Abstract
Collagen peptides (CPs) have been recognized for their potential to enhance skin health, particularly by improving hydration, firmness, and elasticity. However, the molecular mechanisms driving these benefits remain not fully understood, especially regarding their influence on essential extracellular matrix (ECM) components and enzymes that regulate collagen turnover. This study investigated both the clinical efficacy and the underlying preclinical molecular effects of oral collagen oligopeptides (Col-OP) supplementation, aiming to clarify how these peptides contribute to skin improvements. In clinical trials, a double-blind, randomized, placebo-controlled study with 85 women aged 45–60 years examined the effects of daily 2.5 g Col-OP supplementation over 84 days, measuring skin hydration (Corneometer®), firmness, and elasticity (Cutometer®). In addition to clinical studies, preclinical in vitro experiments were conducted on human dermal fibroblast cultures to elucidate the molecular effects of Col-OP. Fibroblasts were treated with noncytotoxic concentrations of Col-OP (10.0, 3.16, and 1.0 mg/mL) for 96 h, assessing the synthesis of type I collagen, matrix metalloproteinase-1 (MMP-1), tissue inhibitor of matrix metalloproteinases (TIMP-1), decorin, versican, biglycan, and hyaluronic acid (HA) through gene expression and enzyme-linked immunosorbent assays. Results showed that Col-OP treatment significantly enhanced gene expression of type I procollagen, decorin, and biglycan while decreasing versican levels (P < .001). It also promoted type I collagen synthesis and TIMP-1 levels, modulating MMP-1 and HA production. Clinically, Col-OP significantly enhanced skin firmness and elasticity compared with placebo (P < .05), while improvements in skin hydration did not achieve statistical significance. Based on the current data, it can be concluded that oral supplementation with Col-OP effectively enhances skin health by promoting key ECM components and modulating collagen turnover, offering a promising approach for improving skin health.
INTRODUCTION
Collagen is the main structural protein in connective tissues, including skin, tendons, cartilage, and bones, constituting 25–30% of total body proteins. It is essential for mechanical stability, elasticity, and tissue resistance. 1 –3 In the skin, collagen accounts for up to 90% of the extracellular matrix (ECM), collaborating with elastin and other components to support cellular functions, enable cell communication, and maintain tissue integrity and hydration. 4 –6
As skin ages, the epidermis and dermis undergo significant changes not only due to the natural aging process but also as a result of environmental factors, epigenetic modifications, and lifestyle influences. 7 –9 The collagen network degrades, resulting in shorter and disorganized fibers, while elastic fibers lose their structure, reducing skin elasticity and strength. 10,11 A decrease in proteoglycans impairs moisture retention, contributing to dryness and making the skin more prone to wrinkles. 6,12 Additionally, aging increases the activity of matrix metalloproteinases (MMPs), which accelerate collagen breakdown and hinder the production of new ECM components, further advancing the aging process. 2,7,9
Oral supplementation with collagen-based nutraceuticals has become increasingly popular. 13 –18 Collagen peptides (CPs) are low-molecular-weight peptides derived from the hydrolysis of native collagen found in bovine, porcine, or marine skin. 19 –22
These peptides are particularly rich in hydroxyproline, proline, and glycine—amino acids crucial for skin health. 23 After ingestion, CPs are broken down into smaller dipeptides and tripeptides, which are absorbed in the digestive tract and transported via the bloodstream to the skin. 24 Once in the dermis, they enhance fibroblast activity by stimulating cellular metabolism, proliferation, and migration. 20,25 CPs also promote the synthesis of collagen and HA, contributing to water retention and improved skin hydration. 16 –18
Several studies have reported that oral collagen supplementation improves skin hydration, enhances elasticity, increases dermal density, and reduces facial wrinkles. These benefits have been observed across various concentrations, sources, formulations, and delivery methods (both liquid and solid). 14 –17 However, while many studies confirm these general benefits, fewer investigations have explored the molecular mechanisms behind these improvements or evaluated the synthesis of individual ECM components affected by CPs. 25 –28
Despite growing evidence on the benefits of CPs for skin health, questions remain about how exactly these peptides influence key components of the ECM and regulate enzymes involved in collagen turnover, such as MMP-1 and tissue inhibitor of MMPs (TIMPs). Additionally, most studies focus on general improvements in hydration and elasticity without exploring their effects on proteoglycans such as decorin, versican, and biglycan—molecules that are essential for collagen fibril formation and tissue repair. 29 –31 Understanding these molecular pathways is critical for developing more targeted and effective collagen-based interventions.
