Abstract
Moringa oleifera Lam. leaves contain various nutrients and bioactive compounds. The present study aimed to assess the anti-fatigue capacity of a flavonoids concentrate purified from M. oleifera Lam. leaves. The total flavonoids in the purified extract were analyzed by ultra-performance liquid chromatography electrospray ionization tandem mass spectrometry (UPLC-MS/MS). The mice were supplemented with purified M. oleifera Lam. leaf flavonoid-rich extract (MLFE) for 14 days. The weight-loaded forced swimming test was used for evaluating exercise endurance. The 90-min non-weight-bearing swimming test was carried out to assess biochemical biomarkers correlated to fatigue and energy metabolism. UPLC-MS/MS analysis identified 83 flavonoids from MLFE. MLFE significantly increased the swimming time by 60%. Serum lactate (9.9 ± 0.9 vs. 8.9 ± 0.7), blood urea nitrogen (BUN) (8.8 ± 0.8 vs. 7.2 ± 0.5), and nonesterified fatty acid (NEFA) (2.4 ± 0.2 vs. 1.7 ± 0.3) were significantly elevated; phosphoenolpyruvate carboxykinase (PEPCK), glucokinase (GCK), and nuclear factor erythroid 2-related factor 2 (Nrf2) mRNA expression were significantly downregulated; and heme oxygenase 1 mRNA expression was significantly upregulated in muscle after swimming. MLFE supplement significantly decreased serum lactate (8.0 ± 1.0 vs. 9.9 ± 0.9), BUN (8.6 ± 0.4 vs. 8.9 ± 0.8), and NEFA (2.3 ± 0.4 vs. 2.4 ± 0.2) and increased the protein and mRNA expression of GCK, PEPCK, and Nrf2. The enhancement of glucose metabolism and antioxidant function by MLFE contributes partly to its anti-fatigue action.
INTRODUCTION
Various nutrients and phytonutrients were identified among the roots, bark, leaves, flowers, seed, and immature pods of Moringa oleifera Lam. (M. oleifera). 1 The extracts from M. oleifera were prepared with different procedures (alcoholic, hydroalcoholic, or aqueous extractions). 2,3 M. oleifera leaves contain various bioactive compounds, including phenolics, flavonoids, glucosinolates, isothiocyanates, tannins, saponins, vitamins, and carotenoids. 4 M. oleifera leaf extract exhibits antioxidant, 5 anti-inflammatory, 6 antiaging, 7 antibacterial, 8 hypolipidemic, hypoglycemic, 9 antihypertensive, 10,11 hepatoprotective, 12 and anticancer activities. 13 Recently, Lamou et al. reported the anti-fatigue properties of the aqueous extract of M. oleifera leaves. The aqueous extract enhanced energy stores and antioxidant capacity and decreased lactate accumulation in male Wistar rats. 14 We previously demonstrated that M. oleifera leaf ethanol extract improved exercise performance through the 5′ adenosine monophosphate-activated protein kinase (AMPK) pathway, which enhances glycogen stores and antioxidant enzyme activity. 15 However, whether the purified concentrate from the ethanol extract shows stronger anti-fatigue properties remains unclear, and the mechanisms involved in are not well described.
Exercise-induced fatigue is a common and complex multidimensional symptom, manifesting as an overwhelming sense of tiredness, feeling of exhaustion, and difficulty in performing voluntary tasks. 16 Physical fatigue is usually characterized by a decreased athletic ability and even difficulty in maintaining their motivation. 17 The required level of strength in the muscle exercise is unable to be maintained during physical fatigue. 18 The prolonged swimming time indicates anti-fatigue action in exhaustive swimming tests. Glucokinase (GCK) and phosphoenolpyruvate carboxykinase (PEPCK) are important enzymes related to energy metabolism. These enzymes increase the utilization rate of glucose, improve exercise endurance, and antagonize exercise fatigue. 19,20 Reactive oxygen species (ROS) that are produced during contractile movement can be scavenged by antioxidants such as superoxide dismutase (SOD), catalase, peroxiredoxins, and heme oxygenase 1 (HO-1). Nuclear factor erythroid 2-related factor 2 (Nrf2) regulates the transcription of HO-1 and SOD. 21,22 We prepared purified extract rich in flavonoids from the M. oleifera leaves through a macroporous adsorption resin method in the present study. Ultra-performance liquid chromatography electrospray ionization tandem mass spectrometry (UPLC-MS/MS) was used to assay flavonoids from the M. oleifera leaves. Swimming tests were performed to assess the anti-fatigue action of the concentrate, and the potential mechanisms were also explored.
