hrs without metabolic activation); and colchicine, 0.075 µg/mL, 24 hrs without metabolic activation).
Chromosomal aberration study
Biokesum® formed a suspension in analytical-grade water and formed a uniform suspension in DMSO. The concentrations tested in the main chromosomal aberration study were 0.15625 mg/mL, 0.3125 mg/mL, and 0.625 mg/mL, based on solubility. The maximum concentration did not affect culture pH, produced slight precipitation, and reduced mitotic index by 20–30% compared to vehicle control. The quality control plates showed no evidence of contamination. 8
Chromosomal aberration test results are shown in Figure 2, with supplementary data provided in Supplementary Tables S4, S5, and S6. There were no statistically significant increases in any type of structural chromosomal aberration (whether excluding or including gaps) in any of the Biokesum® treatment groups, compared to vehicle control. All positive controls produced a statistically significant increase (P < 0.05) in aberration frequency. Cytotoxicity ranged from 15.00% to 30.13% in the 0.625 mg/mL main study cultures. 8,9
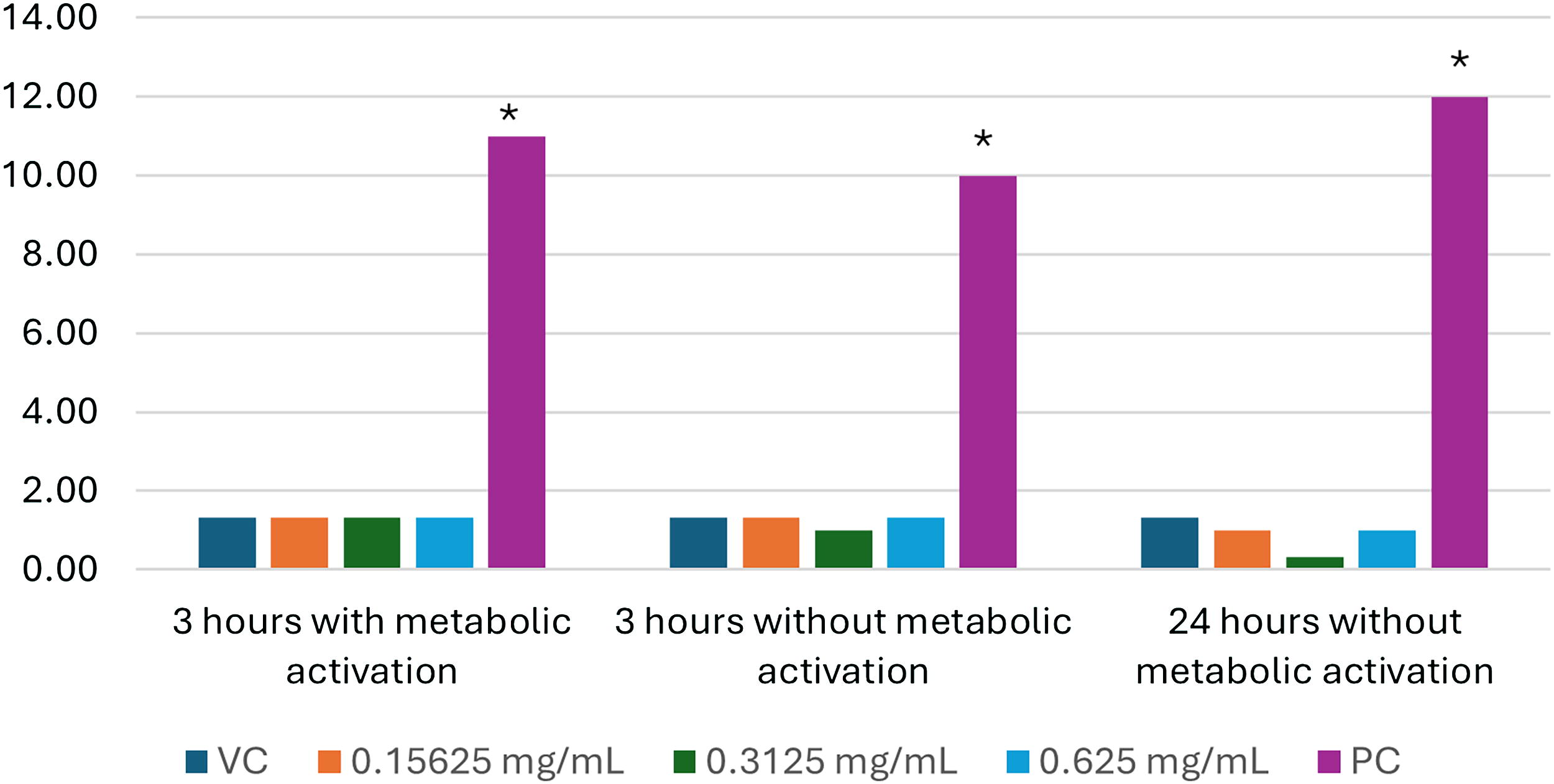
Percentage of human lymphocytes with chromosomal aberrations. *Statistically significant (P < .05). VC—vehicle control (DMSO); PC—positive control (methyl methanesulphonate, 30 µg/mL, incubations without metabolic activation; cyclophosphamide monohydrate, 60 µg/mL, incubations with metabolic activation).
DISCUSSION
The results of these three genotoxicity assays demonstrate that Biokesum® Polygonum minus (Persicaria minor) standardized extract does not pose a concern for genotoxicity.
The evaluation of genotoxicity is most appropriately approached using multiple assays, as there is no single in vitro assay that can accurately identify 100% of substances that are genotoxic and also accurately identify 100% of substances that are not genotoxic (i.e., 100% sensitivity and specificity), thereby limiting their predictive value for carcinogenicity. Even the widely used Ames bacterial reverse mutation assay has only 49–62.3% sensitivity and 73.9–80.3% specificity with respect to predicting rodent carcinogenicity. 