Abstract
HemoHIM is a standardized medicinal herbal preparation consisting of extracts of Angelica gigas Nakai, Cnidium officinale Makino, and Paeonia lactiflora Pallas that possesses immune regulatory activities. This study aimed to research the potential antioxidant effects of HemoHIM and its capacity for reducing fatigue in aged mice subjected to forced exercise. After administering HemoHIM 125 (500 mg/kg orally) for 4 weeks in 8-month-old female C57BL/6 mice (4 groups of 10 mice), various parameters were evaluated. The analyses revealed that HemoHIM enhanced swimming time and grip strength. In addition, it significantly reduced serum lactate levels and increased liver glutathione peroxidase (GPx) levels after exercise challenge. The expression levels of antioxidant enzymes and factors, including nuclear factor erythroid 2-related factor-2 (Nrf-2), heme oxygenase 1, superoxide dismutase, GPx, and glutathione reductase, were significantly higher in liver and muscle tissues of mice treated with HemoHIM. These results indicate that HemoHIM might function as an anti-fatigue and antioxidant agent by modulating the Nrf-2 signaling pathway.
INTRODUCTION
Fatigue is an unstable physiological condition that includes tiredness and weakness and can be physical, mental, or a combination of both. 1 Fatigue can also be caused by illness, but it often occurs in daily life due to lack of sleep or stress, and can be accompanied by physical or mental fatigue. 1,2
Numerous theories regarding the mechanisms related to physical fatigue have been proposed, such as the radical theory and exhaustion. According to the radical theory, the accumulation of reactive free radicals promote oxidative stress. Increased oxidative stress during these processes can impair cellular functions, leading to aging and various physiological abnormalities. 3 Free radicals cause physical fatigue during excessive physical activity. Reactive oxygen species (ROS)-induced lipid peroxidation can impair the integrity of the cell membrane resulting in oxidative fatigue in skeletal muscle, 4,5 which can lead to cell aging and death. Both oxidative stress and damage affect aging-associated diseases, such as cancer, diabetes mellitus, and hypertension. 6,7
Skeletal muscle fibers continuously produce ROS during muscle contractions. In particular, ROS levels increase during intense exercise, which contributes to muscle fatigue. Thus, the antioxidants that regulate ROS levels may be the most important factors in anti-fatigue mechanisms. 8
Nuclear factor erythroid 2-related factor-2 (Nrf-2) is an important marker of antioxidant signaling. Nrf-2 modulates the expression of genes encoding phase II detoxification enzymes and antioxidants such as heme oxygenase 1 (HO-1). Nrf-2 is negatively regulated by Kelch-like ECH-associated protein 1 (Keap-1), which is also associated with the HO-1-mediated antioxidant pathway. 9,10
HO-1 protects cells exposed to oxidizing agents by catalyzing the oxidation of heme to biologically active agents, for example, Fe2+ and CO. The final products produced from heme catabolism neutralizes intracellular ROS and exerts an antioxidant effect. 11 Several studies have reported that the Nrf2 pathway is associated with a protective role against oxidative stress and fatigue.
Hot water extracts of the roots of Angelica gigas Nakai (Angelica Radix), Cnidium officinale Makino (Cnidium Rhizoma), and Paeonia lactiflora Pallas (Paeonia Radix) were used to prepare HemoHIM and a polysaccharide fraction. 12,13 All three herbs are traditional Korean plants used in herbal medicine with several pharmacological activities, such as antioxidant, anti-inflammation, and antimicrobial effect. 14 –16 HemoHIM is a standardized preparation that influences on the immunoregulation and causes the maturation and activation of dendritic cells. 17,18
The key compounds are gallic acid, nodakenin, chlorogenic acid, and paeoniflorin. HemoHIM and its compounds have been studied to control the immunity, 17,18 exert anti-inflammatory effects, 19 and protect against oxidative stress and H2O2-induced apoptosis. 20 In our previous study, HemoHIM improved exercise ability and decreased fatigue and oxidation in ICR mice. 21 However, there are no studies on the anti-fatigue effects of HemoHIM in aged mice. In this study, we investigated the antioxidant and anti-fatigue effects of HemoHIM on fatigue in aged mice.