MATERIALS AND METHODS
Purification of total flavonoids by NKA-II resins from crude extract of M. oleifera leaves
The crude ethanol extract of M. oleifera leaves (Yuanmou New Agricultural Construction Biological Research Co., Ltd.) was prepared by using 70% ethanol (material to liquor was 1:20) and ultrasonic-assisted extraction (800 W, 25°C) followed by freeze-drying. The crude extract of M. oleifera was dissolved in 70% ethanol, left to stand, and filtered. The precipitate was dissolved in distilled water and then purified by NKA-II macroporous resin column chromatography (bed volume [BV], 2 L). The extracted filtrate (10 L) of M. oleifera leaves was first passed through an NKA-II macroporous resin column at a flow rate of 1.5 BV/h, then the NKA-II macroporous resin column was rinsed with 2 BV of distilled water to remove impurities, followed by an NKA-II macroporous resin column rinsed with 11 BV of 70% ethanol at a flow rate of 2.5 BV/h to elute total flavonoids, and the eluate was collected. Then the flavonoids elute was concentrated and freeze-dried.
Analysis of flavonoids in the extract of M. oleifera leaves
UPLC-MS/MS system was used for qualitative analysis of flavonoids in the M. oleifera leaves extract. About 4 μL of purified M. oleifera leaves extracts was injected into the system with HSS T3 C18 (1.8 μm, 2.1 × 100 mm) column held at 40°C. Mobile phase A was water (0.04% acetic acid in water), and mobile phase B was acetonitrile (0.04% acetic acid in acetonitrile). The chromatographic gradient: 0–10 min, linear gradient from 95% to 5% mobile phase A; 10–11 min, hold at 5% mobile phase A; 11–11.1 min, linear gradient from 5% mobile phase A to 95% mobile phase A; 11.1–14 min, hold at 95% mobile phase A. The effluent was connected to the mass spectrometer. We acquired triple quadrupole scans using nitrogen as collision gas. A specific set of multiple reaction monitoring transitions were monitored for each period in accordance with the metabolites eluted during this period.
High-performance liquid chromatography (HPLC; Waters Corporation) was used for determination of quercetin and kaempferol in the purified extract of M. oleifera leaves. 23
Animals
SPF Kunming male mice used in the present study were obtained from Beijing Vital River Laboratory Animal Technology Co., Ltd. The protocol of the animal experiment was approved by the Animal Ethical Committee of Tianjin Institute of Environmental and Operational Medicine (the ethics approval code is IACUC of AMMS-04-2021-004, February 19, 2021).
Weight-loaded swimming test
After acclimation for 5 days, 48 Kunming male mice were randomly divided into control (C), M. oleifera leaves crude extract (MLE), and M. oleifera leaf flavonoid-rich extract (MLFE) groups according to body weight. Distilled water was administrated to the C group. The MLE group and the MLFE group were treated with 340 mg/(kg·bw) MLE and 75 mg/(kg·bw) MLFE by gavage, respectively. On day 15, 30 min after the last dose of MLE and MLFE, the mice were loaded with lead weights (3% of their body weight) tagged to the tail to perform the forced swimming test.
0-min non-weight-bearing swimming test
After acclimation for 5 days, 48 Kunming male mice were randomly divided into C, swimming control (SC), and MLFE groups according to body weight. Distilled water was treated to the C and SC groups, and 75 mg/(kg·bw) MLFE was given to the MLFE group. The experiment lasted for 2 weeks, and body weight and food intake were recorded. On the 15th day, mice in SC and MLFE groups were subjected to a 90-min swimming experiment. Blood samples were collected from anesthetized mice in the end of experiment (retrobulbar venous plexus). The animals were sacrificed immediately after blood collection, and gastrocnemius, flexor digitorum profundus muscle, and adductor medial muscle were removed. The adductor medial muscle and flexor digitorum profundus muscle were stored in liquid nitrogen until use. The experimental flow chart is shown in Figure 1.