10 The mammalian cell chromosomal aberration assay has 48.4–69.4% sensitivity and 44.9–68% specificity and has largely been replaced by the micronucleus assay due to low specificity. 10 The in vitro micronucleus assay, which is among the newest of the validated genotoxicity assays, has higher sensitivity, 67–93.6%, but variable specificity, 16.7–100%. Although the in vivo micronucleus assay has historically been assumed to be more relevant and is required under some regulatory frameworks, its sensitivity and specificity are less than ideal, 44–80% and 67%, respectively. 10
Using multiple assays increases the overall predictive value relative to individual assays. For example, the Ames test and in vitro micronucleus test together correctly identified 73.4% of known rodent carcinogens and 78% of known in vivo genotoxicants. 11 The battery of OECD Guideline assays selected for Biokesum® Polygonum minus (Persicaria minor) standardized extract therefore represents the best available scientific approach for the evaluation of genotoxicity and provides additional safety substantiation for this popular herbal extract.
CONCLUSION
The results of three OECD Guideline genotoxicity assays, performed at accredited laboratory facilities and in compliance with GLPs, confirm that Biokesum® Polygonum minus (Persicaria minor) standardized extract is non-mutagenic and does not induce structural or numerical chromosomal aberrations.
Footnotes
AUTHORS’ CONTRIBUTIONS
J.A.: Writing—original draft, writing—review and editing, visualization. M.K.: Writing—original draft. R.S., R.C.K., and Y.P.S.: Investigation. M.H.W.: Writing—review and editing.
AUTHOR DISCLOSURE STATEMENT
J.A., M.K., and M.H.W. provide scientific consultancy services for Biotropics Malaysia Berhad. R.S., R.C.K., and Y.P.S. have no interests to disclose.
FUNDING INFORMATION
This work was sponsored by
SUPPLEMENTARY MATERIAL
Supplementary Data
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