To address this gap, we designed a study to evaluate both the clinical efficacy and molecular effects of an oral collagen oligopeptide (Col-OP) supplement. Our study aimed to assess the effects of Col-OP on skin hydration, firmness, and elasticity using instrumental techniques. In addition to clinical studies, preclinical in vitro experiments were conducted on human dermal fibroblast cultures to elucidate the molecular effects of Col-OP on gene expression and the synthesis of key ECM components, including type I collagen, MMP-1, TIMP-1, decorin, versican, biglycan, and HA, in human fibroblast cultures.
MATERIALS AND METHODS
Test substance
The hydrolyzed collagen used in this study is an advanced form of CPs known as oligopeptides. The Col-OP provided by Genu-in—JBS S/A is commercially available as Genu-in Skin. This food-grade Col-OP is composed of CPs derived through a complex process of degradation and enzymatic hydrolysis of bovine type I collagen.
The product contains a blend of CPs with an average molecular weight of approximately 4 kDa, with a significant portion (40%) presenting molecular weights <1 kDa.
Preclinical trials
Human dermal fibroblasts were isolated from elective abdominoplasty (approved by the Ethics Committee of the University of São Francisco—SP, CAAE 56005722.8.000.5514, opinion no. 5.503.565) and cultured in Dulbecco’s modified Eagle’s medium containing 4.5 g/L glucose (Thermo Fisher Scientific, Inc., Waltham, MA), supplemented with 10% fetal bovine serum (Thermo Fisher, Inc.) and 1% penicillin/streptomycin/amphotericin (Lonza Walkerville, Inc., Salisbury, MD). Cells were incubated with three noncytotoxic concentrations of Col-OP (10.0, 3.16, and 1.0 mg/mL) for a period of 96 h. These concentrations were selected based on previous results of cytotoxicity assays (data not shown). Total RNA and cell supernatants were collected to assess the expression and quantification of the selected mediators. For the analysis of MMP-1, cells were further exposed to ultraviolet (UV) radiation at a dose of 5 J/cm2 using UVA Cube 400, SOL 500 H2 filter, and UV Meter (Honle UV America, Inc., Marlborough, MA).
For gene expression, total RNA was extracted from the cell cultures using TRIzol® reagent (Thermo Fisher) and quantified with Qubit4 using the Qubit™ RNA IQ Assay Kit (Thermo Fisher, Inc.). Real-time reverse transcription PCR was performed on a QuantStudio™ 3 detection system (Thermo Fisher, Inc.). Gene expression of type I procollagen (Hs01028956_m1), decorin (Hs00754870_m1), versican (Hs00171642_m1), biglycan (Hs00959143_m1), and endogenous control B2M gene (beta-2-microglobulin) was assessed using the TaqMan RNA-to-Ct™ 1-Step Kit and TaqMan® Gene Expression Assays (Thermo Fisher, Inc.). Relative messenger RNA quantification was calculated using 2−ΔΔC(T) method. 32
Protein quantification was performed by enzyme-linked immunosorbent assay kits: type I collagen (ABclonal Biotechnology Co., Ltd., Woburn, MA), MMP-1 (R&D Systems Inc., Minneapolis, MN), TIMP-1 (R&D Systems Inc.), and HA (NeoBiotech, Nanterre, France).
Clinical trials
Study design
A monocentric, double-blind, randomized, placebo-controlled clinical study was carried out in 85 female participants (45–60 years, skin phototypes II–V—according to Fitzpatrick) exhibiting facial wrinkles and expression lines. Exclusion criteria required participants to not be pregnant or nursing, have no history of skin diseases, and not be undergoing medical or cosmetic treatment during the study. They also could not have used retinoic acid, chemical peels, or similar procedures within 4 weeks prior to the study. Participants had to avoid excessive sun exposure, tanning, and using supplements or certain skincare products and could not change contraceptive methods, diet, or cosmetic habits. Noncompliance led to exclusion.