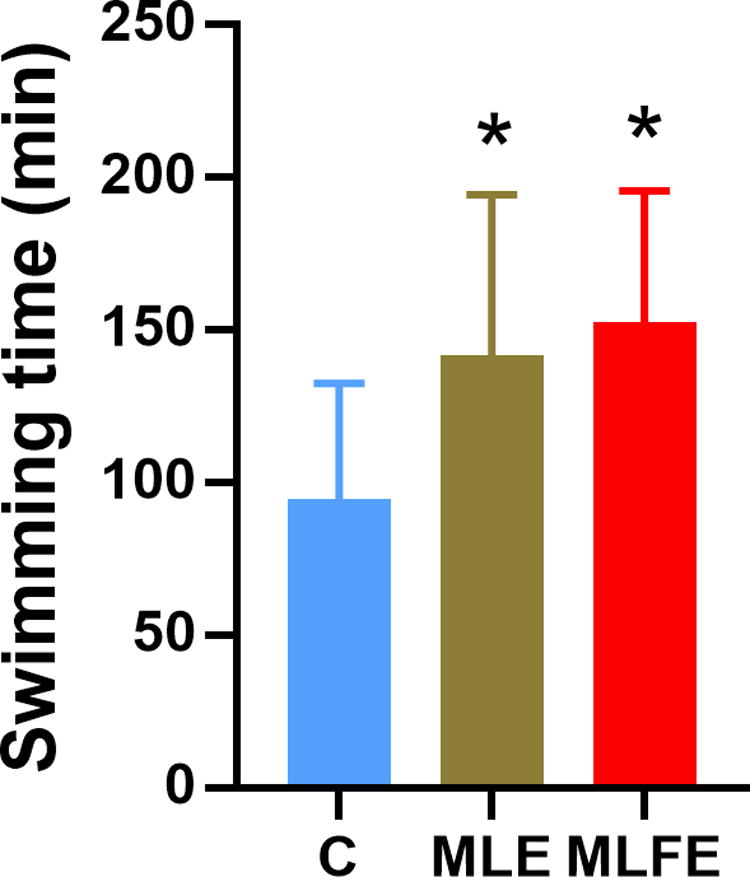
Swimming time of weight-loaded swimming test in mice (n = 16). *P < .05, compared with the control group. C: control group; MLE: M. oleifera leaves crude extract; MLFE: M. oleifera leaf flavonoid-rich extract.
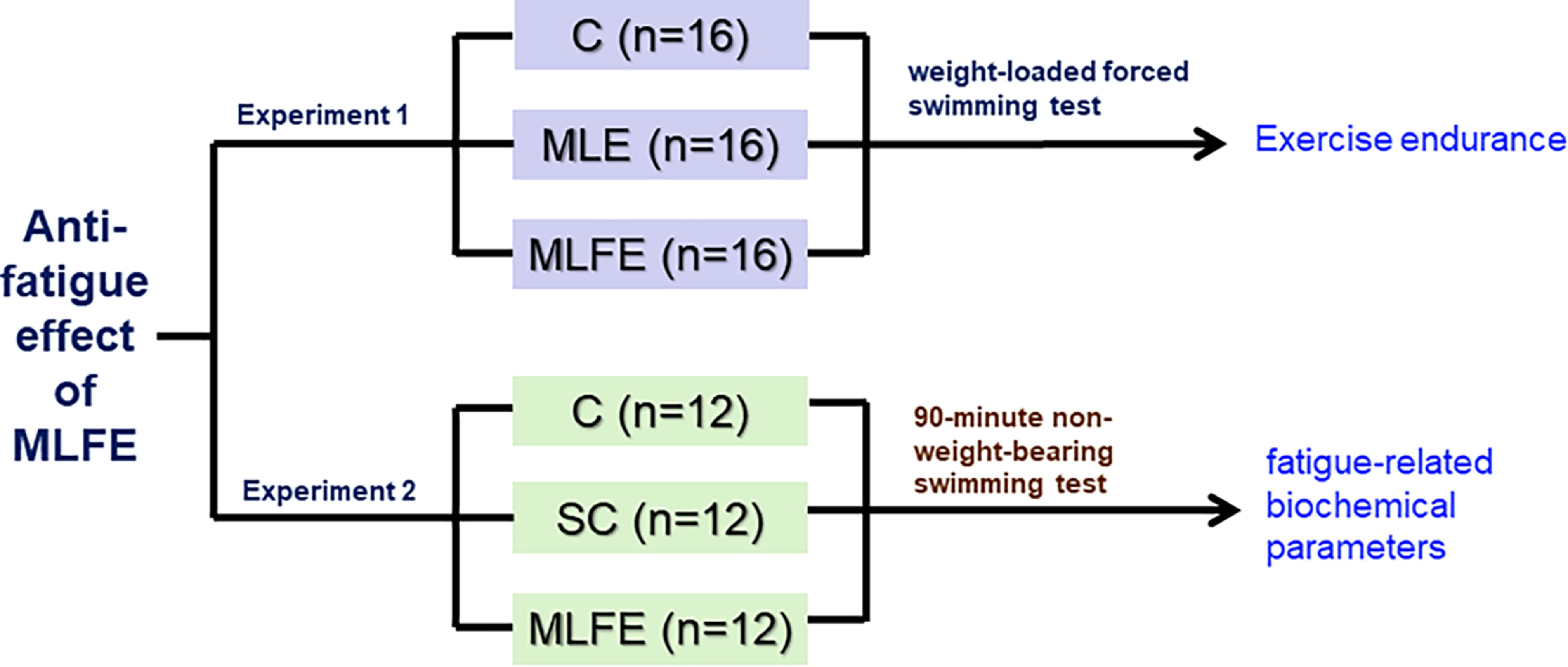
A flow chart of the experimental protocol.