MATERIALS AND METHODS
Preparation of HemoHIM
HemoHIM was manufactured with reference to Jo et al. 22 The standardized HemoHIM contained nodakenin (50–150 mg/100 g), chlorogenic acid (25–60 mg/100 g), and paeoniflorin (200–400 mg/100 g). It was manufactured by HK Kolmar BNH Co., Ltd. (Batch No. HHH010). The roots of A. gigas, C. officinale, and P. lactiflora were extracted by the hot water extraction method. A portion of the extracted material was precipitated with 95% ethanol to obtain a polysaccharide fraction. Finally, the HemoHIM with the polysaccharide fraction was concentrated to a solid content of 30% ± 3% and freeze dried.
Animals and experimental protocol
Eight-month-old female C57BL/6 mice (DooYeol Biotech, Korea) were used (4 groups of 10 mice). The animals were acclimated for 1 week and were housed in a climate-controlled facility with a temperature of 22°C ± 3°C and humidity of 50% ± 5% and a 12 h light–dark cycle. Mice were divided into the following groups: (1) control group, (2) HemoHIM 125 mg/kg, (3) HemoHIM 500 mg/kg, and (4) creatine 625 mg/kg. The control group was orally administered only 0.5% carboxymethylcellulose, whereas the other group was orally administered HemoHIM at the assigned concentration for 4 weeks.
Motor behavior analysis using a grip strength test
The mice were pretrained on a grip strength test twice a week before the test on 0 day. The grip strength test was performed once a week. The grip strengths of the control and the HemoHIM groups were measured using a grip strength meter (model JD-A-22; Jeungdo Bio & Plant, Korea). The mice tails were suspended perpendicular to the grid, and it was able to hold the metal grid of the meter with all four feet until the front foot reached the end of the grid. The tests were performed in triplicate and the results are expressed as the average of the measured data in kilograms-of-force units.
Motor behavior analysis using a forced swimming test
On day 0, the mice received pretraining using the forced swim test (FST) twice a week before the test. The mice were forced to swim in a pool filled with water at 25°C ± 1°C. There were weights added at 5% of their body weight to increase their exercise load. Swimming time was measured until the noses of the mice began to sink below the surface and remained at least 5 sec during exhaustion. FST was performed once a week after the grip test, and tissue analysis was performed on the last day.
Biochemical analysis
Serum was collected by cardiac puncture and centrifuged at 158 g for 15 min (Smart R17 Centrifuge; Hanil Scientific, Inc., Korea). Serum
Hepatic assays
Liver tissue (10 mg) was homogenized (T25 digital Ultra-Turrax; IKA®, Germany) and the homogenized tissues were centrifuged at 158 g and 4°C. Liver malondialdehyde (MDA), glutathione peroxidase (GPx), and catalase (CAT) were measured using a lipid peroxidation (MDA) test kit (ab118970), GPx test kit (ab102530), and CAT activity test kit (ab83464; Abcam).
RNA isolation and reverse transcription–polymerase chain reaction in vivo
Total RNA was extracted (Qiagen, Germany) from liver and muscle samples, and the extracted RNA was quantified using NanoDrop (Thermo Fisher Scientific, Inc., USA). cDNA was synthesized using a high-capacity cDNA reverse transcription kit (Applied Biosystems, USA), and then amplified using AccuPower PCR Premix (Bione, Korea). The primers used are shown in Table 1. Electrophoresis was performed using 1.8% agarose gel containing ethidium bromide. β-Actin was used as a control, and the results were analyzed using ImageJ software (NIH, USA).
Primer Sequences Used for Reverse Transcription–Polymerase Chain Reaction
Statistical analysis
All experimental data are presented as means ± standard errors of the means. Data were analyzed by a one-way analysis of variance (ANOVA) and Duncan's multiple range tests using GraphPad Prism 5.0 (GraphPad Prism Software, Inc., USA). A P value <.05 was considered significant.
RESULTS
HemoHIM enhanced swimming time and grip strength in aged mice
Forced swimming and grip strength tests are among the most frequently used models for evaluating fatigue in animal studies. 23 Swimming time and grip strength were measured in the FST to confirm the anti-fatigue effect of HemoHIM. Compared with the control group, the HemoHIM 500 mg/kg and creatine group had longer swimming times (P < .05) and stronger grip strength (P < .01) (Fig. 1). In addition, there was no significant difference in body weight among all groups (Fig. 2).