The included subjects were randomized for treatment with a daily dose of 2.5 g of Col-OP or placebo (maltodextrin). Both groups were instructed to dissolve 2.5 g of the product in water and consume it once a day in the morning. The study lasted 84 days, with intermediate evaluations conducted at 28 (T28d), 56 (T56d), and 84 (T84d) days to assess skin hydration, firmness, and elasticity, on either the right or left cheek of each participant.
The study was approved by the Ethics Committee of the University of São Francisco—SP (CAAE 70145223.3.0000.5514, opinion no. 6.122.983). All subjects gave signed informed consent after receipt of the written information and having had the possibility for further questions.
Efficacy and safety assessment
Skin hydration was measured using the Corneometer® 825 probe connected to the Multi Probe Adapter, MPA 5 equipment (Courage & Khazaka Electronic GmbH, Köln, Germany). The Corneometer is based on capacitance measurements, which reflect the water content of the skin. The readings, expressed in arbitrary units (a.u.), range from 0 (dry) to 120 (fully hydrated). 33 Skin firmness and elasticity were assessed using a noninvasive equipment that can determine the viscoelasticity properties of skin using the suction method (Cutometer® Dual MPA 580; Courage & Khazaka). Skin firmness was assessed using the Uf parameter (R0), which is the final distension of the first curve and consists of maximum skin deformation after applying a force. The increase in skin firmness is evidenced by the significant reduction of the Uf parameter after the treatment. Skin elasticity was assessed using the Ur/Uf parameter (R7), the ratio of elastic recovery to the total deformation: Ur = immediate retraction/Uf = total deformation. The increase in skin elasticity is evidenced by the increase in Ur/Uf parameter values. 34 Safety and tolerability were evaluated through dermatological assessments performed before, during, and after the study.
Statistical analysis