Biochemical parameters in serum and muscle
The commercial regent kits obtained from the Biosino Bio-Technology and Science Inc. were used to assay the levels of serum glucose. The commercial regent kits were used to assay the levels of glycogen, glutathione (GSH), and superoxide dismutase (SOD) activity in the adductor medial muscle and serum lactate, blood urea nitrogen (BUN), and nonesterified fatty acid (NEFA) in the serum.
Quantitative polymerase chain reaction assay
The mRNA expression of GCK, PEPCK, Nrf2, and HO-1 in the flexor digitorum profundus muscle was detected using the quantitative polymerase chain reaction assay. The diethylpyrocarbonate, SYBR Green PCR Kit (item number, 4368577), and Trizol (item number, 15596018) were purchased from Thermo Fisher Scientific Inc. According to the GAPDH gene and the cycle threshold value of GCK, PEPCK, Nrf2, and HO-1, the relative mRNA expression of GCK, PEPCK, Nrf2, and HO-1 was analyzed according to the 2△Ct (GAPDH) − △Ct (target gene) method. The primers were self-designed by the primer design program Primer5. Primer sequences of GCK, PEPCK, Nrf2, and HO-1 are shown in Table 1.
The Sequences of the Primers
F, forward; R, reverse.
Enzyme-linked immunosorbent assay
Enzyme-linked immunosorbent assay kits obtained from JiangLai Biotechnology were used to determine the protein expression of GCK (item number, JL13834), PEPCK (item number, JL20462), Nrf2 (item number, JL18277), and HO-1 (item number, JL10795) in the adductor medial muscle tissues. Bicinchoninic acid (BCA) reagent kit was used to assay protein concentration of adductor medial muscle tissues.
Statistical analysis
All data in the present study are presented as means ± standard deviations. Significant differences were analyzed among groups using one-way analysis of variance, followed by Tukey’s test (SPSS version 22.0; IBM Corporation). P < .05 is considered a significant difference.
RESULTS
Flavonoids in the extract of M. oleifera leaves
The total flavonoids content of the MLFE was 54.7%, and the yield was (30.5 ± 5.0) % as calculated. The contents of flavonoids in the purified extract increased by 3.4 times in comparison with the crude extract.
UPLC-ESI-MS/MS analysis identified 83 kinds of flavonoids in the purified extracts of M. oleifera leaves, including 41 flavonols, 22 flavonoids, 10 flavonoid carboglycosides, 3 isoflavones, flavanols and dihydroflavanols, and 1 sinensetin. Supplementary Data S1 Among the 10 flavonoids with the highest content, there were 7 flavonols, 2 flavonoid, and 1 flavonoid carboglycoside. In the purified extracts, flavonoids were mainly present as glycosides. Among the flavonols, quercetin and kaempferol showed the highest relative content and existed mainly in the glycoside form (Table 2).
The Top 10 Flavonoids in the Purified Extract of M. oleifera Leaves (n = 3)
MW, molecular weight.
The quercetin and kaempferol aglycon content in MLFE
HPLC analysis showed that the contents of the quercetin and kaempferol aglycon were (304.8 ± 26.3) mg/g and (81.1 ± 3.9) mg/g in MLFE, respectively. However, the contents of quercetin and kaempferol aglycon were (3.4 ± 0.6) mg/g and (1.4 ± 0.2) mg/g in MLE, respectively.
Weight-loaded swimming test of mice after MLFE supplementation
Compared with the C group, the MLFE group exhibited a significantly longer exhaustive swimming time (Fig. 1) changed to Fig. 2. There was no statistically significant difference in the forced swimming time between MLE and MLFE groups. These findings demonstrate the promising anti-fatigue activity of MLFE.
Changes in biochemical parameters in serum and muscle
Swimming exercise significantly increased lactate and BUN concentrations in serum (Fig. 3a and b) in comparison with the C mice. Furthermore, MLFE supplementation reduced lactate and BUN in serum when compared with the SC mice (P < .05).