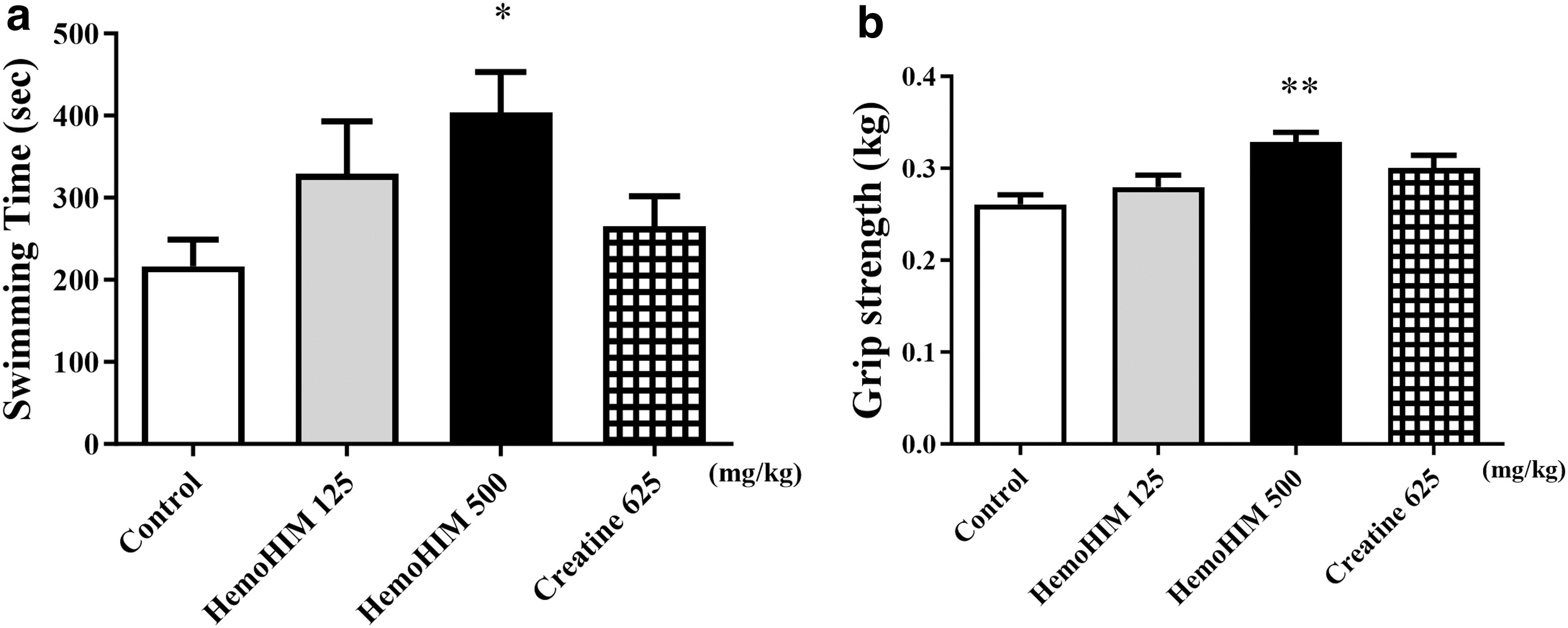
Effect of HemoHIM on swimming time to exhaustion in the FST
Effect of HemoHIM on body weight changes of mice.
HemoHIM decreased serum lactate levels after forced exercise
Serum lactate levels were measured after forced exercise to evaluate the effects of HemoHIM on fatigue. Lactate levels in the HemoHIM-administered mice decreased dose-dependently compared with those in the control group (HemoHIM 125 mg/kg, P < .05; HemoHIM 500 mg/kg, P < .05; Fig. 3).