For preclinical statistical evaluation, an analysis of variance test was used to measure the variation of the results, comparing the data between all the groups. The Bonferroni post-test was applied to enhance the precision of the results. Clinical data analysis and statistics were performed using the paired bimodal Student’s t-test, considering a 95% confidence interval. All statistical analyses were performed using GraphPad™ Prism® 8.0 software (San Diego, California) and considering a 5% significance level.
RESULTS
Preclinical effects of Col-OP on the synthesis of ECM components
Figure 1 shows the effects of Col-OP on gene expression in human fibroblast cultures. Col-OP at 10.0 mg/mL increased type I procollagen expression by 2.05-fold compared with the untreated control (Fig. 1A; P < .01). Decorin expression also significantly increased (P < .01) by 1.43- and 1.36-fold at 10.0 and 3.16 mg/mL, respectively, compared with the untreated control (Fig. 1B). Biglycan expression rose by 2.78-, 2.49-, and 2.27-fold at 10.0, 3.16, and 1.0 mg/mL, respectively, in relation to the control group (Fig. 1C; P < .01). Conversely, when compared with the untreated group, Col-OP decreased versican expression to 20-, 15.6-, and 8.6-fold at 10.0, 3.16, and 1.0 mg/mL, respectively (Fig. 1D; P < .001).
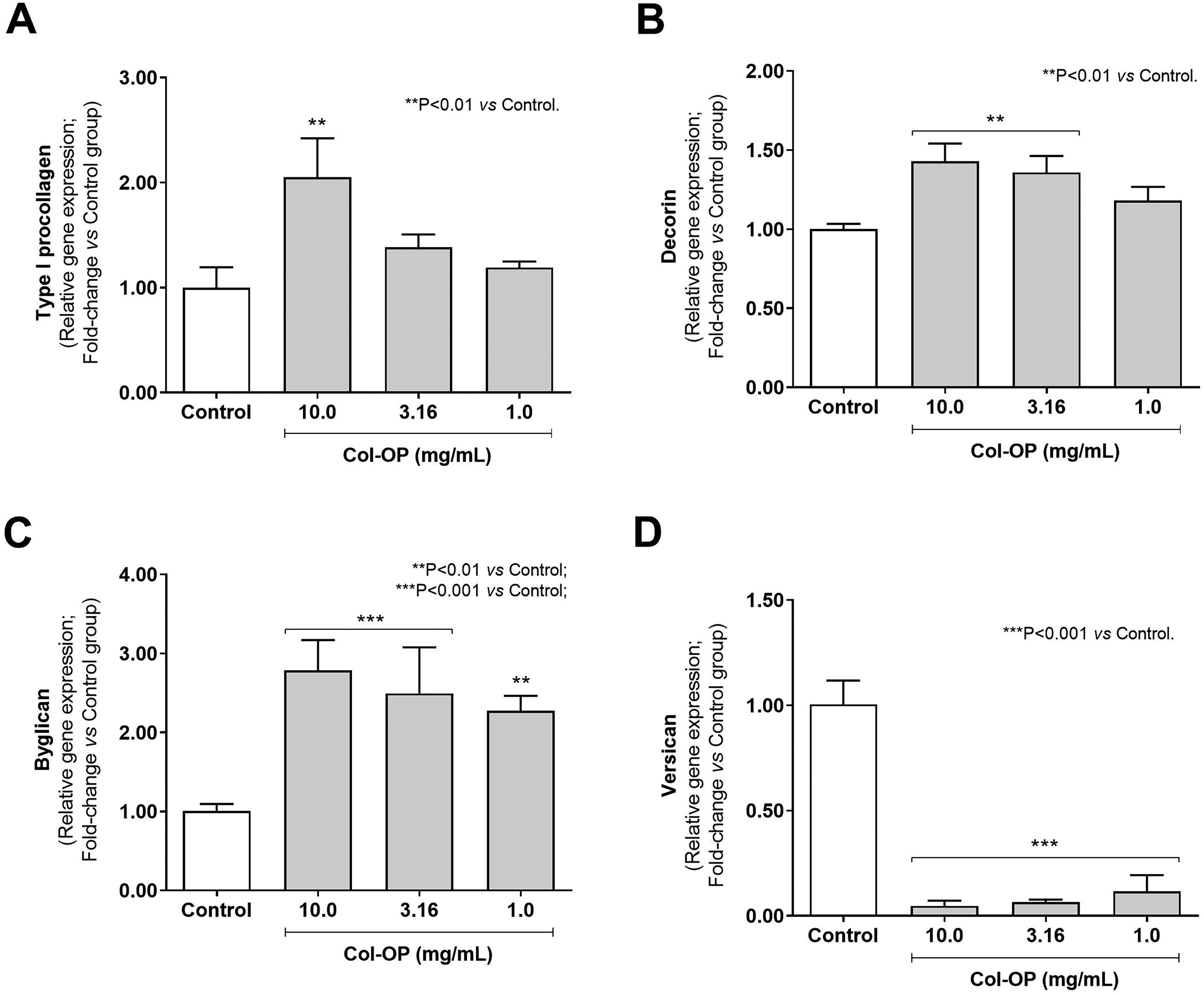
Type I procollagen
Regarding protein quantification, Col-OP modulated the synthesis of type I collagen, TIMP-1, HA, and MMP-1 in human fibroblast cell cultures. Cell incubation with Col-OP at 10.0, 3.16, and 1.0 mg/mL significantly increased type I collagen synthesis by 16.02, 9.78, and 6.86%, respectively, compared with the untreated group (Fig. 2A). TIMP-1 production rose by 1.28- and 1.53-fold (P < .001) at 10.0 and 3.16 mg/mL, respectively, compared with baseline control (Fig. 2B). HA levels were also stimulated by incubation with Col-OP at all evaluated concentrations. Compared with the untreated control group, Col-OP at 10.00, 3.16, and 1.00 mg/mL promoted increases of 2.43-, 1.84-, and 1.79-fold, respectively (Fig. 2C). Fibroblast cultures exposed to UV radiation showed elevated MMP-1 levels by 1.27-fold (P < .001) relative to the nonirradiated untreated control, confirming the occurrence of photodamage. In contrast, incubation with Col-OP reduced MMP-1 production by 20.84%, 18.69%, and 17.72% compared with the UV-only group (Fig. 2D).