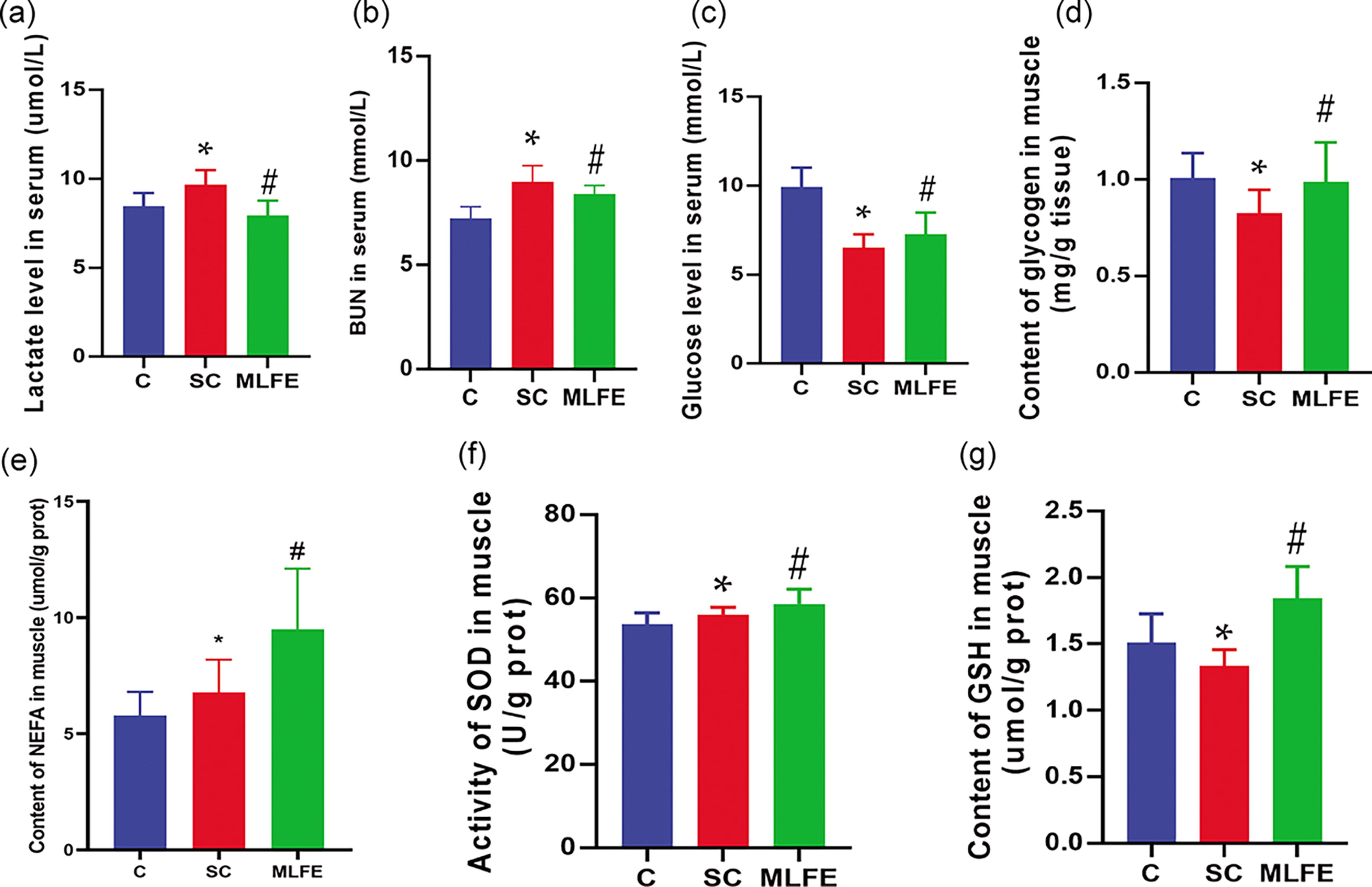
Biochemical indexes in serum and muscle.
Compared with the C mice, swimming exercise decreased serum glucose and muscle glycogen while it increased muscle NEFA in the SC mice (Fig. 3c, d, and e). After treatment with MLFE, serum glucose, muscle glycogen, and muscle NEFA were increased when compared with the SC mice.