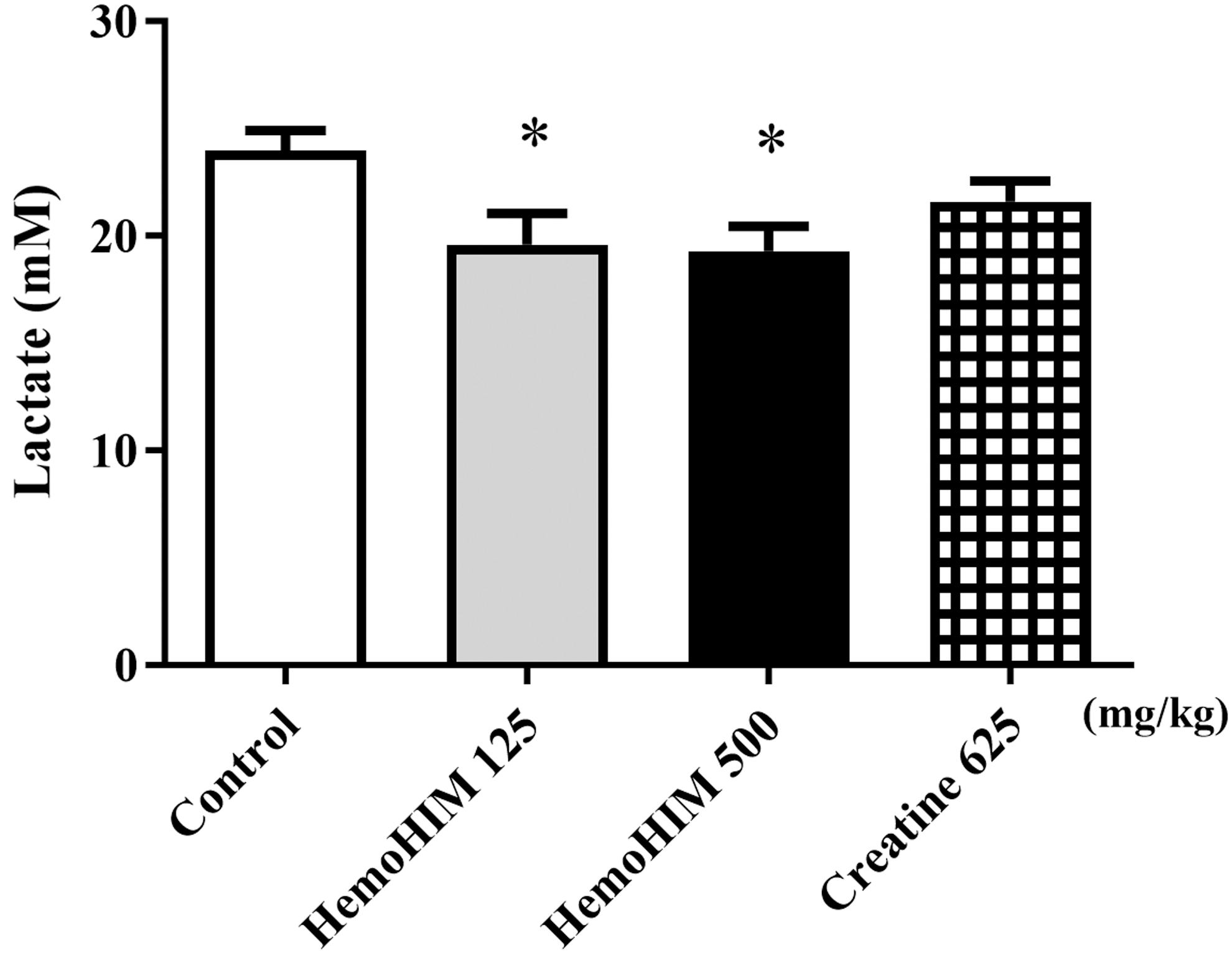
Effect of HemoHIM on lactate levels in serum after exercise challenge. Data are expressed as mean ± SEM. Comparisons were made between control and HemoHIM groups (125 and 500 mg/kg). Significant difference from control group (*P < .05). HemoHIM, standardized fraction of water extracts of Angelica gigas Nakai, Cnidium officinale Makino, and Paeonia lactiflora Pallas.
HemoHIM increased GPx expression in the liver after forced exercise
Antioxidant activity was determined on the basis of liver CAT, MDA, and GPx levels. In the HemoHIM group, the activity of CAT showed a tendency to increase compared with control (Fig. 4a). MDA concentrations were lower in the HemoHIM groups, but the differences were not statistically significant (Fig. 4b). GPx concentrations in the HemoHIM 500 mg/kg group were significantly higher when compared with controls (Fig. 4c).