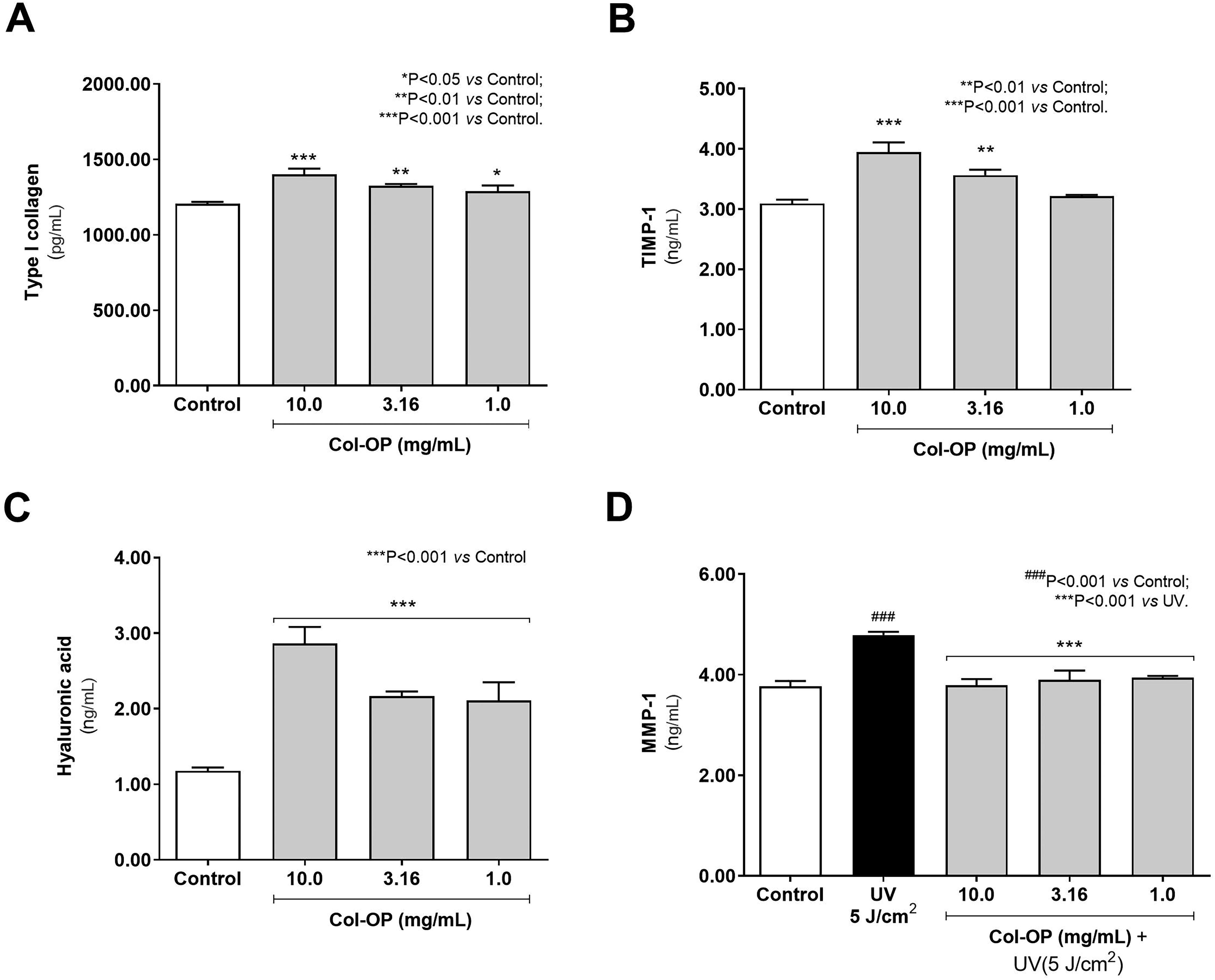
Protein quantification of type I collagen
Clinical effects of Col-OP on skin hydration and viscoelasticity properties
Figure 3 presents the mean capacitance values recorded at baseline and throughout the study period. Both the Col-OP and placebo groups showed significant increases in skin hydration on days 28, 56, and 84 compared with their respective baselines. However, no significant differences were observed between the Col-OP and placebo groups over the study period.