Compared with the C mice, the activity of SOD in muscle was significantly increased, whereas the content of GSH in muscle was significantly decreased in the SC mice (Fig. 3f and g). In this study, compared with the SC mice, MLFE significantly enhanced the SOD activity and GSH in muscle (P < .05). These data suggest the promising anti-fatigue effect of MLFE on fatigue-related indicators.
mRNA expression of GCK, PEPCK, Nrf2, and HO-1 in the muscle
In order to further evaluate the potential mechanism of MLFE of the enhancement of the anti-fatigue activity on mice, GCK, PEPCK, Nrf2, and HO-1 mRNA expressions were assayed. Compared with the C mice, GCK, PEPCK, and Nrf2 mRNA expressions were significantly downregulated, whereas HO-1 was upregulated in SC mice (P < .05) (Fig. 4). GCK, PEPCK, Nrf2, and HO-1 mRNA expressions were increased after MLFE supplementation in comparison with the SC mice (P < .05) (Fig. 4).
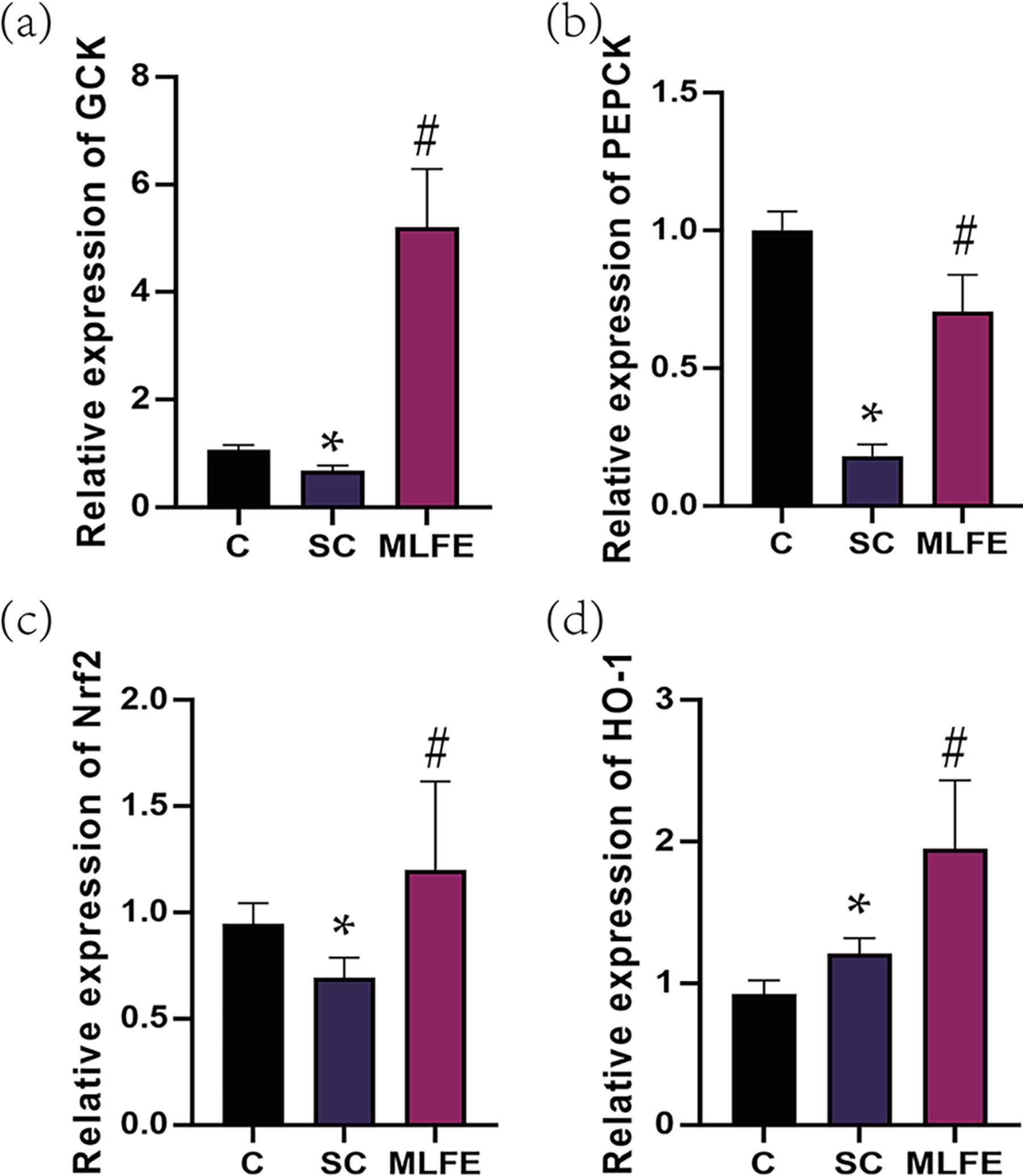
mRNA expression of GCK, PEPCK, Nrf2, and HO-1 in muscle of mice.