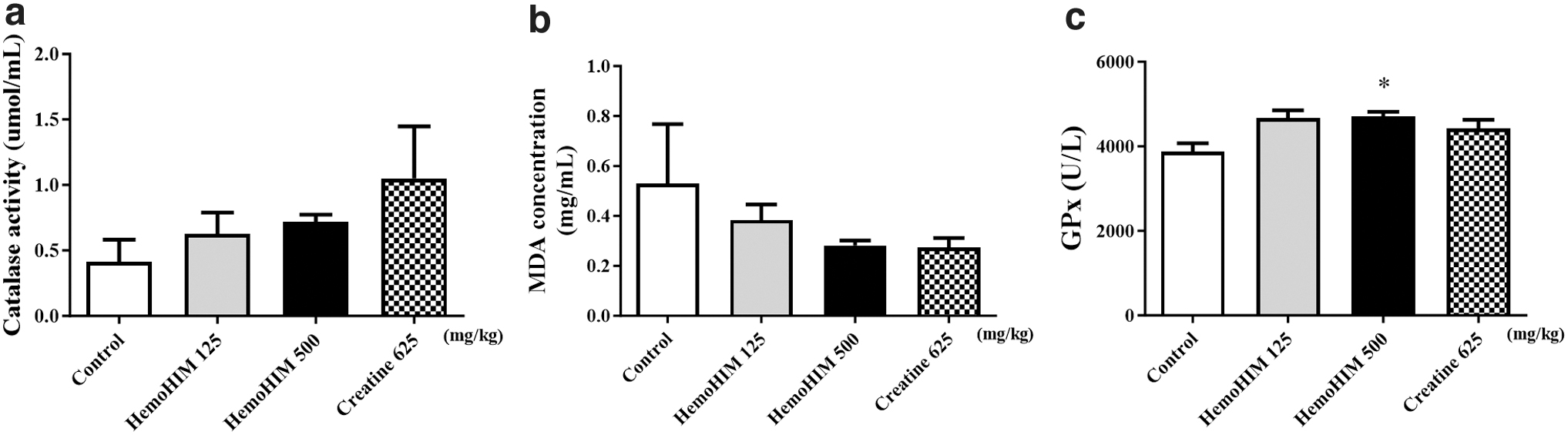
Effect of HemoHIM on CAT activity
HemoHIM upregulated the Nrf-2/HO-1 pathway and regulated other antioxidant factors in the liver after forced exercise
To investigate the antioxidant and anti-fatigue effects of HemoHIM, mRNA expression of genes related to the Nrf-2/HO-1 pathway was measured in liver tissues. Nrf2 levels were elevated in the HemoHIM group compared with the control group (Fig. 5a). Also, HO-1 expressions in the HemoHIM groups were significantly upregulated in liver tissue (HemoHIM 125 mg/kg; HemoHIM 500 mg/kg, P < .01; Fig. 5b).
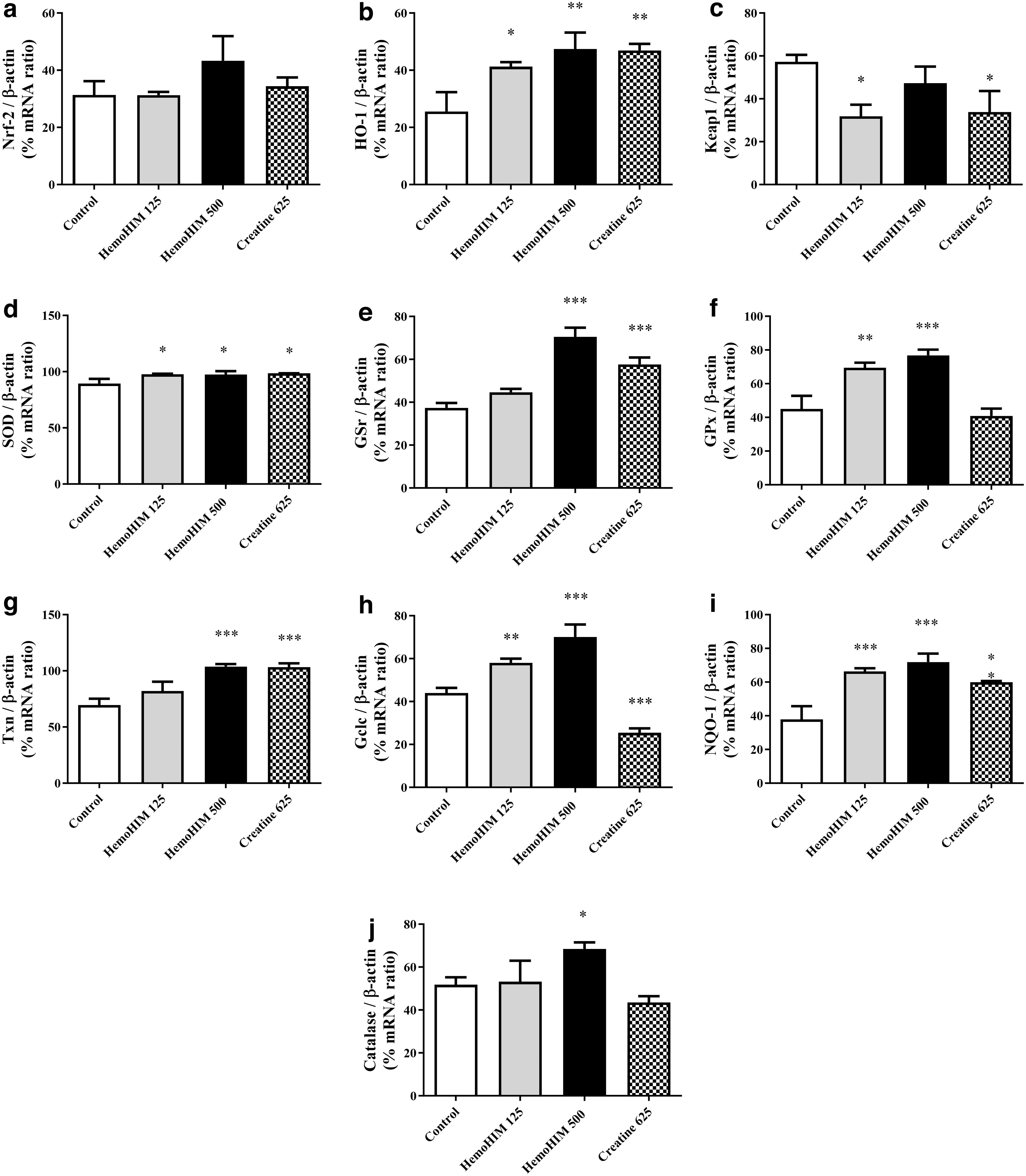
Effects of HemoHIM on
In addition, Keap-1 expression in the HemoHIM 125 mg/kg group was downregulated compared with that in the control group (P < .05; Fig. 5c); however, superoxide dismutase (SOD) levels in both HemoHIM groups were upregulated compared with controls (HemoHIM 125 mg/kg; P < .05, HemoHIM 500 mg/kg; P < .05; Fig. 5d). In addition, there was a statistically significant increase in glutathione reductase (GSr) levels in the HemoHIM 500 mg/kg group compared with the control group (P < .001; Fig. 5e).