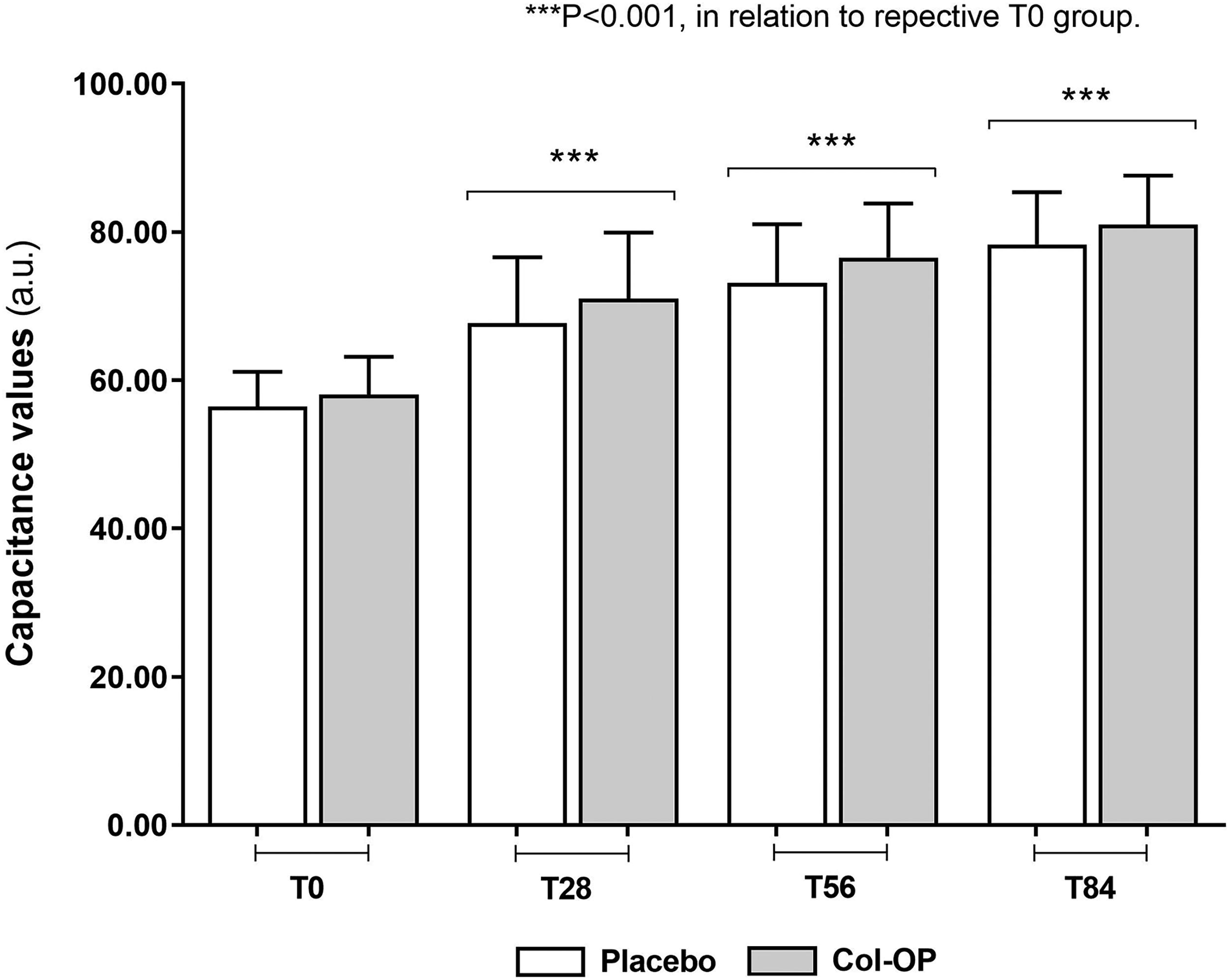
Corneometric index performed in human subjects at baseline (T0), after 28, 56, and 84 days using the oral collagen oligopeptides (Col-OP) supplement and the placebo treatment (maltodextrin). Data represent mean ± standard deviation (n = 42 for Col-OP; n = 43 for placebo; paired bimodal Student’s t-test).
Skin firmness and elasticity, represented by the mean values of R0 and R7 parameters, respectively, are represented in Figure 4A and B. A significant increase in skin firmness was observed after 28, 56, and 84 days in the Col-OP group and after 84 days in the placebo group, compared with respective baseline values (P < .001; Fig. 4A). The collagen supplement resulted in a 3.7% increase in skin firmness after 28 days, 9.7% after 56 days, and 12.2% after 84 days compared with T0. Col-OP significantly improved skin firmness in 86% of participants after 28 days and in 100% of participants after 56 and 84 days. Moreover, the Col-OP supplement provided a significantly greater increase in skin firmness at all time points compared with the placebo (P < .001).