Protein expression of GCK, PEPCK, Nrf2, and HO-1 in the muscle
After swimming, GCK, PEPCK, Nrf2, and HO-1 protein expressions were significantly suppressed in muscle. GCK, PEPCK, and Nrf2 protein expressions were significantly upregulated after supplemented with the purified extract (Fig. 5). Therefore, these data demonstrated that MLFE treatment exerted the anti-fatigue effects possibly by the GCK/PEPCK and Nrf2/HO-1 signaling pathway.
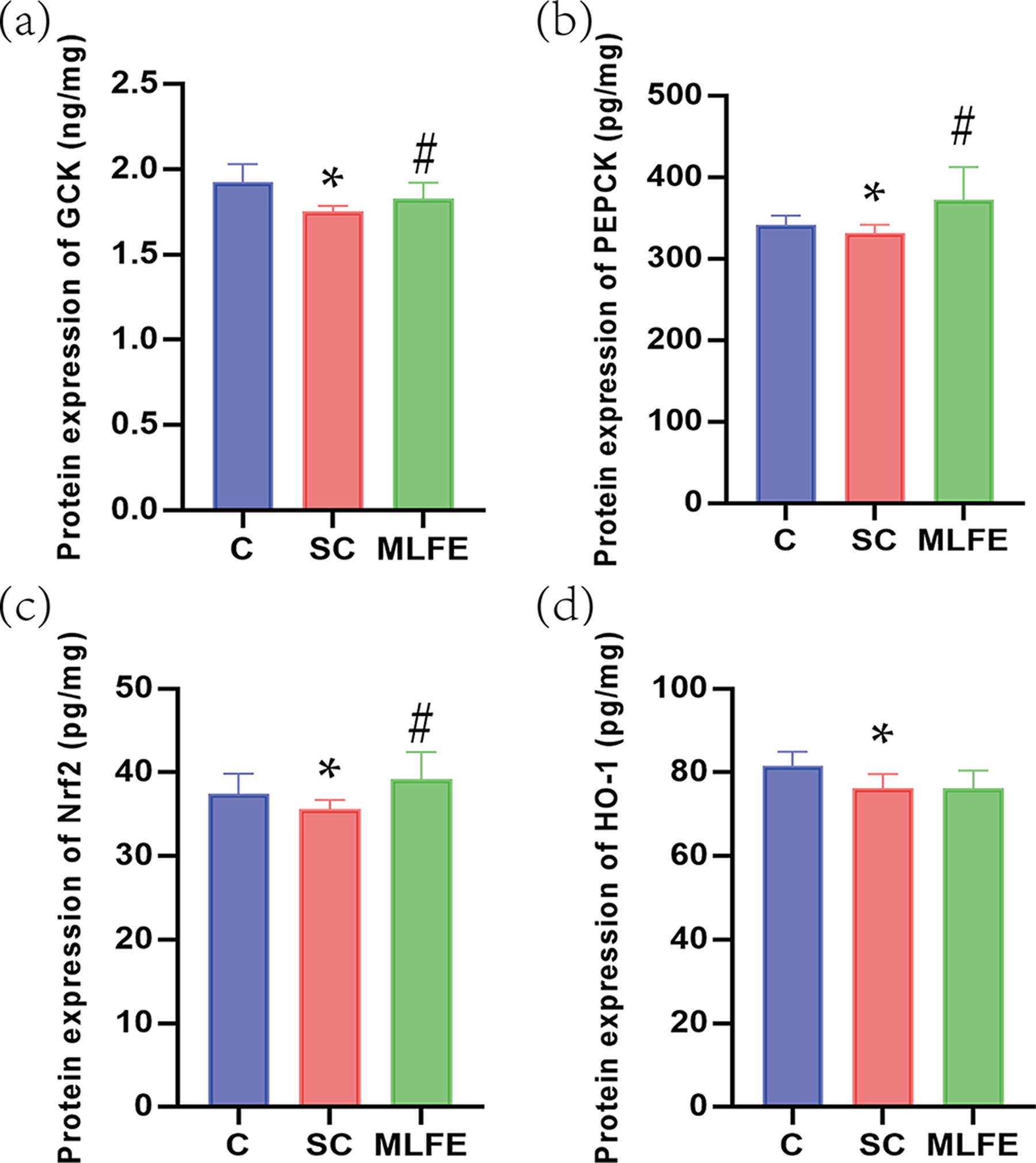
Protein expression of GCK, PEPCK, Nrf2, and HO-1 in the muscle of mice.
DISCUSSION
This study investigated the anti-fatigue capacity and underlying mechanism of MLFE. MLFE extended the forced swimming time and enhanced antioxidants and energy storage, suggesting that MLFE contributes to enhancement of exercise strength and endurance.