The expression of GPx and thioredoxin (Txn) was upregulated compared with that in the control group (GPx: HemoHIM 125 mg/kg, P < .01; HemoHIM 500 mg/kg, P < .001; Txn: HemoHIM 500 mg/kg, P < .001; Fig. 5f, g). Furthermore, the expression of glutamate cysteine ligase catalytic subunit (Gclc) was significantly upregulated compared with that in the control group (HemoHIM 125 mg/kg; P < .01, HemoHIM 500 mg/kg; P < .001; Fig. 5h). HemoHIM 500 mg/kg significantly induced the expression of the antioxidant factors NADPH quinone oxidoreductase-1 (NQO-1; HemoHIM 125 mg/kg; P < .001, HemoHIM 500 mg/kg; P < .001; Fig. 5i) and CAT (HemoHIM 500 mg/kg; P < .05; Fig. 5j) compared with that in the control group.
HemoHIM upregulated the Nrf-2/HO-1 pathway and regulated other antioxidant factors in muscle after forced exercise
To determine the anti-fatigue effect of HemoHIM on exercise performance in mice, Nrf-2/HO-1 pathway genes and antioxidant factors were measured in skeletal muscle tissue after the final exercise. Nrf-2 expression in the HemoHIM groups was significantly elevated compared with that in the control group (HemoHIM 125 mg/kg, P < .001; HemoHIM 500 mg/kg, P < .001) (Fig. 6a). Similarly, HO-1 gene expression was upregulated in the HemoHIM 500 mg/kg group after exercise compared with that in the control group (P < .01) (Fig. 6b).