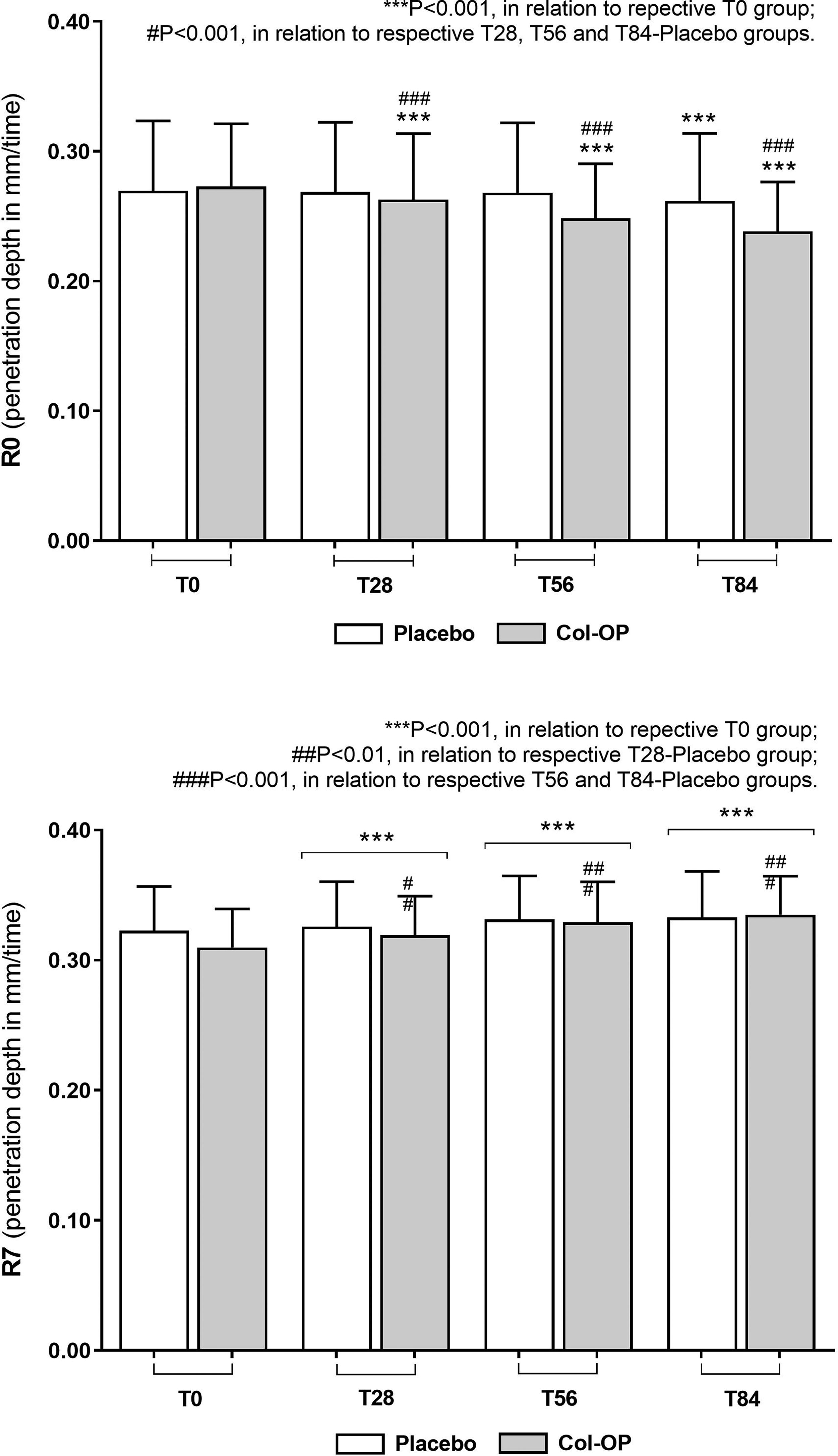
Evaluation of skin firmness (R0) and elasticity (R7) performed in human subjects at baseline (T0), after 28, 56, and 84 days using the oral collagen oligopeptides (Col-OP) supplement and the placebo treatment (maltodextrin). Data represent mean ± standard deviation (n = 42 for Col-OP; n = 43 for placebo; paired bimodal Student’s t-test).