Post-exercise-induced physical fatigue can be assayed by some metabolic wastes, including lactate and BUN in serum, after exercise. 24,25 Lactate is produced during glycolysis, which is the key indicator of physical fatigue. 26 After exercise, anaerobic glycolysis accelerates the production of lactic acid by decomposing muscle glycogen and leads to a rapid decrease in pH value of muscle. 27,28 Carbohydrates and lipids cannot supply enough energy to muscle after strenuous exercise and then protein breakdown and amino acid catabolism to compensate for excess energy expenditure. BUN was produced during proteins and amino acids catabolism. 29 BUN has a positive correlation with athletic performance and endurance, which means that the worse the body is adapted for athletic performance, the more significantly the BUN levels increase. 30 In the present study, MLFE administration attenuated the increases in lactate and BUN levels after exercise. These data indicate that MLFE could decrease fatigue‐related metabolites after exercise.
Glycogen is a direct energy source for exercise and provides enough energy for muscle contraction. Exercise activates skeletal muscle AMPK signaling. 31 Acute activation of AMPK leads to glycogen degradation. 32 Our previous study also showed that exercise activates AMPK in the muscle and reduces muscle glycogen storage. 15 During exercise, muscle glycogen decomposition is accelerated, and glucose is released into blood for performance maintenance. GCK is important for glucose homeostasis and catalyzes the conversion of glucose into glucose-6-phosphate and triggers glucose metabolism in order to provide energy during exercise. PEPCK is a critical enzyme in gluconeogenesis and promotes the conversion of lactate, pyruvic acid, and glucogenic amino acids into glucose. 33 PEPCK also plays an important role during exercise because it prolongs the endurance time in rats. 34,35 The water extracts of Sonchus arvensis L., which was rich in flavonoids, improved hepatic glycogen synthesis via increasing GCK and PEPCK mRNA expression in exercise-trained mice. 36 Silymarin, a flavonoid, ameliorated exercise-induced gluconeogenesis through the regulation of PEPCK in the liver of the exercise-trained mice. 37 During endurance exercise, fatty acid is the primary energy sources for skeletal muscle contraction. 38 NEFA in muscle is the predominant sources of muscle fatty acids, in which NEFA is an important index to assess physical fatigue. 39 In the present study, the results demonstrated that MLFE treatment increased muscle glycogen and PEPCK and GCK mRNA expression, as well as NEFA in muscle, indicating that MLFE may play an antifatigue effect by improving energy storage in mice.
The antioxidant system is also associated with exercise-induced fatigue. 40 GSH, an important antioxidant, attenuates lipolysis and acidification in muscle after exercise, improving exercise performance. 41 Antioxidant enzyme could prevent exhaustive exercise-induced oxidative damage. 42 After exhaustive exercise, reactive oxygen species accumulation induces oxidative stress on muscle. 43 –46 SOD promotes the disruption of superoxide anion to hydrogen peroxide. 47 Enhancement of SOD activity could improve exercise performance and reduce physical fatigue. HO-1 is the rate-limiting enzyme for heme degradation and prevents oxidative stress. 48 –52 Nrf2-mediated expression of SOD contributes to the maintenance of redox homeostasis. 53 –55 In this study, MLFE treatment significantly increased GSH, SOD, and HO-1 and Nrf2 mRNA expressions. These data indicate that MLFE may enhance the antioxidant enzyme activity and upregulate antioxidant-related genes.
Several studies have provided evidence supporting the role of quercetin on anti-fatigue action. A previous study reported that quercetin supplementation promotes anti-fatigue capacity by increasing muscle glycogen and serum NEFA, enhancing SOD activity in gastrocnemius muscle. 56 Another study demonstrated that quercetin improves energy metabolism, especially glucose and NEFA metabolism, which may contribute to enhance exercise tolerance. 57 For these reasons, quercetin would be one of the important constituents for the anti-fatigue capacity of MLFE.
CONCLUSIONS
In the present study, we found that MLFE could upregulate the expression of GCK, PEPCK, and Nrf2 and enhance SOD and GSH in muscle. These findings suggest that this purified extract played its anti-fatigue role via improving energy metabolism and antioxidant function. It is further confirmed that flavonoids, such as quercetin, may be an effective anti-fatigue component. However, it is worth noting that we did not measure the level of flavonoids in serum and muscle, which represents a limitation of our study.
Footnotes
AUTHOR DISCLOSURE STATEMENT
No competing financial interests exist.
FUNDING INFORMATION
This work was supported by
SUPPLEMENTARY MATERIAL
Supplementary Data S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