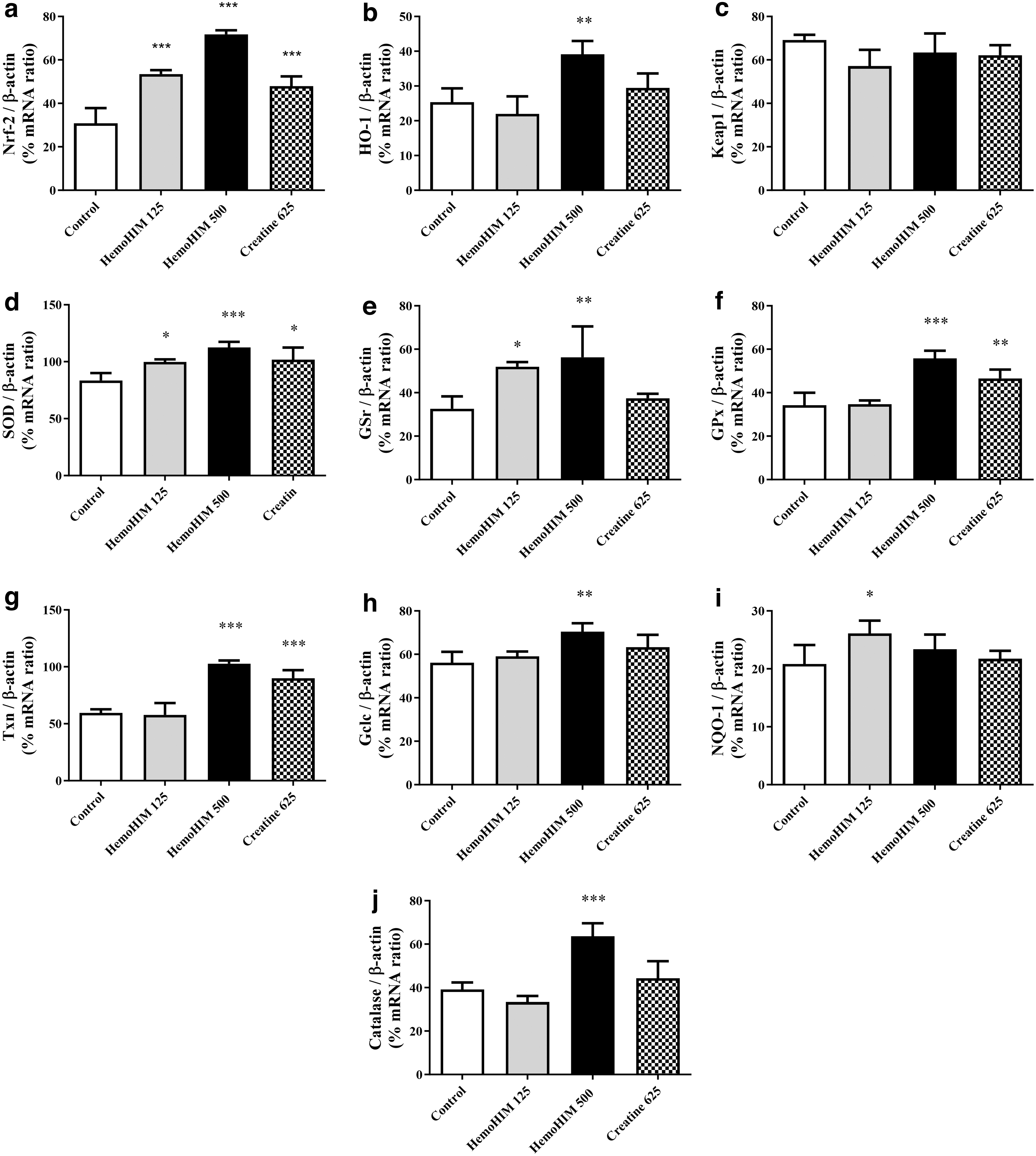
Effects of HemoHIM on
In the HemoHIM group, Keap-1 levels showed a tendency to decrease (Fig. 6c). However, SOD levels were significantly increased in the HemoHIM 125 mg/kg and HemoHIM 500 mg/kg groups compared with those in the control group (HemoHIM 125 mg/kg, P < .05; HemoHIM 500 mg/kg, P < .001) (Fig. 6d). GSr expression was significantly upregulated by HemoHIM (HemoHIM 125 mg/kg, P < .05; HemoHIM 500 mg/kg, P < .01; Fig. 6e).
In addition, GPx levels were higher in the 500 mg/kg HemoHIM group than in the control group (P < .001; Fig. 6f), and Txn levels showed a dose-dependent increase in the HemoHIM groups (500 mg/kg; P < .001; Fig. 6g). Gclc expression was significantly upregulated in the HemoHIM 500 mg/kg group compared with controls (P < .01; Fig. 6h). Finally, NQO-1 expression in the HemoHIM 125 mg/kg group (P < .05; Fig. 6i) and CAT expression in the HemoHIM 500 mg/kg group (P < .001; Fig. 6j) were significantly higher than the corresponding values in the control group.
DISCUSSION
In this study, we explored the effect of HemoHIM on high-intensity exercise-induced fatigue in aged mice. The treated mice exhibited enhanced performance in forced swimming and grip strength tests, thus demonstrating the potential anti-fatigue activity of HemoHIM.