In both the Col-OP and placebo groups, skin elasticity (R7) showed a significant increase after 28, 56, and 84 days compared with their respective baseline values (P < .001; Fig. 4B). Col-OP supplement resulted in a 3.1% increase in skin elasticity after 28 days, 6.8% after 56 days, and 8.3% after 84 days, in relation to T0. Additionally, skin elasticity significantly improved in 100% of participants after 84 days of Col-OP use. Furthermore, collagen supplement provided a statistically greater increase in skin elasticity at all time points compared with placebo.
DISCUSSION
Collagen, the most abundant protein in the human body, is essential for the structure of the dermal ECM, contributing to skin strength, elasticity, and hydration. With aging, collagen production declines, leading to reduced firmness, diminished hydration, and the formation of wrinkles. 2,10,35 Oral supplementation with CPs (Col-OP) has gained prominence for offering long-term benefits for skin health. 3,14 –17,36
This study evaluated the effects of Col-OP supplementation over 84 days, showing progressive improvements in skin hydration, elasticity, and firmness. Hydration increased by up to 39.9%, but no statistically significant difference was observed between the Col-OP and placebo groups, suggesting that other factors, such as skincare habits, may have influenced the results. Nonetheless, the Col-OP group exhibited a consistent trend of improved hydration, aligning with previous research indicating that regular collagen intake can help maintain skin moisture. 11,27,36
Additionally, significant improvements in elasticity and firmness among participants receiving Col-OP were detected, which are clinically relevant for skin integrity and damage prevention. Col-OP supplementation also upregulated key genes involved in ECM organization, including type I procollagen, decorin, and biglycan, which are crucial for collagen fibril formation and tissue repair. At the same time, an increase in TIMP-1 production, which inhibits collagen degradation, and a reduction in MMP-1 levels were observed in the Col-OP group. These findings suggest that Col-OP preserves ECM integrity, thereby contributing to reduced collagen degradation and enhanced tissue structure. The increase in hyaluronic acid (HA) following Col-OP supplementation indicates that CPs promote not only the restoration of structural proteins but also a biochemical environment conducive to sustained skin regeneration.
Zague et al. (2011) demonstrated that CP supplementation increased type I and IV collagen levels while reducing both proenzyme and active forms of MMP-2 in rat skin, suggesting potential benefits against aging-related ECM degradation. 28 Further research by the same group showed that CPs significantly modulate ECM metabolism in human dermal fibroblasts from sun-protected and sun-exposed sites, enhancing procollagen I and collagen I synthesis and reducing collagen breakdown through MMP-1 and MMP-2 inhibition. 37
An intriguing finding was the significant reduction in versican expression following Col-OP supplementation. Versican is a proteoglycan associated with chronic inflammation and ECM disorganization, particularly in aged tissues. 29,38 –40 The decrease in versican expression may suggest that Col-OP promotes more efficient ECM reorganization, reducing the need for high levels of this proteoglycan.
Our findings are consistent with prior clinical studies documenting the benefits of oral collagen supplementation for skin health, showing improvements in hydration and elasticity. Asserin et al. (2015) reported significant improvements in skin hydration and collagen density after 8 weeks of supplementation, with benefits persisting up to 12 weeks. 13 Similarly, Proksch et al. (2014) demonstrated that daily collagen intake improved skin elasticity, with effects remaining noticeable 4 weeks after discontinuation. 24 In another study, Kim et al. (2022) found that low-molecular-weight CPs reduced wrinkles and improved skin hydration and elasticity after 12 weeks of treatment. 36
The combination of clinical and molecular evidence provides a more comprehensive understanding of collagen’s effects, demonstrating that Col-OP supplementation not only improves skin quality but also regulates critical ECM components.
The sustained improvements observed over 84 days suggest that long-term supplementation with Col-OP may contribute to lasting changes in the skin matrix, supporting both functional and esthetic outcomes. Additionally, our findings indicate the potential of Col-OP to aid in wound healing and tissue repair, expanding the possible therapeutic applications of collagen supplementation.
Footnotes
AUTHORS’ CONTRIBUTIONS
V.Z. was responsible for the study conceptualization, methodology, writing original draft preparation, and review and editing. A.L.T.A.P. was responsible for methodology. J.R.P., G.F., and S.E. were responsible for methodology and writing original draft preparation.
AUTHOR DISCLOSURE STATEMENT
V.Z. is an employee of Genu-in—JBS S/A. No competing financial interests exist for the other authors.
FUNDING INFORMATION
This work was supported by grants from