Fatigue is a common physiological response observed in various conditions, including cancer, aging, and depression. 24 It can be categorized as either mental or physical fatigue. Physical fatigue, which arises from intense exercise or demanding physical labor, manifests as physical abnormalities, including sleep disturbances and lethargy. High-intensity exercise can increase the production of ROS and oxygen consumption, resulting in elevated oxidative stress leading to oxidative damage. 25
Studies such as those by Ma et al. 26 and Oliveira et al. 27 have explored drugs such as melatonin and carvedilol, which possess strong antioxidant properties but may entail serious side effects. Recently, several studies have highlighted the potential of natural antioxidant medicinal plants and herbs in reducing fatigue and exercise-induced oxidative damage. 28,29 Therefore, one strategy to suppress fatigue involves eliminating or inhibiting the production of fatigue-related metabolites during exercise.
In this study, we found increased CAT and GPx levels and decreased MDA levels in the liver of HemoHIM-treated mice, suggesting that HemoHIM could alleviate fatigue. Exercise-induced protein oxidation leads to the production of ROS and MDA, accompanied by an increase in the levels of primary antioxidant enzymes such as CAT and GPx. 30 CAT can reduce fatigue and alleviate other physiological abnormalities by breaking down H2O2 into H2O and O2, whereas GPx is responsible for reducing H2O2 into water and alcohol. 31,32
Accumulation of ROS and MDA, as well as reduced antioxidant enzyme levels, has been reported in chronic fatigue. Therefore, it can be reasoned that reversing these effects can help combat fatigue. We also found that HemoHIM reduced blood lactate levels, potentially enhancing the anti-fatigue effect. Blood lactate accumulation can lower the pH value of the muscle tissue or blood, potentially causing side effects in physiological responses. 23
Furthermore, HemoHIM regulated the expression activities of Nrf-2, HO-1, NQO-1, Txn, and Gclc by reverse transcription–polymerase chain reaction (PCR) analysis of liver and muscle tissue. In addition, HemoHIM activated the expression of antioxidant-related enzymes and factors such as SOD, GSr, GPx, and CAT. The Nrf-2 signaling pathway is crucially involved in oxidative stress. 33 Under normal conditions, Nrf-2-dependent transcription is repressed by the negative regulator Keap-1.
However, when cells or tissues are exposed to oxidative stress environments, Nrf-2 avoids Keap-1 mediated inhibition and enters the nucleus, activating genes encoding antioxidant and phase II detoxifying enzymes (Gclc), detoxifying enzymes (GSr and NQO-1), and antioxidant enzymes (CAT, GPx, SOD, and HO-1). 34 Therefore, activation of the Nrf-2 signaling pathway is vital for antioxidation.
In summary, this study demonstrated that HemoHIM exhibits an anti-fatigue effect in aged mice subjected to forced exercise. This anti-fatigue effect was most likely caused by the increased antioxidant activity of the enzyme system, and the removal free radicals resulting in decreased oxidative damage.
In conclusion, this study confirmed that HemoHIM exhibits anti-fatigue effects in aged mice. The observed effects included a reduction in lactate and MDA concentrations, increased GPx and CAT activity, and improved exercise performance. The underlying mechanism of these anti-fatigue effects is thought to be associated with the antioxidant properties of HemoHIM, exerted through the activation of the Nrf-2/HO-1 signaling pathway and various antioxidant factors such as SOD, GSr, GPx, Txn, Gclc, NQO-1, and CAT.
The potential anti-fatigue activity of HemoHIM could benefit both the general population and elderly individuals experiencing fatigue. Although the exact mechanisms of HemoHIM are yet to be elucidated, this study suggests the use of HemoHIM as an anti-fatigue agent by showing scientific evidence supporting anti-fatigue outcomes with HemoHIM consumption.
ETHICAL APPROVAL STATEMENT
All animal experiments were approved by the Institutional Animal Care and Use Committee (IACUC) of the HK Korea Kolmar Animal Ethics Committee (19-KBH-F-01) and were followed by national and institutional guidelines.
Footnotes
AUTHORs' CONTRIBUTIONS
Conceptualization and writing—review and editing by H.S.L. Supervision and methodology by S.-K.K. Methodology, formal analysis, investigation, resources, data curation, and writing—original draft by D.-A.K. Methodology by Y.S.K. Writing—original draft by S.-B.B.
AUTHOR DISCLOSURE STATEMENT
No competing financial interests exist.
FUNDING INFORMATION
This study was supported by a grant from HK Kolmar BNH Co., Ltd., Korea.
